ORGANIC CHEMISTRY Organic compounds always contain carbon The

ORGANIC CHEMISTRY

• Organic compounds always contain carbon • The compounds can be straight chains, branched, or rings • 4 groups are carbohydrates, lipids, proteins, and nucleic acids

Carbohydrates (sugars) • Made of C, H, and O • 1: 2: 1 ratio of C: H: O • Ex: C 6 H 12 O 6 (glucose) • Names usually end in the suffix -ose

3 Types • Monosaccharides (simple sugars) • Formula C 6 H 12 O 6 • Isomers-same formula, different structure • Ex: glucose, fructose, galactose

Disaccharides (double sugars) • Formula C 12 H 22 O 11 • Formed through condensation/dehydration synthesis reactions-producing a waste water molecule. Taking small things to make a large compound • Ex: maltose, lactose, sucrose

• C 6 H 12 O 6 + C 6 H 12 O 6 = C 12 H 22 O 11 + H 2 O (Dehydration Synthesis/Condensation Reaction)

Polysaccharides • • • 3 or more monomers Cellulose- plant structure, cell wall Glycogen- animal energy storage Starch- plant energy storage Chitin- animal shells (exoskeletons)

Can you figure it out?

Lipids (fats, oils, & waxes) • Solid lipids- fats, liquid lipidsoils • Contains C, H, O • C: H: O ratio is very large • Ex: C 57 H 110 O 6 (no 1: 2: 1 ratio)

• Provides the most energy because they have the most chemical bonds • Used for long term energy storage, insulation, protective coatings, cell membranes • Formed by 3 fatty acids + glycerol lipids+ water (condensation reaction)

Proteins (large complex polymers) • Made of C, H, O, N • 20 different amino acids make hundred of different proteins • Amino acids are linked by peptide bonds forming protein & water

• Used for growth, repair of cells, limited amounts of energy, not stored for energy because Nitrogen is harmful to the body

Enzymes • Special proteins • Used as a catalyst, speeds up chemical reactions • Boost metabolic processes (digestion), releasing energy • Allows reactions to occur more quickly & at a lower temp.

• Lock & key theory (specific substrates only fit specific active sites) • Enzymes end in the suffix –ase • Ex: lactase, sucrase, peroxidase • Active Site-part of the enzyme where the substrate attaches • Substrate-compound that the enzymes acts on
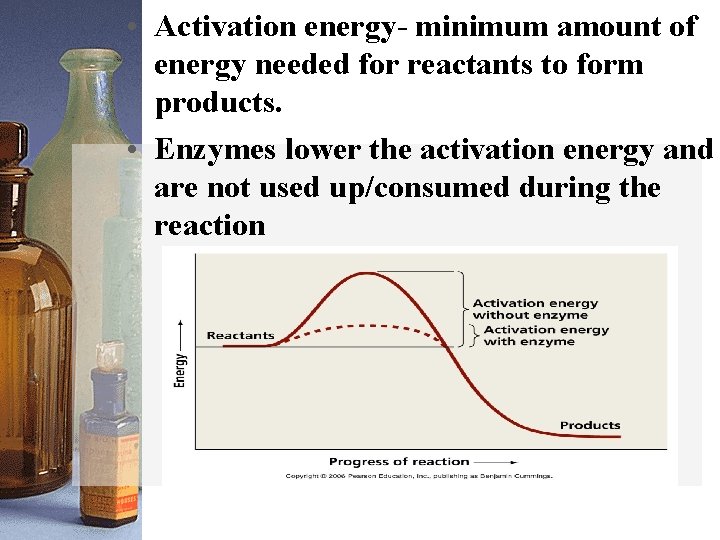
• Activation energy- minimum amount of energy needed for reactants to form products. • Enzymes lower the activation energy and are not used up/consumed during the reaction

Nucleic Acids • Macromolecule that stores information in the cells in the form of a code • Composed of nucleotides- made of C, H, O, N, P • Arranged as a base, simple sugar, & a phosphate group • DNA, RNA

• Organic compounds that are used for energy are : 1. Lipids 2. Carbohydrates 3. Proteins • Energy is made available by breaking down polymers through a reaction called hydrolysis • Hydrolysis- uses water to break up polymer to release energy (reverse of condensation/dehydration synthesis reaction)
- Slides: 17