Organic Chemistry William H Brown Christopher S Foote

Organic Chemistry William H. Brown Christopher S. Foote Brent L. Iverson 13 -1

Nuclear Magnetic Resonance Chapter 13 13 -2

Molecular Spectroscopy u Nuclear magnetic resonance (NMR) spectroscopy: a spectroscopic technique that gives us information about the number and types of atoms in a molecule, for example, about the number and types of • hydrogen atoms using 1 H-NMR spectroscopy • carbon atoms using 13 C-NMR spectroscopy • phosphorus atoms using 31 P-NMR spectroscopy 13 -3

Nuclear Spin States u An electron has a spin quantum number of 1/2 with allowed values of +1/2 and -1/2 • this spinning charge creates an associated magnetic field • in effect, an electron behaves as if it is a tiny bar magnet and has what is called a magnetic moment u The same effect holds for certain atomic nuclei • any atomic nucleus that has an odd mass number, an odd atomic number, or both also has a spin and a resulting nuclear magnetic moment • the allowed nuclear spin states are determined by the spin quantum number, I, of the nucleus 13 -4

Nuclear Spin States • a nucleus with spin quantum number I has 2 I + 1 spin states; if I = 1/2, there are two allowed spin states • Table 13. 1 gives the spin quantum numbers and allowed nuclear spin states for selected isotopes of elements common to organic compounds 13 -5

Nuclear Spins in B 0 • within a collection of 1 H and 13 C atoms, nuclear spins are completely random in orientation • when placed in a strong external magnetic field of strength B 0, however, interaction between nuclear spins and the applied magnetic field is quantized, with the result that only certain orientations of nuclear magnetic moments are allowed 13 -6
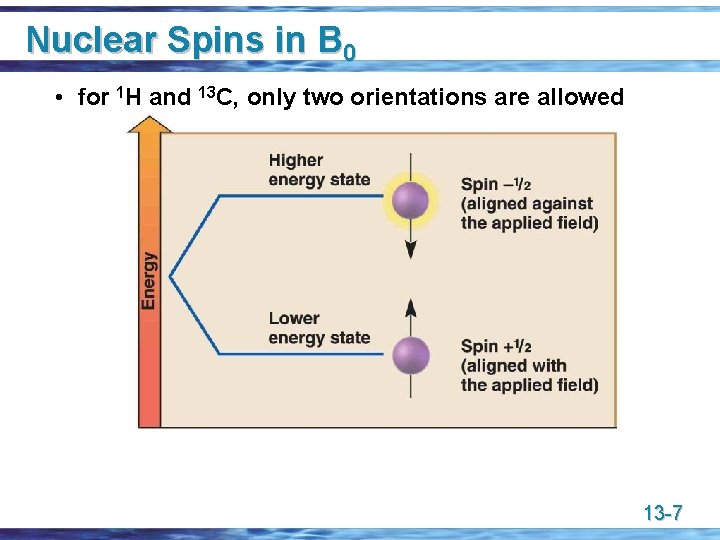
Nuclear Spins in B 0 • for 1 H and 13 C, only two orientations are allowed 13 -7

Nuclear Spins in B 0 u In an applied field strength of 7. 05 T, which is readily available with present-day superconducting electromagnets, the difference in energy between nuclear spin states for • • 1 H is approximately 0. 120 J (0. 0286 cal)/mol, which corresponds to electromagnetic radiation of 300 MHz (300, 000 Hz) 13 C is approximately 0. 030 J (0. 00715 cal)/mol, which corresponds to electromagnetic radiation of 75 MHz (75, 000 Hz) 13 -8
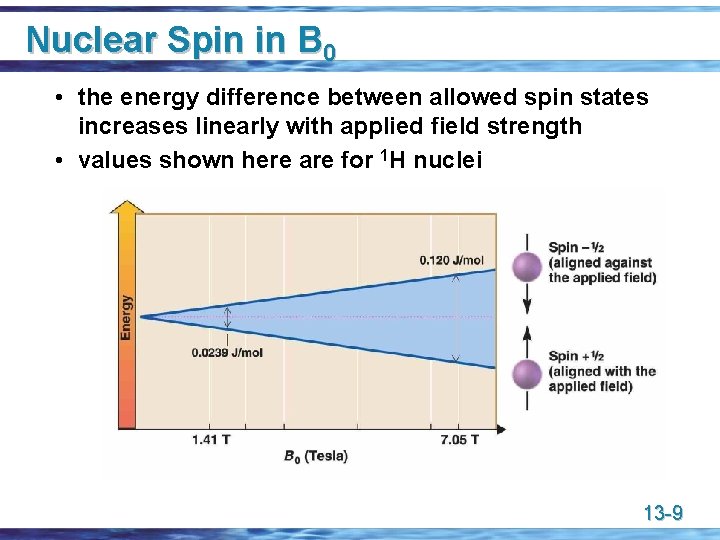
Nuclear Spin in B 0 • the energy difference between allowed spin states increases linearly with applied field strength • values shown here are for 1 H nuclei 13 -9

Nuclear Magnetic Resonance • when nuclei with a spin quantum number of 1/2 are placed in an applied field, a small majority of nuclear spins are aligned with the applied field in the lower energy state • the nucleus begins to precess and traces out a coneshaped surface, in much the same way a spinning top or gyroscope traces out a cone-shaped surface as it precesses in the earth’s gravitational field • we express the rate of precession as a frequency in hertz 13 -10

Nuclear Magnetic Resonance u If the precessing nucleus is irradiated with electromagnetic radiation of the same frequency as the rate of precession, • the two frequencies couple, • energy is absorbed, and • the nuclear spin is flipped from spin state +1/2 (with the applied field) to -1/2 (against the applied field) 13 -11
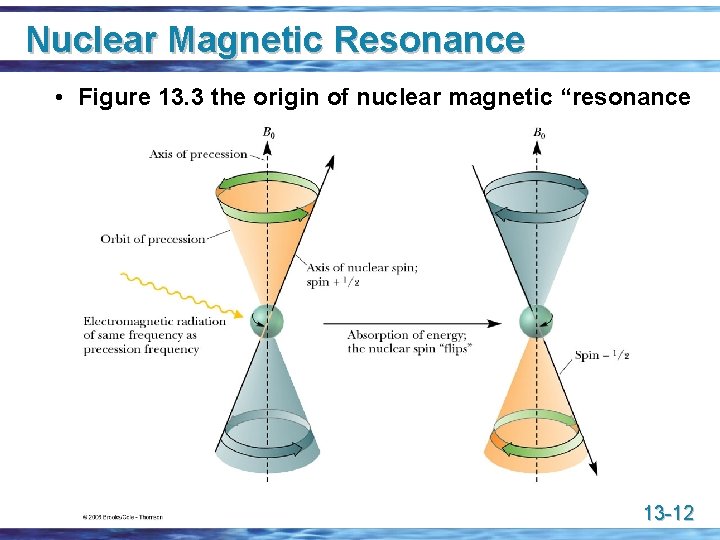
Nuclear Magnetic Resonance • Figure 13. 3 the origin of nuclear magnetic “resonance 13 -12

Nuclear Magnetic Resonance u Resonance: in NMR spectroscopy, resonance is the absorption of electromagnetic radiation by a precessing nucleus and the resulting “flip” of its nuclear spin from a lower energy state to a higher energy state u The instrument used to detect this coupling of precession frequency and electromagnetic radiation records it as a signal • signal: a recording in an NMR spectrum of a nuclear magnetic resonance 13 -13

Nuclear Magnetic Resonance • if we were dealing with 1 H nuclei isolated from all other atoms and electrons, any combination of applied field and radiation that produces a signal for one 1 H would produce a signal for all 1 H. The same is true of 13 C nuclei • but hydrogens in organic molecules are not isolated from all other atoms; they are surrounded by electrons, which are caused to circulate by the presence of the applied field • the circulation of electrons around a nucleus in an applied field is called diamagnetic current and the nuclear shielding resulting from it is called diamagnetic shielding 13 -14

Nuclear Magnetic Resonance • the difference in resonance frequencies among the various hydrogen nuclei within a molecule due to shielding/deshielding is generally very small • the difference in resonance frequencies for hydrogens in CH 3 Cl compared to CH 3 F under an applied field of 7. 05 T is only 360 Hz, which is 1. 2 parts per million (ppm) compared with the irradiating frequency 13 -15

Nuclear Magnetic Resonance • signals are measured relative to the signal of the reference compound tetramethylsilane (TMS) • for a 1 H-NMR spectrum, signals are reported by their shift from the 12 H signal in TMS • for a 13 C-NMR spectrum, signals are reported by their shift from the 4 C signal in TMS • Chemical shift ( ): the shift in ppm of an NMR signal from the signal of TMS 13 -16
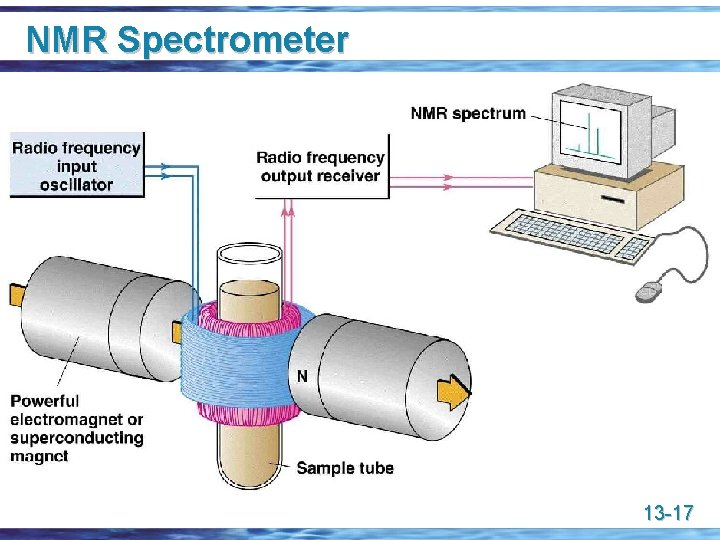
NMR Spectrometer 13 -17

NMR Spectrometer u Essentials of an NMR spectrometer are a powerful magnet, a radio-frequency generator, and a radio-frequency detector u The sample is dissolved in a solvent, most commonly CDCl 3 or D 2 O, and placed in a sample tube which is then suspended in the magnetic field and set spinning u Using a Fourier transform NMR (FT-NMR) spectrometer, a spectrum can be recorded in about 2 seconds 13 -18
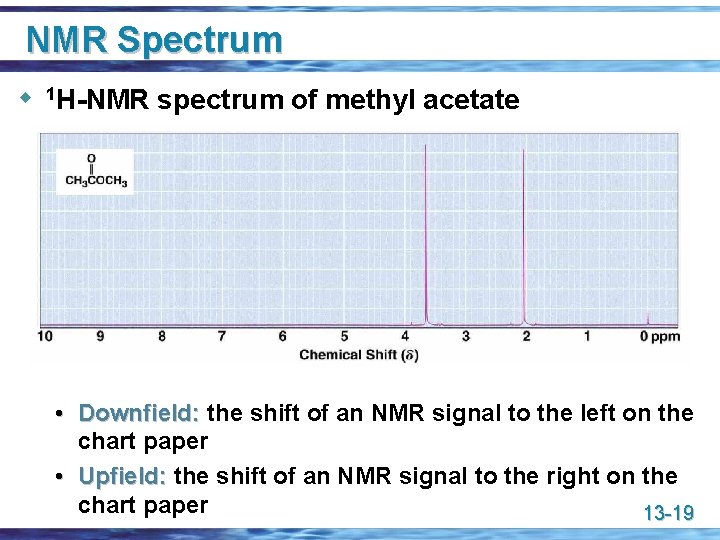
NMR Spectrum u 1 H-NMR spectrum of methyl acetate • Downfield: the shift of an NMR signal to the left on the chart paper • Upfield: the shift of an NMR signal to the right on the chart paper 13 -19

Equivalent Hydrogens u Equivalent hydrogens: have the same chemical environment • a molecule with 1 set of equivalent hydrogens gives 1 NMR signal 13 -20

Equivalent Hydrogens • a molecule with 2 or more sets of equivalent hydrogens gives a different NMR signal for each set 13 -21
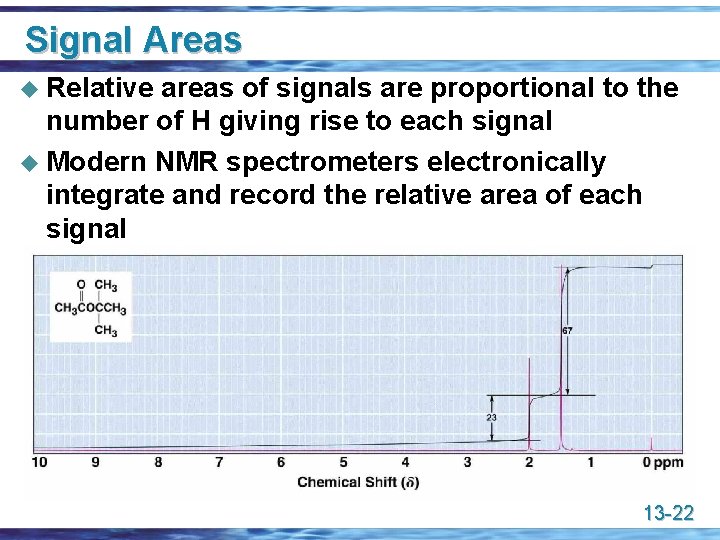
Signal Areas u Relative areas of signals are proportional to the number of H giving rise to each signal u Modern NMR spectrometers electronically integrate and record the relative area of each signal 13 -22

Chemical Shifts 1 H-NMR 13 -23
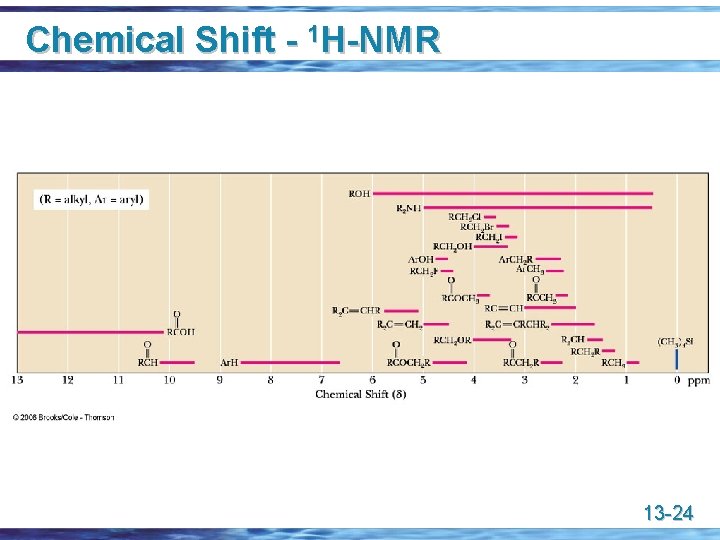
Chemical Shift - 1 H-NMR 13 -24

Chemical Shift u u Depends on (1) electronegativity of nearby atoms, (2) the hybridization of adjacent atoms, and (3) diamagnetic effects from adjacent pi bonds Electronegativity 13 -25

Chemical Shift u Hybridization of adjacent atoms 13 -26

Chemical Shift u Diamagnetic effects of pi bonds • a carbon-carbon triple bond shields an acetylenic hydrogen and shifts its signal upfield (to the right) to a smaller value • a carbon-carbon double bond deshields vinylic hydrogens and shifts their signal downfield (to the left) to a larger value 13 -27
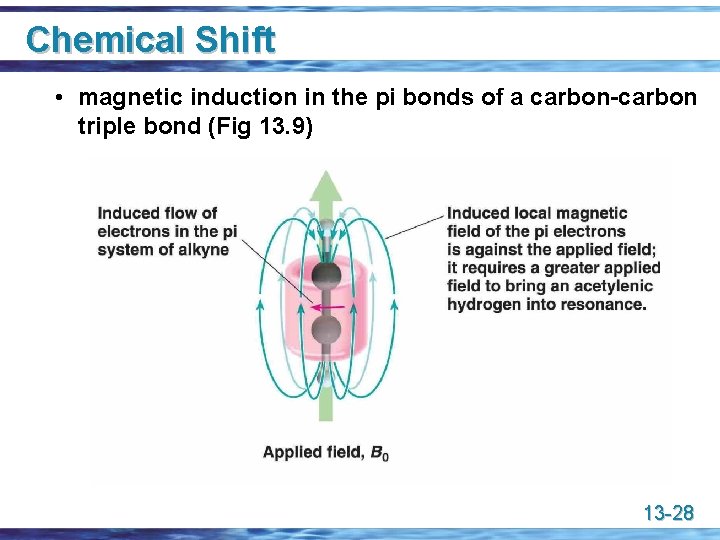
Chemical Shift • magnetic induction in the pi bonds of a carbon-carbon triple bond (Fig 13. 9) 13 -28
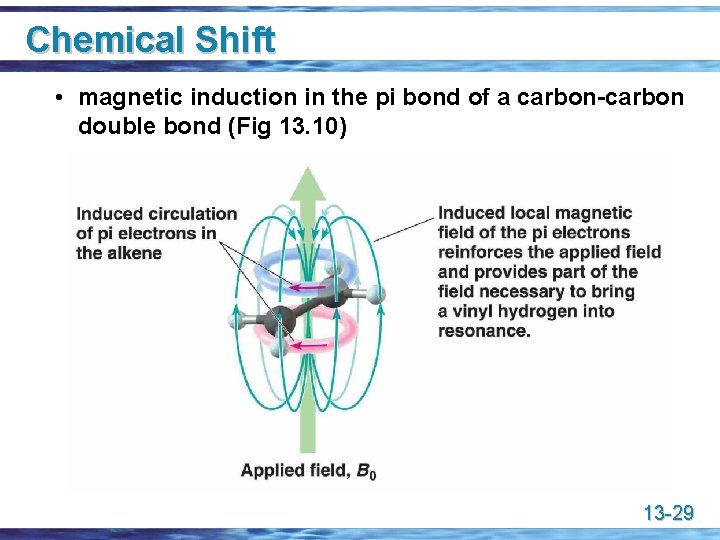
Chemical Shift • magnetic induction in the pi bond of a carbon-carbon double bond (Fig 13. 10) 13 -29
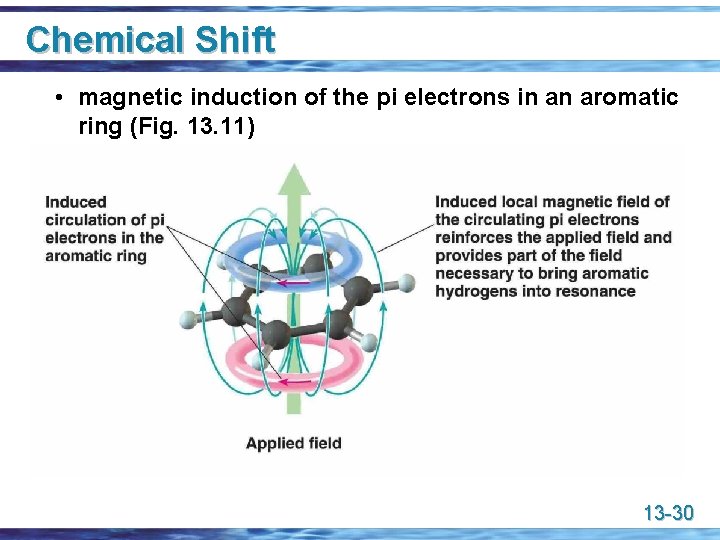
Chemical Shift • magnetic induction of the pi electrons in an aromatic ring (Fig. 13. 11) 13 -30

Signal Splitting; the (n + 1) Rule u Peak: the units into which an NMR signal is split; doublet, triplet, quartet, etc. u Signal splitting: splitting of an NMR signal into a set of peaks by the influence of neighboring nonequivalent hydrogens u (n + 1) rule: if a hydrogen has n hydrogens nonequivalent to it but equivalent among themselves on the same or adjacent atom(s), its 1 H-NMR signal is split into (n + 1) peaks 13 -31
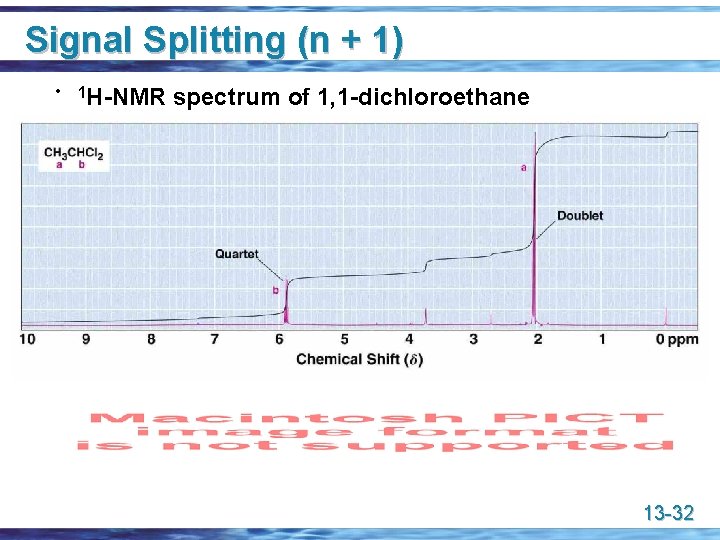
Signal Splitting (n + 1) • 1 H-NMR spectrum of 1, 1 -dichloroethane 13 -32

Signal Splitting (n + 1) Problem: Problem predict the number of 1 H-NMR signals and the splitting pattern of each 13 -33

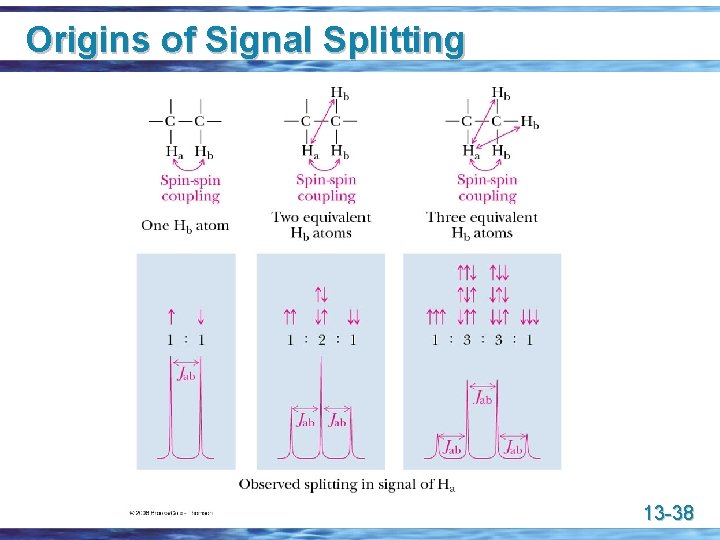
Origins of Signal Splitting u Signal coupling: an interaction in which the nuclear spins of adjacent atoms influence each other and lead to the splitting of NMR signals u Coupling constant (J): the separation on an NMR spectrum (in hertz) between adjacent peaks in a multiplet; • a quantitative measure of the influence of the spin-spin coupling with adjacent nuclei 13 -34
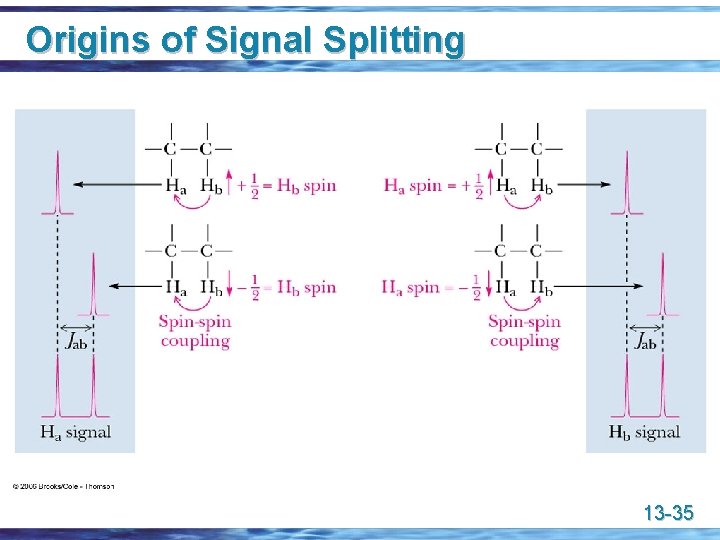
Origins of Signal Splitting 13 -35
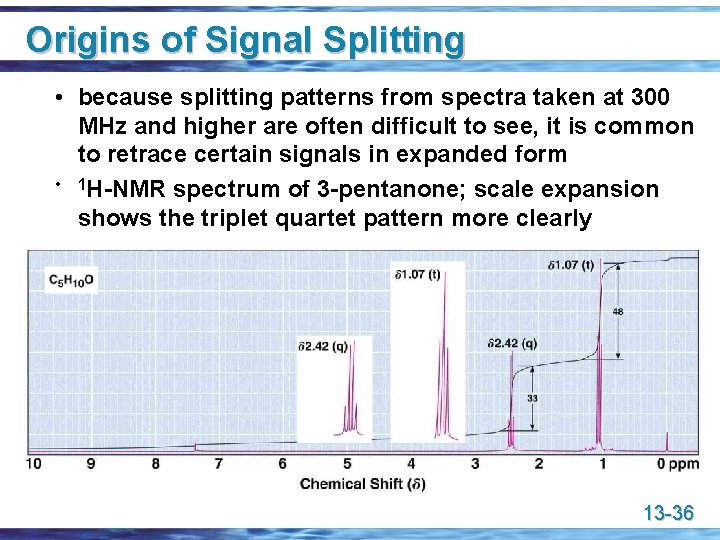
Origins of Signal Splitting • because splitting patterns from spectra taken at 300 MHz and higher are often difficult to see, it is common to retrace certain signals in expanded form • 1 H-NMR spectrum of 3 -pentanone; scale expansion shows the triplet quartet pattern more clearly 13 -36

Coupling Constants u Coupling constant (J): the distance between peaks in a split signal, expressed in hertz • the value is a quantitative measure of the magnetic interaction of nuclei whose spins are coupled 13 -37
Origins of Signal Splitting 13 -38
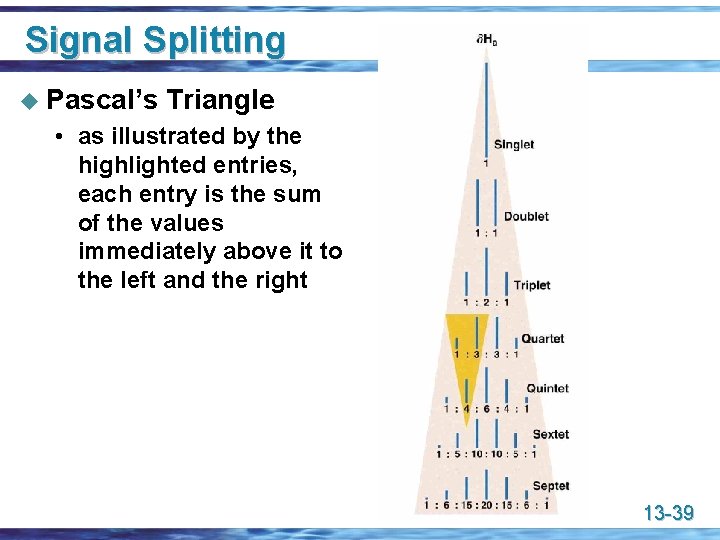
Signal Splitting u Pascal’s Triangle • as illustrated by the highlighted entries, each entry is the sum of the values immediately above it to the left and the right 13 -39
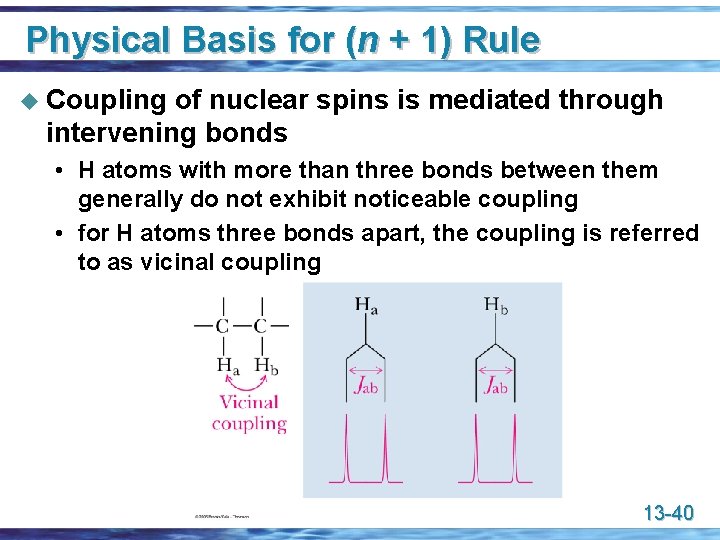
Physical Basis for (n + 1) Rule u Coupling of nuclear spins is mediated through intervening bonds • H atoms with more than three bonds between them generally do not exhibit noticeable coupling • for H atoms three bonds apart, the coupling is referred to as vicinal coupling 13 -40
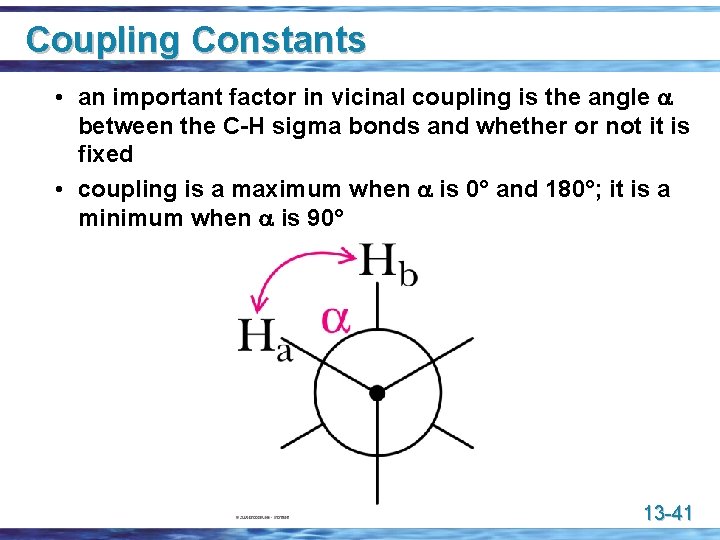
Coupling Constants • an important factor in vicinal coupling is the angle a between the C-H sigma bonds and whether or not it is fixed • coupling is a maximum when a is 0° and 180°; it is a minimum when a is 90° 13 -41
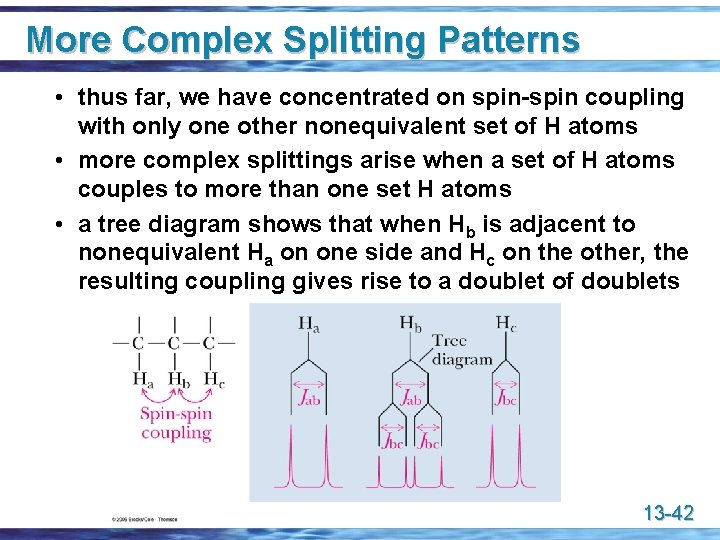
More Complex Splitting Patterns • thus far, we have concentrated on spin-spin coupling with only one other nonequivalent set of H atoms • more complex splittings arise when a set of H atoms couples to more than one set H atoms • a tree diagram shows that when Hb is adjacent to nonequivalent Ha on one side and Hc on the other, the resulting coupling gives rise to a doublet of doublets 13 -42
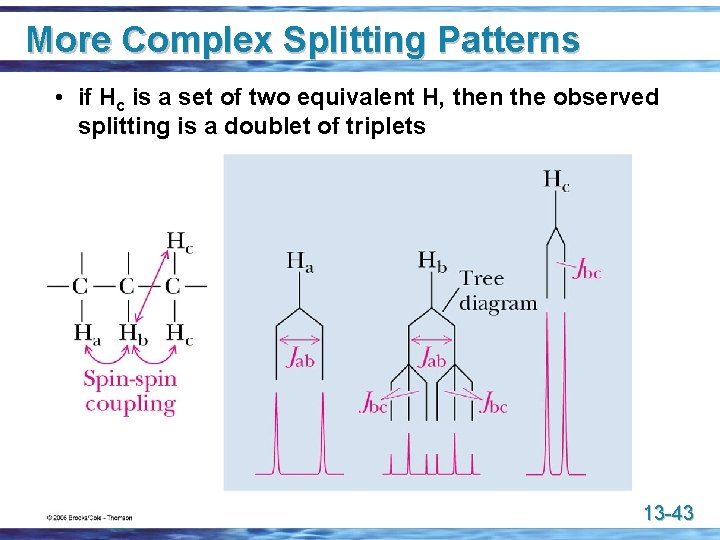
More Complex Splitting Patterns • if Hc is a set of two equivalent H, then the observed splitting is a doublet of triplets 13 -43

More Complex Splitting Patterns • because the angle between C-H bond determines the extent of coupling, bond rotation is a key parameter • in molecules with relatively free rotation about C-C sigma bonds, H atoms bonded to the same carbon in CH 3 and CH 2 groups generally are equivalent • if there is restricted rotation, as in alkenes and cyclic structures, H atoms bonded to the same carbon may not be equivalent • nonequivalent H on the same carbon will couple and cause signal splitting • this type of coupling is called geminal coupling 13 -44
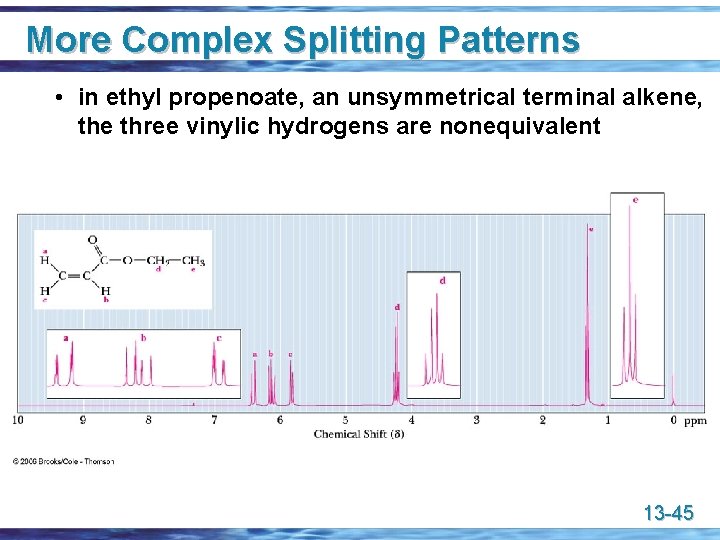
More Complex Splitting Patterns • in ethyl propenoate, an unsymmetrical terminal alkene, the three vinylic hydrogens are nonequivalent 13 -45
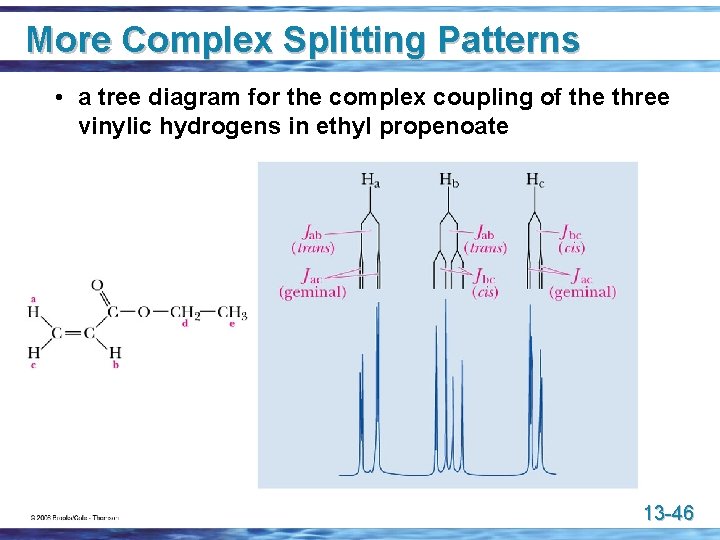
More Complex Splitting Patterns • a tree diagram for the complex coupling of the three vinylic hydrogens in ethyl propenoate 13 -46
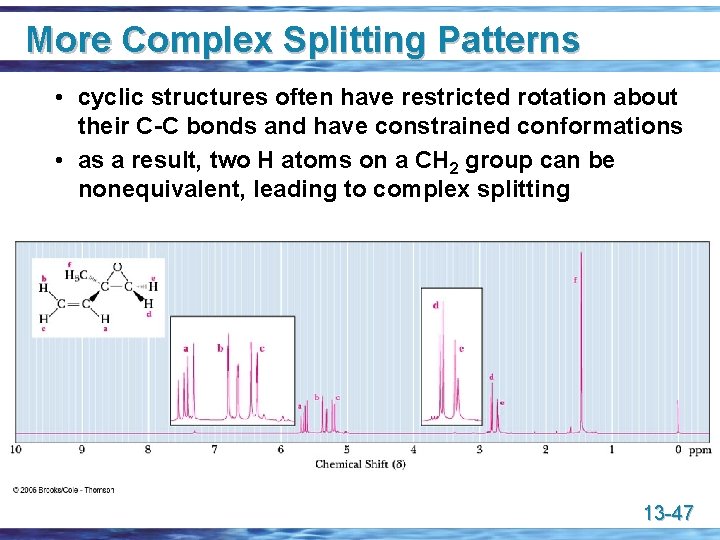
More Complex Splitting Patterns • cyclic structures often have restricted rotation about their C-C bonds and have constrained conformations • as a result, two H atoms on a CH 2 group can be nonequivalent, leading to complex splitting 13 -47
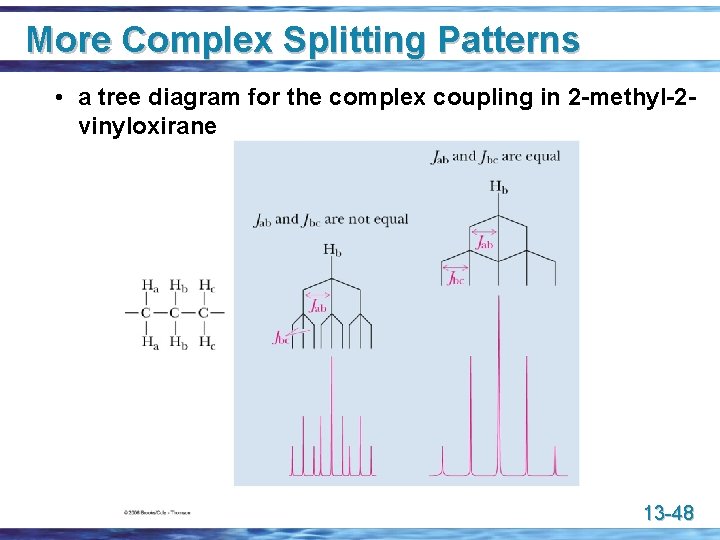
More Complex Splitting Patterns • a tree diagram for the complex coupling in 2 -methyl-2 vinyloxirane 13 -48

More Complex Splitting Patterns u Complex coupling in flexible molecules • coupling in molecules with unrestricted bond rotation often gives only m + n + I peaks • that is, the number of peaks for a signal is the number of adjacent hydrogens + 1, no matter how many different sets of equivalent H atoms that represents • the explanation is that bond rotation averages the coupling constants throughout molecules with freely rotation bonds and tends to make them similar; for example in the 6 - to 8 -Hz range for H atoms on freely rotating sp 3 hybridized C atoms 13 -49
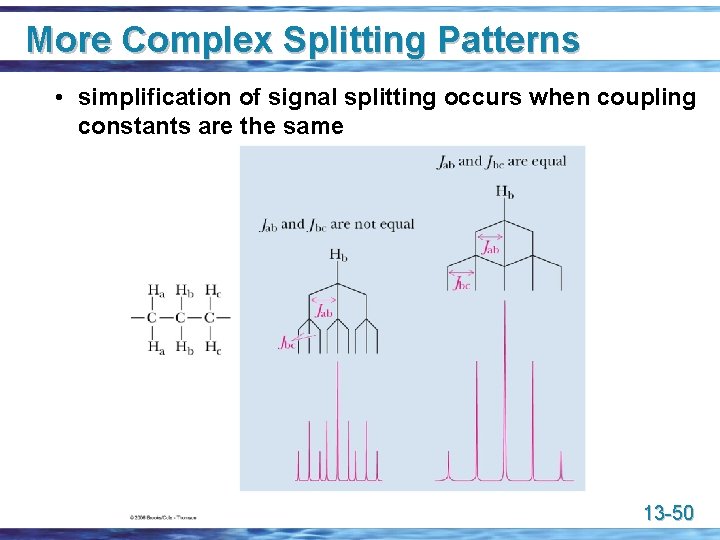
More Complex Splitting Patterns • simplification of signal splitting occurs when coupling constants are the same 13 -50
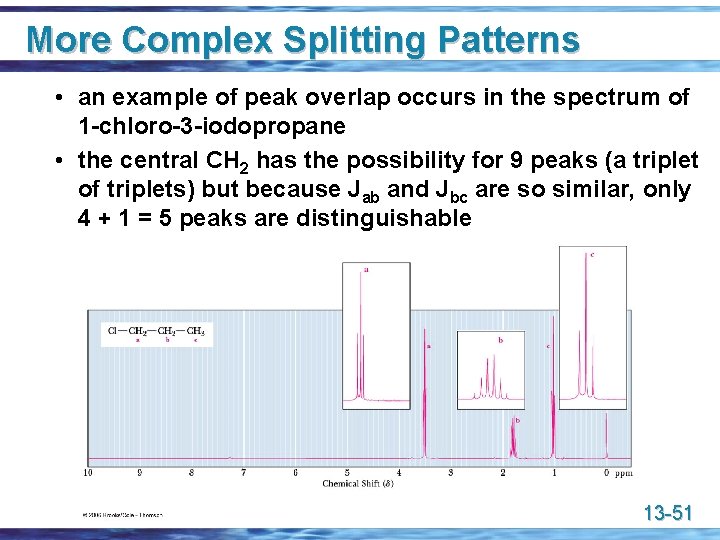
More Complex Splitting Patterns • an example of peak overlap occurs in the spectrum of 1 -chloro-3 -iodopropane • the central CH 2 has the possibility for 9 peaks (a triplet of triplets) but because Jab and Jbc are so similar, only 4 + 1 = 5 peaks are distinguishable 13 -51

Stereochemistry & Topicity u Depending on the symmetry of a molecule, otherwise equivalent hydrogens may be • homotopic • enantiotopic • diastereotopic u The simplest way to visualize topicity is to substitute an atom or group by an isotope; is the resulting compound • the same as its mirror image • different from its mirror image • are diastereomers possible 13 -52
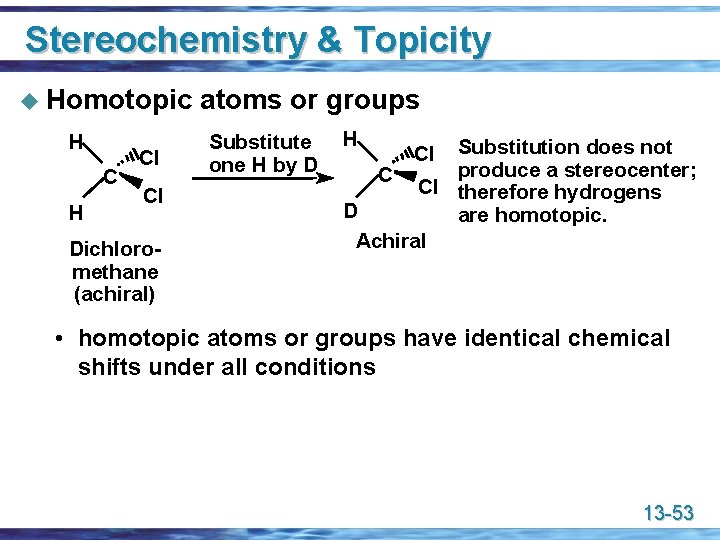
Stereochemistry & Topicity u Homotopic H Cl Cl Dichloromethane (achiral) atoms or groups Substitute one H by D H Substitution does not produce a stereocenter; C Cl therefore hydrogens D are homotopic. Achiral Cl • homotopic atoms or groups have identical chemical shifts under all conditions 13 -53
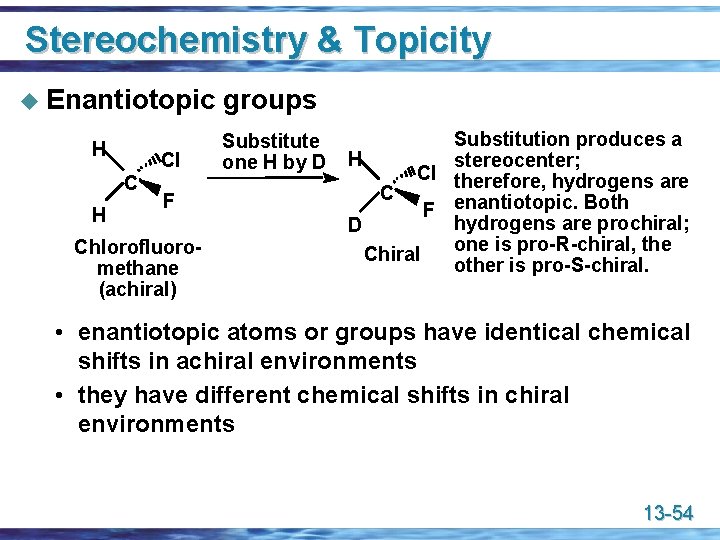
Stereochemistry & Topicity u Enantiotopic H Cl C H F Chlorofluoromethane (achiral) groups Substitute one H by D Substitution produces a H stereocenter; Cl therefore, hydrogens are C F enantiotopic. Both hydrogens are prochiral; D one is pro-R-chiral, the Chiral other is pro-S-chiral. • enantiotopic atoms or groups have identical chemical shifts in achiral environments • they have different chemical shifts in chiral environments 13 -54

Stereochemistry & Topicity u Diastereotopic groups • H atoms on C-3 of 2 -butanol are diastereotopic • substitution by deuterium creates a chiral center • because there is already a chiral center in the molecule, diastereomers are now possible • diastereotopic hydrogens have different chemical shifts under all conditions 13 -55

Stereochemistry & Topicity u The methyl groups on carbon 3 of 3 -methyl-2 butanol are diastereotopic • if a methyl hydrogen of carbon 4 is substituted by deuterium, a new chiral center is created • because there is already one chiral center, diastereomers are now possible OH 3 -Methyl-2 -butanol • protons of the methyl groups on carbon 3 have different chemical shifts 13 -56
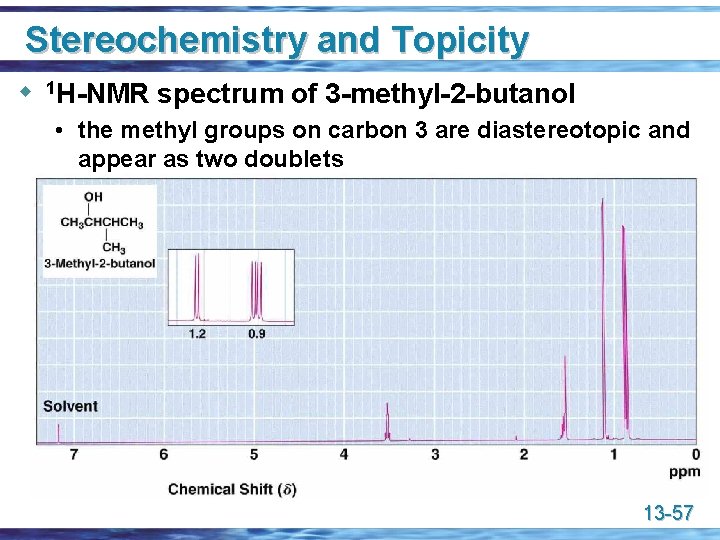
Stereochemistry and Topicity u 1 H-NMR spectrum of 3 -methyl-2 -butanol • the methyl groups on carbon 3 are diastereotopic and appear as two doublets 13 -57

13 C-NMR u Each Spectroscopy nonequivalent 13 C gives a different signal • a 13 C signal is split by the 1 H bonded to it according to the (n + 1) rule • coupling constants of 100 -250 Hz are common, which means that there is often significant overlap between signals, and splitting patterns can be very difficult to determine u The most common mode of operation of a 13 CNMR spectrometer is a hydrogen-decoupled mode 13 -58

13 C-NMR Spectroscopy u In a hydrogen-decoupled mode, a sample is irradiated with two different radio frequencies • one to excite all 13 C nuclei • a second broad spectrum of frequencies to cause all hydrogens in the molecule to undergo rapid transitions between their nuclear spin states u On the time scale of a 13 C-NMR spectrum, each hydrogen is in an average or effectively constant nuclear spin state, with the result that 1 H-13 C spin -spin interactions are not observed; they are decoupled 13 -59
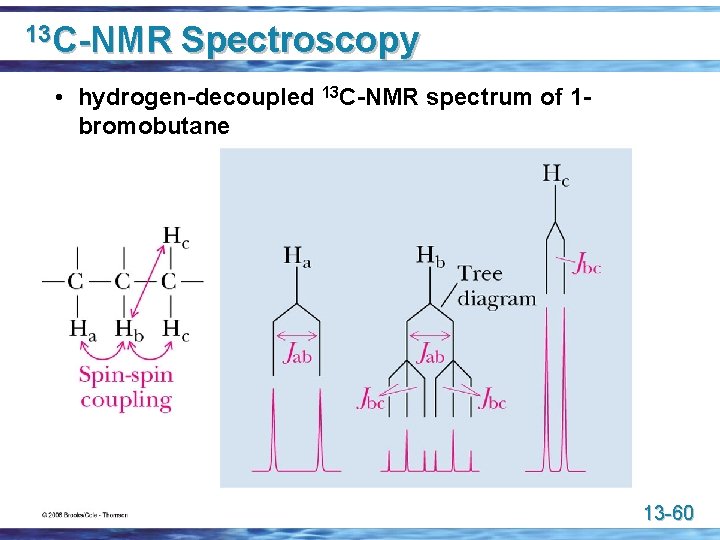
13 C-NMR Spectroscopy • hydrogen-decoupled 13 C-NMR spectrum of 1 bromobutane 13 -60

Chemical Shift - 13 C-NMR 13 -61
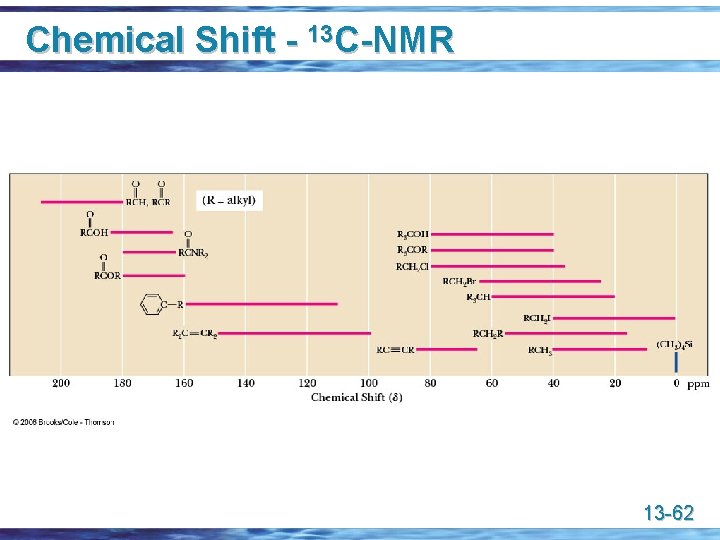
Chemical Shift - 13 C-NMR 13 -62

The DEPT Method u In the hydrogen-decoupled mode, information on spin-spin coupling between 13 C and hydrogens bonded to it is lost u The DEPT method is an instrumental mode that provides a way to acquire this information • Distortionless Enhancement by Polarization Transfer (DEPT): an NMR technique for distinguishing among 13 C signals for CH , CH, and quaternary carbons 3 2 13 -63

The DEPT Method u The DEPT methods uses a complex series of pulses in both the 1 H and 13 C ranges, with the result that CH 3, CH 2, and CH signals exhibit different phases; • signals for CH 3 and CH carbons are recorded as positive signals • signals for CH 2 carbons are recorded as negative signals • quaternary carbons give no signal in the DEPT method 13 -64
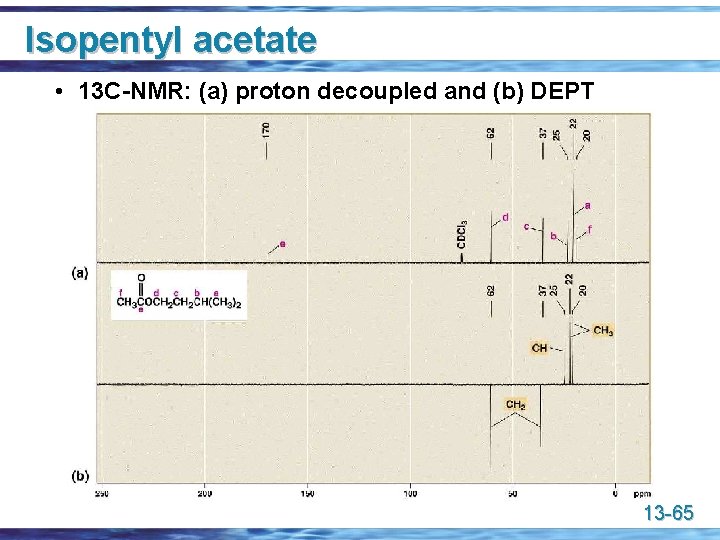
Isopentyl acetate • 13 C-NMR: (a) proton decoupled and (b) DEPT 13 -65

Interpreting NMR Spectra u Alkanes • • signals appear in the range of 0. 8 -1. 7 13 C-NMR signals appear in the considerably wider range of 10 -60 1 H-NMR u Alkenes • • • signals appear in the range 4. 6 -5. 7 1 H-NMR coupling constants are generally larger for trans vinylic hydrogens (J= 11 -18 Hz) compared with cis vinylic hydrogens (J= 5 -10 Hz) 13 C-NMR signals for sp 2 hybridized carbons appear in the range 100 -160, which is downfield from the signals of sp 3 hybridized carbons 1 H-NMR 13 -66
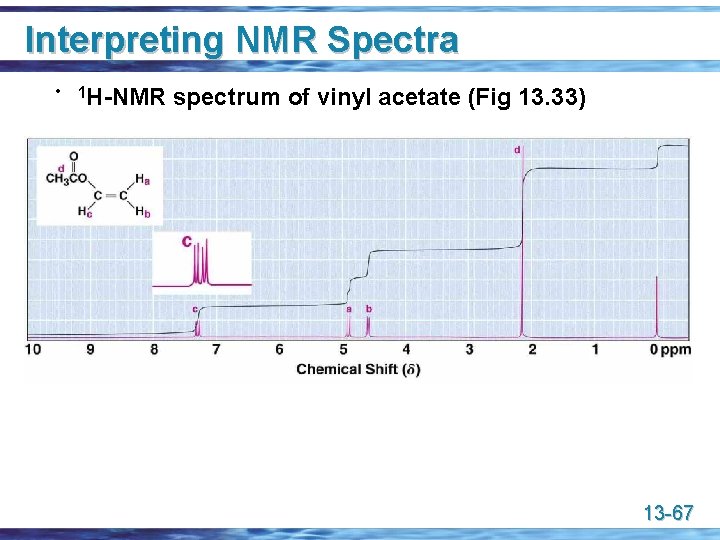
Interpreting NMR Spectra • 1 H-NMR spectrum of vinyl acetate (Fig 13. 33) 13 -67

Interpreting NMR Spectra u Alcohols u 1 H-NMR O-H chemical shifts often appears in the range 3. 0 -4. 0, but may be as low as 0. 5. • 1 H-NMR chemical shifts of hydrogens on the carbon bearing the -OH group are deshielded by the electronwithdrawing inductive effect of the oxygen and appear in the range 3. 0 -4. 0 u Ethers • a distinctive feature in the 1 H-MNR spectra of ethers is the chemical shift, 3. 3 -4. 0, of hydrogens on carbon attached to the ether oxygen 13 -68
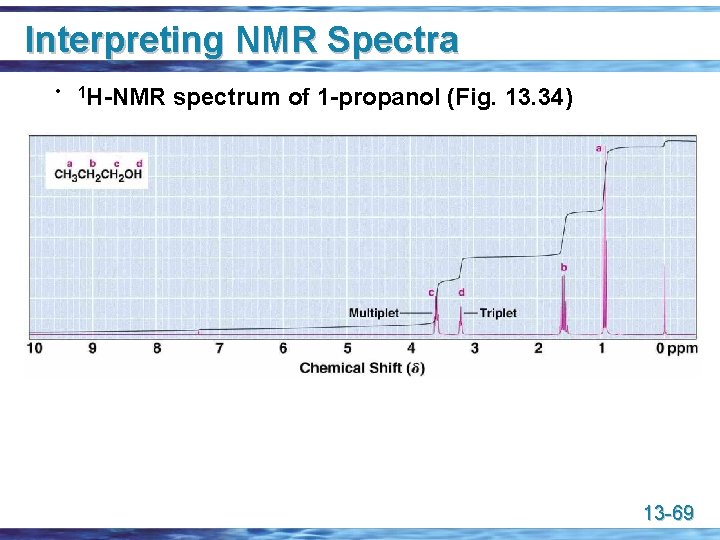
Interpreting NMR Spectra • 1 H-NMR spectrum of 1 -propanol (Fig. 13. 34) 13 -69

Interpreting NMR Spectra u Aldehydes • • • and ketones aldehyde hydrogens appear at 9. 5 -10. 1 1 H-NMR: a-hydrogens of aldehydes and ketones appear at 2. 2 -2. 6 13 C-NMR: carbonyl carbons appear at 180 -215 1 H-NMR: u Amines • amine hydrogens appear at 0. 5 -5. 0 depending on conditions 1 H-NMR: 13 -70
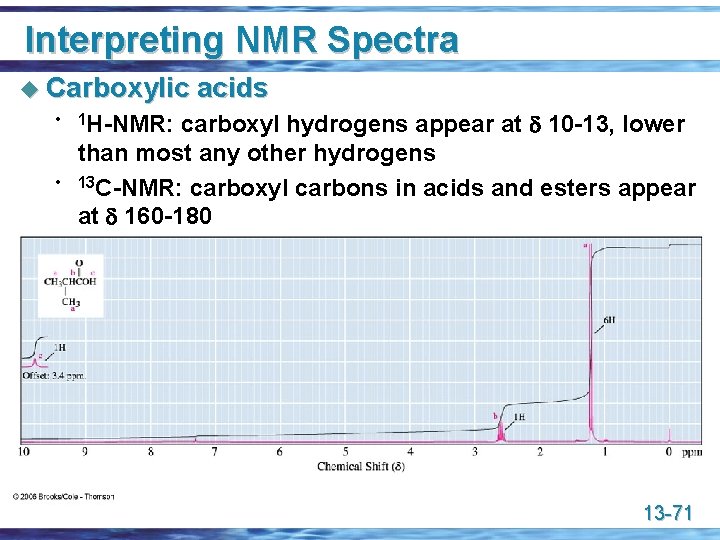
Interpreting NMR Spectra u Carboxylic • • acids carboxyl hydrogens appear at 10 -13, lower than most any other hydrogens 13 C-NMR: carboxyl carbons in acids and esters appear at 160 -180 1 H-NMR: 13 -71
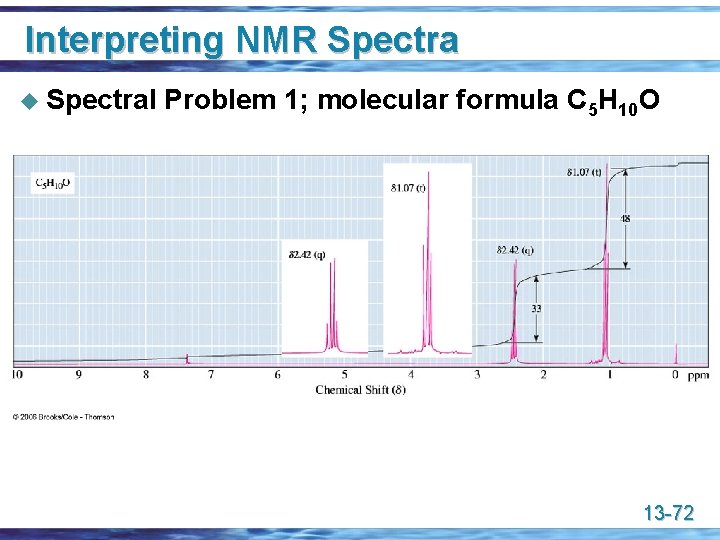
Interpreting NMR Spectra u Spectral Problem 1; molecular formula C 5 H 10 O 13 -72
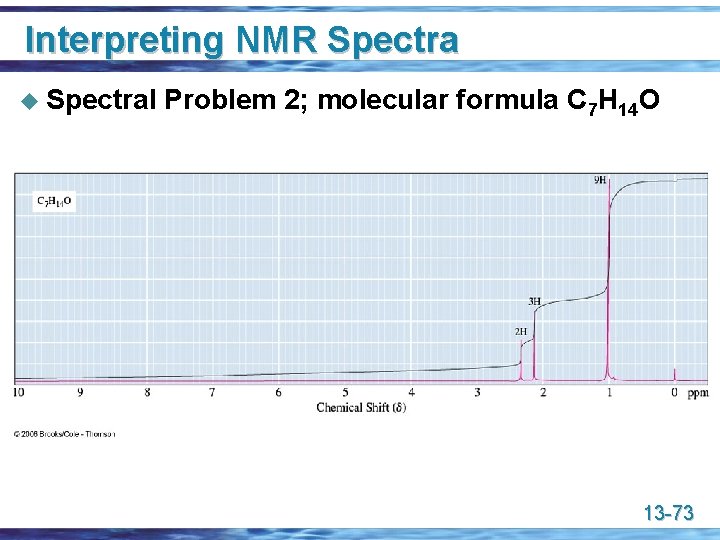
Interpreting NMR Spectra u Spectral Problem 2; molecular formula C 7 H 14 O 13 -73

Nuclear Magnetic Resonance End Chapter 13 13 -74
- Slides: 74