Organic Chemistry Organic molecules that have a carbon

Organic Chemistry • Organic – molecules that have a carbon skeleton and also contain some hydrogen atoms • Carbon is versatile because can form many bonds allowing for vast array of organic molecules • Functional groups – groups of atoms attached to the carbon backbone that determine the characteristics and chemical reactivity of the molecule

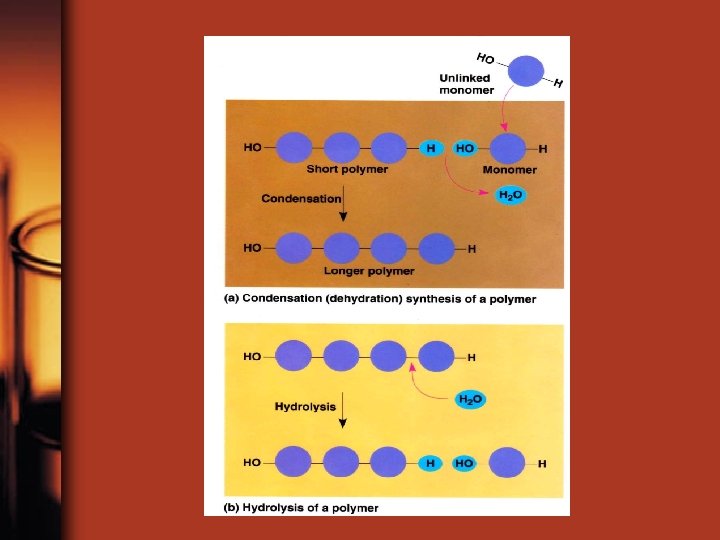
• Organic molecules are synthesized by linking small organic molecule subunits (monomers) together to form a long chain called a polymer • Monomers are linked together by a condensation reaction (dehydration synthesis) • Monomers are broken apart by hydrolysis – a water molecule is added to the reaction and broken apart to separate monomers

Carbohydrates • Composed of carbon, hydrogen, and oxygen (CH 2 O)n • Important source of energy • Important structural support for single cells, plants, fungi, bacteria, and insects

Simple Sugars monosaccharides • Composed of 3 to 7 carbon atoms – “circle up” into a ring when dissolved in water • Glucose (C 6 H 12 O 6) most common monosaccharide in living organisms • Body’s main source of energy • Other monosaccharides include fructose, galactose, ribose, and deoxyribose

Disaccharides and Polysaccharides • • 1. 2. 3. 4. Disaccharides – two single sugars linked together (examples: maltose, sucrose, lactose) Polysaccharides – long chains of single sugars Starch – long term energy (glucose) storage for plants Glycogen – energy storage (glucose) for animals Cellulose – structural support for plants (makes up cell walls) a. Highly insoluble b. Bonds btw glucose units are β linkages instead of α linkages c. Animals cannot digest cellulose

Lipids • • 1. 2. 3. 4. 5. Contain carbon, hydrogen, and oxygen and may contain phosphorus and nitrogen – highly insoluble (hydrophobic) Functions: Energy storage molecules Provide insulation Forms waterproof covering Make up the bulk of cell membranes Act as hormones

Three types of lipids 1. Oils, fats and waxes • • Contain only carbon, hydrogen, and oxygen Contain glycerol bonded to 3 fatty acid subunits

2. Phospholipids • Similar to oil except one fatty acid is replaced with a polar phosphate group – primary component of cell membranes
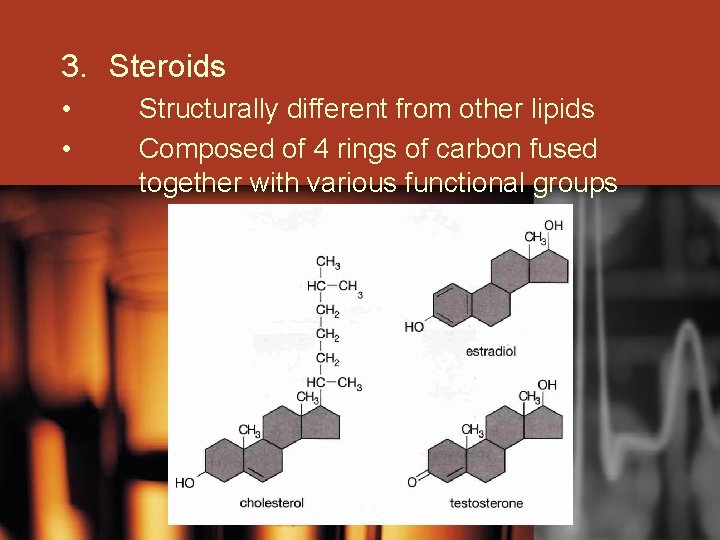
3. Steroids • • Structurally different from other lipids Composed of 4 rings of carbon fused together with various functional groups

Proteins • • 1. 2. 3. 4. 5. 6. Molecules composed of one or more chains of amino acids Functions of proteins: Structure (skin, hair, nails, horns) Movement (muscles) Defense (antibodies) Storage of energy and materials (albumin in egg whites) Transport (hemoglobin to carry O 2) Signals/hormones (neurotransmitters, protein hormones)
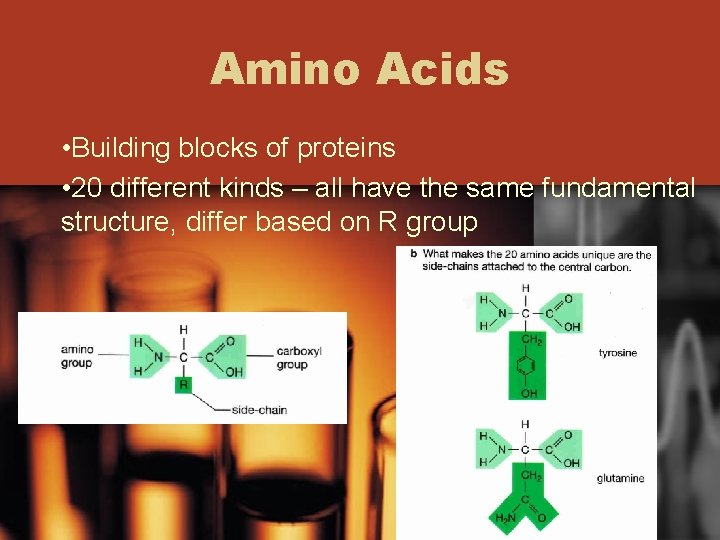
Amino Acids • Building blocks of proteins • 20 different kinds – all have the same fundamental structure, differ based on R group

• R group gives each amino acid it’s distinctive properties (size, water solubility, electrical charge) • Cysteine has sulfur in it’s R group causing it to form bonds with other cysteines forming disulfide bridges
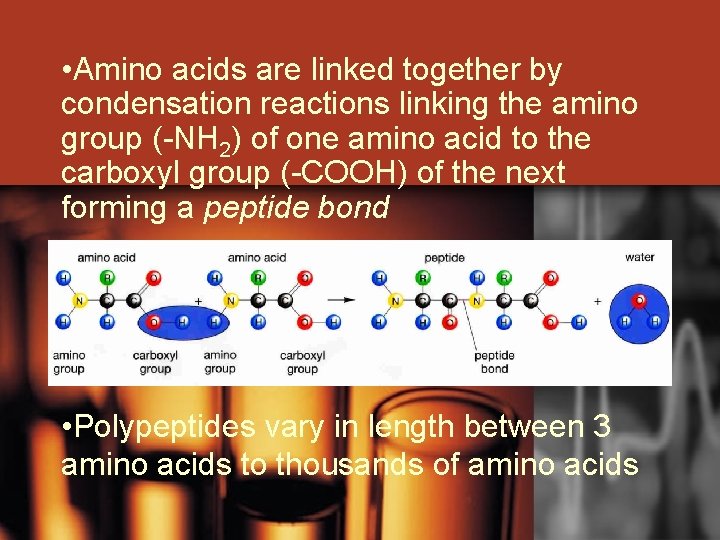
• Amino acids are linked together by condensation reactions linking the amino group (-NH 2) of one amino acid to the carboxyl group (-COOH) of the next forming a peptide bond • Polypeptides vary in length between 3 amino acids to thousands of amino acids
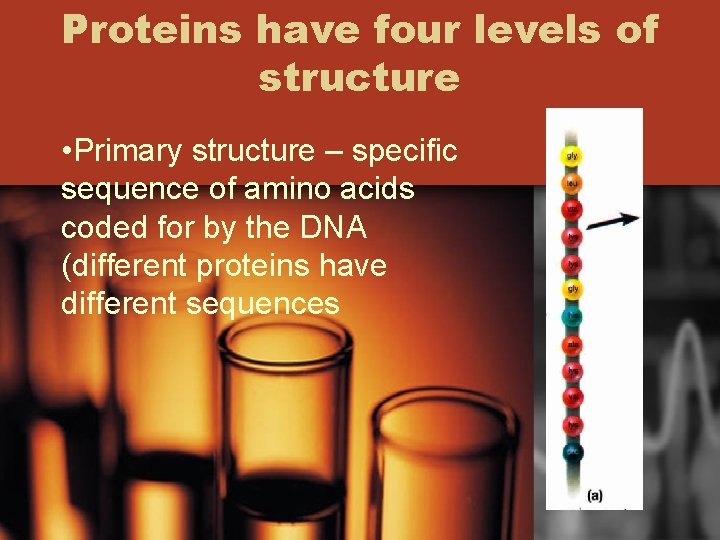
Proteins have four levels of structure • Primary structure – specific sequence of amino acids coded for by the DNA (different proteins have different sequences
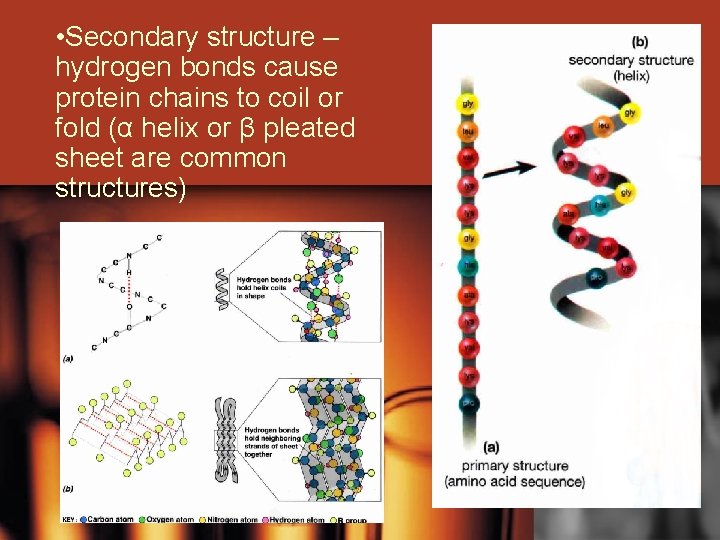
• Secondary structure – hydrogen bonds cause protein chains to coil or fold (α helix or β pleated sheet are common structures)
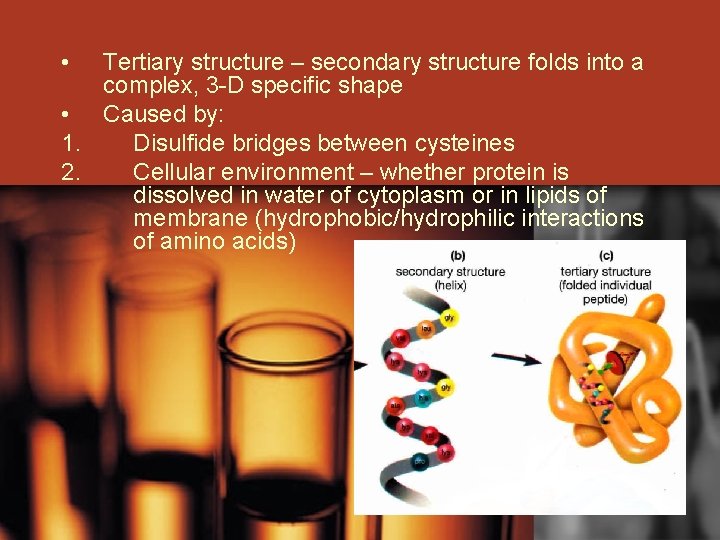
• Tertiary structure – secondary structure folds into a complex, 3 -D specific shape • Caused by: 1. Disulfide bridges between cysteines 2. Cellular environment – whether protein is dissolved in water of cytoplasm or in lipids of membrane (hydrophobic/hydrophilic interactions of amino acids)
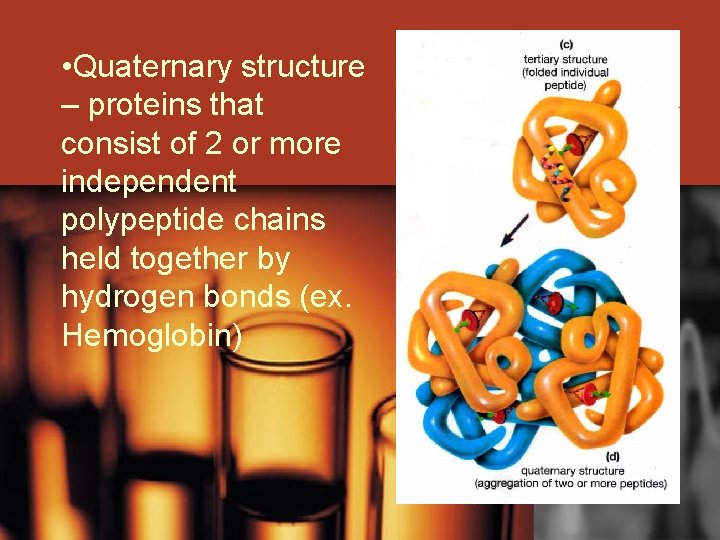
• Quaternary structure – proteins that consist of 2 or more independent polypeptide chains held together by hydrogen bonds (ex. Hemoglobin)

The shape of a protein allows it to perform it’s function • • • a. b. c. d. e. Exact type, position and number of amino acids with specific R groups determines both the structure (shape) of the protein and it’s biological function If the shape is changed, the protein will NO LONGER function the same way Denatured proteins – have lost most of their secondary, tertiary, and quaternary structure (THE SHAPE HAS CHANGED – protein will no longer function the same way) 1. Causes: Extremes in temperature (over 60 o. C) Extremes in p. H Excessive radiation Electricity Certain chemicals
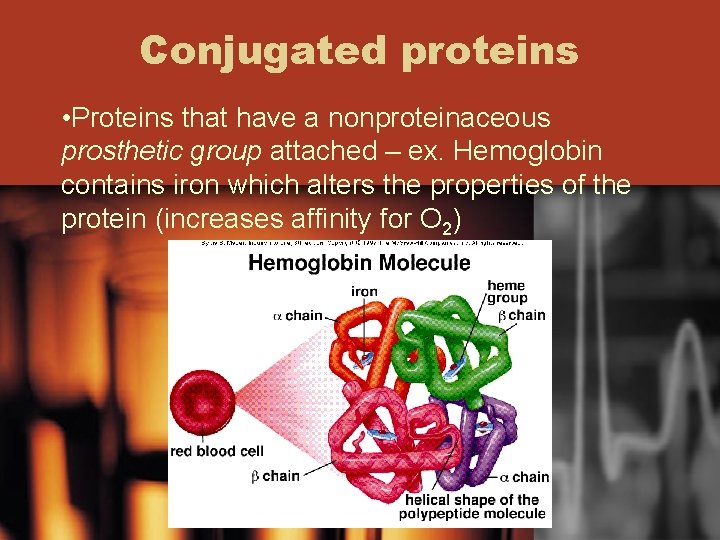
Conjugated proteins • Proteins that have a nonproteinaceous prosthetic group attached – ex. Hemoglobin contains iron which alters the properties of the protein (increases affinity for O 2)
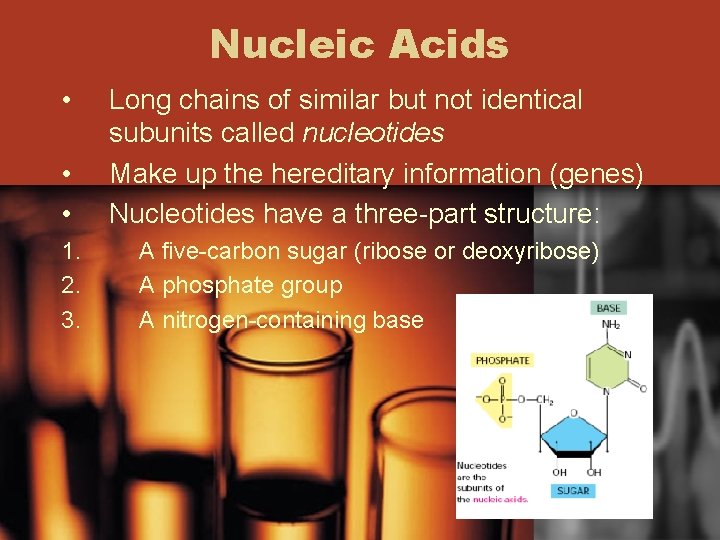
Nucleic Acids • • • 1. 2. 3. Long chains of similar but not identical subunits called nucleotides Make up the hereditary information (genes) Nucleotides have a three-part structure: A five-carbon sugar (ribose or deoxyribose) A phosphate group A nitrogen-containing base

There are two types of nucleotides • Ribose nucleotides (contain the sugar, ribose) § bond to four types of nitrogen bases: adenine, guanine, cytosine, and uracil • Deoxyribose nucleotides (contain the sugar, deoxyribose) § bond to adenine, guanine, cytosine, and thymine
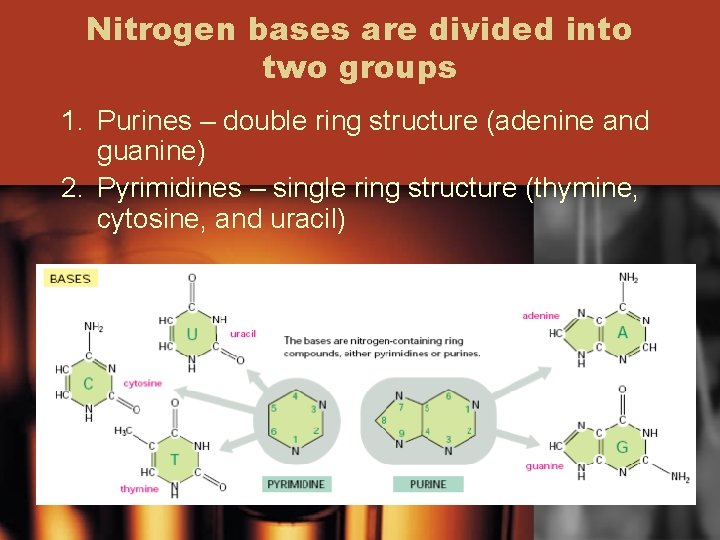
Nitrogen bases are divided into two groups 1. Purines – double ring structure (adenine and guanine) 2. Pyrimidines – single ring structure (thymine, cytosine, and uracil)
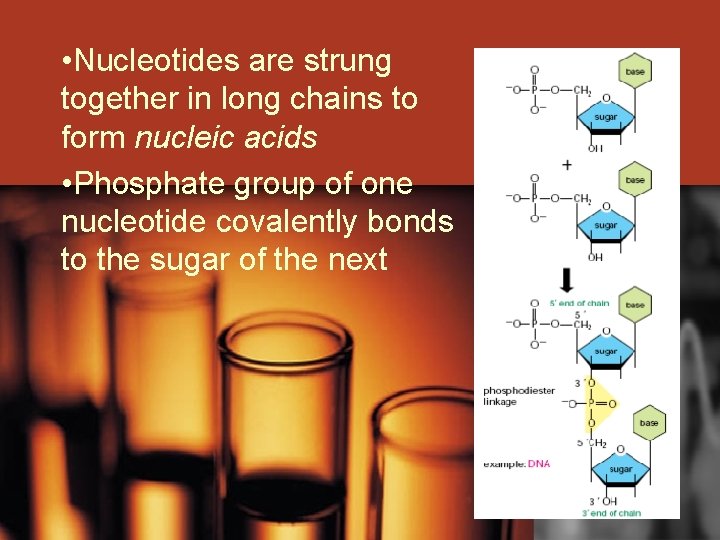
• Nucleotides are strung together in long chains to form nucleic acids • Phosphate group of one nucleotide covalently bonds to the sugar of the next

Two types of Nucleic Acids • Deoxyribonucleic Acid (DNA) 1. Double helix 2. Deoxyribose sugar 3. Contains A, C, T, and G (A-T, C-G) 4. Makes up chromosomes of all living things 5. Sequence of nucleotides spells out information to construct proteins • Ribonucleic Acid (RNA) 1. Single helix 2. Ribose sugar 3. Contains A, C, U, and G (A-U, C-G) 4. RNAs are copies of DNA – carries message to cell to direct synthesis of proteins

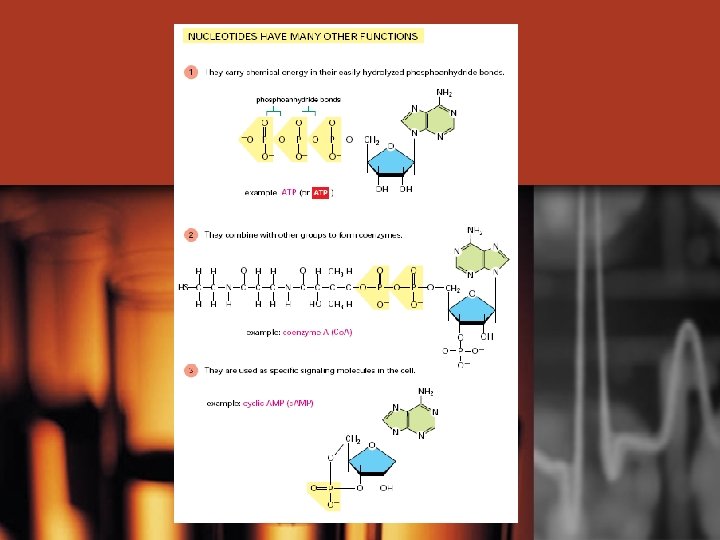
Nucleotides have many functions – not all are part of nucleic acids • • • 1. May exist singly or occur as parts of other molecules Some act as intracellular messengers to carry information from cell membrane to other molecules in cell – ex. cyclic AMP Some nucleotides have extra phosphates groups – ex. Adenosine triphosphate (ATP), Adenosine diphosphate (ADP) Unstable molecules that carry energy from place to place – pick up energy where it is produced (cellular respiration) and give up energy to drive energy demanding reactions elsewhere • Certain nucleotides assist enzyme in their role of promoting chemical rxns – coenzymes – usually consist of a nucleotide combined with a vitamin
- Slides: 27