Organic Chemistry The Chemistry of Carbon Table of
Organic Chemistry The Chemistry of Carbon

Table of Contents ‘Organic Chemistry’ Distilling Crude Oil Methane Ethane Propane Butane Pentane Alkanes Alcohols Aldehydes and Ketones Ethers Functional Groups Alkenes & Alkynes Cycloalkanes Benzene Aromatic Hydrocarbons Carboxylic Acids Classes of Organic Compounds Chirality Polymers

Organic Chemistry: The chemistry of carbon and carbon-based compounds Organic Chemistry in everyday life: Smells & tastes: fruits, chocolate, fish, mint Medications: aspirin, Tylenol, decongestants, sedatives Addictive substances: caffeine, nicotine, alcohol, narcotics Hormones/Neurotransmitters: adrenaline, epinephrine Food: carbohydrates, protein, fat Genetics: DNA, RNA Consumer products: plastics, nylon, rayon, polyester

Friedrich Wöhler Made the first organic compound from non-living substances. Shot down the Vital Force idea of organic substances.

Distilling Crude Oil
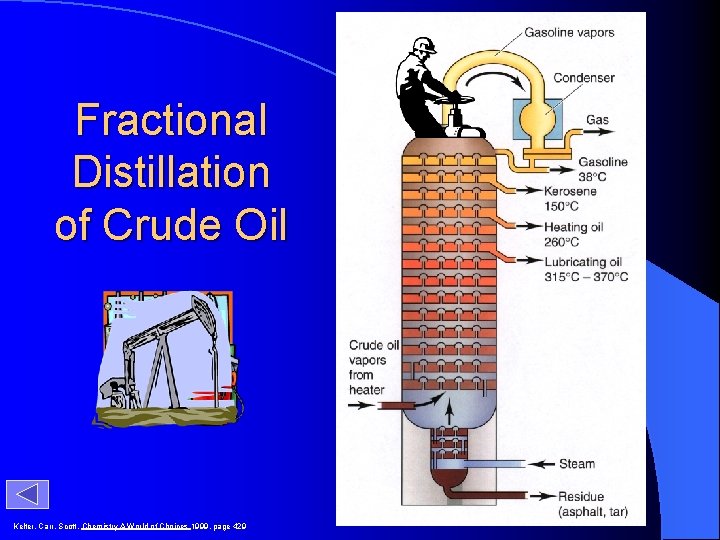
Fractional Distillation of Crude Oil Kelter, Carr, Scott, Chemistry A World of Choices 1999, page 429
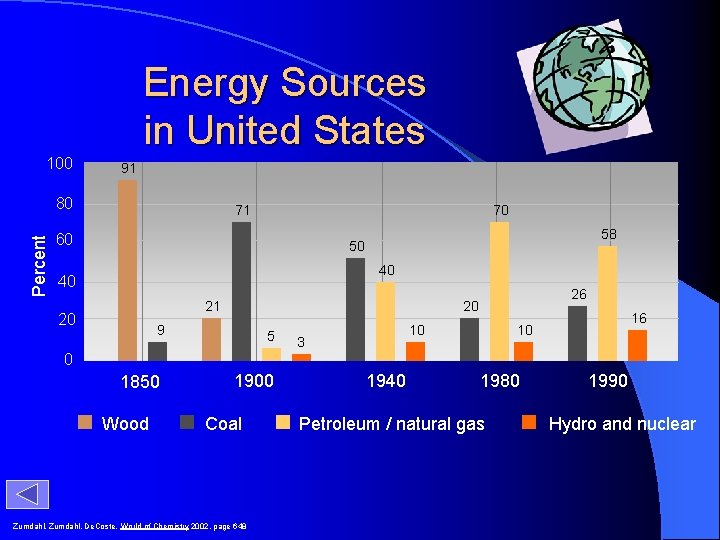
Energy Sources in United States 100 91 Percent 80 71 70 60 58 50 40 40 21 20 26 20 9 5 10 3 16 10 0 1850 Wood 1900 Coal Zumdahl, De. Coste, World of Chemistry 2002, page 648 1940 1980 Petroleum / natural gas 1990 Hydro and nuclear

Distilling Crude Oil Separate fractions based on differences in boiling point.

American Petroleum Usage AMERICAN PETROLEUM USAGE A Lesson in Economics 1 Barrel of Petroleum 93% Energy Marketplace 7% Materials Marketplace (42 gallons) 42 Gallons of Crude Petroleum 36 Gallons Fuel At $3. 00 per gallon 36 Gallons Fuel 19 Gallons Gasoline (11 Gallons in 1920) $108. 00 OR 7% (3 Gallons Crude) At $20. 00 per shirt 100 Nylon Shirts $2000. 00 Entrepreneurs: [Put an alligator on the shirt and make even more $$$$$]
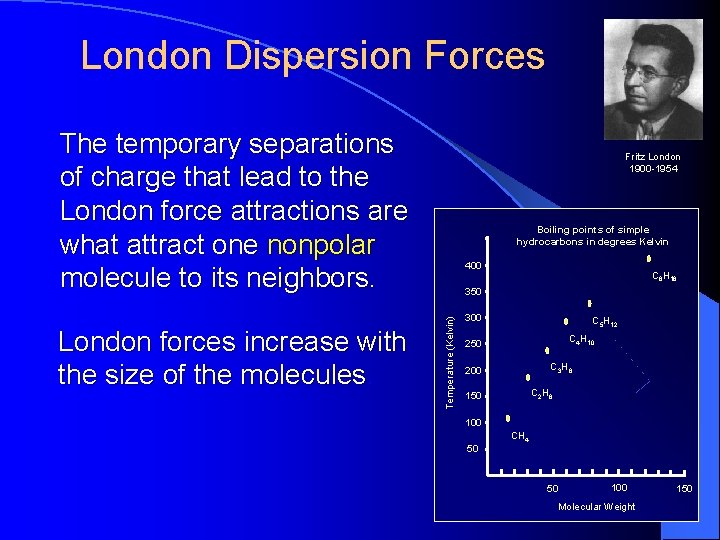
London Dispersion Forces The temporary separations of charge that lead to the London force attractions are what attract one nonpolar molecule to its neighbors. Boiling points of simple hydrocarbons in degrees Kelvin 400 C 8 H 18 350 Temperature (Kelvin) London forces increase with the size of the molecules. Fritz London 1900 -1954 300 C 5 H 12 C 4 H 10 250 C 3 H 8 200 C 2 H 6 150 100 CH 4 50 50 100 Molecular Weight 150
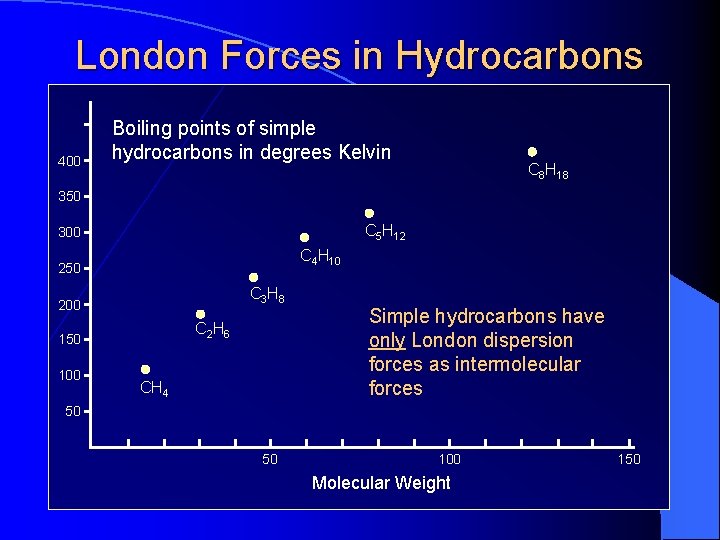
London Forces in Hydrocarbons 400 Boiling points of simple hydrocarbons in degrees Kelvin C 8 H 18 350 C 5 H 12 300 C 4 H 10 250 C 3 H 8 200 C 2 H 6 150 100 CH 4 Simple hydrocarbons have only London dispersion forces as intermolecular forces 50 50 100 Molecular Weight 150
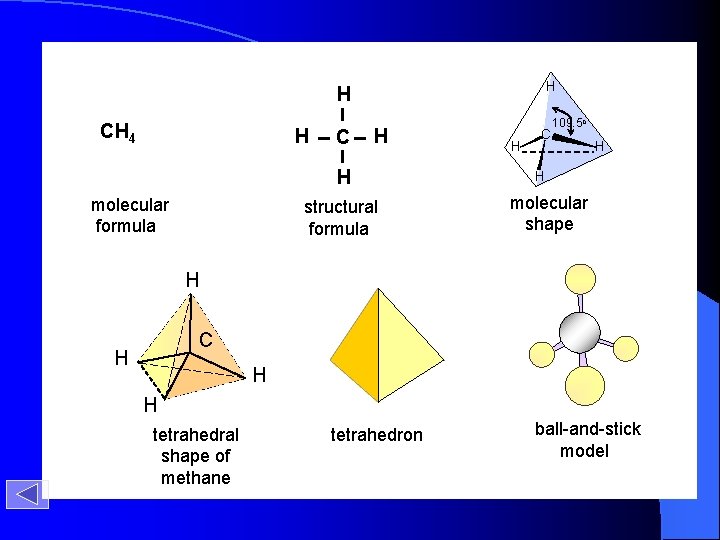
H H CH 4 H C H H molecular formula structural formula H C 109. 5 o H H molecular shape H C H H H tetrahedral shape of methane tetrahedron ball-and-stick model
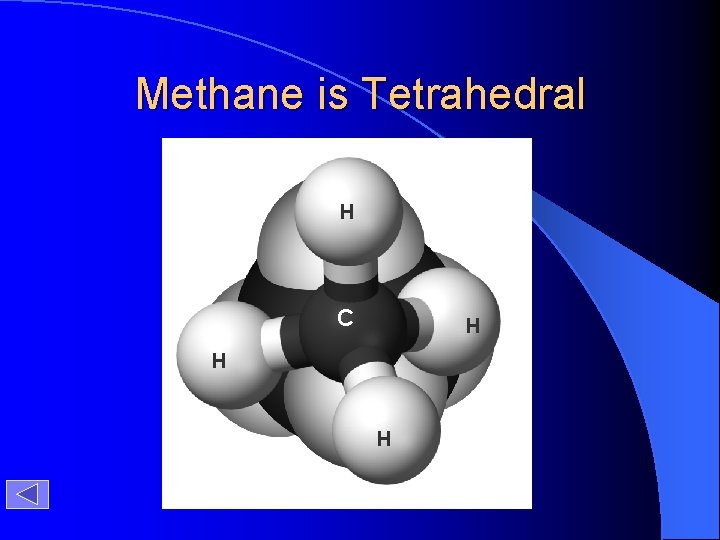
Methane is Tetrahedral H C H H H
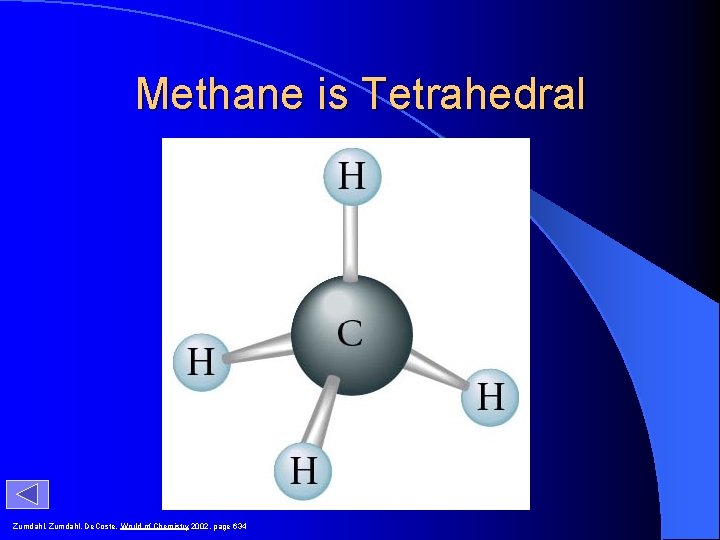
Methane is Tetrahedral Zumdahl, De. Coste, World of Chemistry 2002, page 634
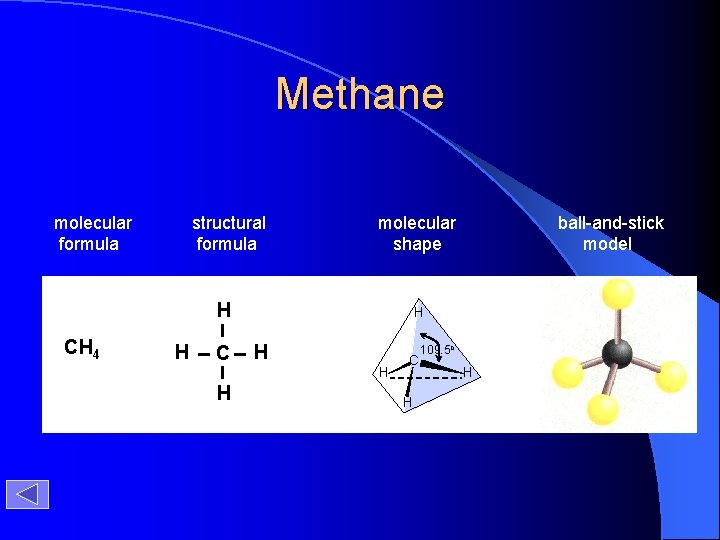
Methane molecular formula structural formula molecular shape H CH 4 H C H H ball-and-stick model C H 109. 5 o H
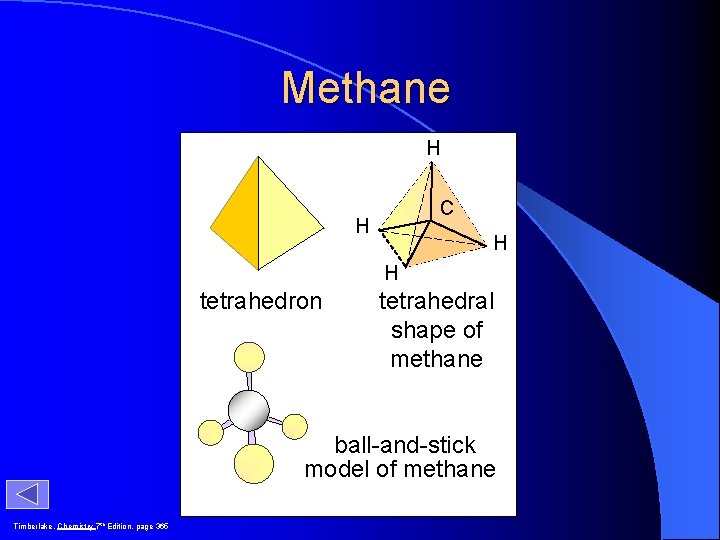
Methane H C H H H tetrahedron tetrahedral shape of methane ball-and-stick model of methane Timberlake, Chemistry 7 th Edition, page 365

Lewis structure of Ethane H H H C C H H H

Space filling model of Ethane Zumdahl, De. Coste, World of Chemistry 2002, page 635
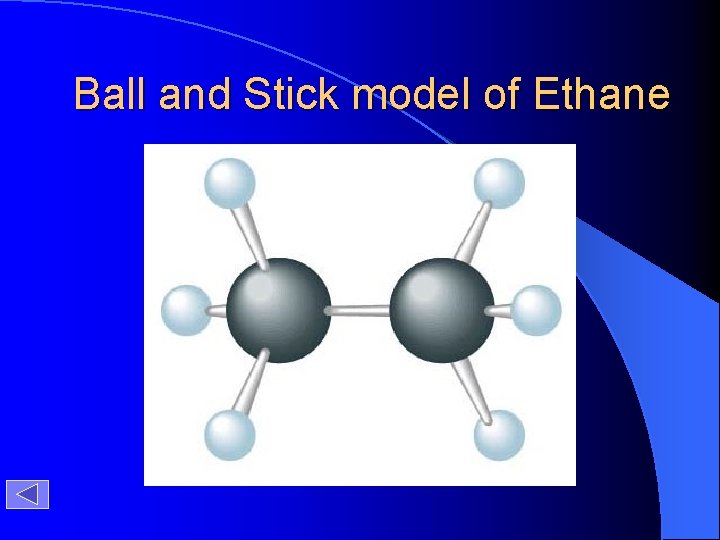
Ball and Stick model of Ethane
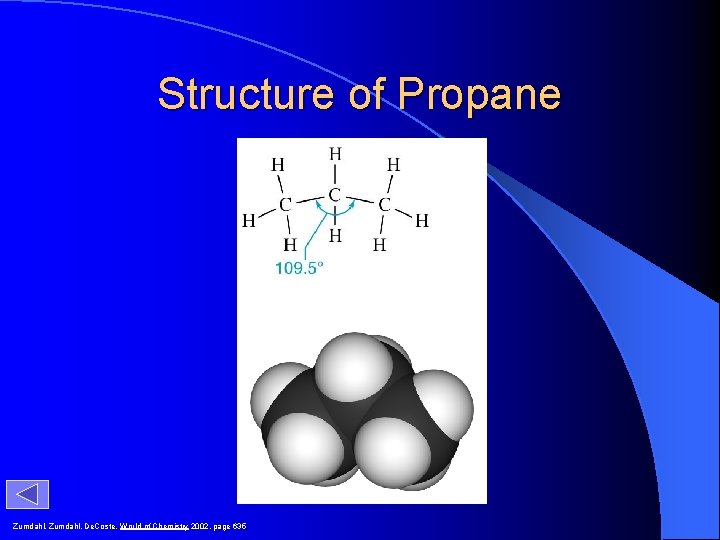
Structure of Propane Zumdahl, De. Coste, World of Chemistry 2002, page 635
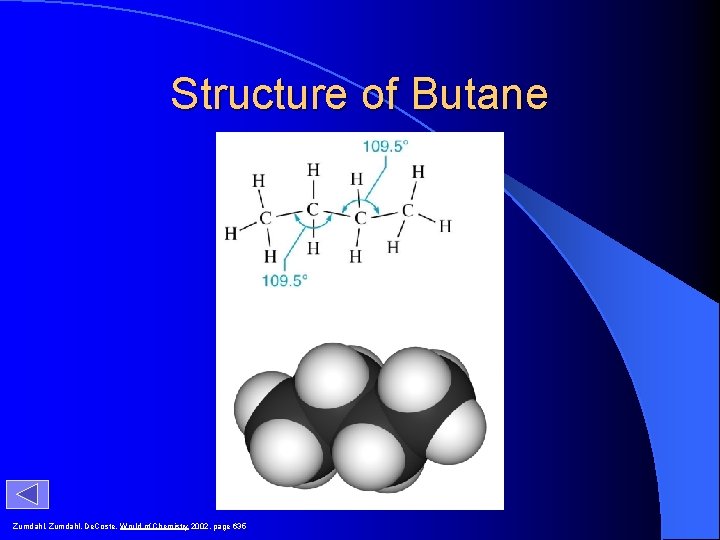
Structure of Butane Zumdahl, De. Coste, World of Chemistry 2002, page 635
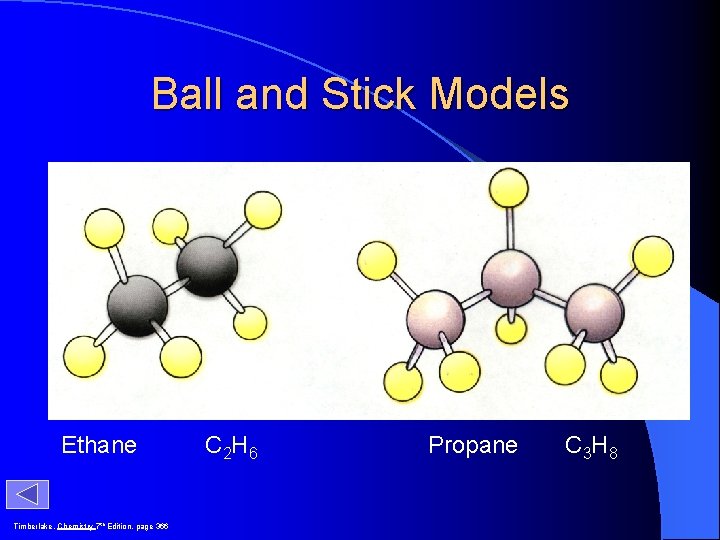
Ball and Stick Models Ethane Timberlake, Chemistry 7 th Edition, page 366 C 2 H 6 Propane C 3 H 8

First Ten Hydrocarbons Name Number of Carbon Atoms Molecular Formula Condensed Structural Formula Methane 1 CH 4 Ethane 2 C 2 H 6 CH 3 Propane 3 C 3 H 8 CH 3 CH 2 CH 3 Butane 4 C 4 H 10 CH 3 CH 2 CH 3 Pentane 5 C 5 H 12 CH 3 CH 2 CH 2 CH 3 Hexane 6 C 6 H 14 CH 3 CH 2 CH 2 CH 3 Heptane 7 C 7 H 16 CH 3 CH 2 CH 2 CH 2 CH 3 Octane 8 C 8 H 18 CH 3 CH 2 CH 2 CH 2 CH 3 Nonane 9 C 9 H 20 CH 3 CH 2 CH 2 CH 3 Decane 10 C 10 H 22 CH 3 CH 2 CH 2 CH 3
Hydrocarbons (alkanes)

First Ten Hydrocarbons Name Number of Carbon Atoms Molecular Formula Melting Point, o. C Boiling Point, o. C # of Isomers Methane 1 CH 4 -182. 5 -161. 5 0 Ethane 2 C 2 H 6 -183. 2 -88. 6 0 n-Propane 3 C 3 H 8 -187. 7 -42. 1 0 n-Butane 4 C 4 H 10 -138. 3 -0. 5 2 n-Pentane 5 C 5 H 12 -129. 7 36. 1 3 n-Hexane 6 C 6 H 14 -95. 3 68. 7 5 n-Heptane 7 C 7 H 16 -90. 6 98. 4 9 n-Octane 8 C 8 H 18 -56. 8 125. 7 18 n-Nonane 9 C 9 H 20 -53. 6 150. 8 35 n-Decane 10 C 10 H 22 -29. 7 174. 0 75

Increasing mass and boiling point Methane 16 g/mol -161. 5 o. C Ethane 30 g/mol -88. 6 o. C Propane 44 g/mol -42. 1 o. C n-Butane 58 g/mol -0. 5 o. C
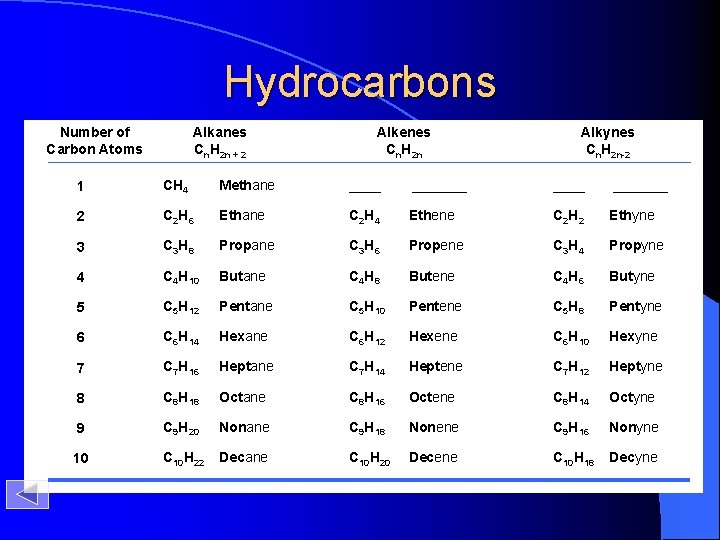
Hydrocarbons Number of Carbon Atoms Alkanes Cn. H 2 n + 2 Alkenes Cn. H 2 n Alkynes Cn. H 2 n-2 1 CH 4 Methane _______ 2 C 2 H 6 Ethane C 2 H 4 Ethene C 2 H 2 Ethyne 3 C 3 H 8 Propane C 3 H 6 Propene C 3 H 4 Propyne 4 C 4 H 10 Butane C 4 H 8 Butene C 4 H 6 Butyne 5 C 5 H 12 Pentane C 5 H 10 Pentene C 5 H 8 Pentyne 6 C 6 H 14 Hexane C 6 H 12 Hexene C 6 H 10 Hexyne 7 C 7 H 16 Heptane C 7 H 14 Heptene C 7 H 12 Heptyne 8 C 8 H 18 Octane C 8 H 16 Octene C 8 H 14 Octyne 9 C 9 H 20 Nonane C 9 H 18 Nonene C 9 H 16 Nonyne 10 C 10 H 22 Decane C 10 H 20 Decene C 10 H 18 Decyne

Isomers The fat dog shook himself, and then rolled over on the wet rug. The dog shook the fat rug, then rolled over and wet on himself. (These two statements use the same words. . . but have very different meanings. )
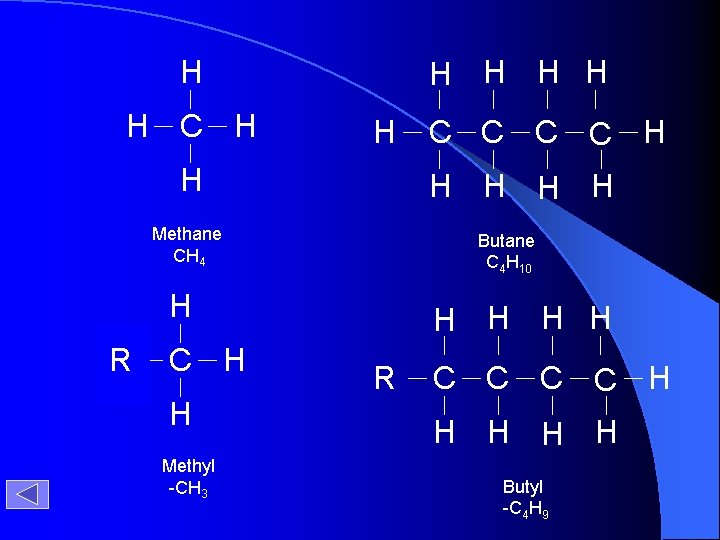
H H H C H H H H Methane CH 4 Butane C 4 H 10 H ? R C H H Methyl -CH 3 C C H H H R C C H H H Butyl -C 4 H 9

Naming Alkanes 1. Find the longest continuous chain of carbon atoms in the molecule 2. Start numbering the chain at the carbon nearer the first branching point (the substituents should have the lowest numbers possible) 3. Name and number the substituents If there are two substituents attached to the same carbon, assign both of them the same number 4. Write out the name of the molecule • List the substitutents names in alphabetical order if there are two identical substituents on the molecule, use the prefix di; if there are three identical substituents, use the prefix tri-; if there are four identical substituents, use the prefix tetra(alphabetize the substituents by the main name of the substituent, not by any prefixes the substituents may have) • Separate the substituents names with hyphens • Tag the name of the parent chain onto the end of the substituent names

A Few Extra Rules to Follow Use commas to separate numbers Use hyphens to separate numbers from the substituent names Never name alkanes after drinking Don’t allow children to name alkanes unattended
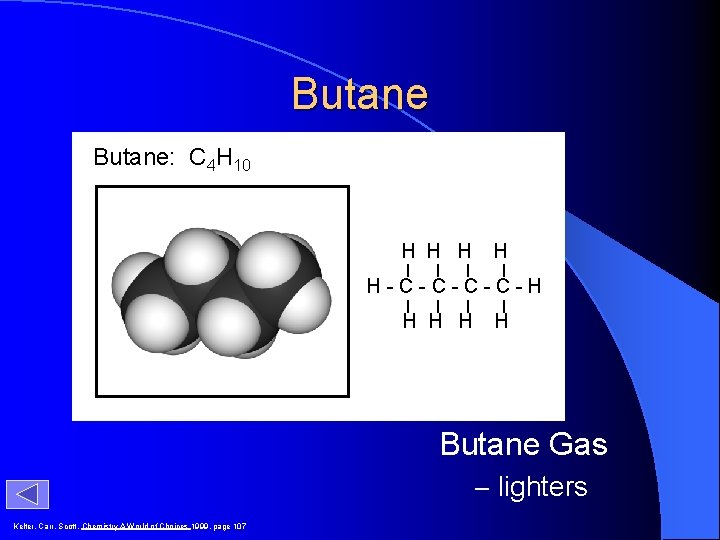
Butane: C 4 H 10 H H H-C-C-H H H Butane Gas – lighters Kelter, Carr, Scott, Chemistry A World of Choices 1999, page 107
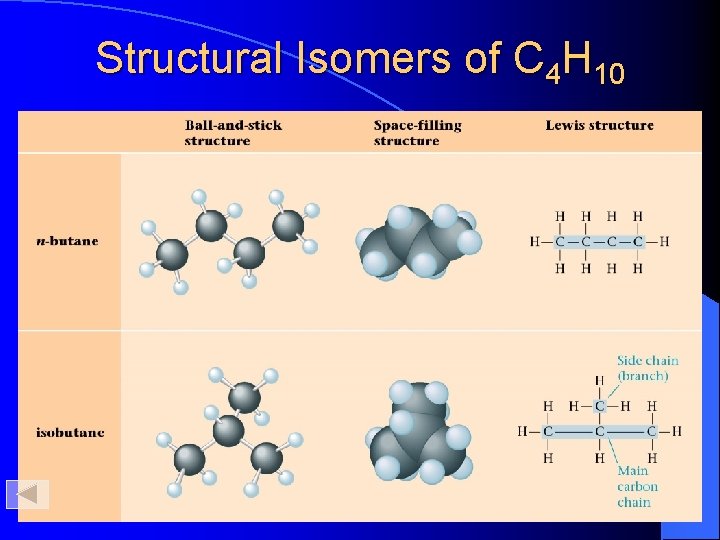
Structural Isomers of C 4 H 10
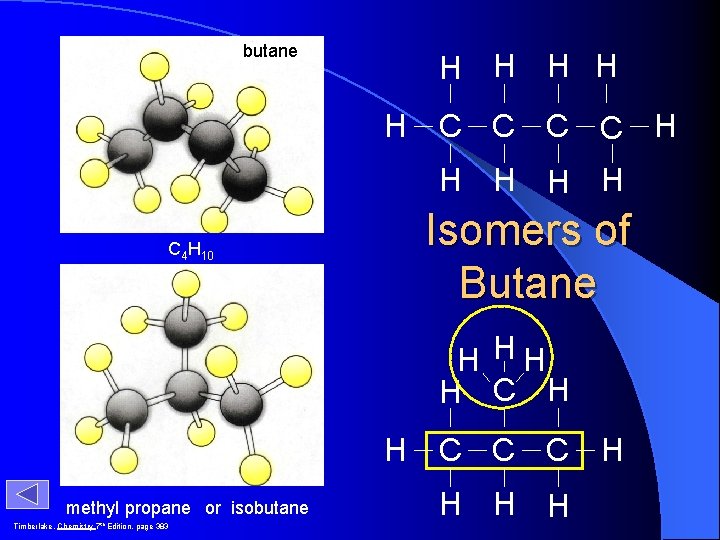
butane H H H C C H H H Isomers of Butane C 4 H 10 H HH H C H H methyl propane or isobutane Timberlake, Chemistry 7 th Edition, page 383 C C C H H
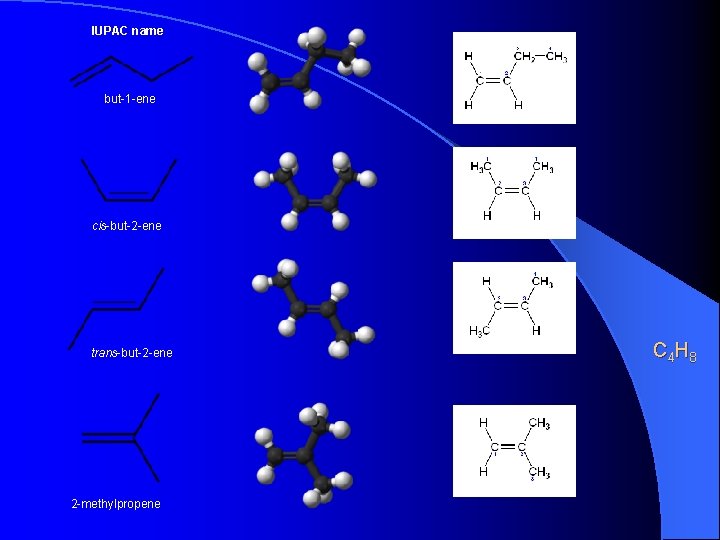
IUPAC name but-1 -ene cis-but-2 -ene trans-but-2 -ene 2 -methylpropene C 4 H 8
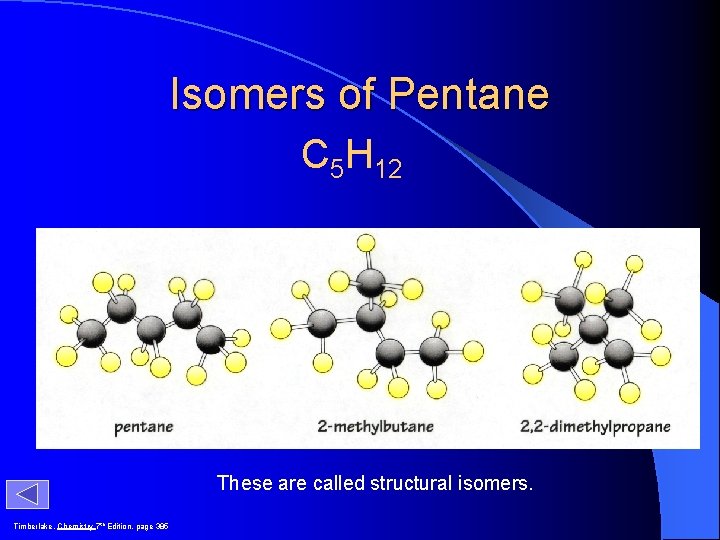
Isomers of Pentane C 5 H 12 These are called structural isomers. Timberlake, Chemistry 7 th Edition, page 385
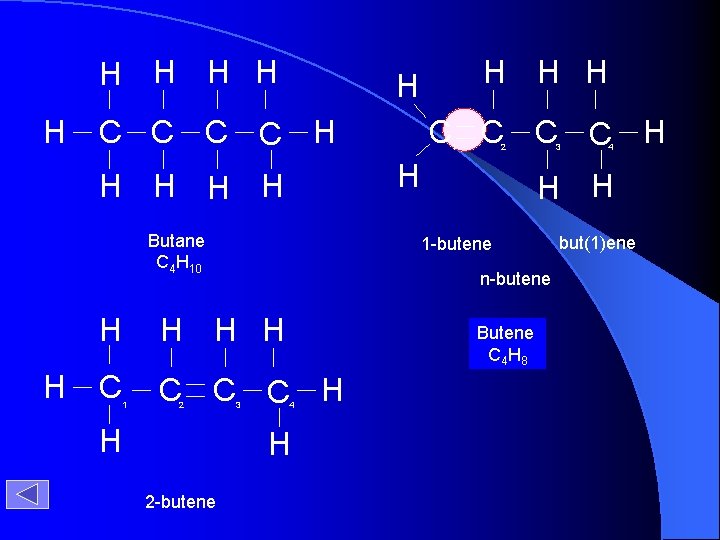
H H H C C H Butane C 4 H 10 C C H H 3 3 4 4 H H Butene Butane C 4 H 8 H 2 -butene 2 n-butene H H H 2 1 1 -butene H 1 C C H H H H H but(1)ene
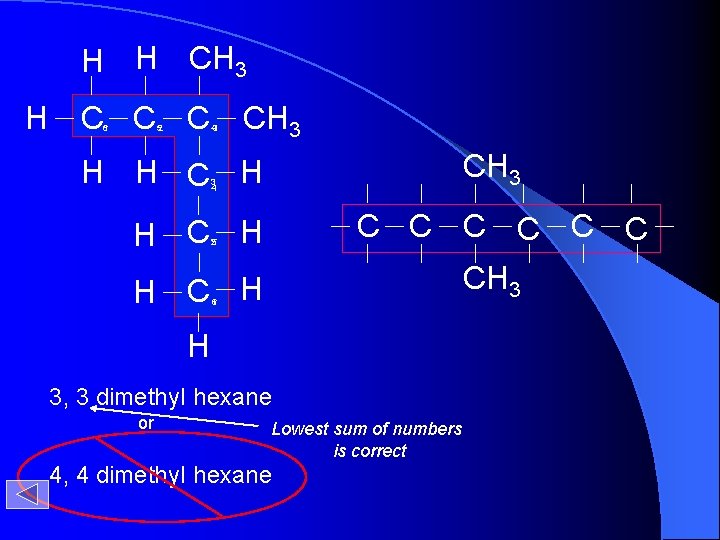
H H CH 3 H C C C 6 1 52 43 CH 3 H H C H 3 4 C C C H C H 25 CH 3 H C H 16 H 3, 3 dimethyl hexane or Lowest sum of numbers is correct 4, 4 dimethyl hexane
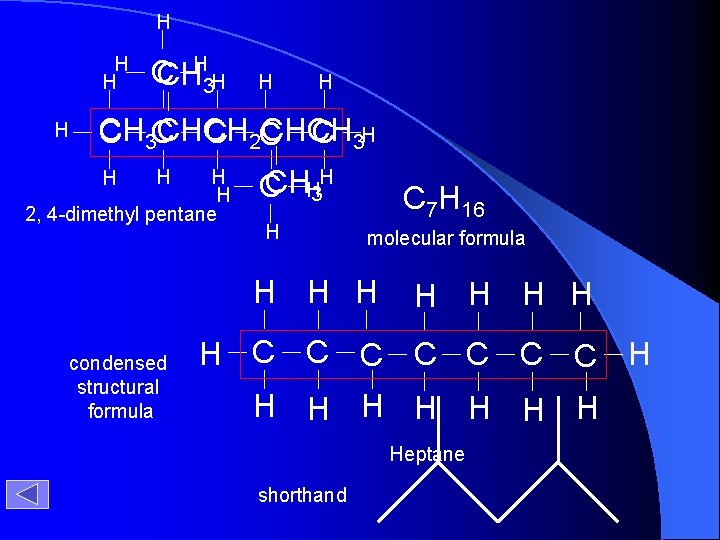
H H H C CH 3 H H H CH CHCH C 3 C C 2 C C 3 H H H 2, 4 -dimethyl pentane H H H CH C H 3 H C 7 H 16 molecular formula H H H condensed structural formula H H H C C C C H H H H Heptane shorthand
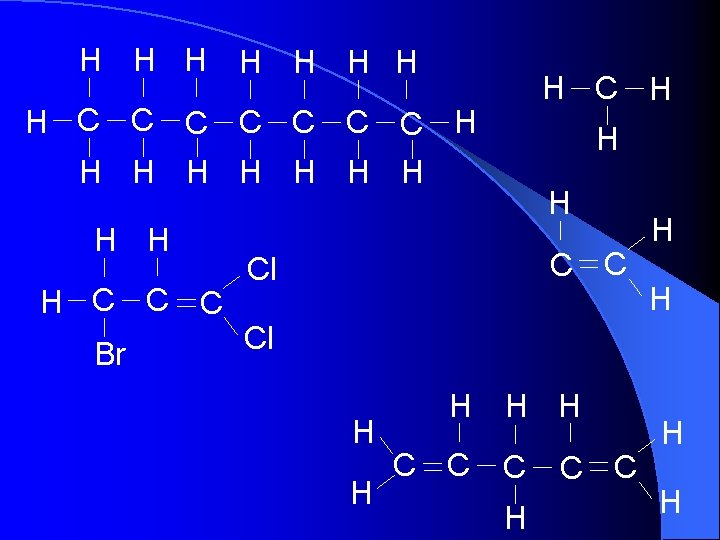
H H H H C C C C H H H C C C Br H H C C Cl H H H C C H H
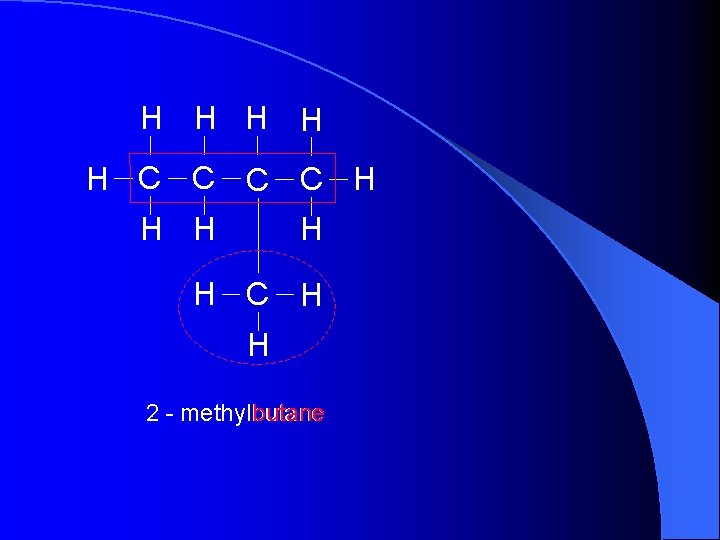
H H H C C H H H C H H butane 2 - methylbutane
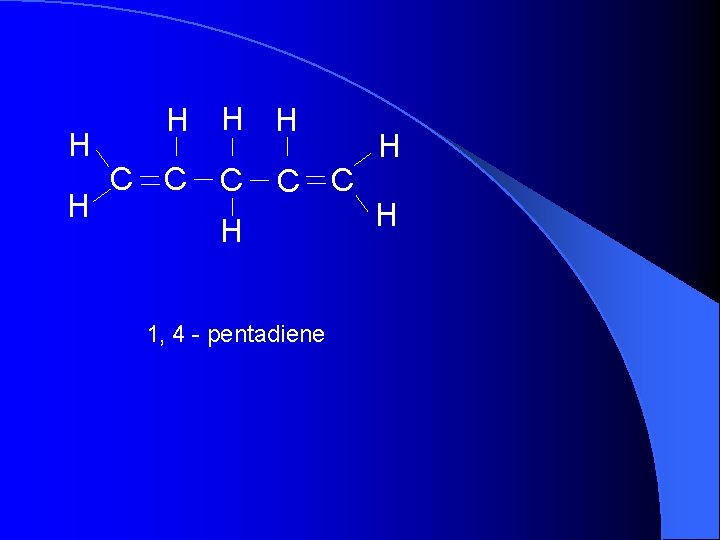
H H H C C C H 1, 4 - pentadiene H H
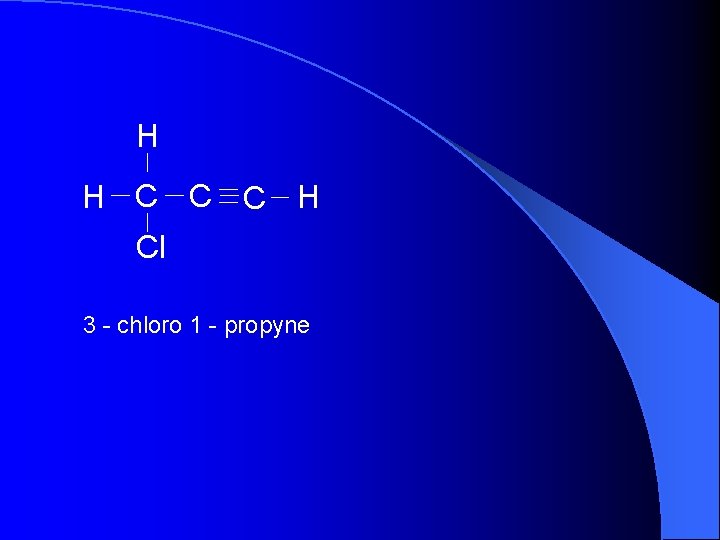
H H C C C H Cl 3 - chloro 1 - propyne
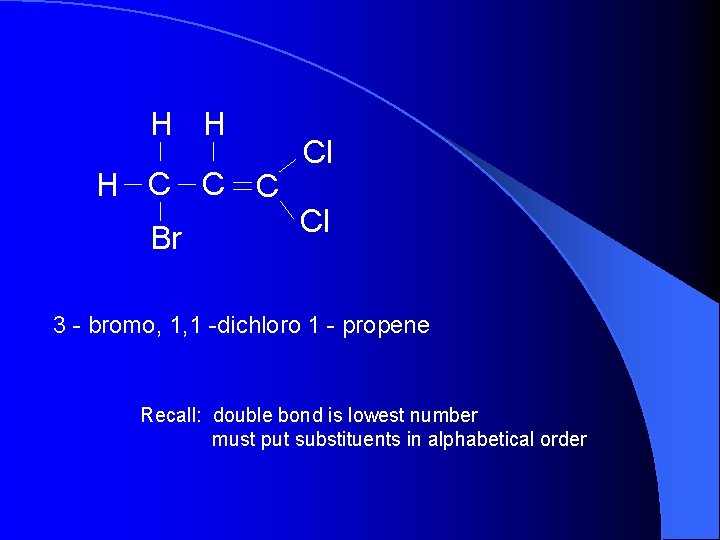
H H H C C C Br Cl Cl 3 - bromo, 1, 1 -dichloro 1 - propene Recall: double bond is lowest number must put substituents in alphabetical order
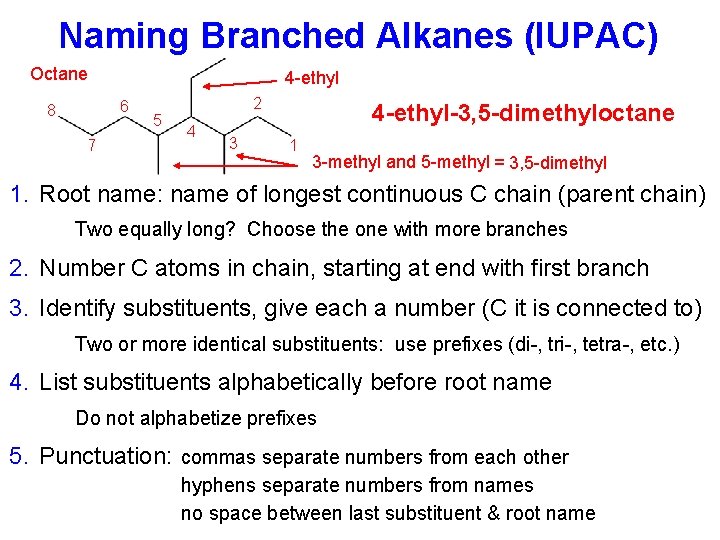
Naming Branched Alkanes (IUPAC) Octane 4 -ethyl 6 8 7 5 2 4 3 4 -ethyl-3, 5 -dimethyloctane 1 3 -methyl and 5 -methyl = 3, 5 -dimethyl 1. Root name: name of longest continuous C chain (parent chain) Two equally long? Choose the one with more branches 2. Number C atoms in chain, starting at end with first branch 3. Identify substituents, give each a number (C it is connected to) Two or more identical substituents: use prefixes (di-, tri-, tetra-, etc. ) 4. List substituents alphabetically before root name Do not alphabetize prefixes 5. Punctuation: commas separate numbers from each other hyphens separate numbers from names no space between last substituent & root name
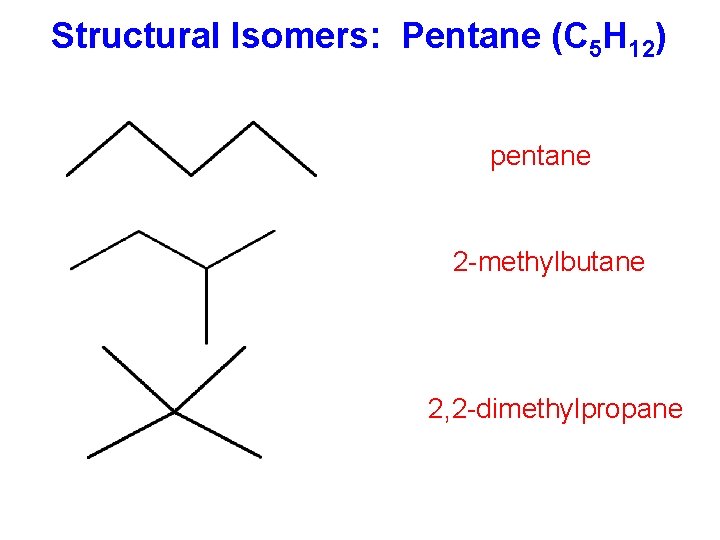
Structural Isomers: Pentane (C 5 H 12) pentane 2 -methylbutane 2, 2 -dimethylpropane
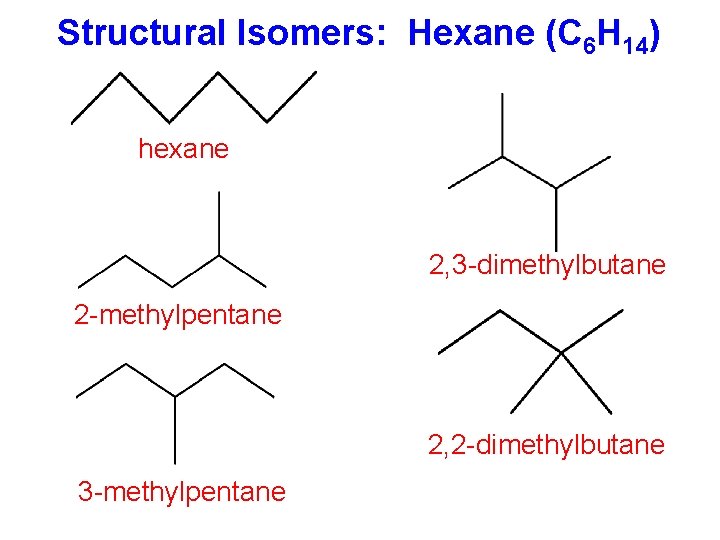
Structural Isomers: Hexane (C 6 H 14) hexane 2, 3 -dimethylbutane 2 -methylpentane 2, 2 -dimethylbutane 3 -methylpentane
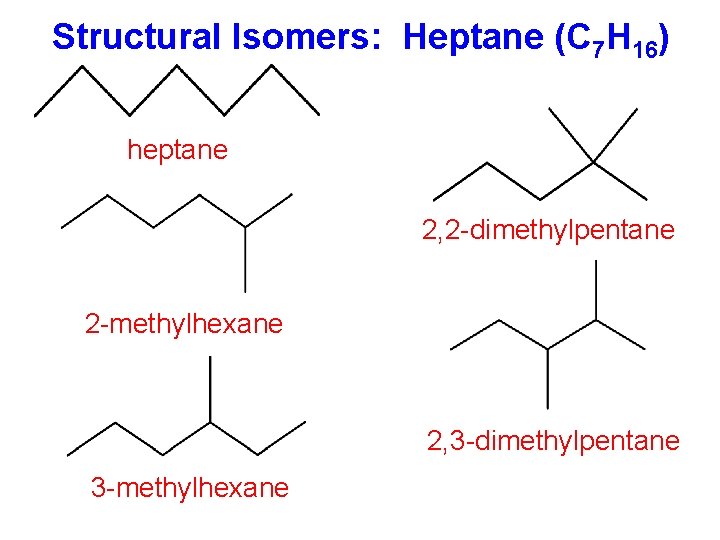
Structural Isomers: Heptane (C 7 H 16) heptane 2, 2 -dimethylpentane 2 -methylhexane 2, 3 -dimethylpentane 3 -methylhexane
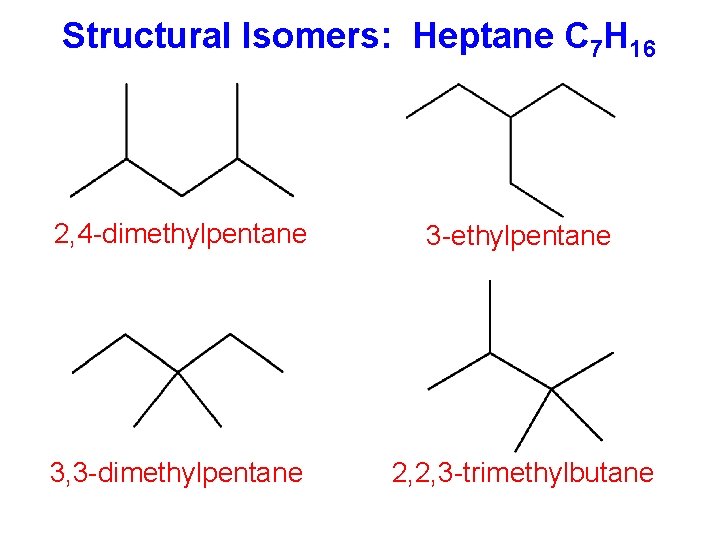
Structural Isomers: Heptane C 7 H 16 2, 4 -dimethylpentane 3 -ethylpentane 3, 3 -dimethylpentane 2, 2, 3 -trimethylbutane
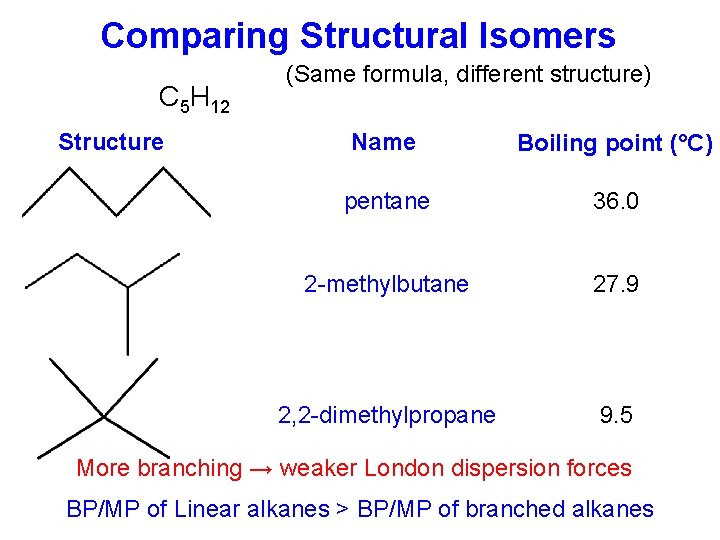
Comparing Structural Isomers C 5 H 12 Structure (Same formula, different structure) Name Boiling point (°C) pentane 36. 0 2 -methylbutane 27. 9 2, 2 -dimethylpropane 9. 5 More branching → weaker London dispersion forces BP/MP of Linear alkanes > BP/MP of branched alkanes
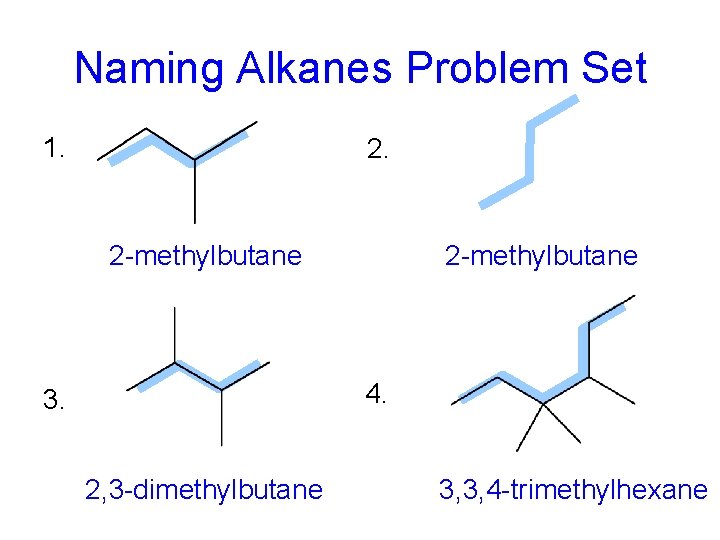
Naming Alkanes Problem Set 1. 2 -methylbutane 4. 3. 2, 3 -dimethylbutane 3, 3, 4 -trimethylhexane
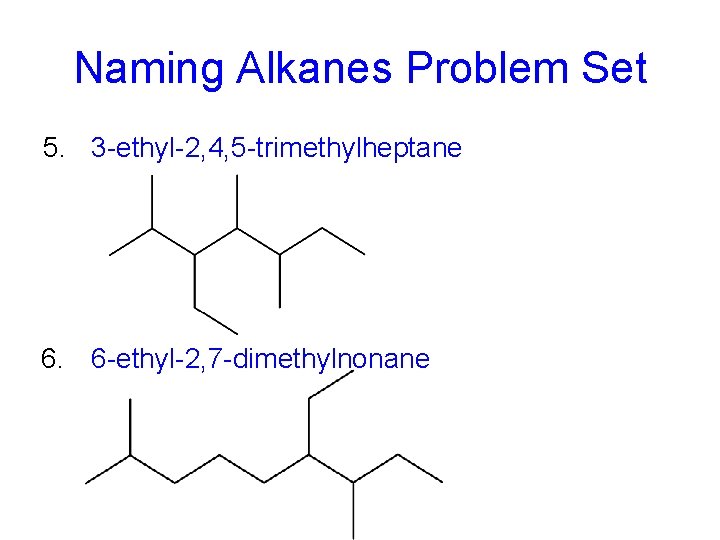
Naming Alkanes Problem Set 5. 3 -ethyl-2, 4, 5 -trimethylheptane 6. 6 -ethyl-2, 7 -dimethylnonane
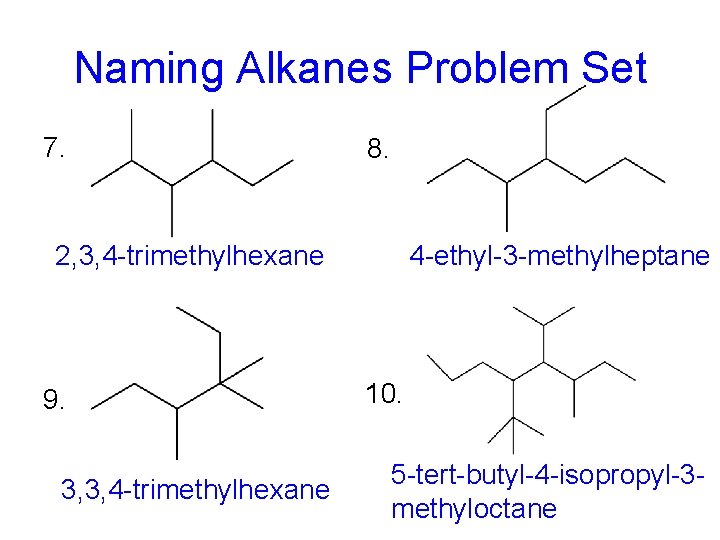
Naming Alkanes Problem Set 7. 8. 2, 3, 4 -trimethylhexane 9. 3, 3, 4 -trimethylhexane 4 -ethyl-3 -methylheptane 10. 5 -tert-butyl-4 -isopropyl-3 methyloctane
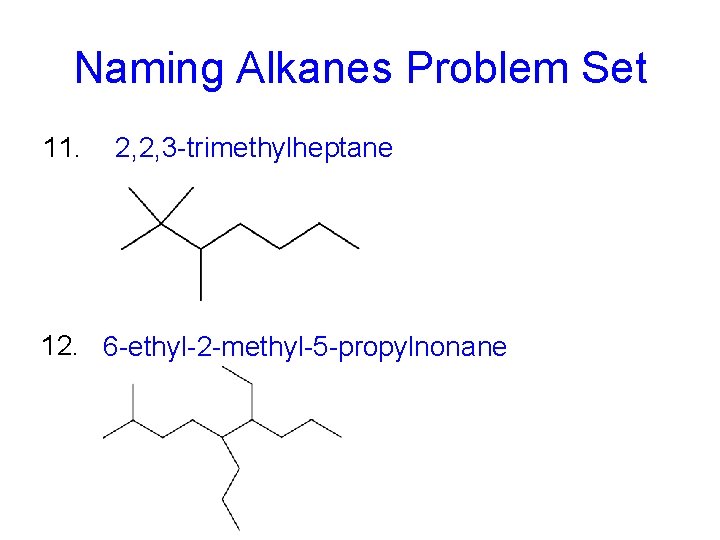
Naming Alkanes Problem Set 11. 2, 2, 3 -trimethylheptane 12. 6 -ethyl-2 -methyl-5 -propylnonane
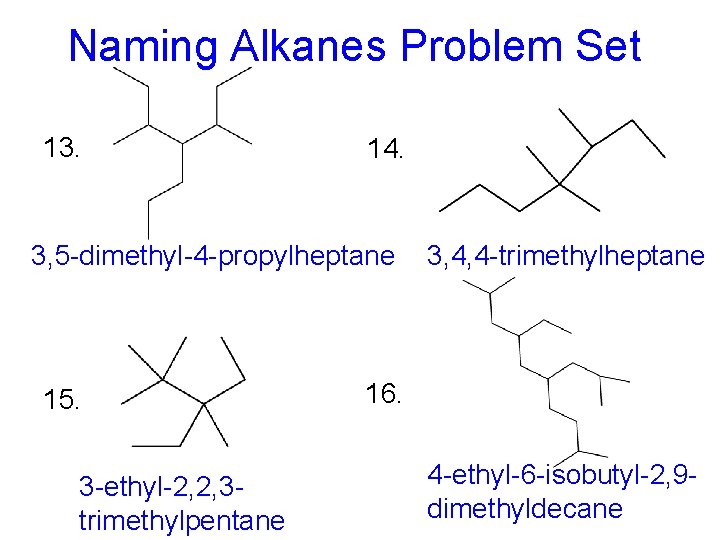
Naming Alkanes Problem Set 13. 14. 3, 5 -dimethyl-4 -propylheptane 15. 3 -ethyl-2, 2, 3 trimethylpentane 3, 4, 4 -trimethylheptane 16. 4 -ethyl-6 -isobutyl-2, 9 dimethyldecane
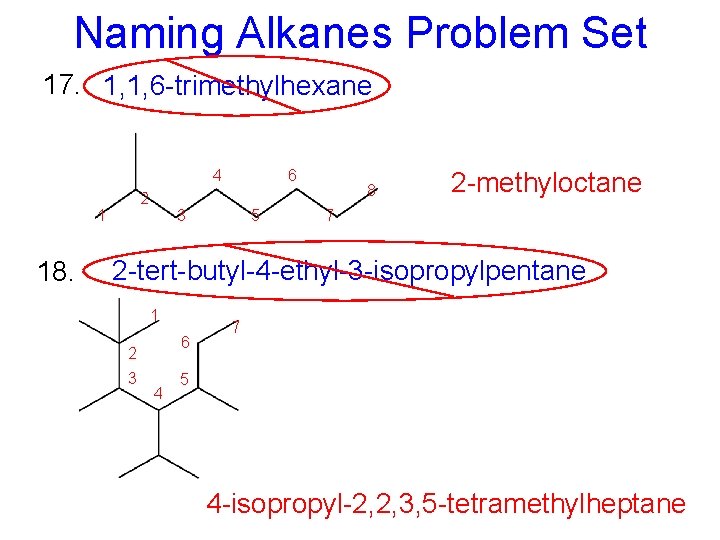
Naming Alkanes Problem Set 17. 1, 1, 6 -trimethylhexane 4 2 1 18. 6 3 5 8 2 -methyloctane 7 2 -tert-butyl-4 -ethyl-3 -isopropylpentane 1 6 2 3 4 7 5 4 -isopropyl-2, 2, 3, 5 -tetramethylheptane
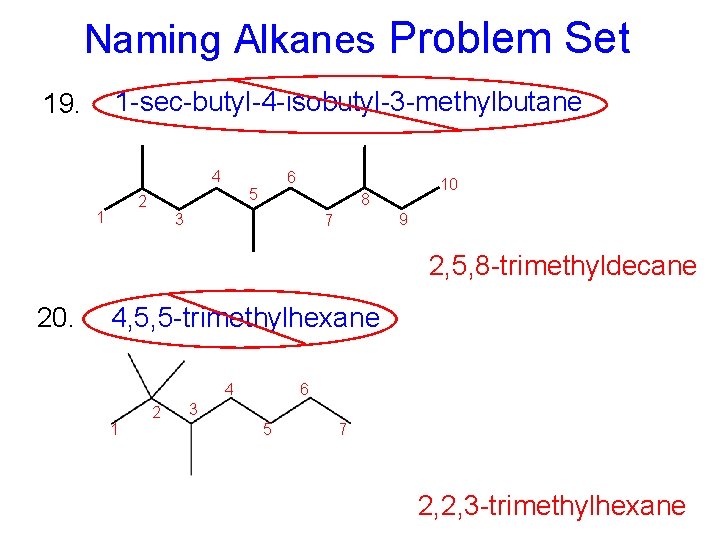
Naming Alkanes Problem Set 1 -sec-butyl-4 -isobutyl-3 -methylbutane 19. 4 5 2 1 6 10 8 3 9 7 2, 5, 8 -trimethyldecane 20. 4, 5, 5 -trimethylhexane 4 1 2 6 3 5 7 2, 2, 3 -trimethylhexane
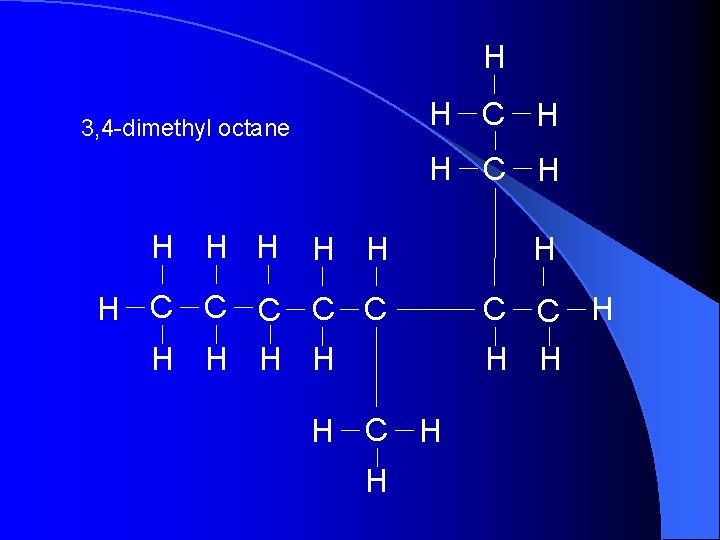
H H C H 3, 4 -dimethyl octane H C H H H H C C C H H H C C H H H
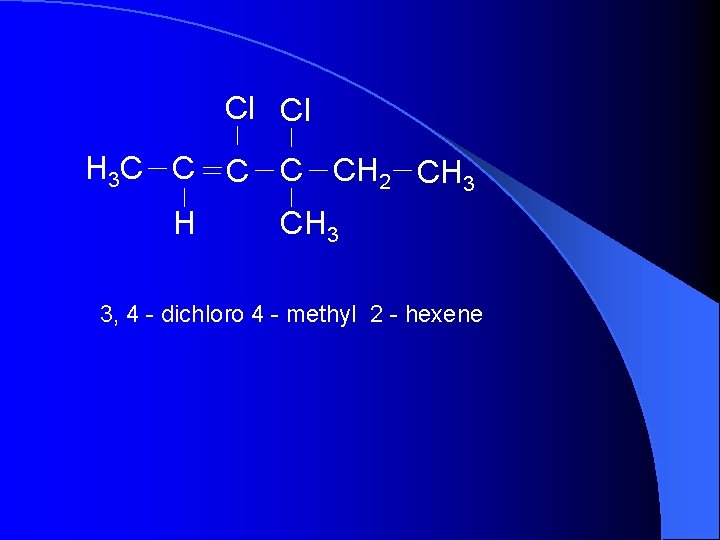
Cl Cl H 3 C C CH 2 CH 3 H CH 3 3, 4 - dichloro 4 - methyl 2 - hexene

Functional Groups Hydrocarbons in which some hydrogen atoms have been replaced can be compared to an electric drill with attachments. Inferring: What determines the function of the drill, the drill itself or the attachments? Electric drill Forstner drill bit hole saw bit Twist drill bit Philips screwdriver bit drum sander
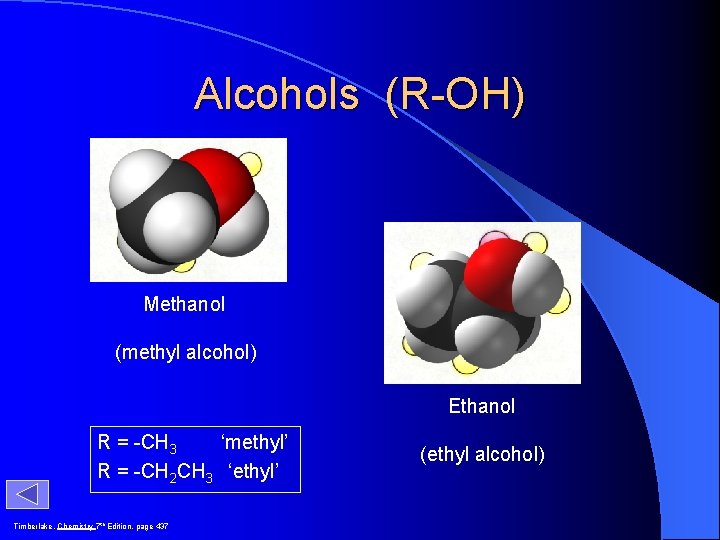
Alcohols (R-OH) Methanol (methyl alcohol) Ethanol R = -CH 3 ‘methyl’ R = -CH 2 CH 3 ‘ethyl’ Timberlake, Chemistry 7 th Edition, page 437 (ethyl alcohol)
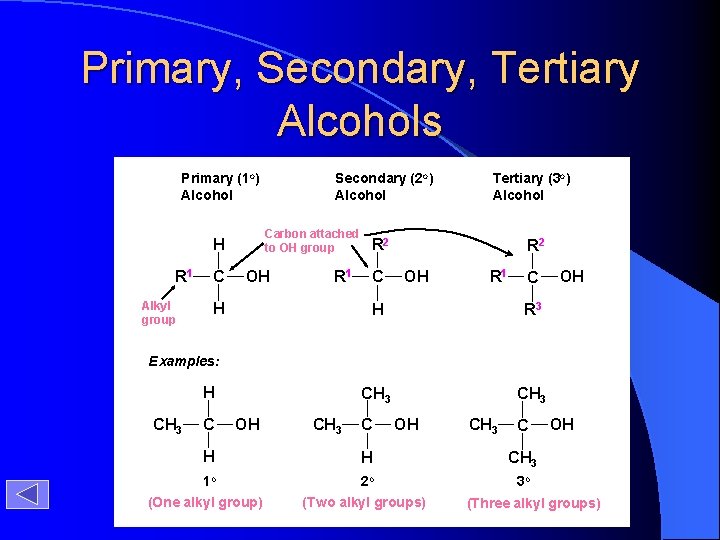
Primary, Secondary, Tertiary Alcohols Primary (1 o) Alcohol Carbon attached to OH group H R 1 Alkyl group C Secondary (2 o) Alcohol OH R 1 H Tertiary (3 o) Alcohol R 2 C R 2 OH R 1 C H R 3 CH 3 OH Examples: H CH 3 C OH CH 3 C H H CH 3 1 o 2 o 3 o (One alkyl group) (Two alkyl groups) OH (Three alkyl groups)
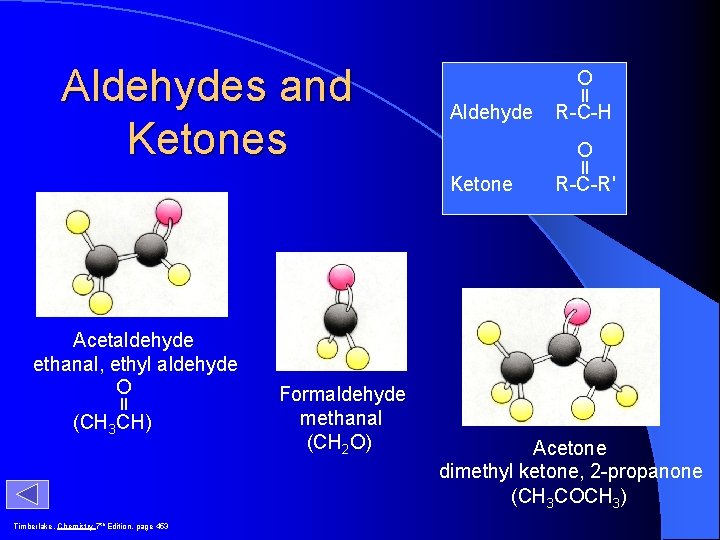
Aldehydes and Ketones O Aldehyde O Ketone Acetaldehyde ethanal, ethyl aldehyde O (CH 3 CH) Timberlake, Chemistry 7 th Edition, page 453 Formaldehyde methanal (CH 2 O) R-C-H R-C-R' Acetone dimethyl ketone, 2 -propanone (CH 3 COCH 3)
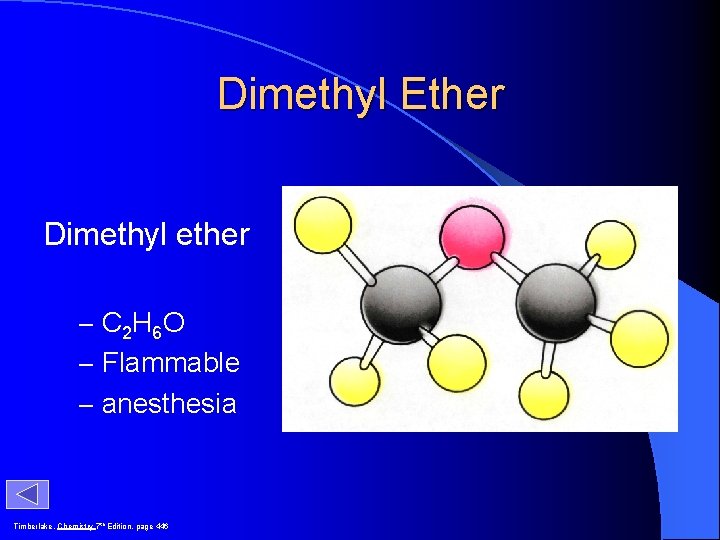
Dimethyl Ether Dimethyl ether – C 2 H 6 O – Flammable – anesthesia Timberlake, Chemistry 7 th Edition, page 446

R- Functional Groups -CH 3 methyl -CH 2 CH 3 ethyl -CH 2 CH 2 CH 3 H CH 3 CHCH 2 CH 3 -CH 2 CH 3 propyl CH 3 CCH 3 isopropyl sec-butyl H – CH 2 – CH 3 H butyl isobutyl CH 3 – CH 3 tert-butyl
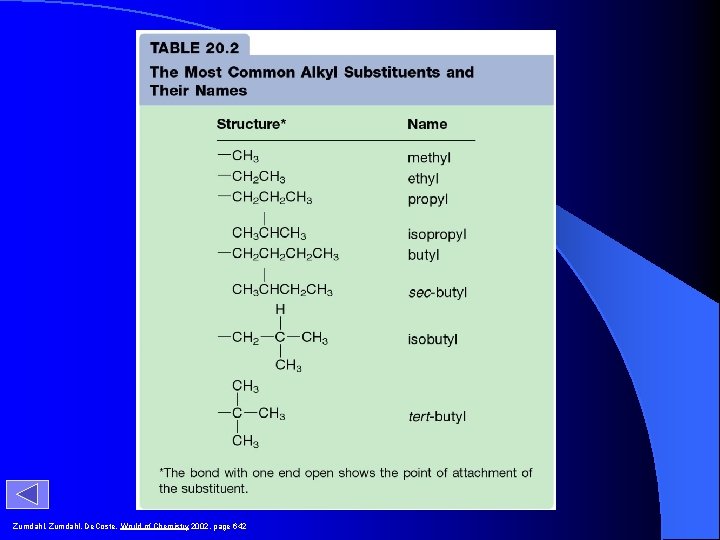
Zumdahl, De. Coste, World of Chemistry 2002, page 642
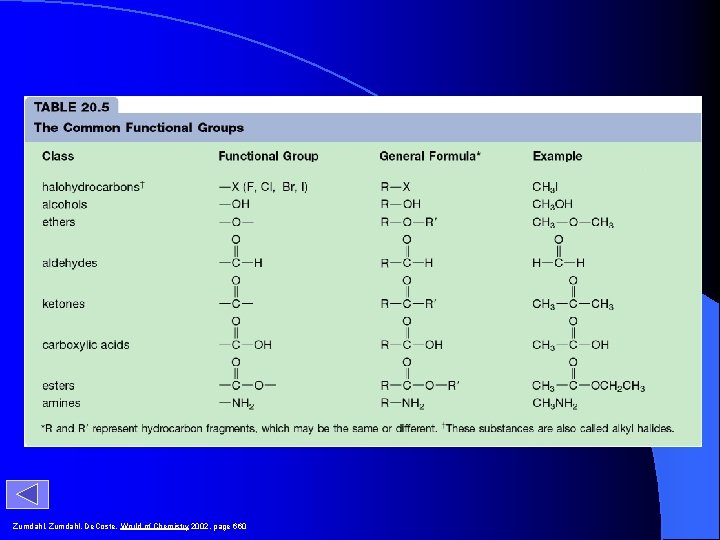
Zumdahl, De. Coste, World of Chemistry 2002, page 660
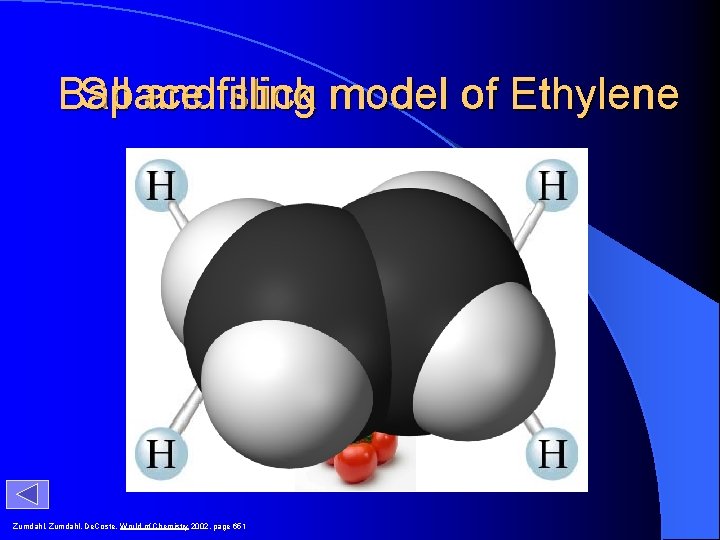
Ball Space andfilling stick model of Ethylene Zumdahl, De. Coste, World of Chemistry 2002, page 651
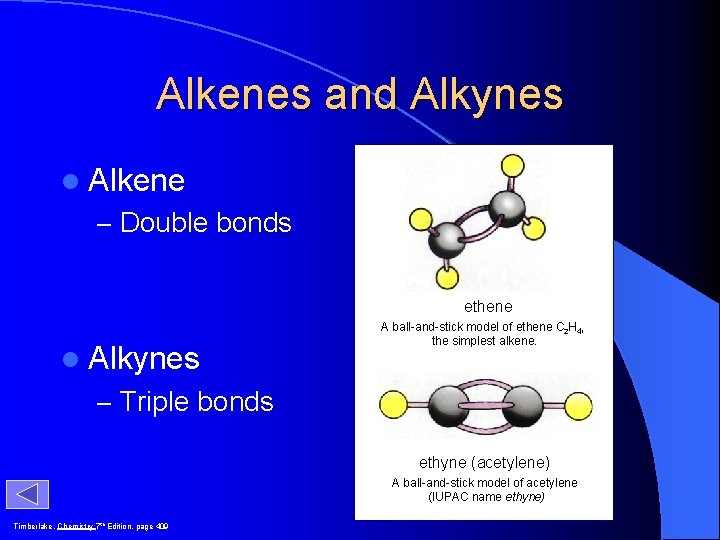
Alkenes and Alkynes l Alkene – Double bonds ethene l Alkynes A ball-and-stick model of ethene C 2 H 4, the simplest alkene. – Triple bonds ethyne (acetylene) A ball-and-stick model of acetylene (IUPAC name ethyne) Timberlake, Chemistry 7 th Edition, page 409

Saturated vs. Unsaturated Hydrocarbons l Saturated – Single bonds l Example: l Unsaturated – Double & triple bonds l Example:
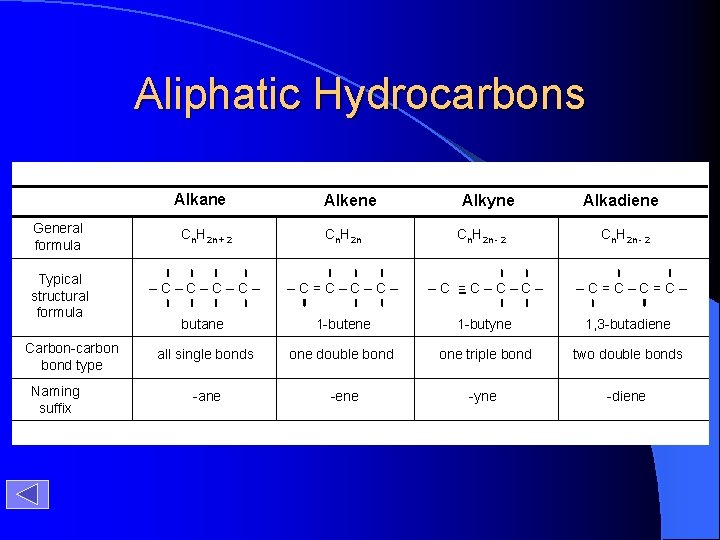
Aliphatic Hydrocarbons Alkane General formula Typical structural formula Carbon-carbon bond type Naming suffix Alkene Alkyne Alkadiene Cn. H 2 n + 2 Cn. H 2 n - 2 –C–C– –C=C–C–C– –C=C– butane 1 -butene 1 -butyne 1, 3 -butadiene all single bonds one double bond one triple bond two double bonds -ane -ene -yne -diene
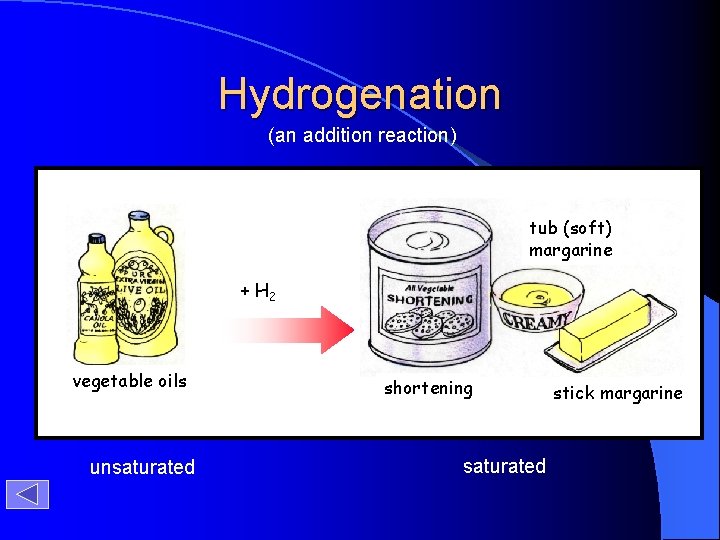
Hydrogenation (an addition reaction) tub (soft) margarine + H 2 vegetable oils unsaturated shortening saturated stick margarine
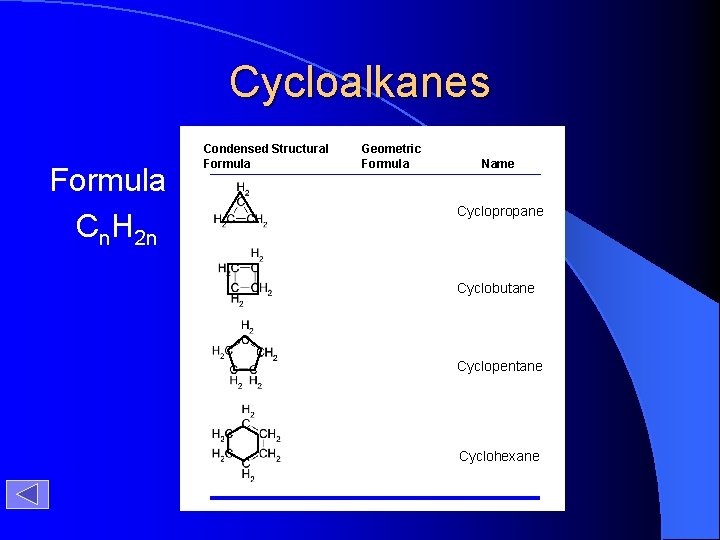
Cycloalkanes Formula Cn. H 2 n Condensed Structural Formula Geometric Formula Name Cyclopropane Cyclobutane Cyclopentane Cyclohexane

Cycloalkanes Formula Cn. H 2 n Condensed Structural Formula Geometric Formula Name Cyclopropane Cyclobutane Cyclopentane Cyclohexane Timberlake, Chemistry 7 th Edition, page 388
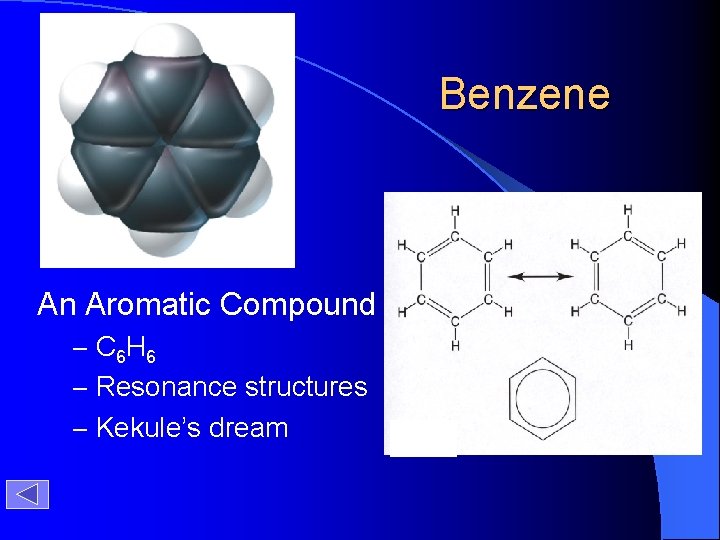
Benzene An Aromatic Compound – C 6 H 6 – Resonance structures – Kekule’s dream
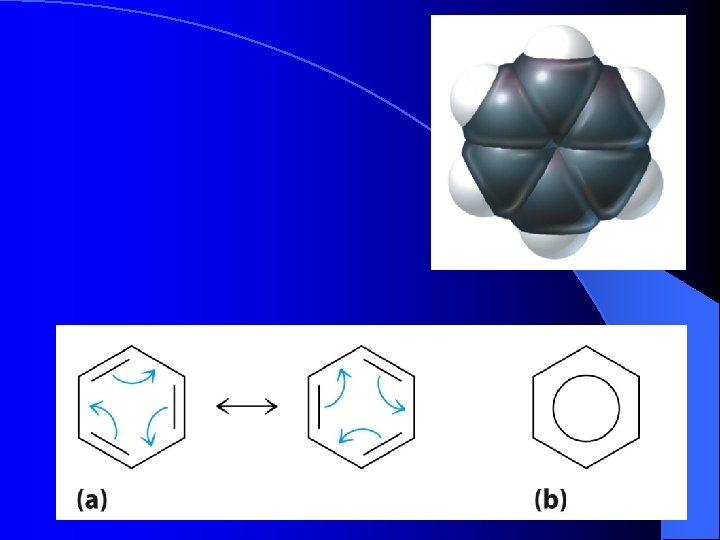
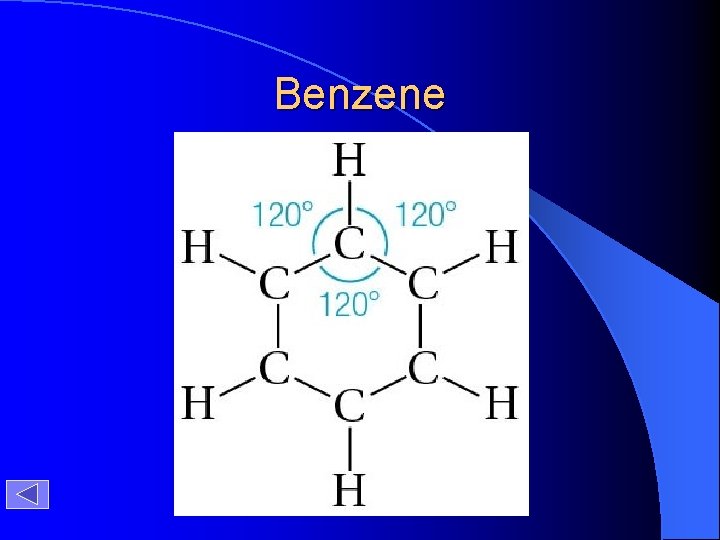
Benzene
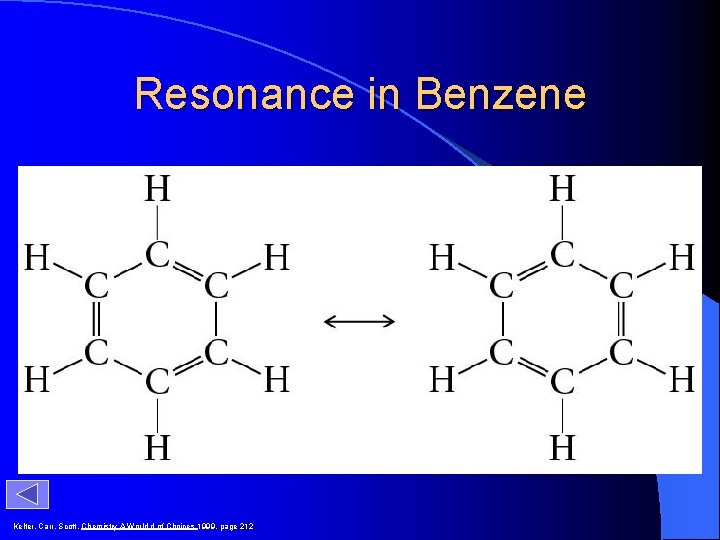
Resonance in Benzene Kelter, Carr, Scott, Chemistry A World d of Choices 1999, page 212
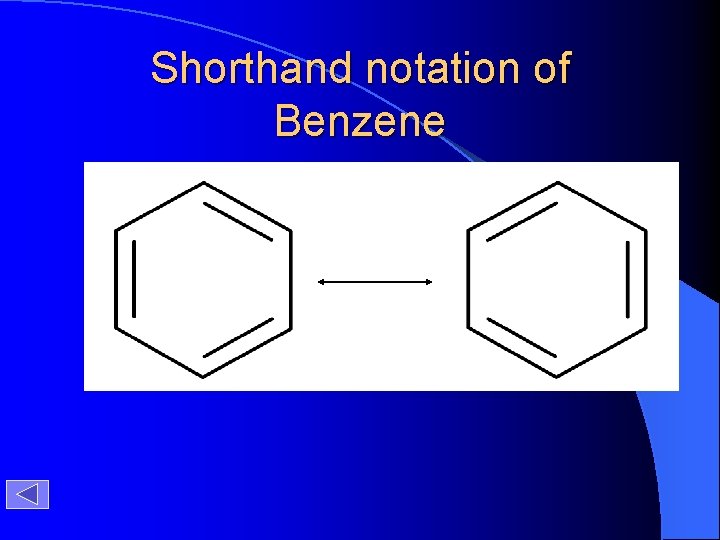
Shorthand notation of Benzene
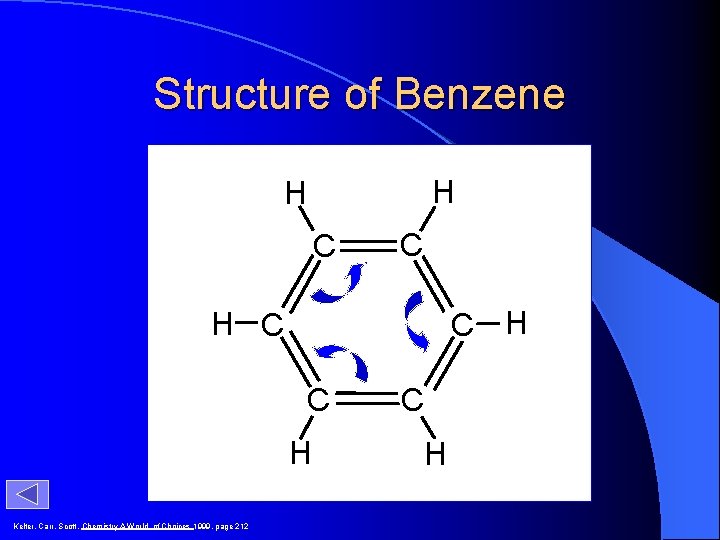
Structure of Benzene H H C C C H H C C H Kelter, Carr, Scott, Chemistry A World of Choices 1999, page 212 C H
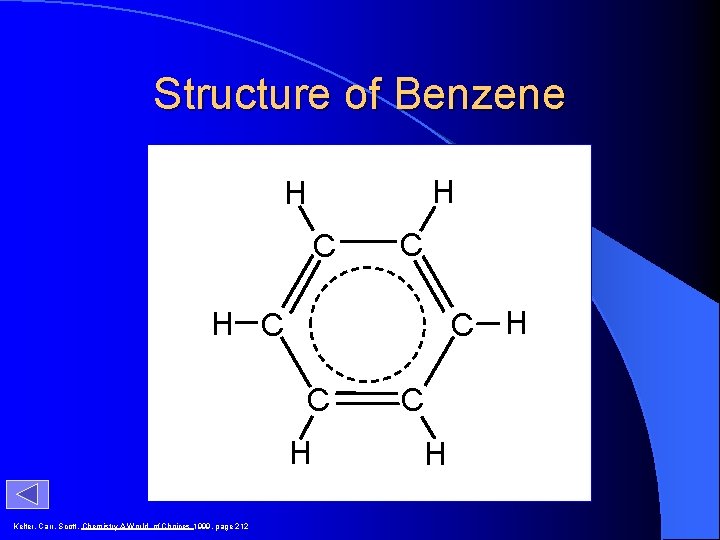
Structure of Benzene H H C C C H H C C H Kelter, Carr, Scott, Chemistry A World of Choices 1999, page 212 C H
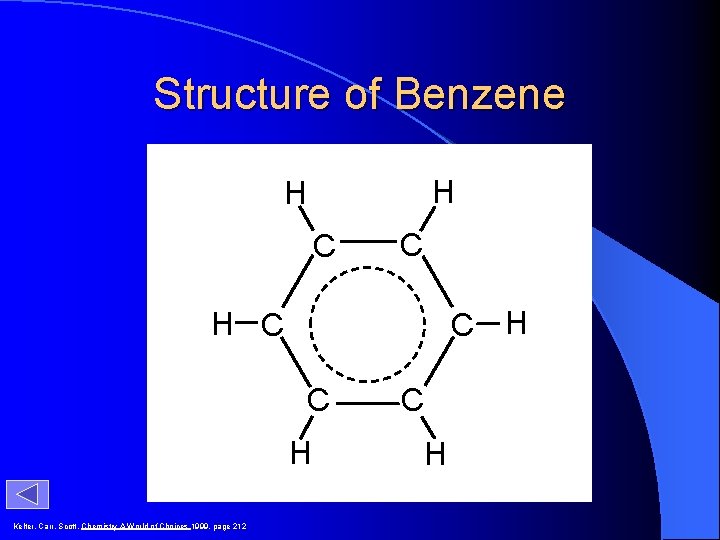
Structure of Benzene H H C C C H H C C H Kelter, Carr, Scott, Chemistry A World of Choices 1999, page 212 C H

Benzene 3 -D – VSEPR Diagram Kelter, Carr, Scott, Chemistry A World of Choices 1999, page 212
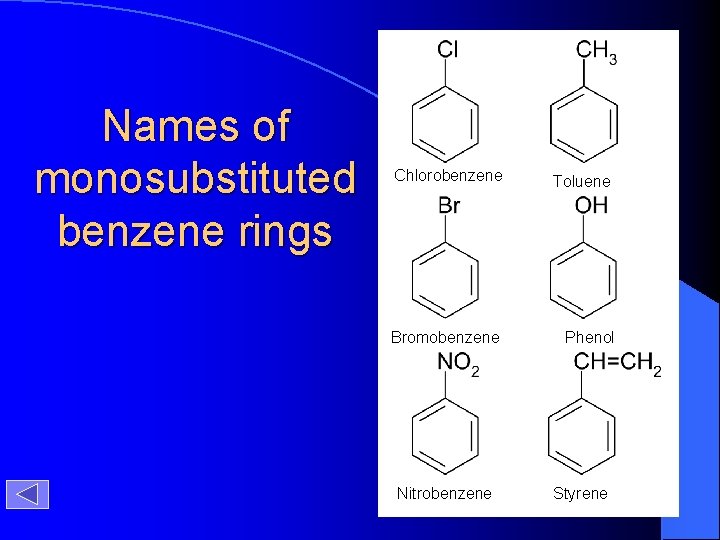
Names of monosubstituted benzene rings Chlorobenzene Toluene Bromobenzene Phenol Nitrobenzene Styrene
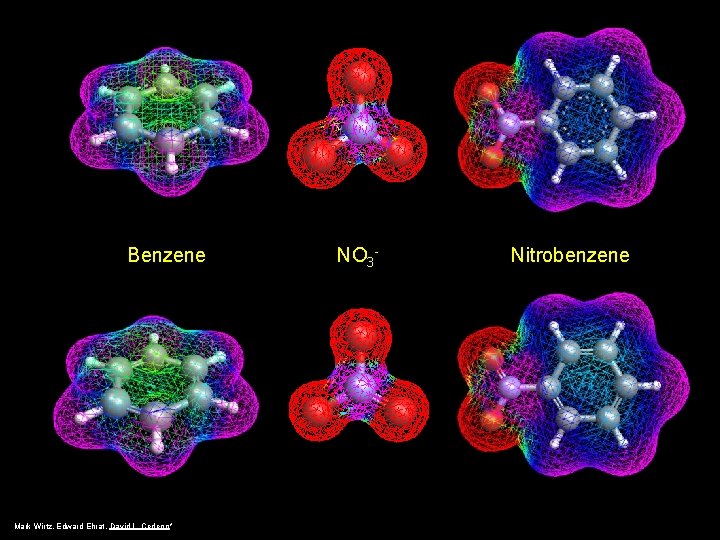
Benzene Mark Wirtz, Edward Ehrat, David L. Cedeno* NO 3 - Nitrobenzene
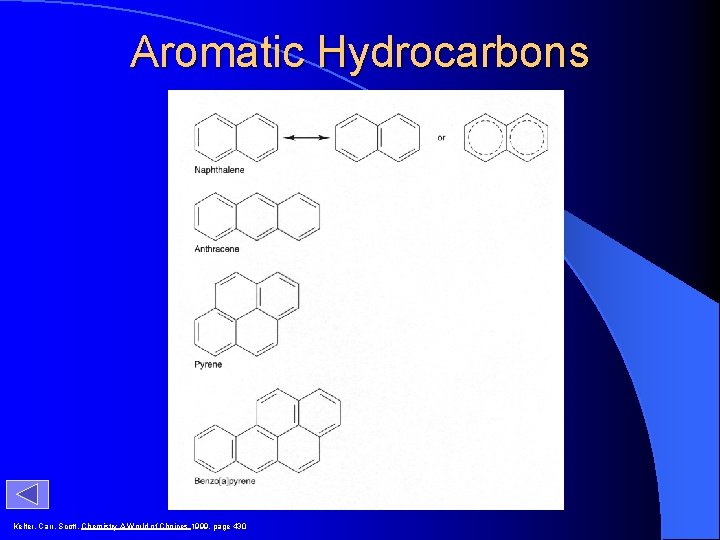
Aromatic Hydrocarbons Kelter, Carr, Scott, Chemistry A World of Choices 1999, page 430

Cholesterol Compounds
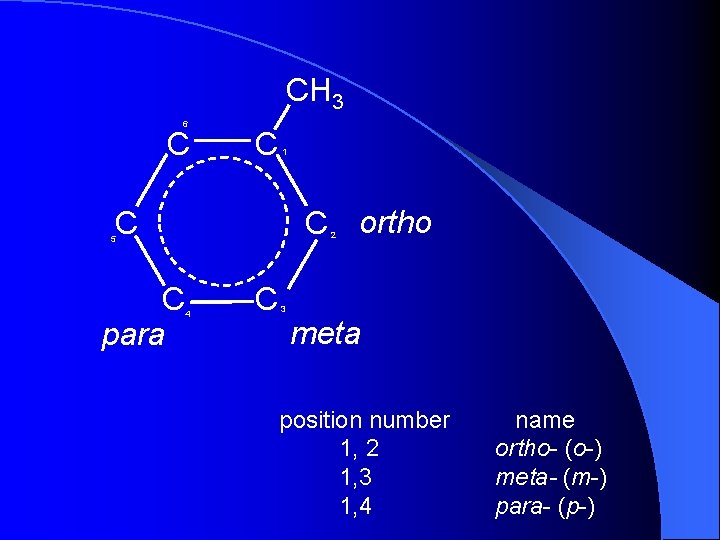
CH 3 6 C C 1 C ortho C 2 5 C para 4 C 3 meta position number 1, 2 1, 3 1, 4 name ortho- (o-) meta- (m-) para- (p-)
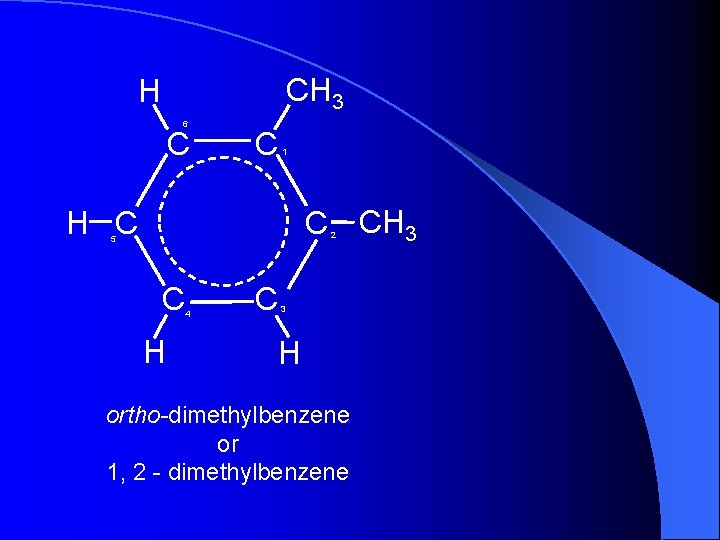
CH 3 H 6 C C 1 C CH 3 H C 2 5 C H 4 C 3 H ortho-dimethylbenzene or 1, 2 - dimethylbenzene
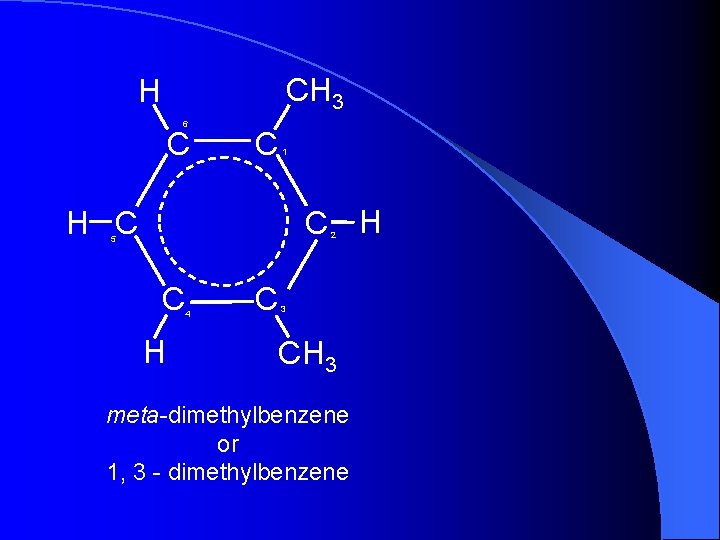
CH 3 H 6 C C 1 C H H C 2 5 C H 4 C 3 CH 3 meta-dimethylbenzene or 1, 3 - dimethylbenzene
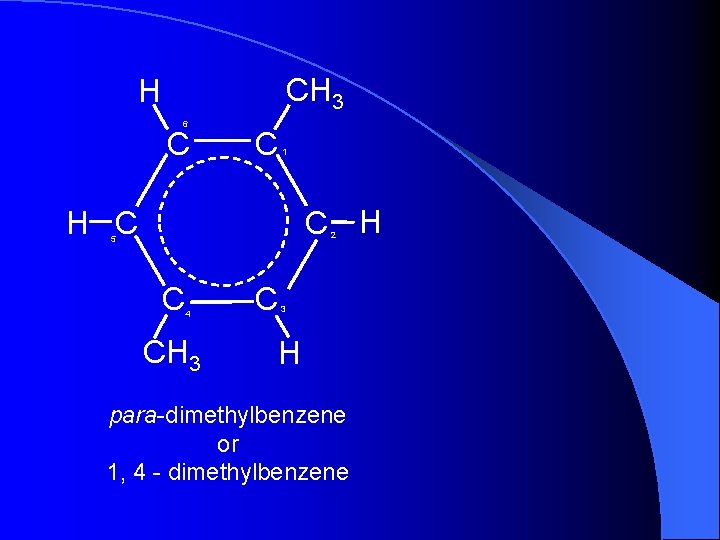
CH 3 H 6 C C 1 C H H C 2 5 C 4 CH 3 C 3 H para-dimethylbenzene or 1, 4 - dimethylbenzene
MD H 6 C C 1 C H H C 2 5 C 4 MD C 3 H para-docs or paradox
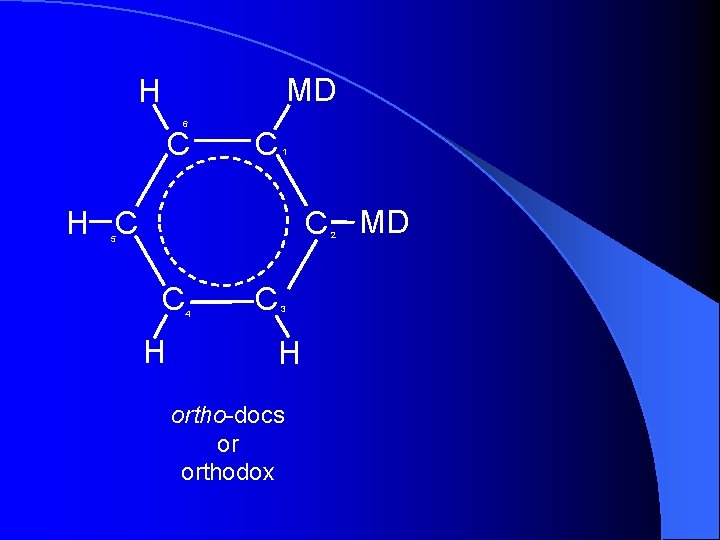
MD H 6 C C 1 C MD H C 2 5 C H 4 C 3 H ortho-docs or orthodox
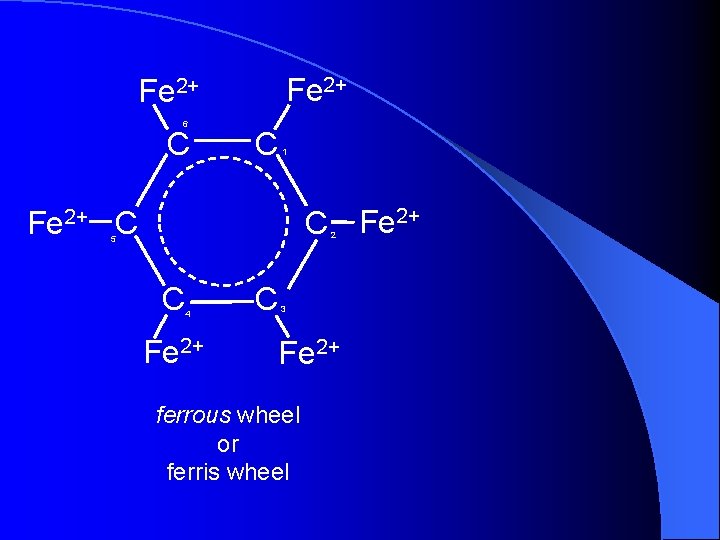
Fe 2+ 6 C C 1 C Fe 2+ C 2 5 C 4 Fe 2+ C 3 Fe 2+ ferrous wheel or ferris wheel
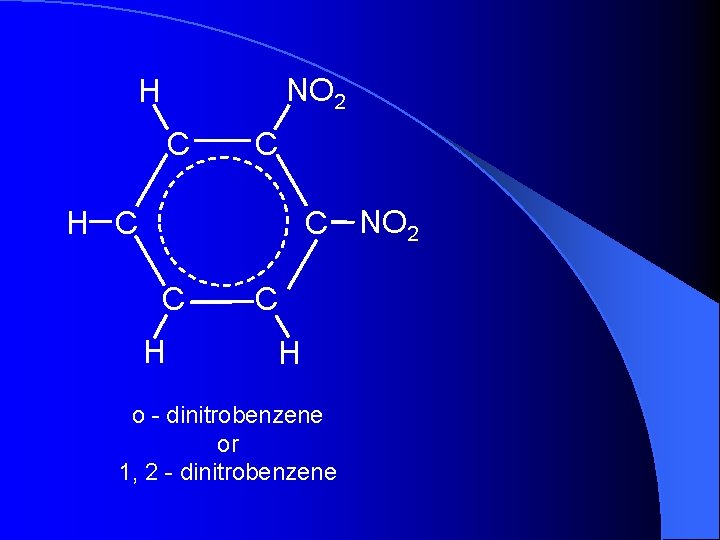
NO 2 H C C C NO 2 H C C H o - dinitrobenzene or 1, 2 - dinitrobenzene
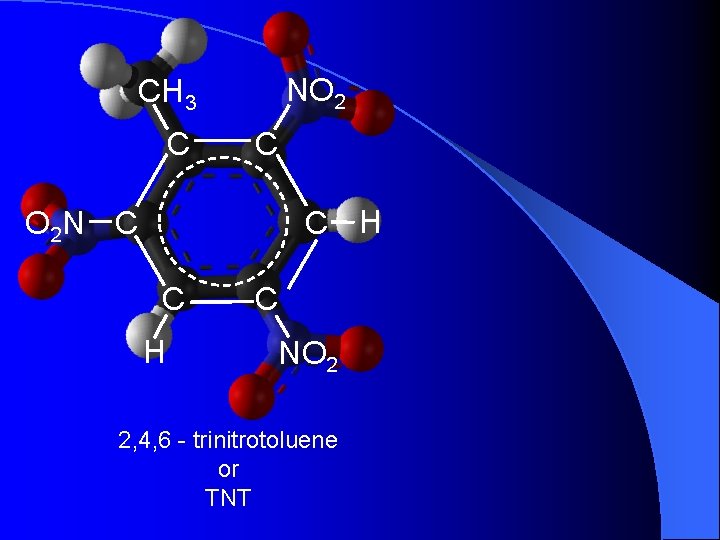
NO 2 CH 3 C C C H O 2 N C C H C NO 2 2, 4, 6 - trinitrotoluene or TNT
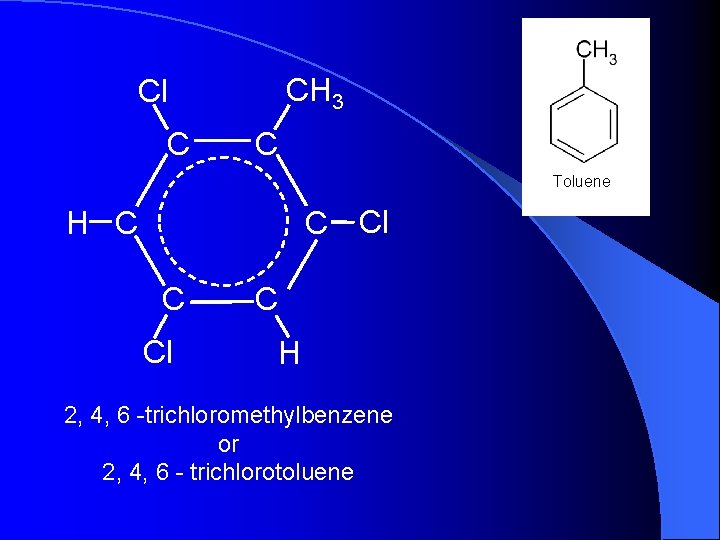
CH 3 Cl C C Toluene C Cl H C C Cl C H 2, 4, 6 -trichloromethylbenzene or 2, 4, 6 - trichlorotoluene
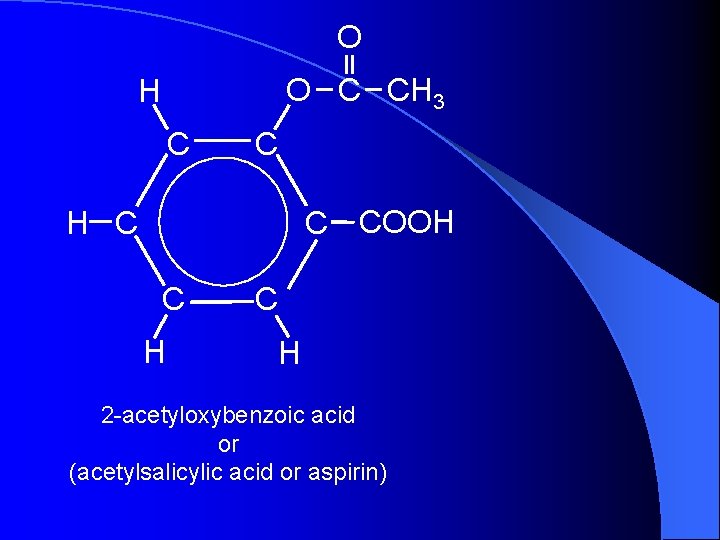
O O C CH 3 H C COOH H C C H 2 -acetyloxybenzoic acid or (acetylsalicylic acid or aspirin)
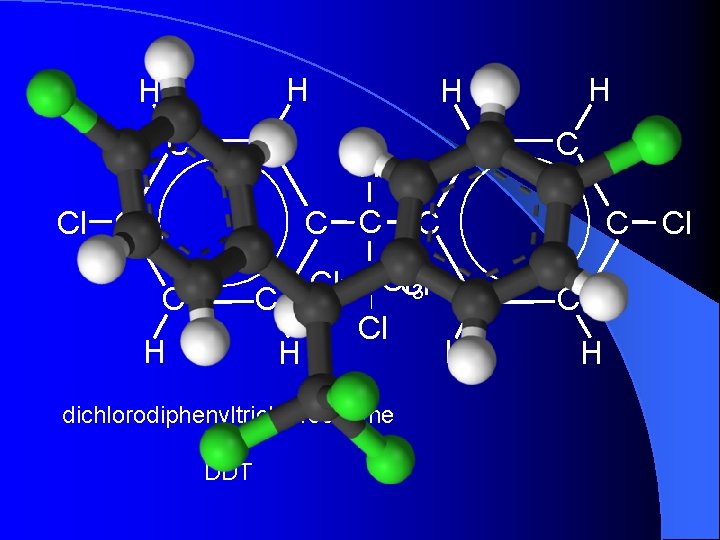
H H C C Cl C H H C CCl. Cl Cl C 3 C C Cl H H dichlorodiphenyltrichloroethane or DDT C C C H Cl
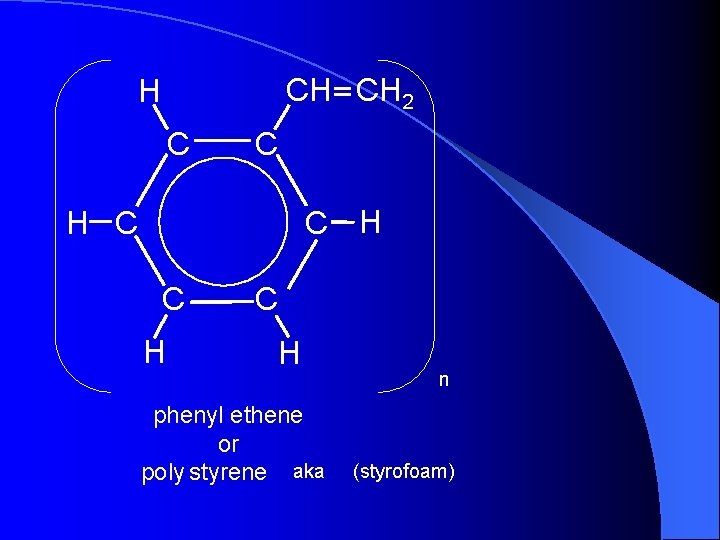
CH CH 2 H C C C H H C C H phenyl ethene or poly styrene aka n (styrofoam)
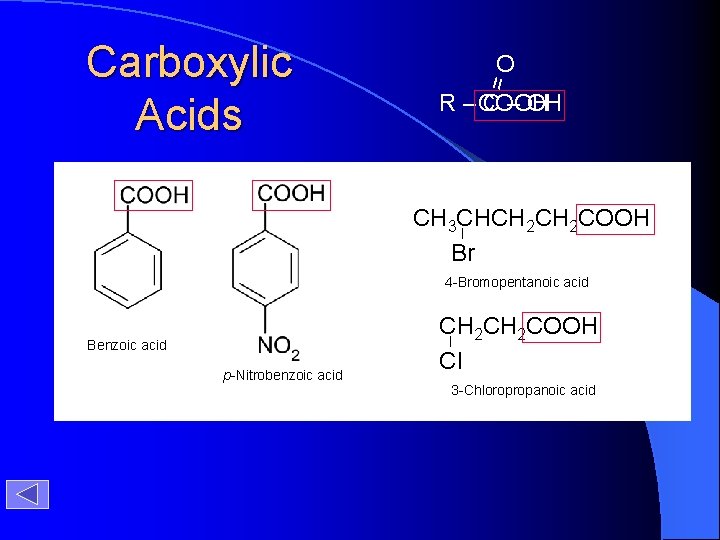
O = Carboxylic Acids - COOH R– C – OH CH 3 CHCH 2 COOH Br 4 -Bromopentanoic acid Benzoic acid p-Nitrobenzoic acid CH 2 COOH Cl 3 -Chloropropanoic acid
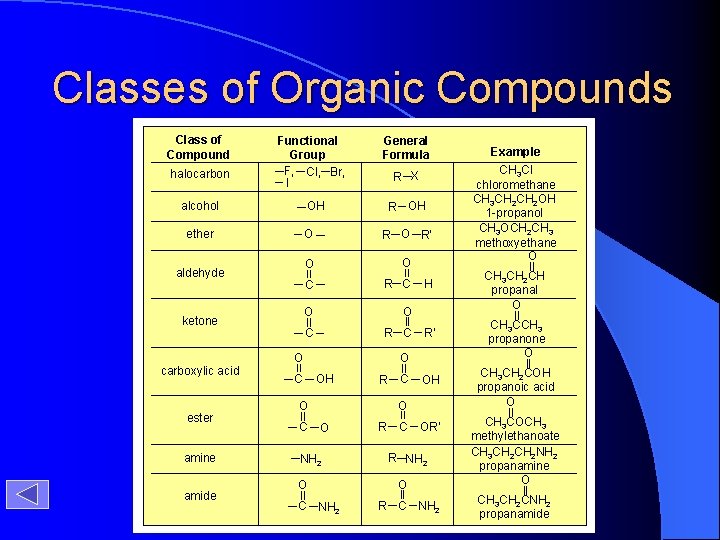
Classes of Organic Compounds Class of Compound halocarbon Functional Group F, Cl, Br, I alcohol aldehyde ketone ester amine amide R X R OH OH ether carboxylic acid General Formula O R’ O O C R C O C R’ O OH R C OH O O C H O NH 2 O C NH 2 R C OR’ R NH 2 O R C NH 2 Example CH 3 Cl chloromethane CH 3 CH 2 OH 1 -propanol CH 3 OCH 2 CH 3 methoxyethane O CH 3 CH 2 CH propanal O CH 3 CCH 3 propanone O CH 3 CH 2 COH propanoic acid O CH 3 COCH 3 methylethanoate CH 3 CH 2 NH 2 propanamine O CH 3 CH 2 CNH 2 propanamide
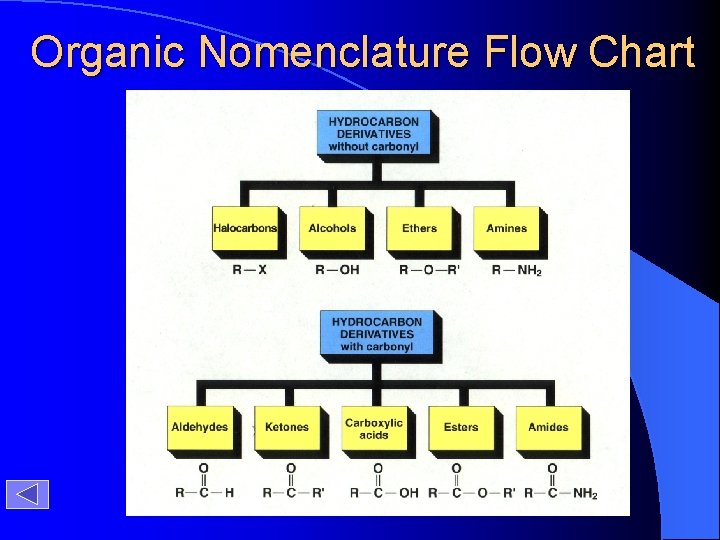
Organic Nomenclature Flow Chart
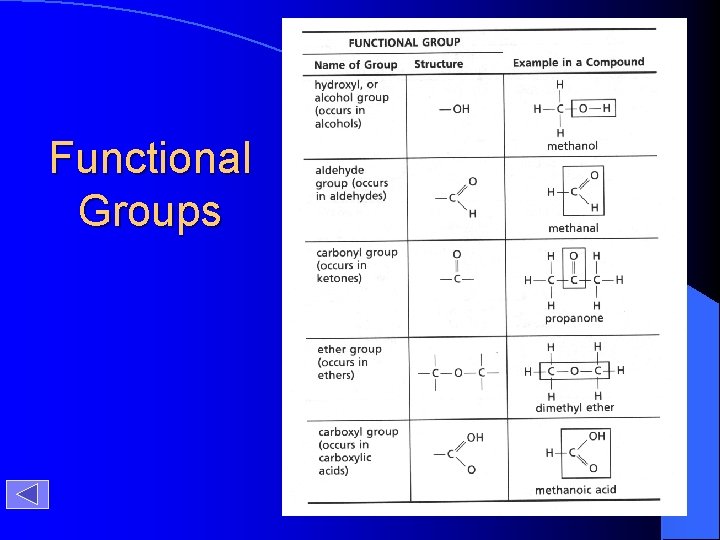
Functional Groups
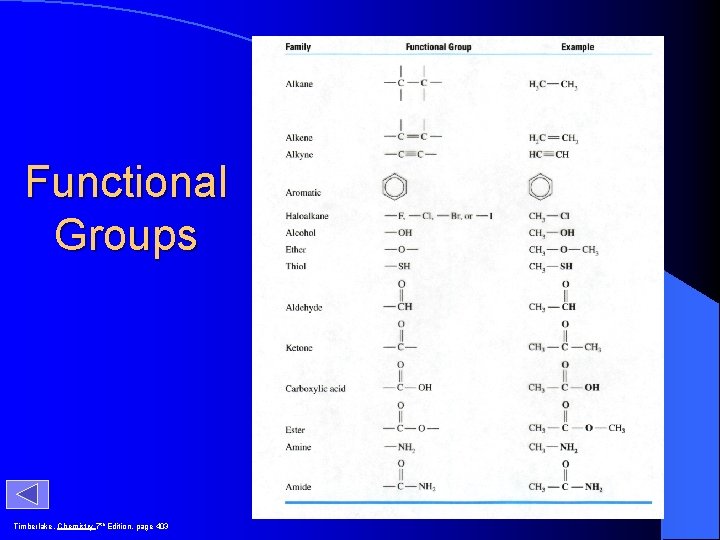
Functional Groups Timberlake, Chemistry 7 th Edition, page 403
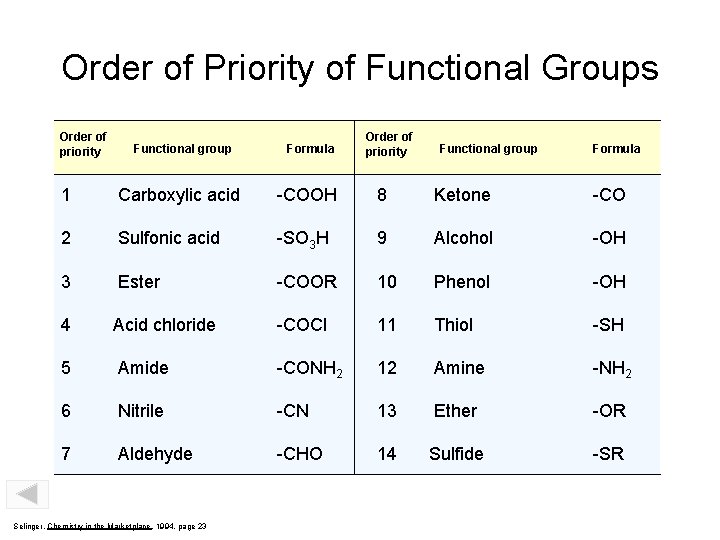
Order of Priority of Functional Groups Order of priority Functional group Formula 1 Carboxylic acid -COOH 8 Ketone -CO 2 Sulfonic acid -SO 3 H 9 Alcohol -OH 3 Ester -COOR 10 Phenol -OH 4 Acid chloride -COCl 11 Thiol -SH 5 Amide -CONH 2 12 Amine -NH 2 6 Nitrile -CN 13 Ether -OR 7 Aldehyde -CHO 14 Sulfide -SR Selinger, Chemistry in the Marketplace, 1994, page 23
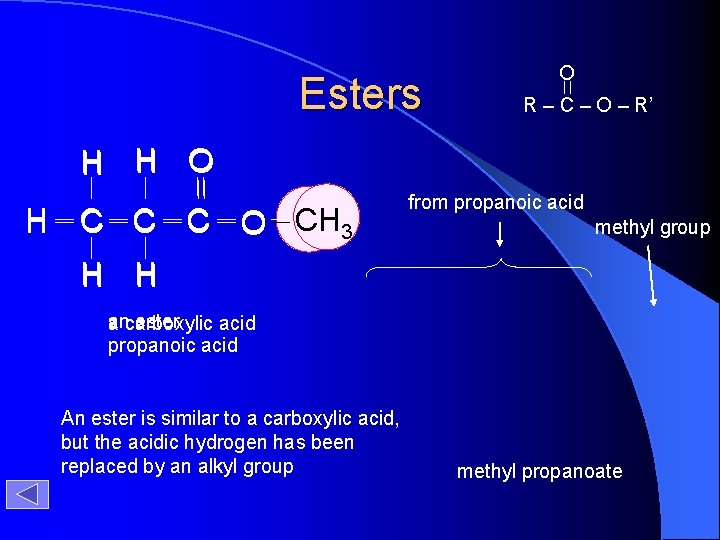
Esters O R – C – O – R’ H H O H C C C O CH H 3 from propanoic acid methyl group H H an ester a carboxylic acid propanoic acid An ester is similar to a carboxylic acid, but the acidic hydrogen has been replaced by an alkyl group methyl propanoate
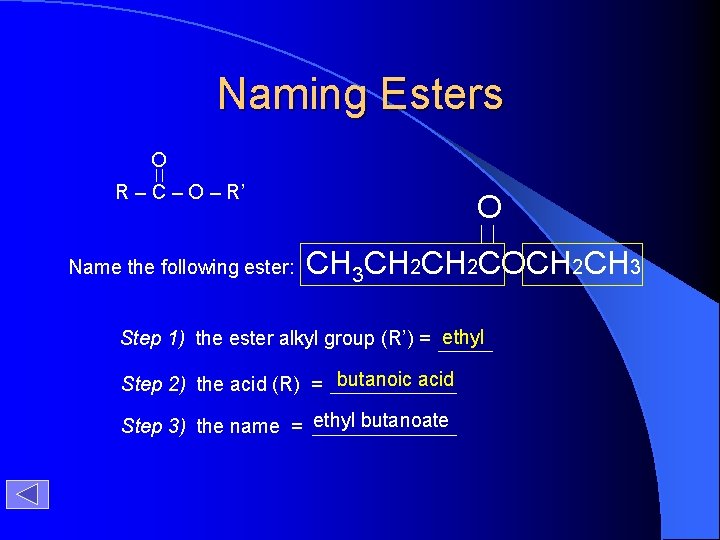
Naming Esters O R – C – O – R’ Name the following ester: O CH 3 CH 2 COCH 2 CH 3 Step 1) the ester alkyl group (R’) = ethyl Step 2) the acid (R) = butanoic acid Step 3) the name = ethyl butanoate
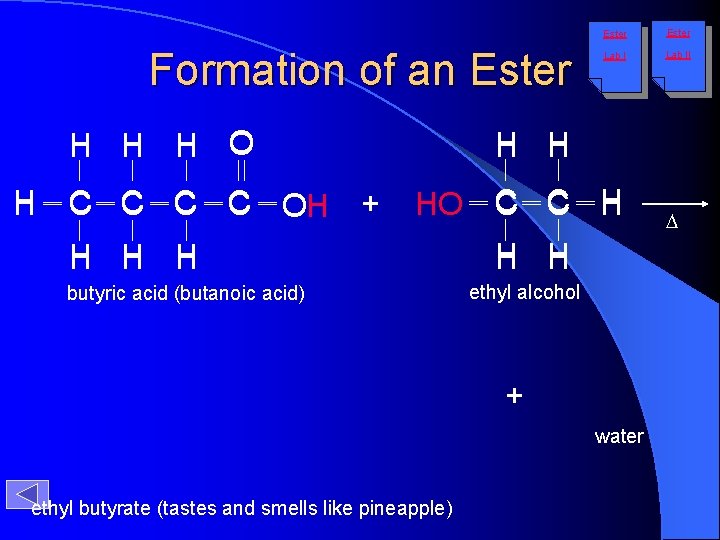
Formation of an Ester H H H O H C C O OH H + Ester Lab II H H HO C C H H butyric acid (butanoic acid) H H ethyl alcohol + water ethyl butyrate (tastes and smells like pineapple) D

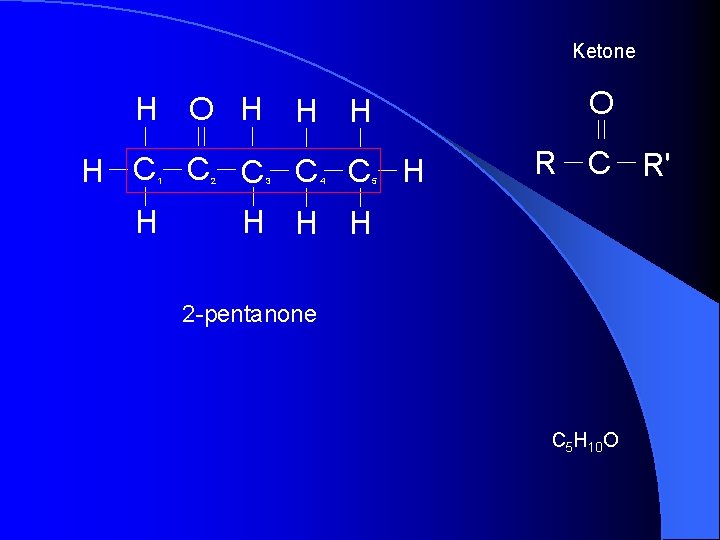
Ketone H O H H C C C H 1 H 2 3 4 5 O R C R' H H H 2 -pentanone C 5 H 10 O
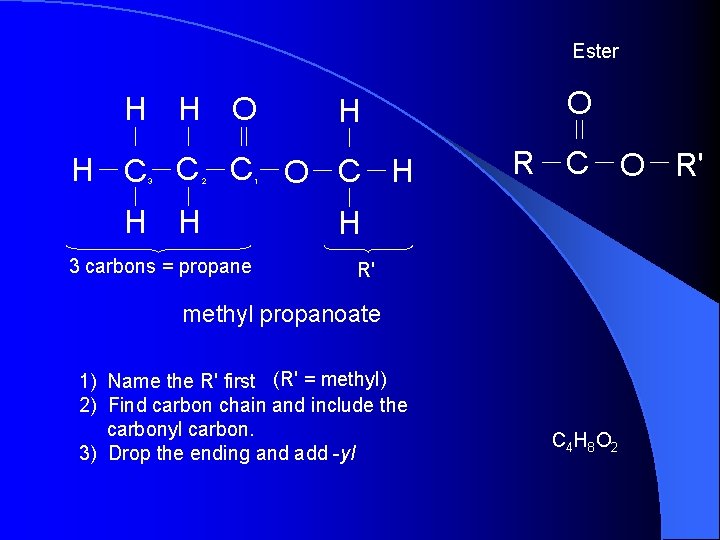
Ester H H O H H C C C O C H 2 3 H H 3 carbons = propane 1 O R C O R' H R' methyl propanoate 1) Name the R' first (R' = methyl) 2) Find carbon chain and include the carbonyl carbon. 3) Drop the ending and add -yl C 4 H 8 O 2
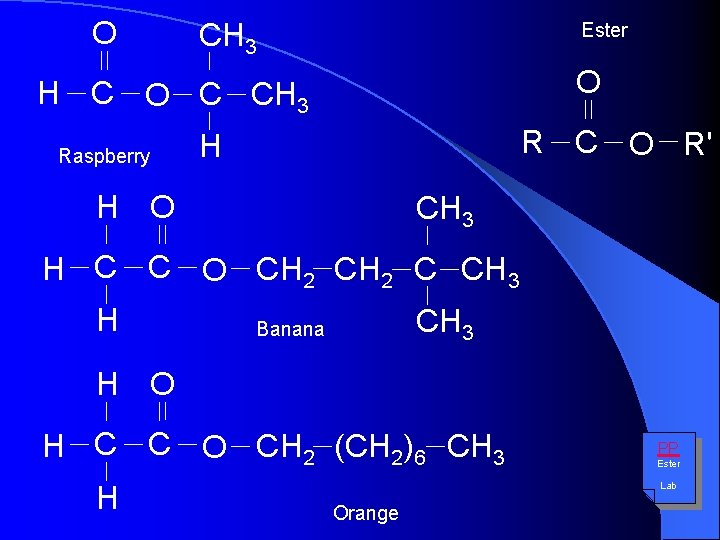
O CH 3 Ester O H C O C CH 3 Raspberry R C O R' H H O CH 3 H C C O CH 2 C CH 3 H CH 3 Banana H O H C C O CH 2 (CH 2)6 CH 3 H PP Ester Lab Orange
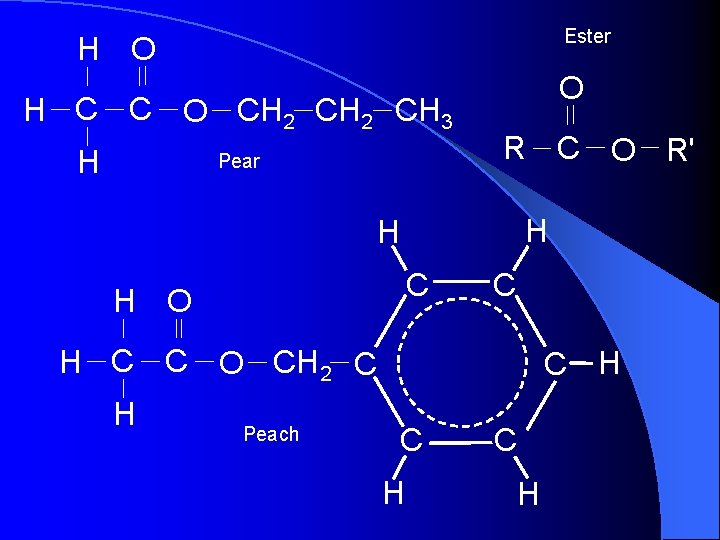
Ester H O H C C O CH 2 CH 3 H Pear O R C O R' H H C H O C H C C O CH 2 C H Peach C H C H

Carboxylic acid H R C H H H H C C 5 4 H H H C C C H H 3 2 1 O OH 3 -methylpentanoic acid C 6 H 12 O 2 O OH
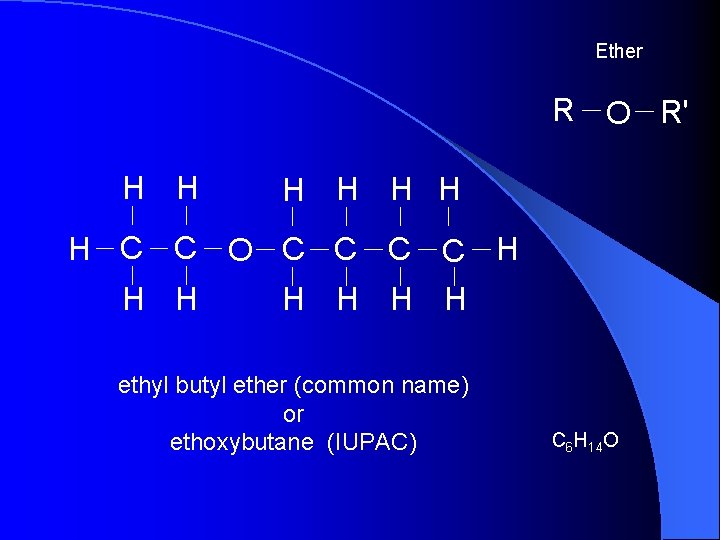
Ether R O R' H H H H C C O C C H H H H ethyl butyl ether (common name) or ethoxybutane (IUPAC) C 6 H 14 O
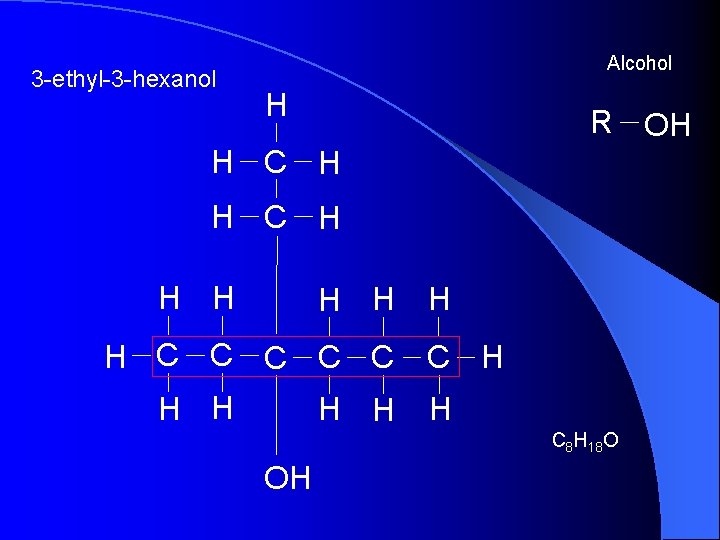
3 -ethyl-3 -hexanol Alcohol H R OH H C H H H C C C H H H C 8 H 18 O OH
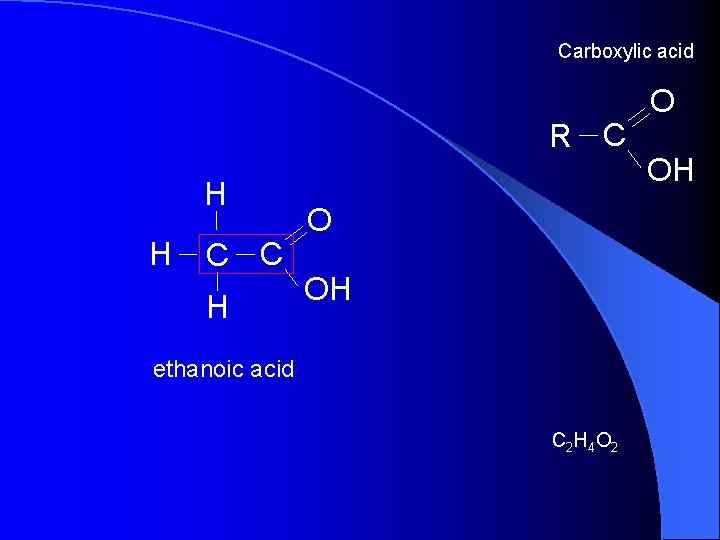
Carboxylic acid R C H H C C H O OH ethanoic acid C 2 H 4 O 2 O OH
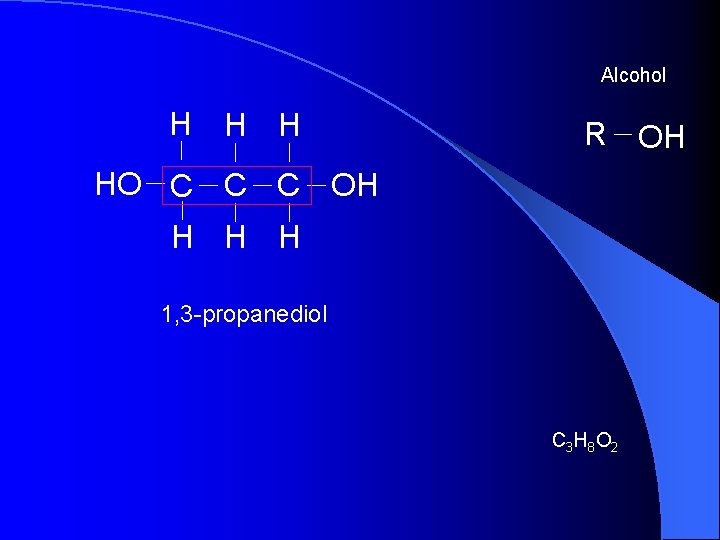
Alcohol H H H R OH HO C C C OH H 1, 3 -propanediol C 3 H 8 O 2

Draw structural formulas for the following: a. 3 -heptene b. trichloromethane c. 2 -chloro-3 -phenylhexane d. 1, 3 -cyclopentadiene e. toluene (methylbenzene) f. 1, 4 -dibromobenzene g. 2 -bromo-3 -methyl-2 -butene

Write the condensed formulas for the following haloalkanes: a. ethyl chloride (common name) or chloroethane (IUPAC) b. bromomethane c. 1 -bromo-3 -chlorocyclopentane d. 1, 1 -dichlorocyclohexane e. 2, 2, 3 -trichlorobutane f. 2, 4 -dibromo-2, 4 -dichloropentane
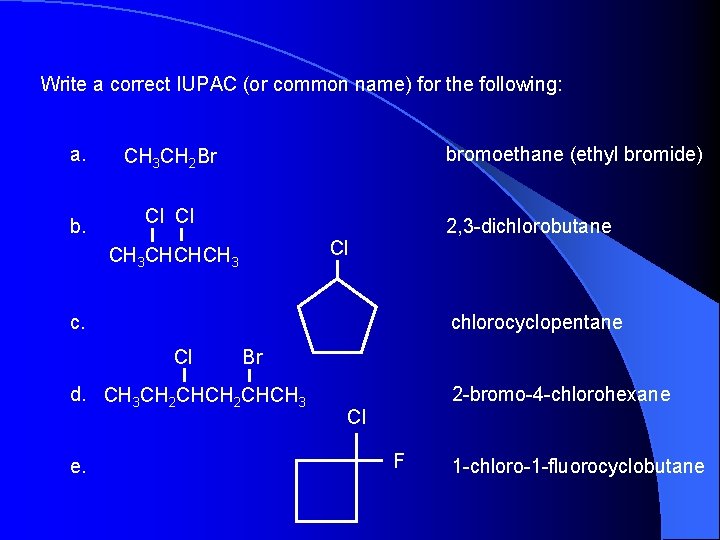
Write a correct IUPAC (or common name) for the following: a. CH 3 CH 2 Br b. Cl Cl bromoethane (ethyl bromide) 2, 3 -dichlorobutane Cl CH 3 CHCHCH 3 c. chlorocyclopentane Cl Br d. CH 3 CH 2 CHCH 3 e. 2 -bromo-4 -chlorohexane Cl F 1 -chloro-1 -fluorocyclobutane

Draw structures for the following: 1, 1, 1 -trifluoroethane cis-2 -butene 1 -heptyne 2 -chloro-4, 5 -diethylnonane cyclohexane ethylcyclopropane meta-dichlorobenzene 2, 4, 6 -trinitrotoluene ortho-iodotoluene ethyl pentanoate 2 -bromohexanal methyl propanamide propoxypropane (propylpentyl ether) m-iodophenol 1, 3 -propanedioic acid propylamine 1, 4 -butandiol 3 -chloropentanoic acid 1, 3 -dibromo-2 -propanone methyl-3 -chloropropyl ether 3 -hydroxy-1 -pentyne
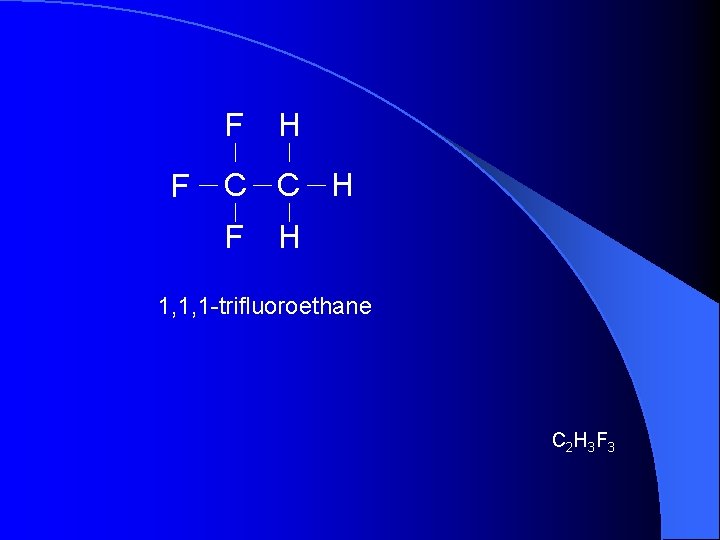
F F H C C H F H 1, 1, 1 -trifluoroethane C 2 H 3 F 3

H H C C H 3 C CH 3 cis-2 -butene H 3 C H H C C CH 3 trans-2 -butene C 4 H 8
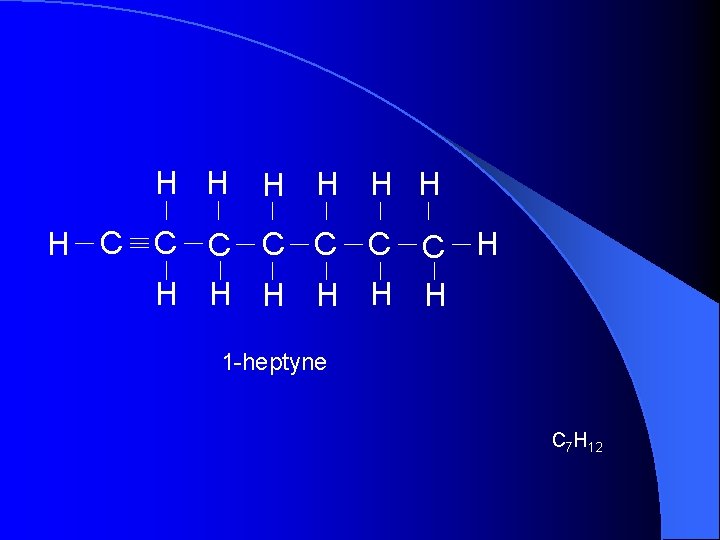
H H H H C C C C H H H H 1 -heptyne C 7 H 12

H H H H C C C C H H H C C C Br H H C C Cl H H H C C H H
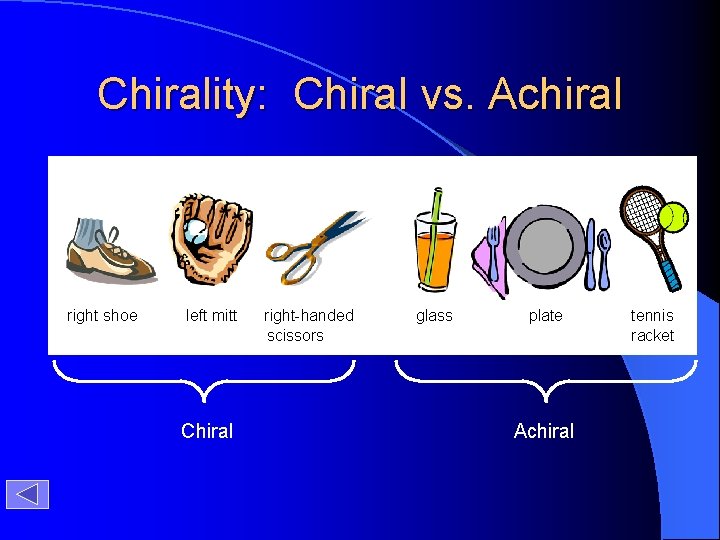
Chirality: Chiral vs. Achiral right shoe left mitt Chiral right-handed scissors glass plate Achiral tennis racket
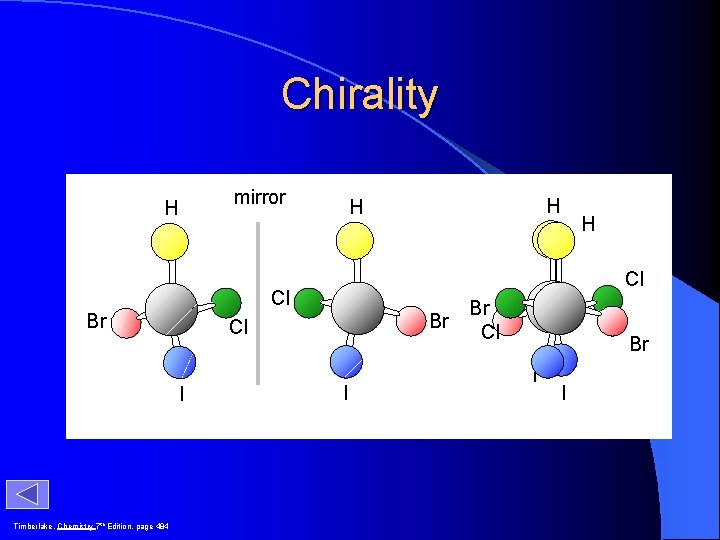
Chirality H mirror H H Cl Cl Br Timberlake, Chemistry 7 th Edition, page 484 Br Cl I H I Br Cl Br I I
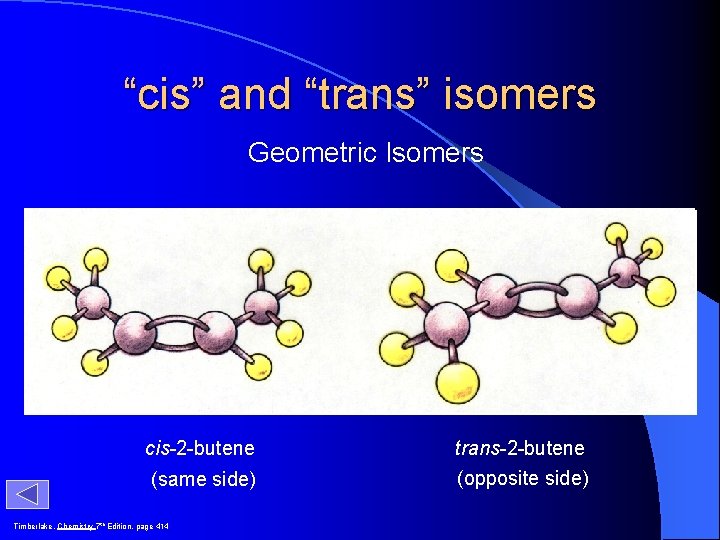
“cis” and “trans” isomers Geometric Isomers H H 3 C H C=C CH 3 H 3 C C=C H cis-2 -butene (same side) Timberlake, Chemistry 7 th Edition, page 414 trans-2 -butene (opposite side) CH 3 H
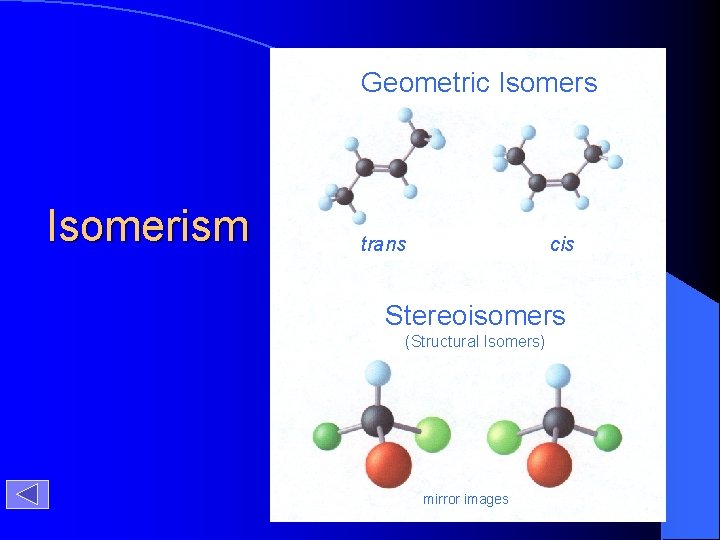
Geometric Isomers Isomerism trans cis Stereoisomers (Structural Isomers) mirror images
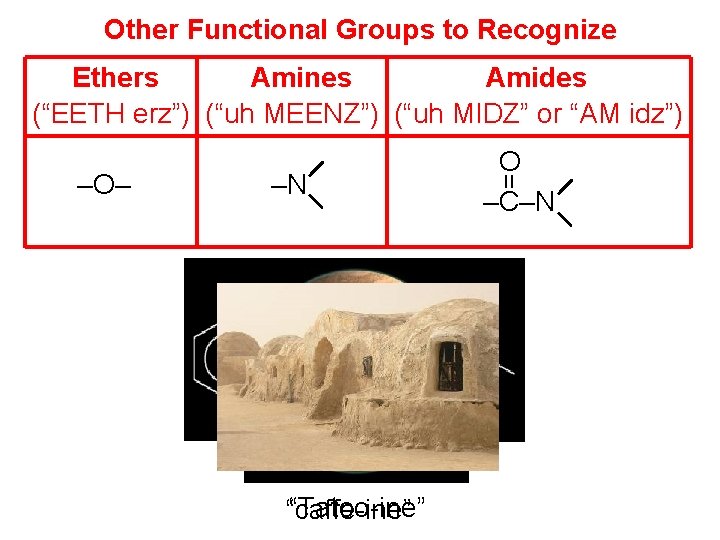
Other Functional Groups to Recognize Ethers Amines Amides (“EETH erz”) (“uh MEENZ”) (“uh MIDZ” or “AM idz”) –N “coca-ine” “Tatoo-ine” “caffe-ine” = –O– O –C–N
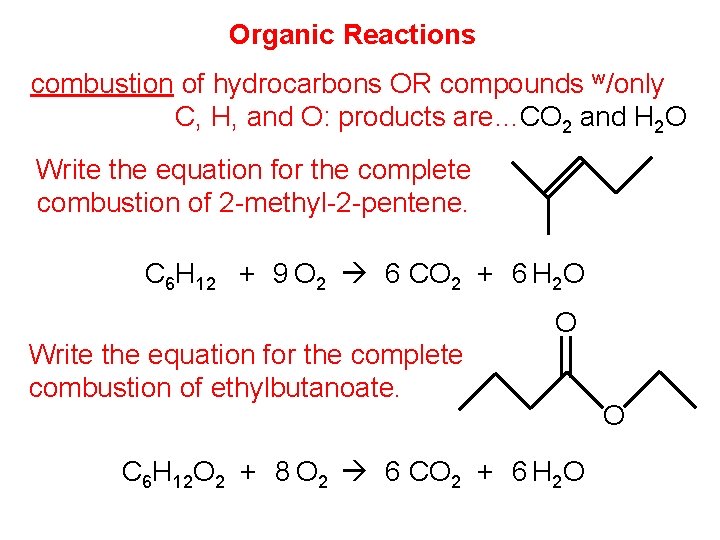
Organic Reactions combustion of hydrocarbons OR compounds w/only C, H, and O: products are…CO 2 and H 2 O Write the equation for the complete combustion of 2 -methyl-2 -pentene. C 6 H 12 + 9 O 2 6 CO 2 + 6 H 2 O Write the equation for the complete combustion of ethylbutanoate. O C 6 H 12 O 2 + 8 O 2 6 CO 2 + 6 H 2 O O
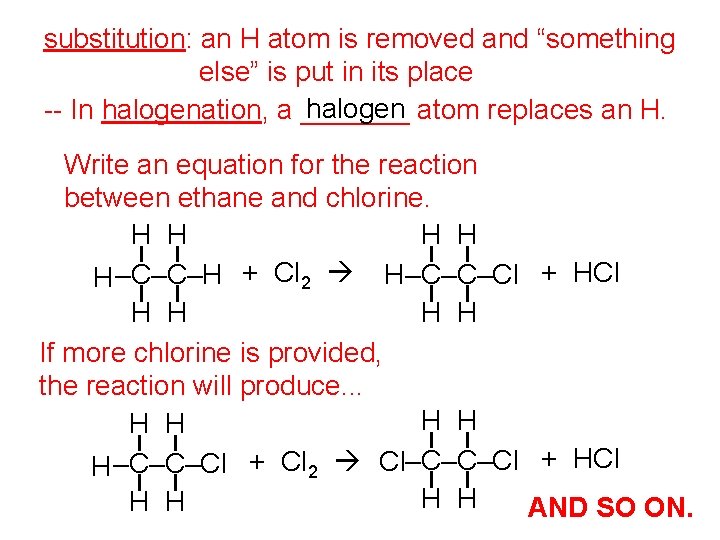
substitution: an H atom is removed and “something else” is put in its place halogen atom replaces an H. -- In halogenation, a _______ Write an equation for the reaction between ethane and chlorine. H H H –C–C–H + Cl 2 H–C–C–Cl + HCl H H If more chlorine is provided, the reaction will produce. . . H H H –C–C–Cl + Cl 2 Cl–C–C–Cl + HCl H H AND SO ON.
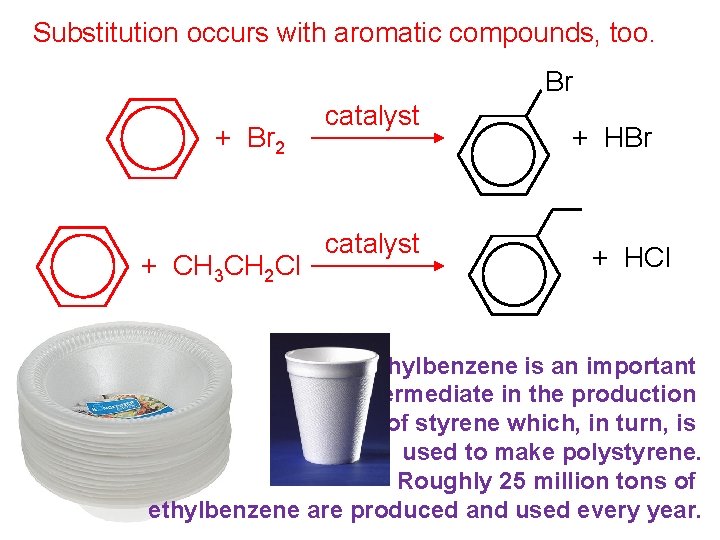
Substitution occurs with aromatic compounds, too. Br + Br 2 + CH 3 CH 2 Cl catalyst + HBr + HCl Ethylbenzene is an important intermediate in the production of styrene which, in turn, is used to make polystyrene. Roughly 25 million tons of ethylbenzene are produced and used every year.
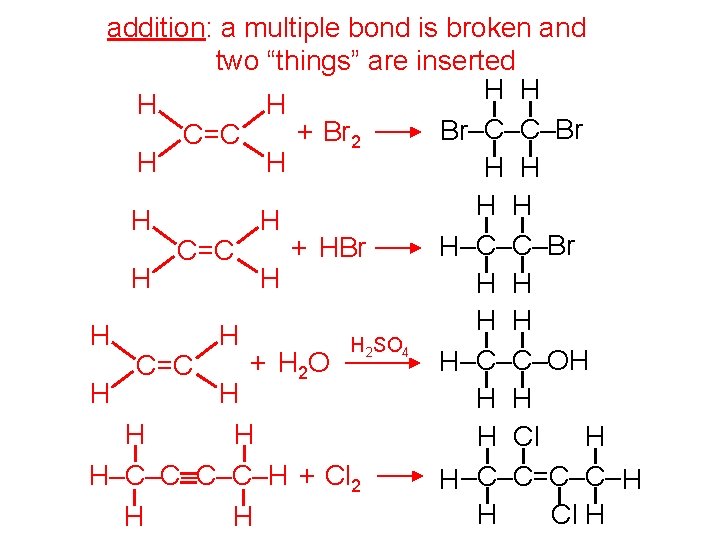
addition: a multiple bond is broken and two “things” are inserted H H Br–C–C–Br + Br 2 C=C H H H H H–C–C–Br + HBr C=C H H H H H 2 SO 4 H–C–C–OH + H 2 O C=C H H H H Cl H H–C–C–H + Cl 2 H –C–C=C–C– H H Cl H H H
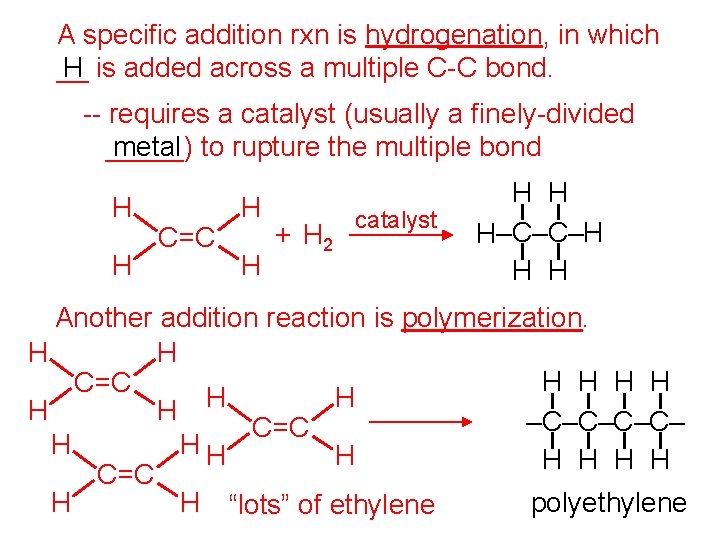
A specific addition rxn is hydrogenation, in which __ H is added across a multiple C-C bond. -- requires a catalyst (usually a finely-divided _____) metal to rupture the multiple bond H H C=C H H + H 2 catalyst H H H–C–C–H H H Another addition reaction is polymerization. H H H C=C H H –C–C– C=C H HH H H C=C H H “lots” of ethylene polyethylene
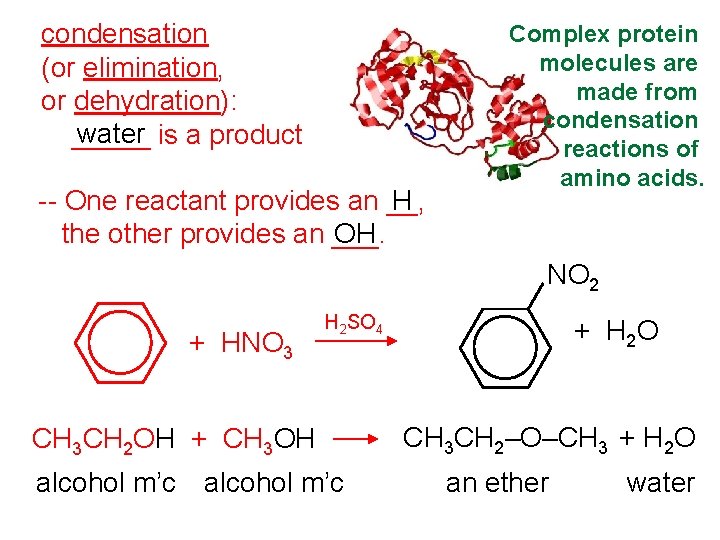
condensation (or elimination, or dehydration): water is a product _____ -- One reactant provides an __, H the other provides an ___. OH Complex protein molecules are made from condensation reactions of amino acids. NO 2 + HNO 3 H 2 SO 4 CH 3 CH 2 OH + CH 3 OH alcohol m’c + H 2 O CH 3 CH 2–O–CH 3 + H 2 O an ether water

-- Amides can be formed in condensation rxns between carboxylic acids and amines. Write the equation for the reaction between butanoic acid and nitrogen trihydride. Ammonia is the simplest amine. O O + NH 3 + H 2 O OH carboxylic acid amine NH 2 amide water
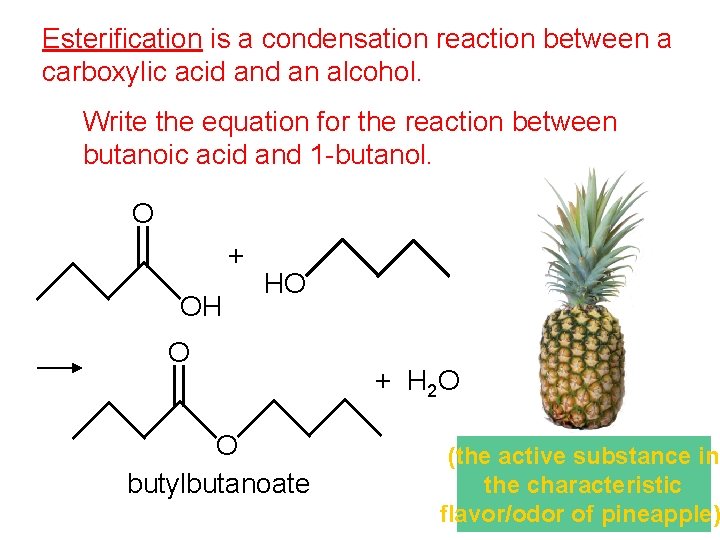
Esterification is a condensation reaction between a carboxylic acid an alcohol. Write the equation for the reaction between butanoic acid and 1 -butanol. O + OH HO O O butylbutanoate + H 2 O (the active substance in the characteristic flavor/odor of pineapple)

Write the equation for the reaction between 3 -phenyl-2 -propenoic acid and ethanol. O + HO OH O + H 2 O (the active substance in O ethyl-3 -phenyl-2 -propenoate (i. e, ethyl cinnamate ) the characteristic flavor/odor of cinnamon) “When in doubt, make water. ” Mr. B
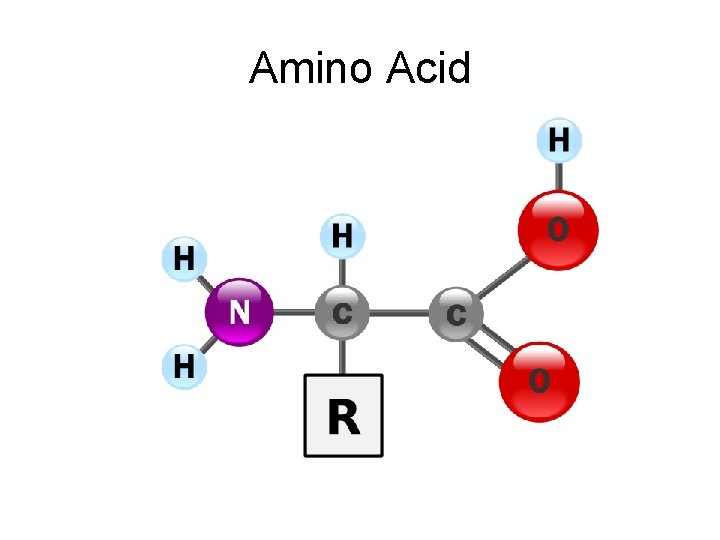
Amino Acid
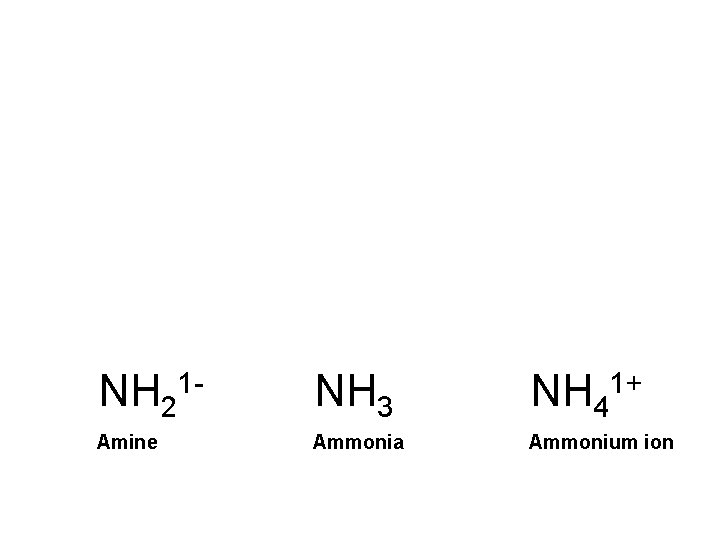
NH 21 - NH 3 NH 41+ Amine Ammonia Ammonium ion
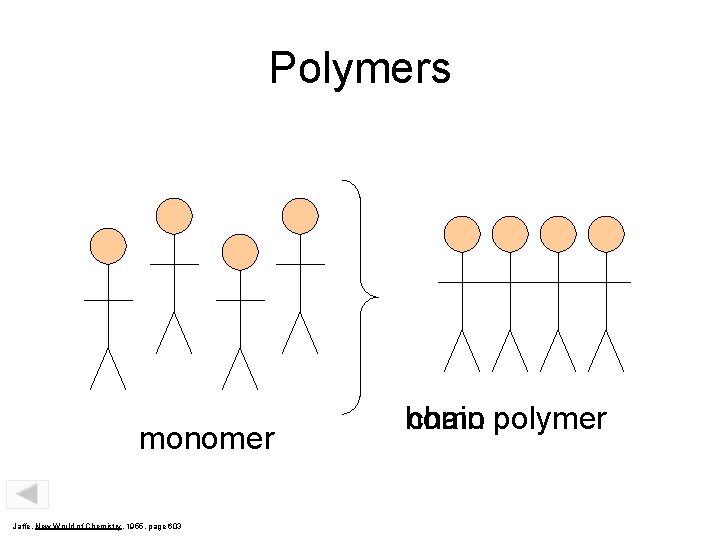
Polymers monomer Jaffe, New World of Chemistry, 1955, page 603 homo chain polymer
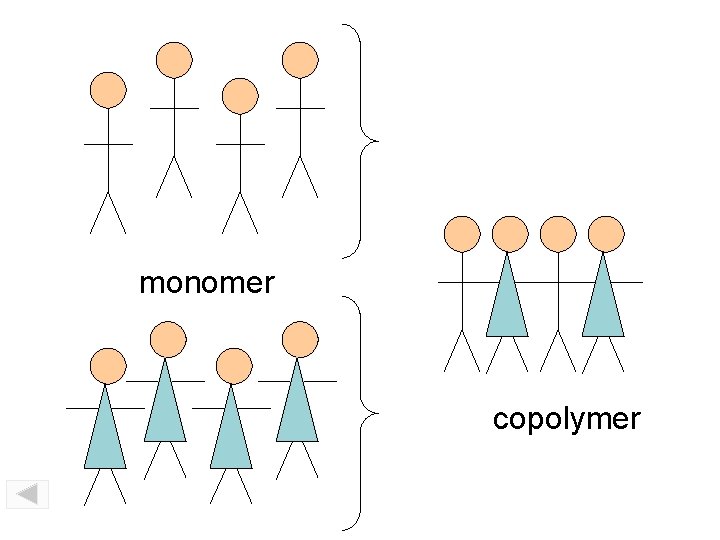
monomer copolymer
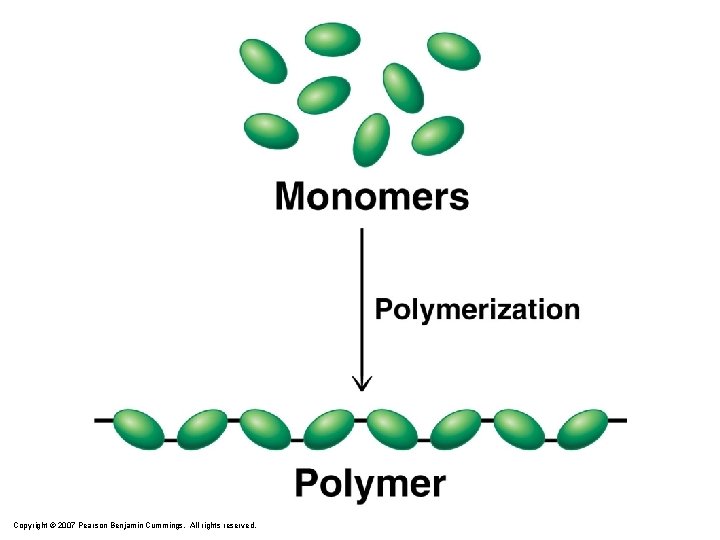
Copyright © 2007 Pearson Benjamin Cummings. All rights reserved.

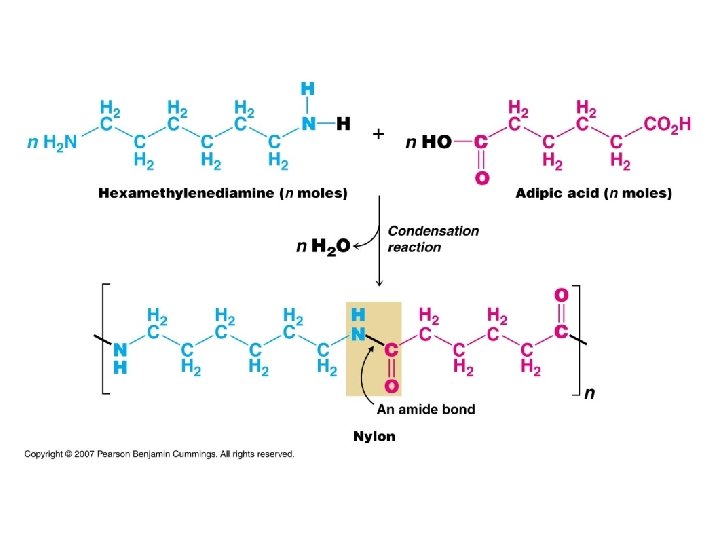
Polymerization Polymers = Monomer + … l Synthetic Polymers – – l Nylon “plastics” Vulcanized rubber polyethylene Natural Polymers – Silk – Proteins (amino acids) – Starch (sugars)
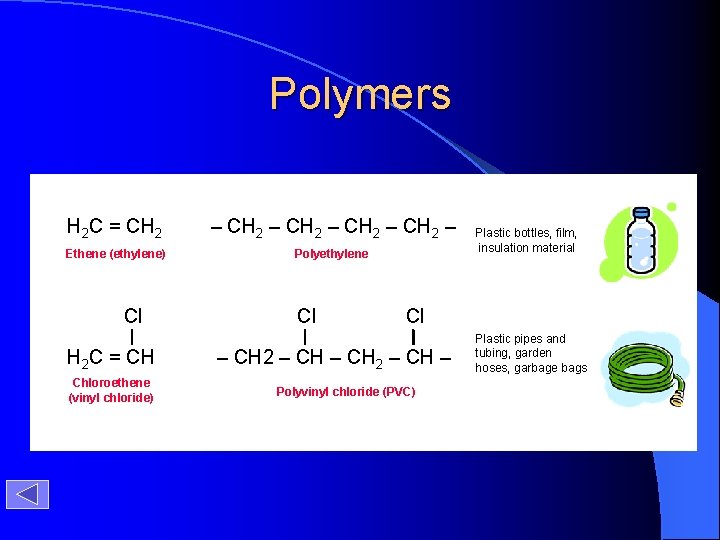
Polymers H 2 C = CH 2 – CH 2 – Ethene (ethylene) Polyethylene Cl H 2 C = CH Chloroethene (vinyl chloride) Cl Plastic bottles, film, insulation material Cl – CH 2 – CH – Polyvinyl chloride (PVC) Plastic pipes and tubing, garden hoses, garbage bags
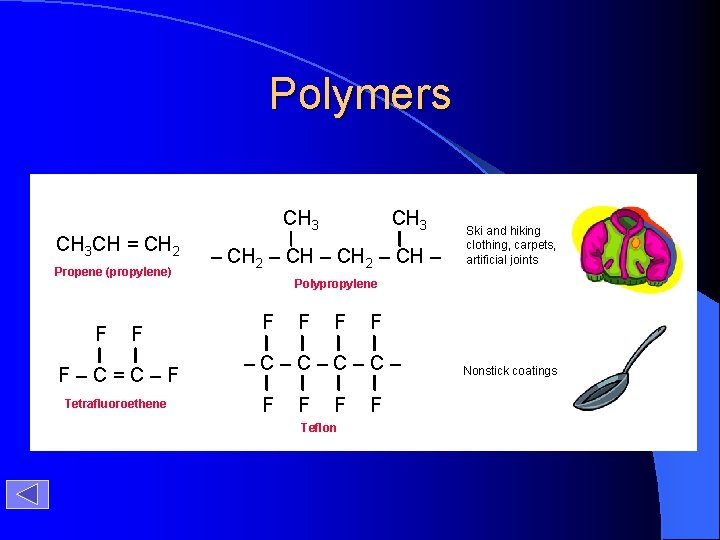
Polymers CH 3 CH = CH 2 Propene (propylene) F F F–C=C–F Tetrafluoroethene CH 3 – CH 2 – CH – Ski and hiking clothing, carpets, artificial joints Polypropylene F F –C–C– F F F Teflon F Nonstick coatings
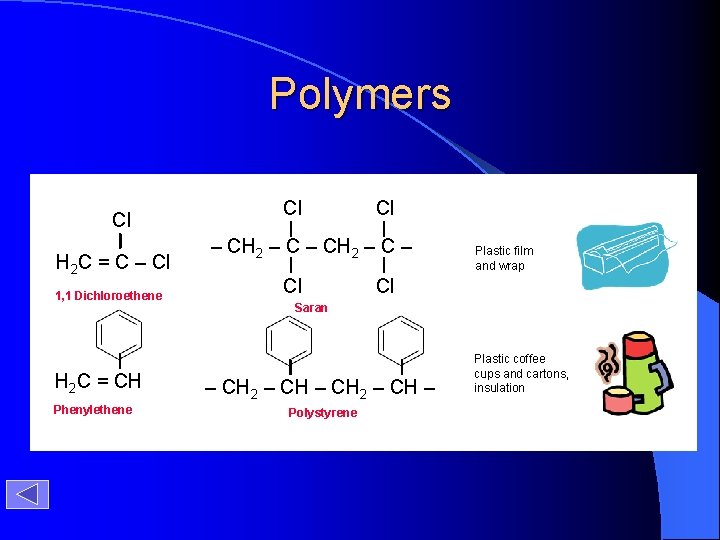
Polymers Cl H 2 C = C – Cl 1, 1 Dichloroethene H 2 C = CH Phenylethene Cl Cl – CH 2 – C – Cl Plastic film and wrap Cl Saran – CH 2 – CH – Polystyrene Plastic coffee cups and cartons, insulation

Slime l PVA + PVA – With cross-linking agent (borax solution)

What are some things you think of when I say organic? • • Medicine Plastics Soil Soap Petroleum Grass Clothing PRISM fellow: Mr. Kevin J. Hodel

• • Organic - the study of carbon containing compounds that their properties Why carbon? ? Strong bonds between carbons Low reactivity of carbon compounds Geometry of carbon compounds PRISM fellow: Mr. Kevin J. Hodel

Diamonds PRISM fellow: Mr. Kevin J. Hodel

Graphite PRISM fellow: Mr. Kevin J. Hodel

Polymers • Polymer - a large organic molecule composed of smaller units bonded together – These smaller units are called monomers. • Tires – Vulcanized rubber was discovered by mistake!! • soft and weak until heated then forms disulfide linkages between the rubber polymers. PRISM fellow: Mr. Kevin J. Hodel

Natural Polymers • • • Rubber Cotton Paper – • Cotton and paper are made of cellulose which is the most abundant natural polymer in the world. Starch – Potatoes, corn, bread PRISM fellow: Mr. Kevin J. Hodel

Synthetic Polymers • • Half of the industrial chemists work in some area of polymer chemistry. In 1996, more than 270 lbs of plastic was produced person in the US. PRISM fellow: Mr. Kevin J. Hodel

Types of Synthetic Polymers • Elastomers - can be highly stretched and returns to their original shape. • Fibers - polymers that exhibit little or no elasticity (threadlike) • Plastics - synthetic polymers that are more elastic than fibers but less elastic the elastomers. PRISM fellow: Mr. Kevin J. Hodel
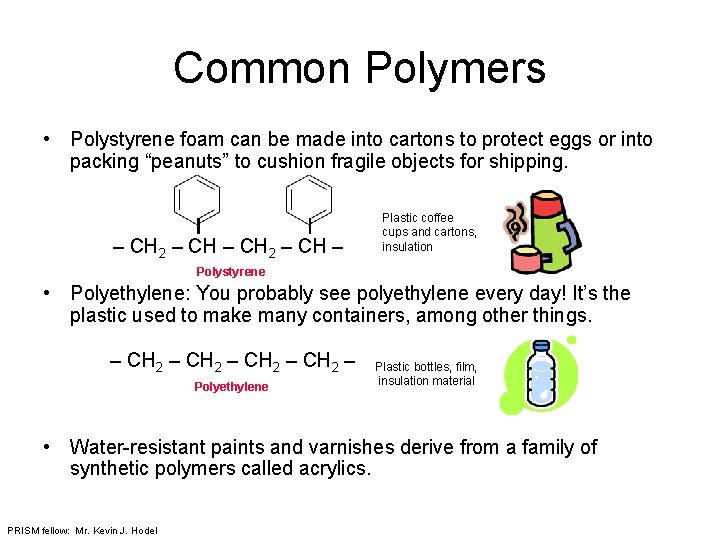
Common Polymers • Polystyrene foam can be made into cartons to protect eggs or into packing “peanuts” to cushion fragile objects for shipping. – CH 2 – CH – Plastic coffee cups and cartons, insulation Polystyrene • Polyethylene: You probably see polyethylene every day! It’s the plastic used to make many containers, among other things. – CH 2 – Polyethylene Plastic bottles, film, insulation material • Water-resistant paints and varnishes derive from a family of synthetic polymers called acrylics. PRISM fellow: Mr. Kevin J. Hodel

Recycling • Before: Milk jug. After: Picnic table. • Before: Bread bag. After: Trash can. • Before: Sandwich box. After: Frisbee. PRISM fellow: Mr. Kevin J. Hodel
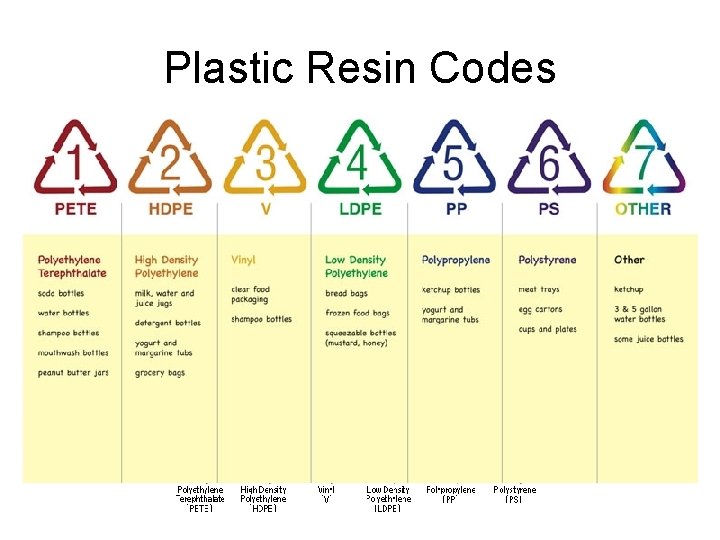
Plastic Resin Codes
Copyright © 2007 Pearson Benjamin Cummings. All rights reserved.
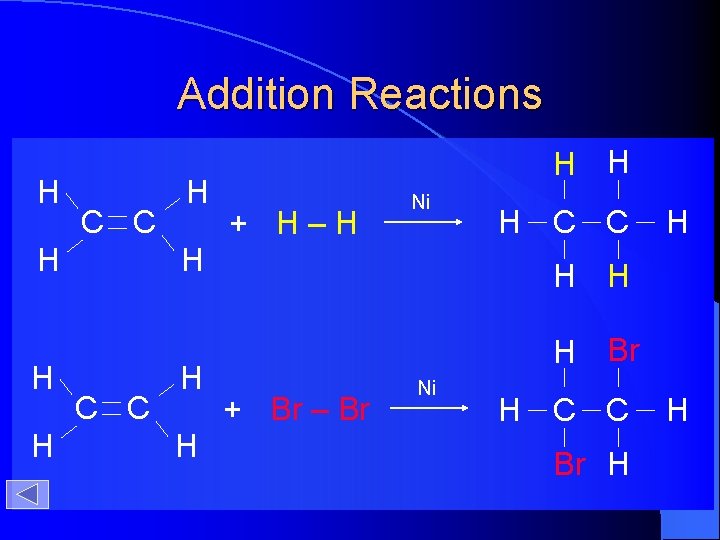
Addition Reactions H C C H H H + H–H Ni H H C C H H Br + Br – Br Ni H C C Br H H
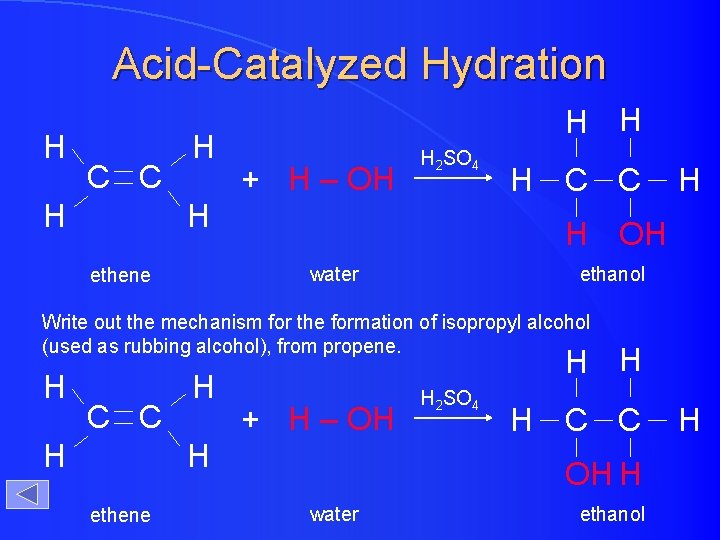
Acid-Catalyzed Hydration H C C H H + H – OH H 2 SO 4 H H OH water ethene C C ethanol Write out the mechanism for the formation of isopropyl alcohol (used as rubbing alcohol), from propene. H C C H H + H – OH H ethene H 2 SO 4 H C C OH H water ethanol H
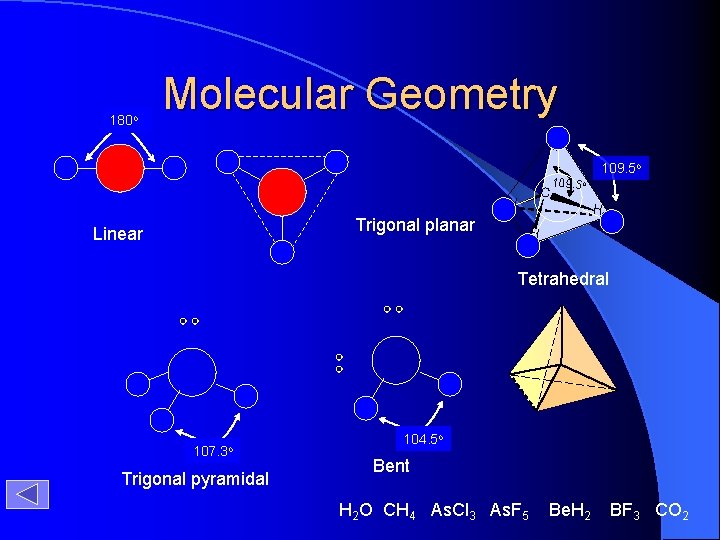
180 o Molecular Geometry H C H Trigonal planar Linear 109. 5 o H H Tetrahedral 107. 3 o Trigonal pyramidal 104. 5 o Bent H 2 O CH 4 As. Cl 3 As. F 5 Be. H 2 BF 3 CO 2

Order of Priority of Functional Groups Order of priority Functional group Formula 1 Carboxylic acid -COOH 8 Ketone -CO 2 Sulfonic acid -SO 3 H 9 Alcohol -OH 3 Ester -COOR 10 Phenol -OH 4 Acid chloride -COCl 11 Thiol -SH 5 Amide -CONH 2 12 Amine -NH 2 6 Nitrile -CN 13 Ether -OR 7 Aldehyde -CHO 14 Sulfide -SR Selinger, Chemistry in the Marketplace, 1994, page 23
- Slides: 168