Organic Compounds Carbohydrates Include sugars glycogen starches and

Organic Compounds

Carbohydrates Include sugars, glycogen, starches and cellulose Made from Carbon, Hydrogen and Oxygen Usually 1: 2: 1 ratio (CH 2 O) Divided into groups based on their size

Monosaccharides AKA simple sugars Building block of carbohydrates Source of chemical energy for generating ATP Part of the structure of nucleic acids (DNA and RNA)

Disaccharides AKA simple sugars 2 monosaccharides joined by dehydration synthesis EX – sucrose (table sugar) is glucose and fructose

Polysaccharides AKA complex carbohydrates Contain tens or hundreds of monosaccharides Can be broken down by hydrolysis Main polysaccharide in the human body is glycogen
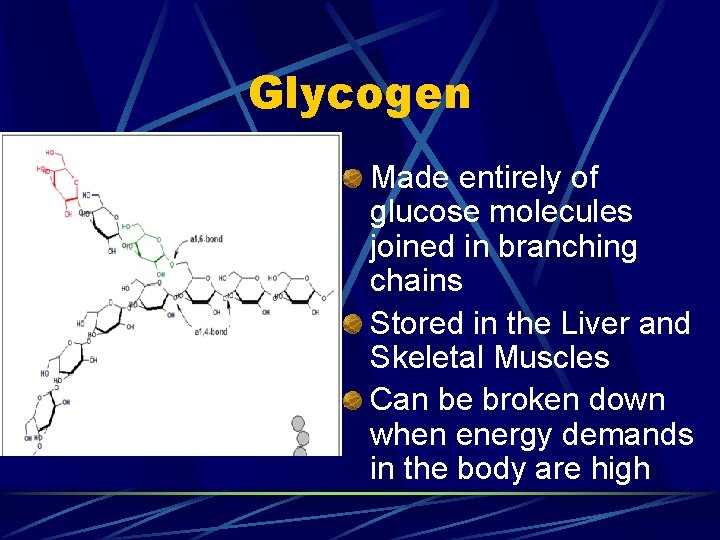
Glycogen Made entirely of glucose molecules joined in branching chains Stored in the Liver and Skeletal Muscles Can be broken down when energy demands in the body are high

Lipids (Fats) Made of Carbon, Hydrogen and Oxygen Proportion of Oxygen is usually lower than in carbohydrates Not as many polar bonds so they are not soluble in water Include triglycerides, phospholipids, steroids, fatty acids and fat soluble vitamins (A, D, E and K)

Triglycerides Most plentiful lipid in the body Most highly concentrated form of energy Adipose Tissue = Fat Tissue l Excess dietary carbohydrates, fats and protein are all deposited in adipose tissue
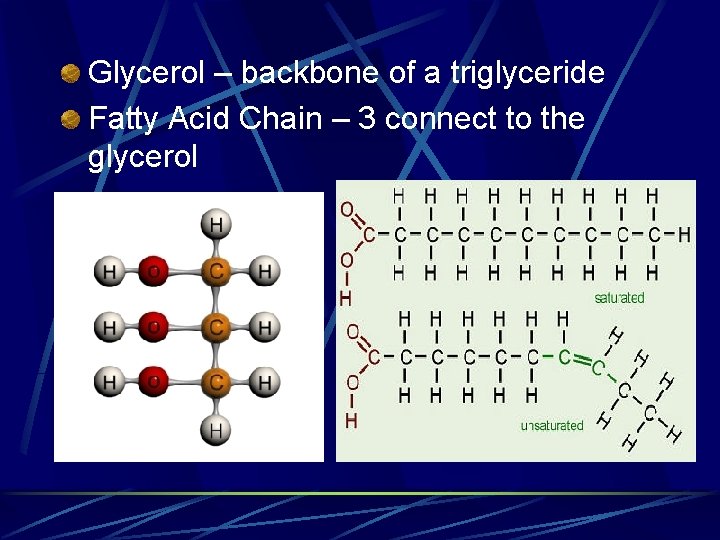
Glycerol – backbone of a triglyceride Fatty Acid Chain – 3 connect to the glycerol

Fatty Acids Saturated – contain only single covalent bonds between carbons Unsaturated contain 1 or more double bonds between carbons Monounsaturated – only 1 double bond l Polyunsaturated – more than 1 double bond l
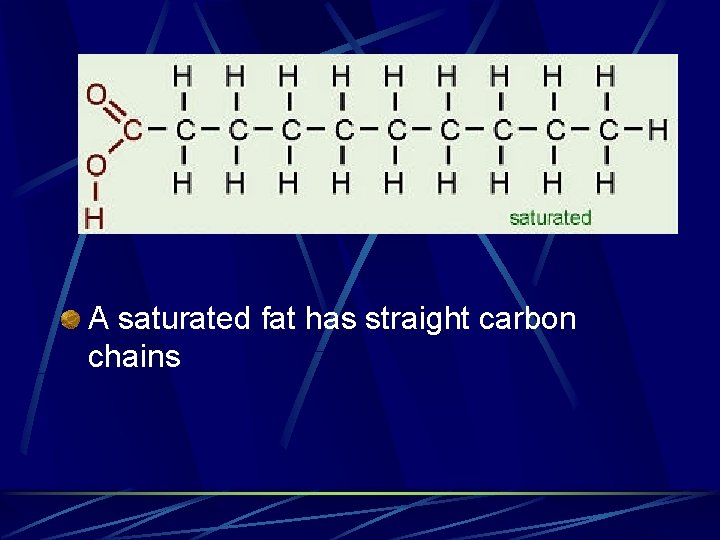
A saturated fat has straight carbon chains
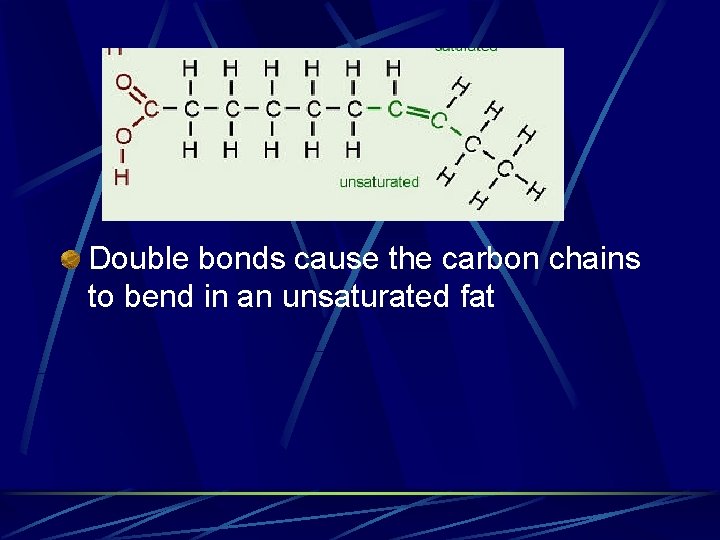
Double bonds cause the carbon chains to bend in an unsaturated fat
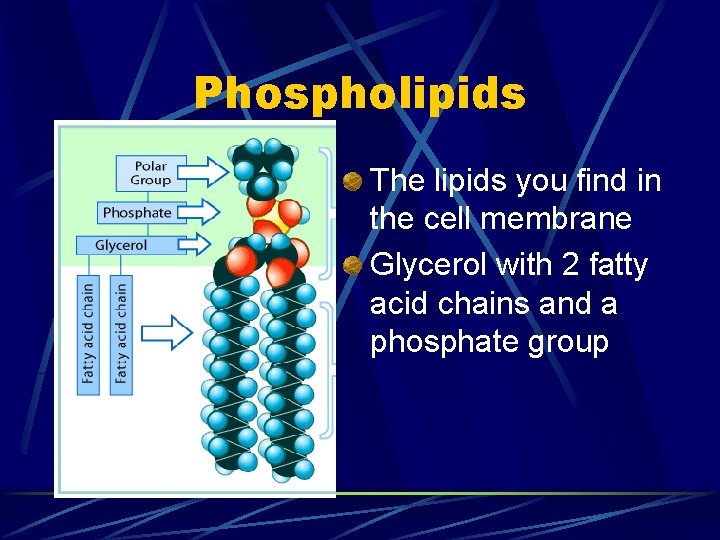
Phospholipids The lipids you find in the cell membrane Glycerol with 2 fatty acid chains and a phosphate group

Steroids Rings of 4 carbons rather than chains Cholesterol is the steroid from which other steroids are made Examples: esterdiol, testosterone, cortisol and Vitamin D

Protein Large molecules containing Carbon, Hydrogen, Oxygen, Nitrogen and sometimes Sulfur Building Blocks are Amino Acids
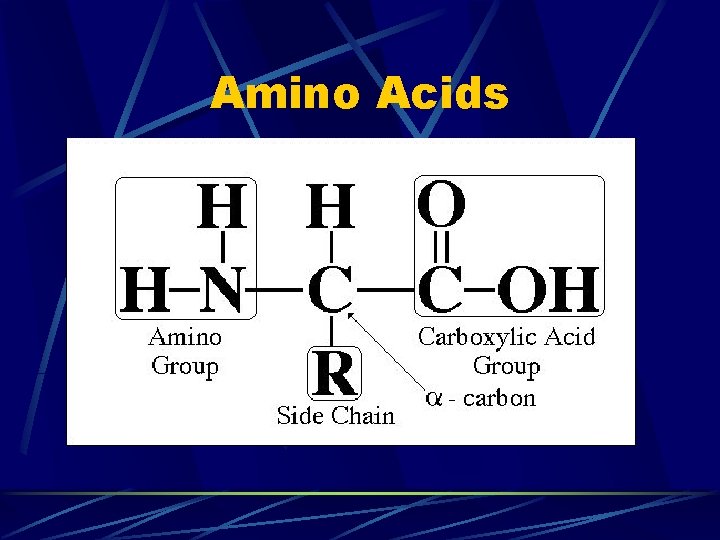
Amino Acids

Amino Group --NH 2

Carboxyl Group --COOH

“R” Group (Radical Group) 20 different side chains that determine which amino acid the molecule will be

Peptide The union of 2 or more amino acids Dipeptide = 2 aa Tripeptide = 3 aa Polypeptide = lots of aa
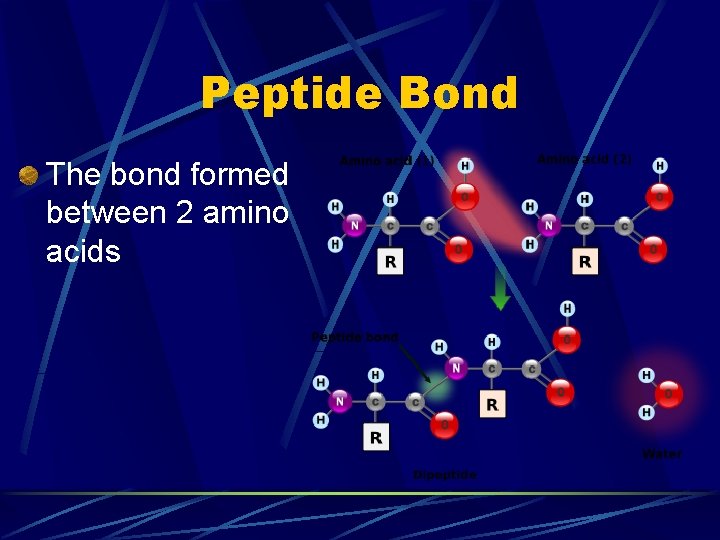
Peptide Bond The bond formed between 2 amino acids

Protein A protein is a polypeptide that can contain as few as 50 aa’s or as many as 2000 aa’s The variation in the number and sequence of aa’s produces a different protein
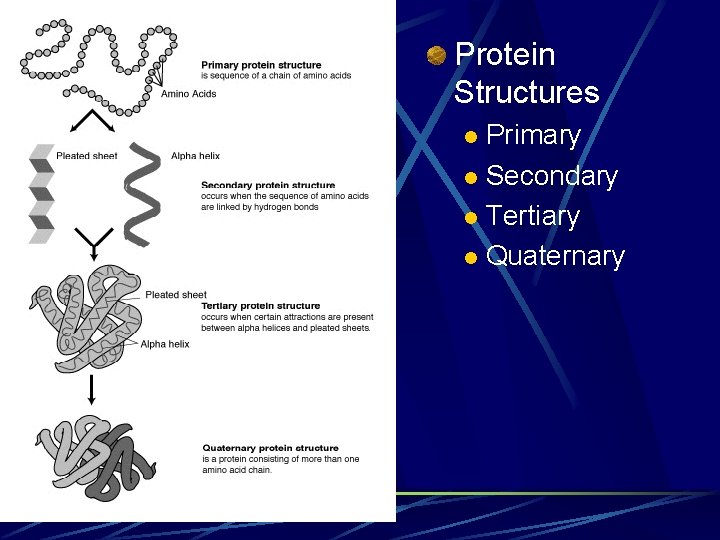
Protein Structures Primary l Secondary l Tertiary l Quaternary l
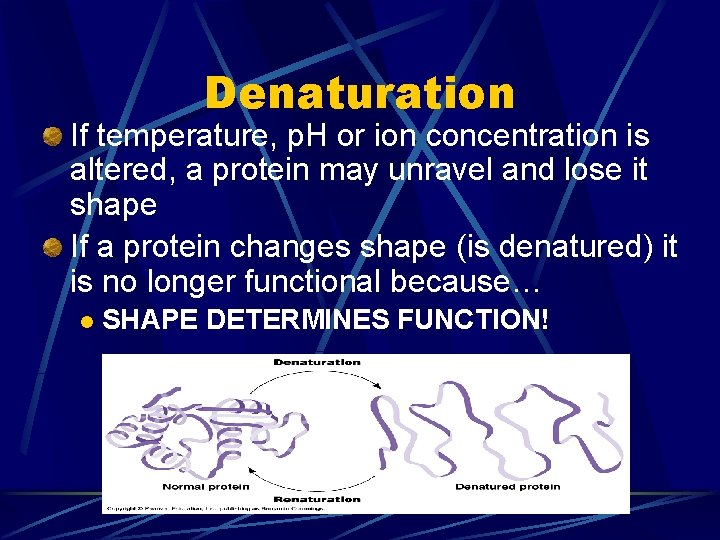
Denaturation If temperature, p. H or ion concentration is altered, a protein may unravel and lose it shape If a protein changes shape (is denatured) it is no longer functional because… l SHAPE DETERMINES FUNCTION!

Enzymes Protein Catalyst A substance that can speed up a chemical reaction without themselves being altered Names of catalysts generally end in -ase l Ex. Kinase, dehydrogenase, amylase

Properties of Enzymes Specificity – each particular enzyme catalyze a particular chemical reaction l Lock and Key Efficiency – under optimal conditions, enzymes can catalyze reactions at rates millions of times that of an uncatalyzed reation. Control – rate of synthesis is under the control of the cell’s genes
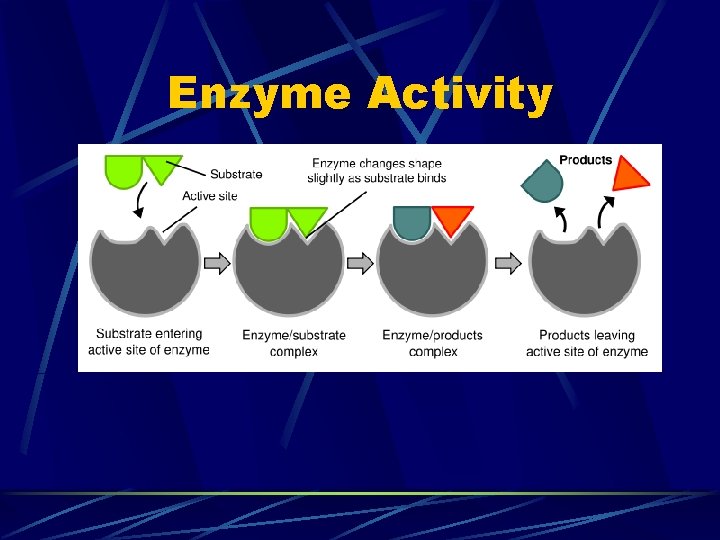
Enzyme Activity

Enzyme Vocab Substrate – the molecule on which the enzyme acts Products – molecule(s) produced by the reaction Cofactor/Coenzyme – a nonprotein substance that some enzymes require to operate properly Active Site – the particular spot on the enzyme molecule that catalyzes the reaction Enzyme-Substrate Complex – a temporary compound formed when the enzyme and substrate bind

Review of Organic Compounds Carbohydrates l Building blocks = monosaccharides Lipids (Triglycerides) l Building Blocks = glycerol and fatty acids Proteins l Building Blocks = amino acids
- Slides: 30