Organic Compounds Carbohydrates Lipids Proteins Nucleic Acids Organic

Organic Compounds Carbohydrates, Lipids, Proteins & Nucleic Acids
Organic Chemistry • Organic compound = compound that contains carbon (and Hydrogen) • Not Organic: CO 2 • CO •

Why Carbon? • Abundant • Small, stable atom • Bond angles at 90 degrees forming 3 -D rings • Can form isomers (or different structures of molecules that have the same formula)

Why Carbon? 4 valence electrons… Carbon easily forms covalent bonds with up to four other atoms, including other carbon atoms, by usingle, double and triple bonds. Means Carbon can make chains

Chemical Formula vs. Structural Formula Chemical Formula: CH 4 CO 2 H 20 NH 3 CH 3 CH 2 CH 3 Structural Formula:

Terms • Macromolecules: The 4 molecules of life are called macromolecules (large) • Carbohydrates, Proteins, Lipids, Nucleic acids • Monomer = building blocks • Polymer = large compound made up of smaller monomers
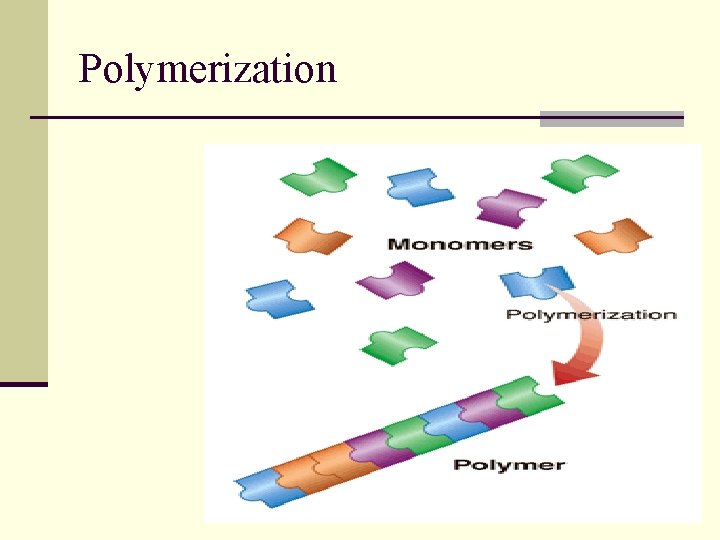
Polymerization
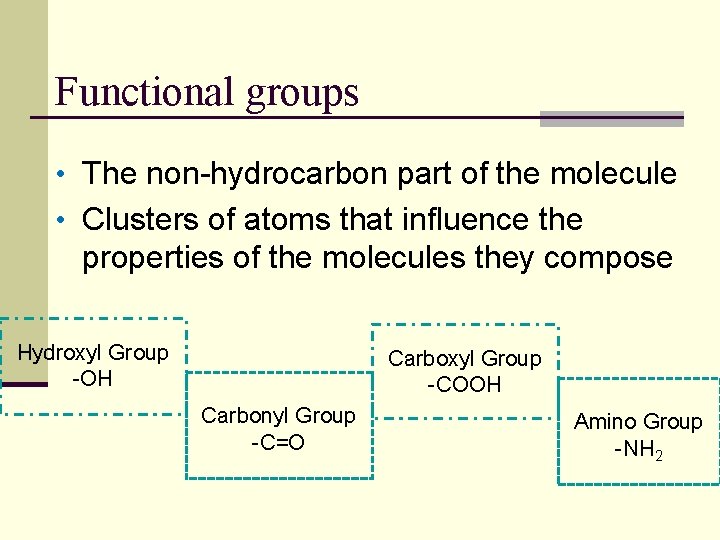
Functional groups • The non-hydrocarbon part of the molecule • Clusters of atoms that influence the properties of the molecules they compose Hydroxyl Group -OH Carboxyl Group -COOH Carbonyl Group -C=O Amino Group -NH 2
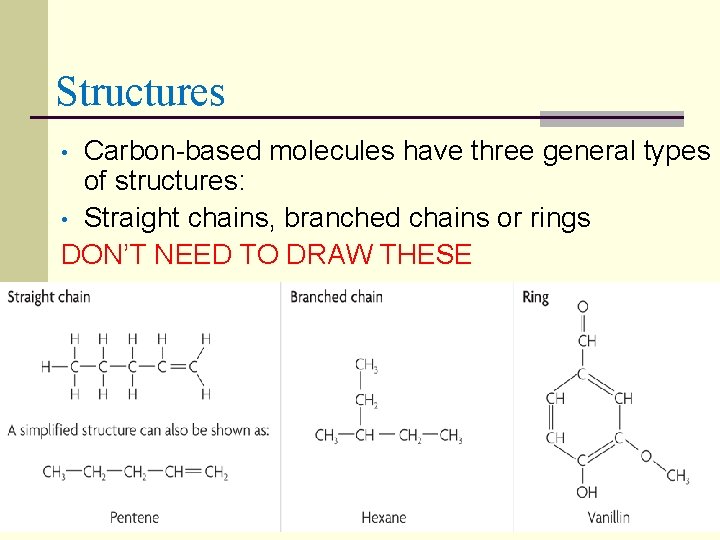
Structures Carbon-based molecules have three general types of structures: • Straight chains, branched chains or rings DON’T NEED TO DRAW THESE •

Organic Molecules 1. Carbohydrates 2. Proteins 3. Lipids 4. Nucleic Acids
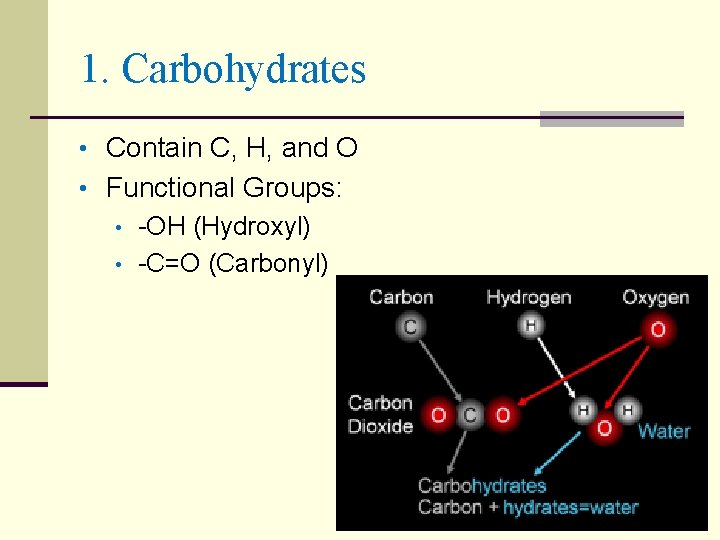
1. Carbohydrates • Contain C, H, and O • Functional Groups: • -OH (Hydroxyl) • -C=O (Carbonyl)

Carbohydrates: Function • Short-term energy source for organisms (glucose) • Long-term energy storage (starch in plants, glycogen in animals) • Structural molecules (cellulose in rigid cell walls)

Carbohydrates n Examples n n Sugars (glucose, sucrose) Starch Cellulose (plants) Glycogen (animals)

Carbohydrates: Subunits n Monomer: monosaccharide (simple sugars) n Contain 3 -7 carbons (often in a ring/ but can be straight chain) n Contain C, H & O in 1: 2: 1 ratio n 3 most common: n Glucose: fuel for cells n Fructose: fruit, sweetest sugar n Galactose: milk sugar
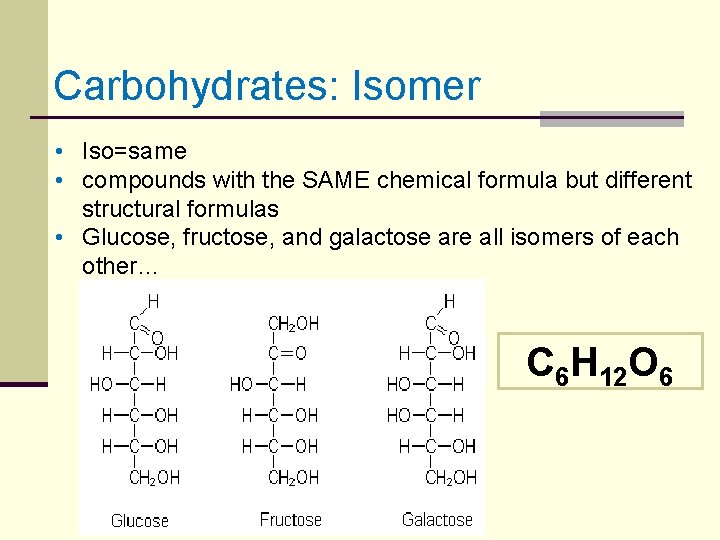
Carbohydrates: Isomer • Iso=same • compounds with the SAME chemical formula but different structural formulas • Glucose, fructose, and galactose are all isomers of each other… C 6 H 12 O 6
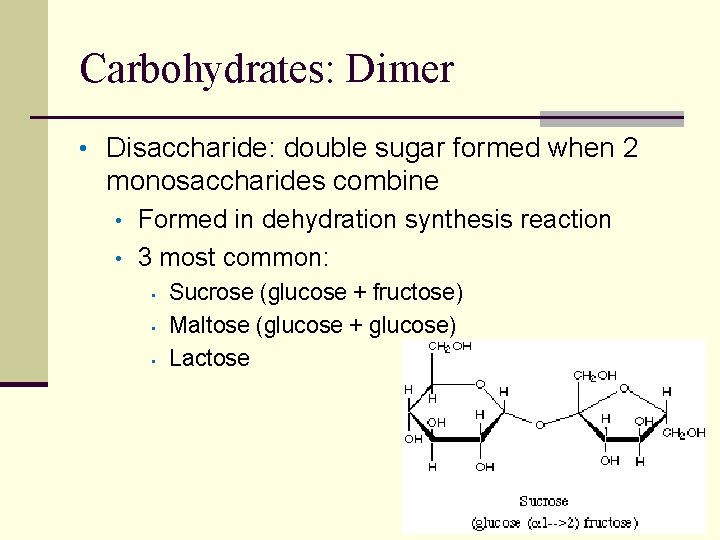
Carbohydrates: Dimer • Disaccharide: double sugar formed when 2 monosaccharides combine • • Formed in dehydration synthesis reaction 3 most common: • • • Sucrose (glucose + fructose) Maltose (glucose + glucose) Lactose
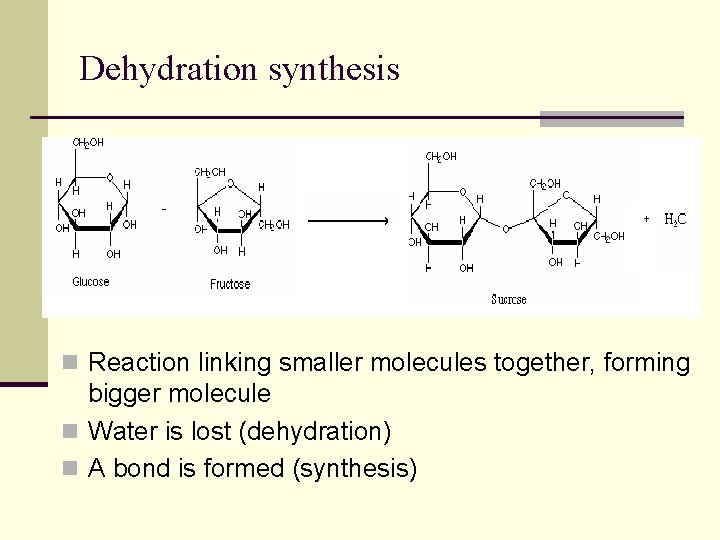
Dehydration synthesis n Reaction linking smaller molecules together, forming bigger molecule n Water is lost (dehydration) n A bond is formed (synthesis)
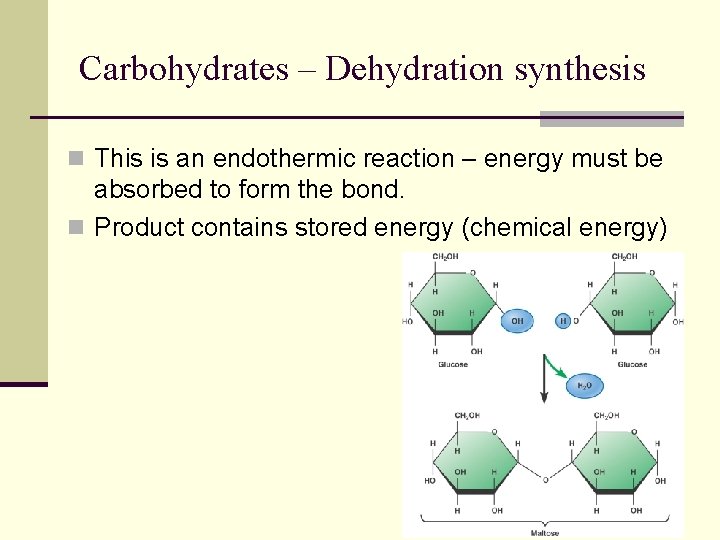
Carbohydrates – Dehydration synthesis n This is an endothermic reaction – energy must be absorbed to form the bond. n Product contains stored energy (chemical energy)
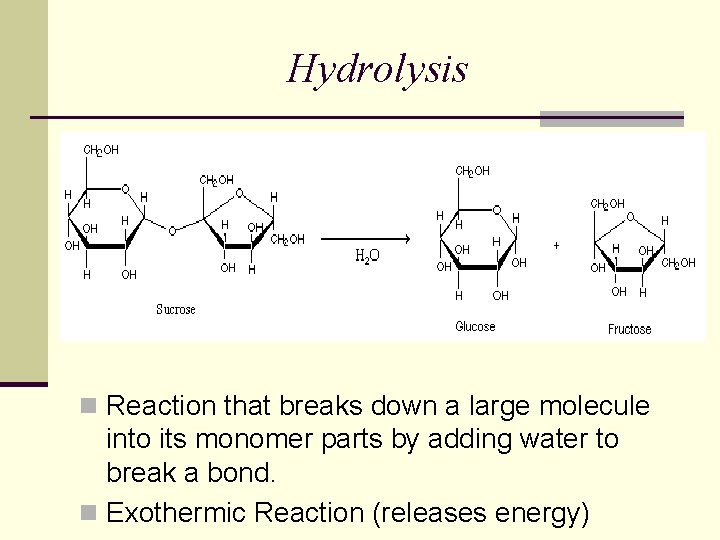
Hydrolysis n Reaction that breaks down a large molecule into its monomer parts by adding water to break a bond. n Exothermic Reaction (releases energy)
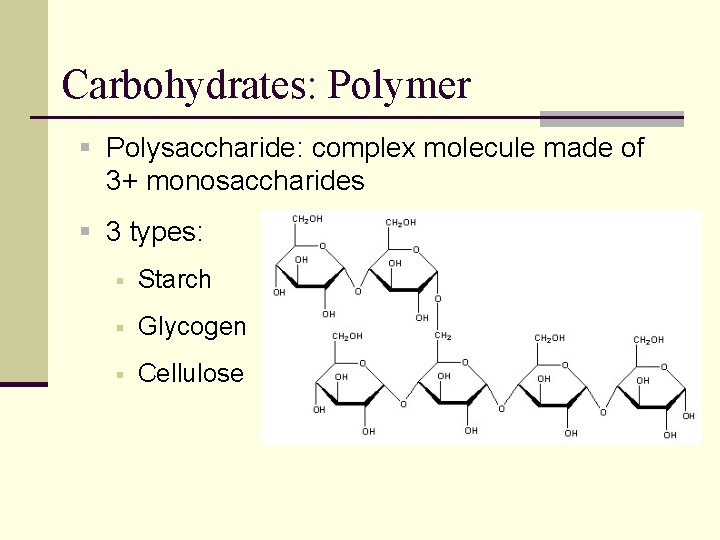
Carbohydrates: Polymer § Polysaccharide: complex molecule made of 3+ monosaccharides § 3 types: § Starch § Glycogen § Cellulose

Proteins • Contain C, H, O and N • Sometimes S (Sulfur) • Functions • Enzymes that catalyze (speed up) reactions • Structural & Mechanical functions (muscles) • Cell signaling, immune response, & cell division
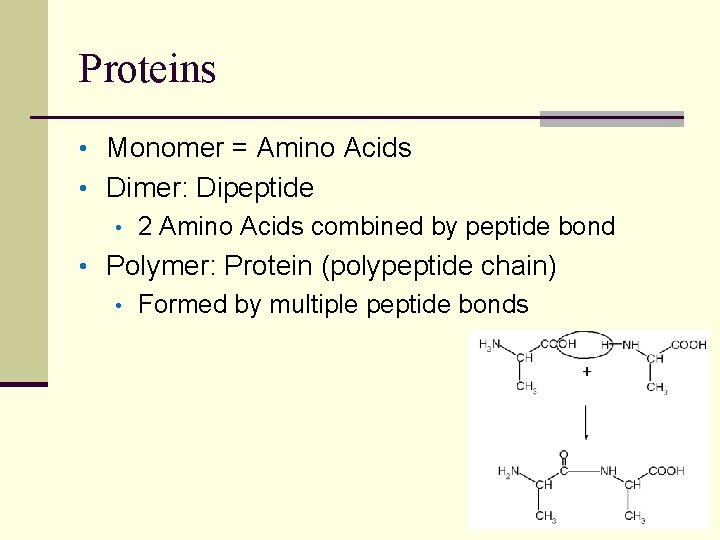
Proteins • Monomer = Amino Acids • Dimer: Dipeptide • 2 Amino Acids combined by peptide bond • Polymer: Protein (polypeptide chain) • Formed by multiple peptide bonds
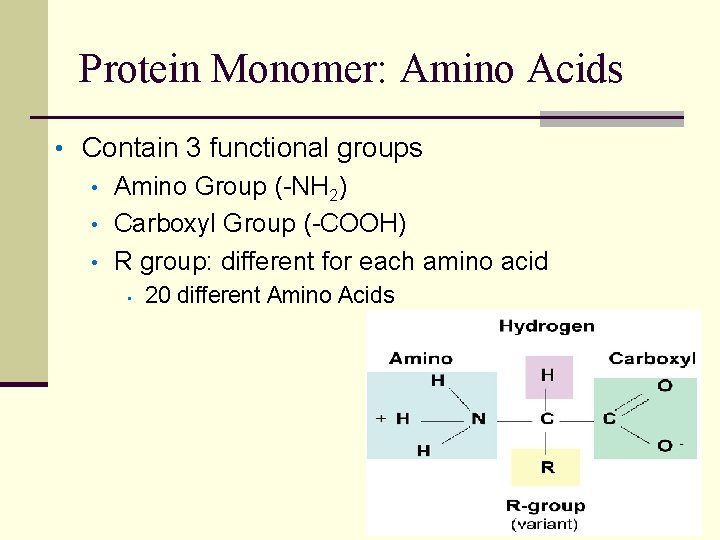
Protein Monomer: Amino Acids • Contain 3 functional groups • Amino Group (-NH 2) • Carboxyl Group (-COOH) • R group: different for each amino acid • 20 different Amino Acids
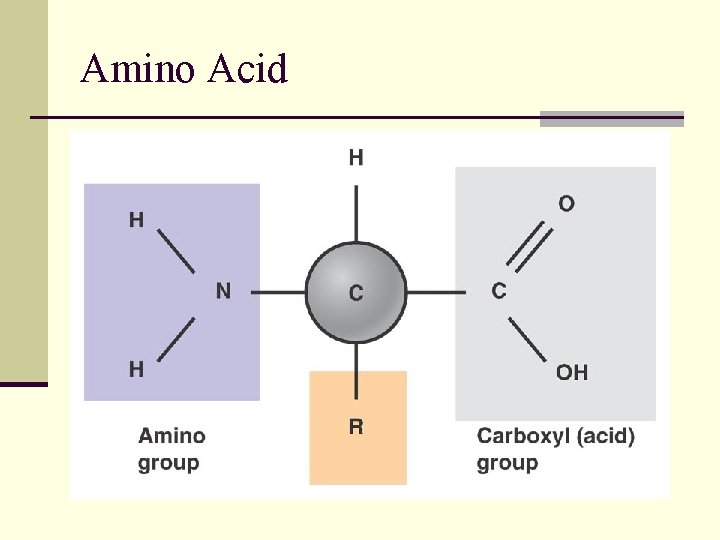
Amino Acid
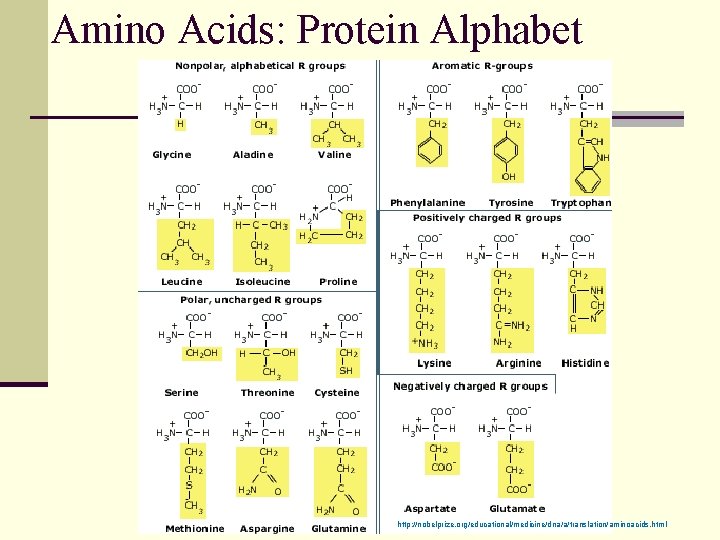
Amino Acids: Protein Alphabet http: //nobelprize. org/educational/medicine/dna/a/translation/aminoacids. html
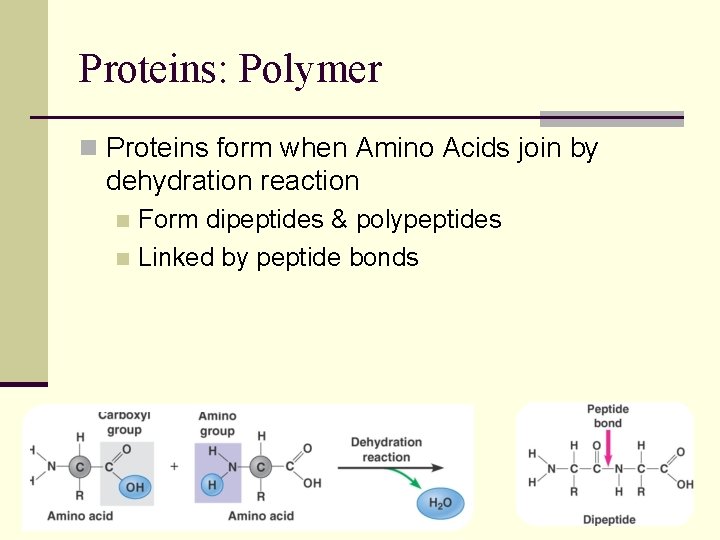
Proteins: Polymer n Proteins form when Amino Acids join by dehydration reaction Form dipeptides & polypeptides n Linked by peptide bonds n
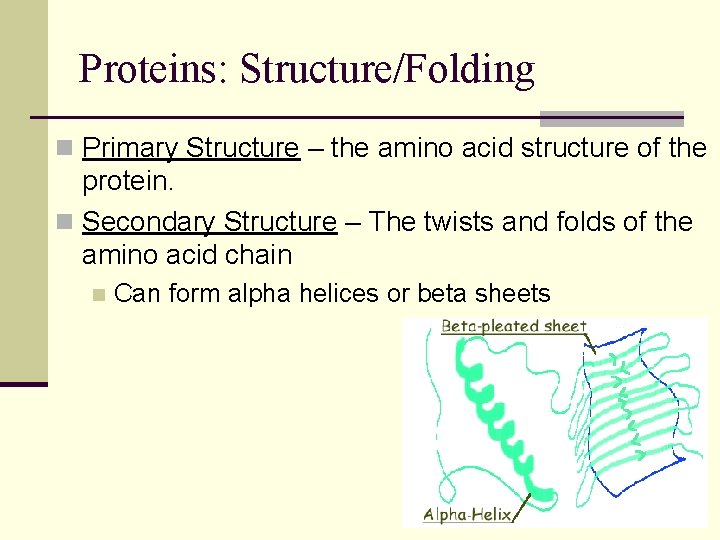
Proteins: Structure/Folding n Primary Structure – the amino acid structure of the protein. n Secondary Structure – The twists and folds of the amino acid chain n Can form alpha helices or beta sheets
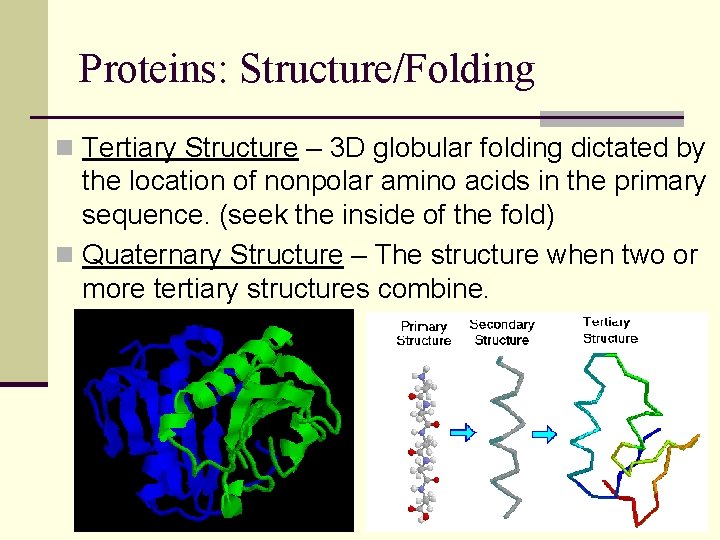
Proteins: Structure/Folding n Tertiary Structure – 3 D globular folding dictated by the location of nonpolar amino acids in the primary sequence. (seek the inside of the fold) n Quaternary Structure – The structure when two or more tertiary structures combine.

Proteins: Structure/Folding n Correct folding is critical to protein’s function n Tertiary and Quaternary structure are influenced by temperature and p. H If conditions are not right, process will not take place or make mistakes n Cannot be reversed (denaturation) n Protein won’t function n

3. Lipids n Contain C, H, O n Fats & Oils n Function depends on type n No specific & typical monomer /polymer http: //www. red-spirit-energy-healing. com/essential-fats. html
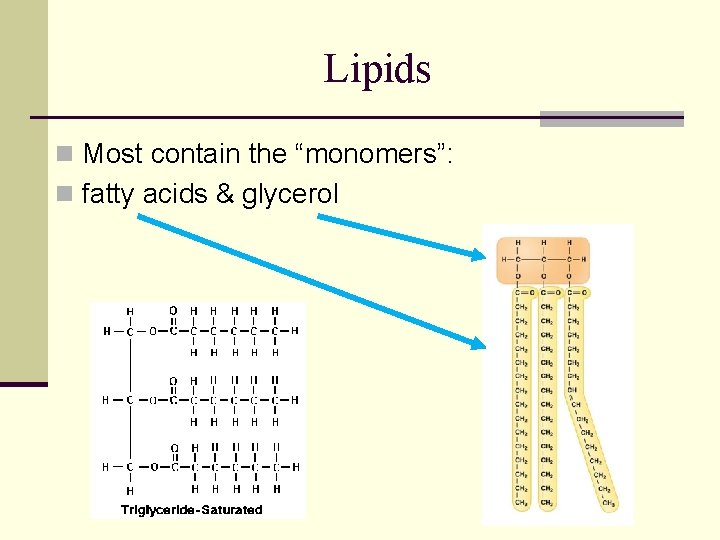
Lipids n Most contain the “monomers”: n fatty acids & glycerol
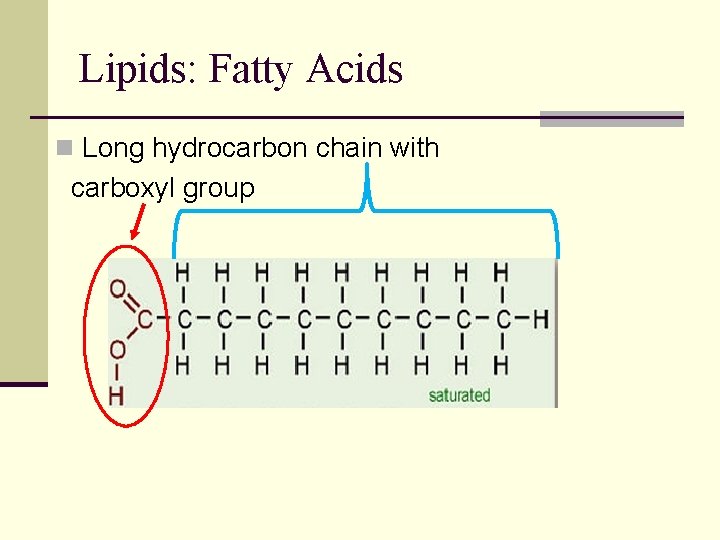
Lipids: Fatty Acids n Long hydrocarbon chain with carboxyl group
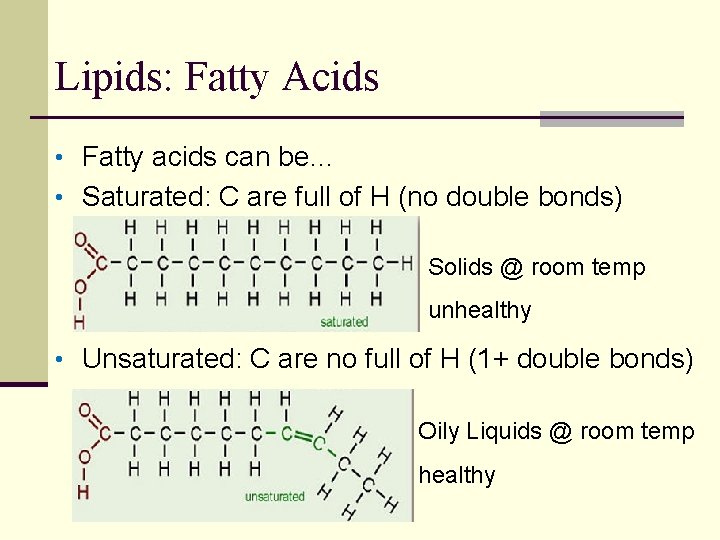
Lipids: Fatty Acids • Fatty acids can be… • Saturated: C are full of H (no double bonds) Solids @ room temp unhealthy • Unsaturated: C are no full of H (1+ double bonds) Oily Liquids @ room temp healthy
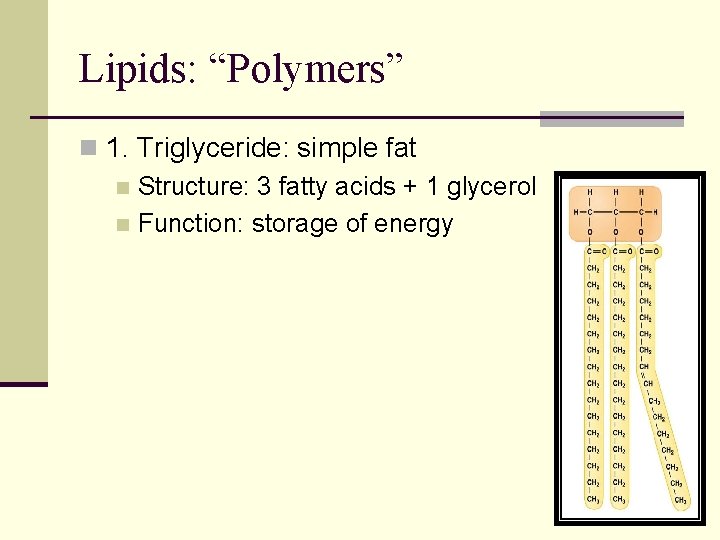
Lipids: “Polymers” n 1. Triglyceride: simple fat n Structure: 3 fatty acids + 1 glycerol n Function: storage of energy
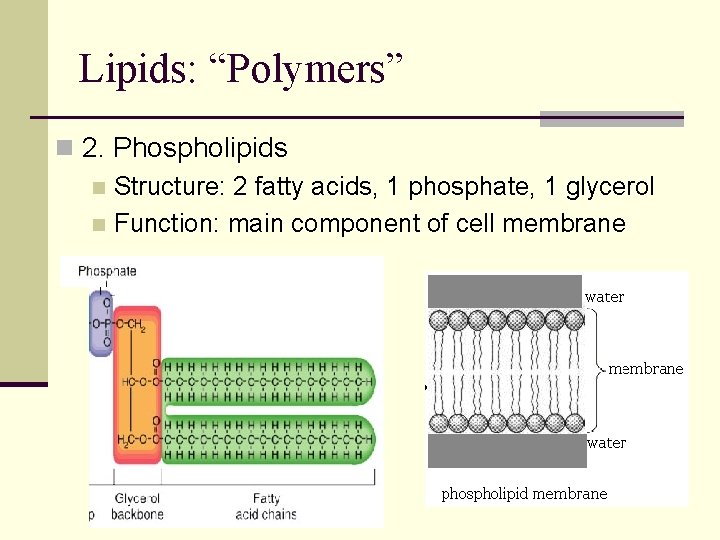
Lipids: “Polymers” n 2. Phospholipids n Structure: 2 fatty acids, 1 phosphate, 1 glycerol n Function: main component of cell membrane
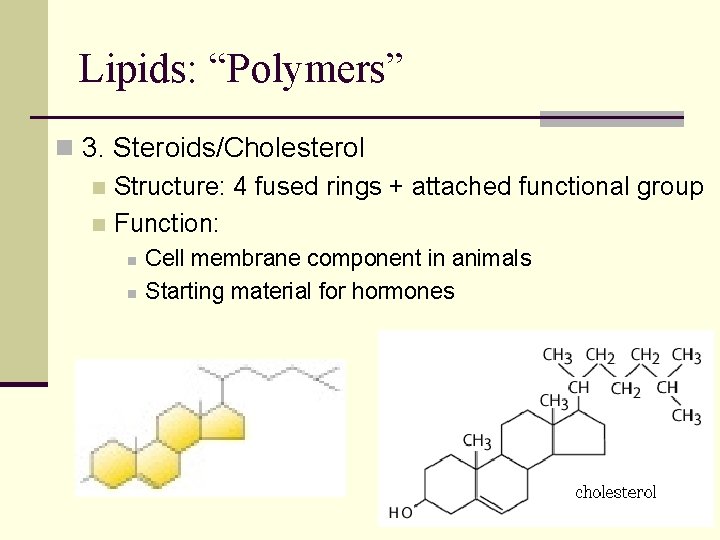
Lipids: “Polymers” n 3. Steroids/Cholesterol n Structure: 4 fused rings + attached functional group n Function: n n Cell membrane component in animals Starting material for hormones

4. Nucleic Acids n Contain C, H, O, N, P n Function: n Source of genetic information n Store & transmit into to carry out cell processes n Determine protein’s amino acid sequence
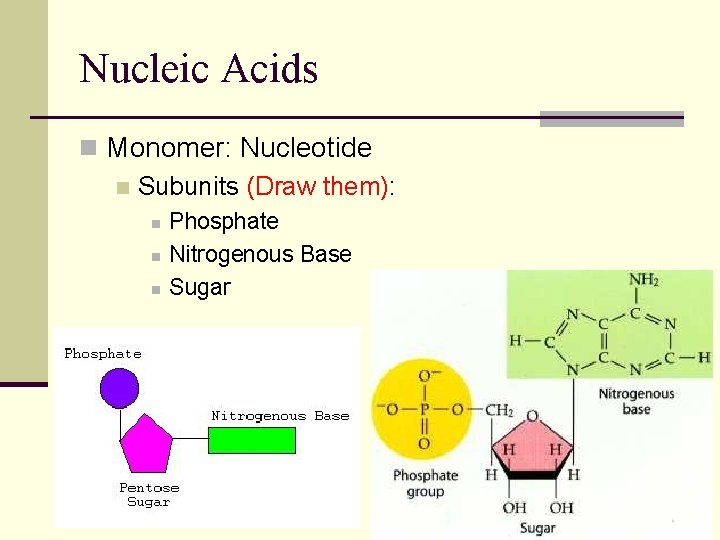
Nucleic Acids n Monomer: Nucleotide n Subunits (Draw them): n n n Phosphate Nitrogenous Base Sugar
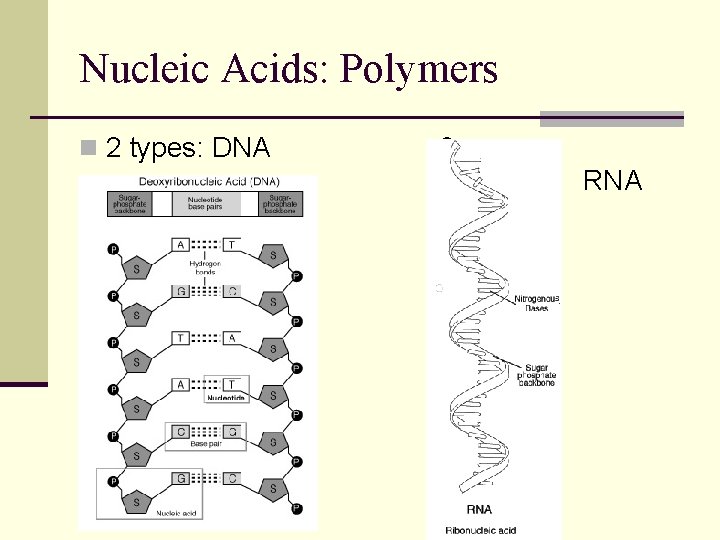
Nucleic Acids: Polymers n 2 types: DNA & RNA

Nucleic Acids: DNA vs. RNA Double helix RNA Single-stranded Nitrogen bases: Adenine, Cytosine, Guanine, Thymine Nitrogen bases: Adenine, Cytosine, Guanine, Uracil Sugar = Deoxyribose Sugar = Ribose
- Slides: 40