Organic Compounds Compounds that contain CARBON are called

Organic Compounds ¡ Compounds that contain CARBON are called organic ¡ Macromolecules are large organic molecules 1

Carbon (C) ¡ Carbon has 4 electrons in outer shell. ¡ Carbon can form covalent bonds with as many as 4 other atoms (elements). ¡ Usually with C, H, O or N. N ¡ Example: CH 4(methane) 2

Hydrocarbons ¡ The simplest carbon compounds … Contain only carbon & hydrogen atoms

Macromolecules ¡ Large organic molecules. ¡ Also called POLYMERS ¡ Made up of smaller “building blocks” called MONOMERS Examples: ¡ 1. Carbohydrates ¡ 2. Lipids/Fats ¡ 3. Proteins ¡ 4. Nucleic acids (DNA and copyright cmassengale RNA) ¡ 4
Giant Molecules - Polymers ¡ Large molecules are called polymers Polymers are built from smaller molecules called monomers Biologists call them all macromolecules
Examples of Polymers ¡ Proteins Lipids Carbohydrates Nucleic Acids

Carbohydrates ¡ Carbohydrates include: ¡ Small sugar molecules in soft drinks ¡ Long starch molecules in pasta and potatoes

I. Carbohydrates ~Carbohydrates are sugars~ *Hint: if you see “ose” on the end of a word, it’s probably a sugar (example – glucose) Categories of Sugars: 1. Monosaccharides: single sugars (mono = one, saccharide = sugar) Ex: Glucose (C 6 H 12 O 6) 2. Disacharcides: two sugars that are covalently bonded Ex: Lactose (found in milk) is galactose + glucose
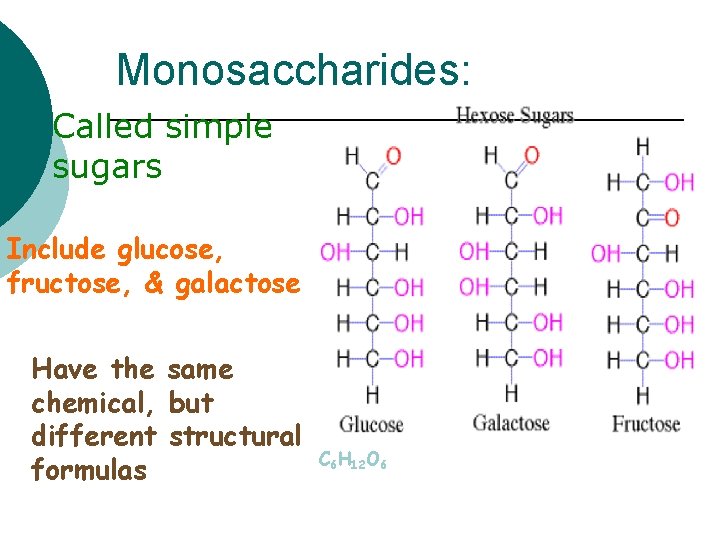
Monosaccharides: ¡ Called sugars simple Include glucose, fructose, & galactose Have the same chemical, but different structural formulas C 6 H 12 O 6

Monosaccharides Glucose is found in sports drinks Fructose is found in fruits Honey contains both glucose & fructose Galactose is called “milk sugar” -OSE ending means SUGAR
Function of Carbohydrates ¡ Monosaccharides are the main fuel that cells use for cellular work ¡ Cellular energy ATP

Proteins ¡ Proteins are polymers made of monomers called amino acids All proteins are made of 20 different amino acids linked in different orders Proteins are used to build cells, act as hormones & enzymes, and do much of the work in a cell

Types of Proteins
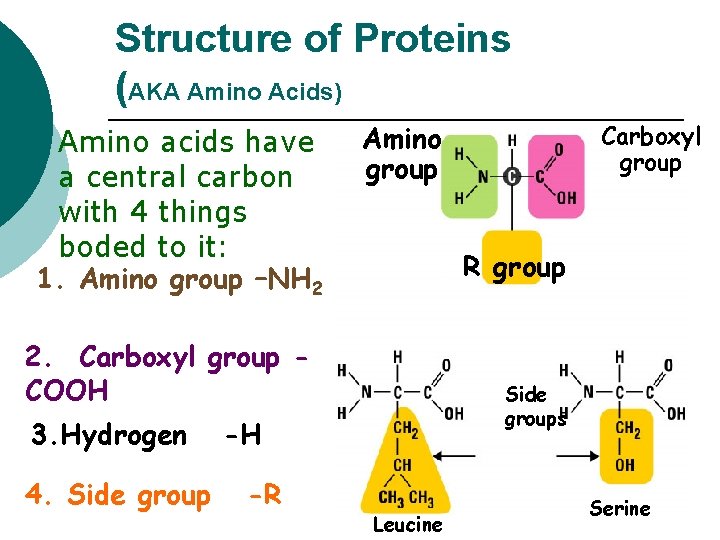
Structure of Proteins (AKA Amino Acids) Amino acids have a central carbon with 4 things boded to it: 1. Amino group –NH 2 ¡ Amino group R group 2. Carboxyl group COOH 3. Hydrogen 4. Side groups -H -R Carboxyl group Leucine Serine

PROTEIN Mmm, beefy! Vegetarians have more fun!
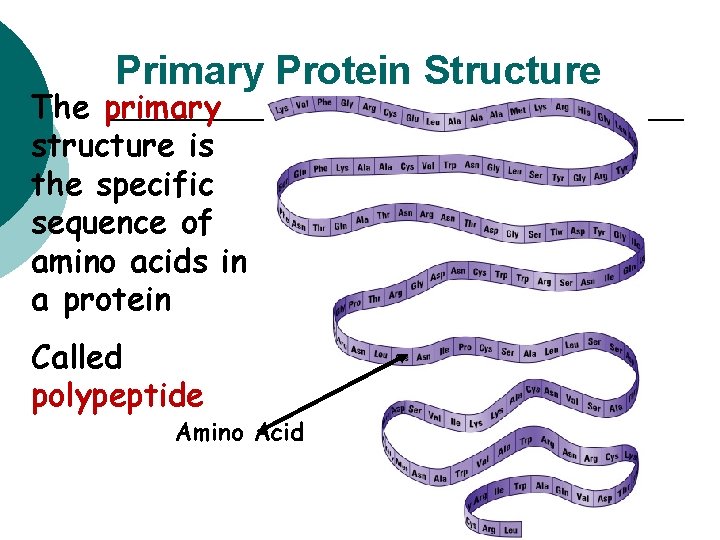
Primary Protein Structure The primary structure is the specific sequence of amino acids in a protein Called polypeptide Amino Acid

Proteins Continued… There are twenty amino acids, acids and thousands of proteins. Q: How can that be? A: It’s like the alphabet – 26 letters, but millions of words. If we combine the amino acids in different ways we can form different proteins. Amino acids form proteins
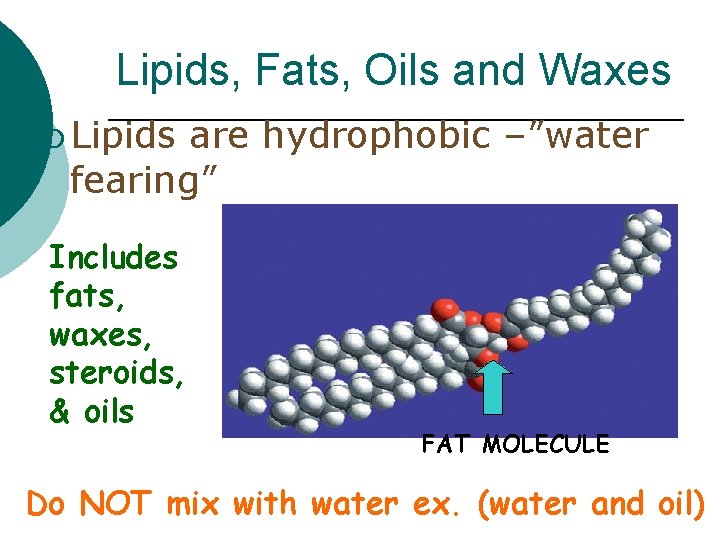
Lipids, Fats, Oils and Waxes ¡ Lipids are hydrophobic –”water fearing” Includes fats, waxes, steroids, & oils FAT MOLECULE Do NOT mix with water ex. (water and oil)

Fats from animal ¡ Most animal fats have a high proportion of saturated fatty acids & exist as solids at room temperature (butter, margarine, shortening)

Fats from plants ¡ Most plant oils tend to be low in saturated fatty acids & exist as liquids at room temperature (oils)

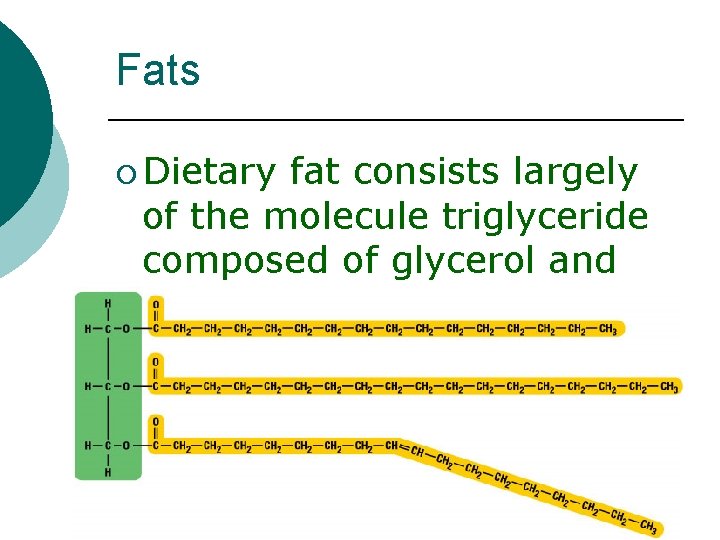
Fats ¡ Dietary fat consists largely of the molecule triglyceride composed of glycerol and three fatty acid chains

Function of Lipids ¡ Fats store energy, help to insulate the body, and cushion and protect organs, make up a cell membrane.
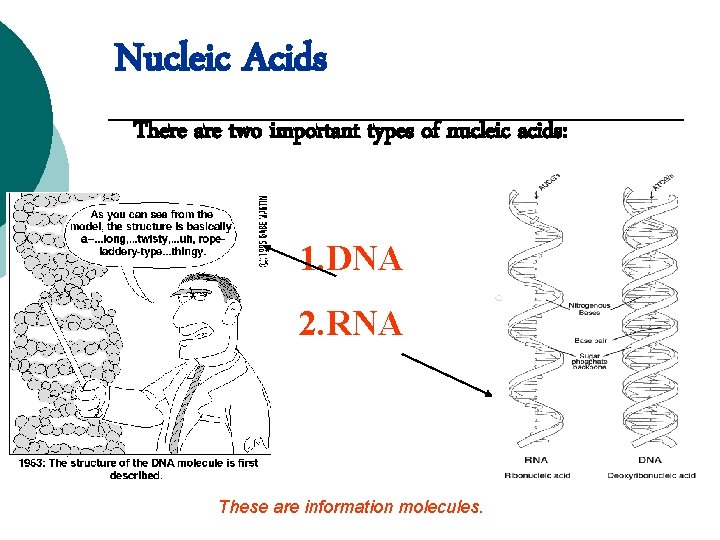
Nucleic Acids There are two important types of nucleic acids: 1. DNA 2. RNA These are information molecules.

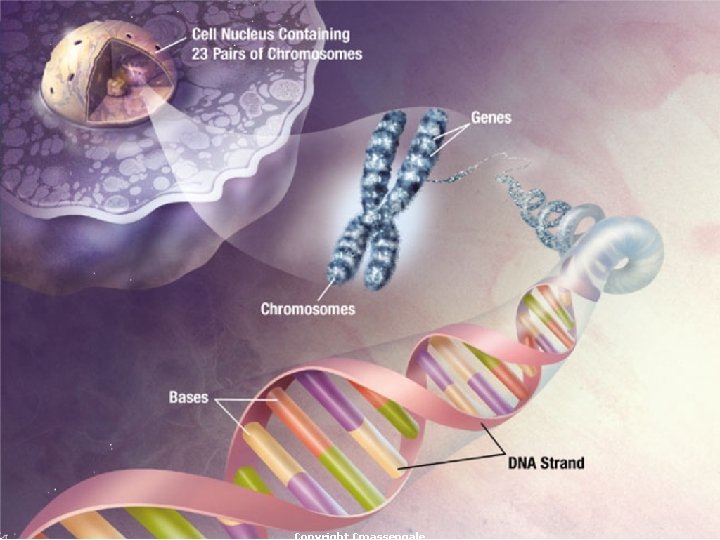
Functions of Nucleic Acids ¡Store hereditary information Contain information for making all the body’s proteins Two types exist --- DNA & RNA



- Slides: 29