Organic Chemistry Organic compound compound that contains carbon
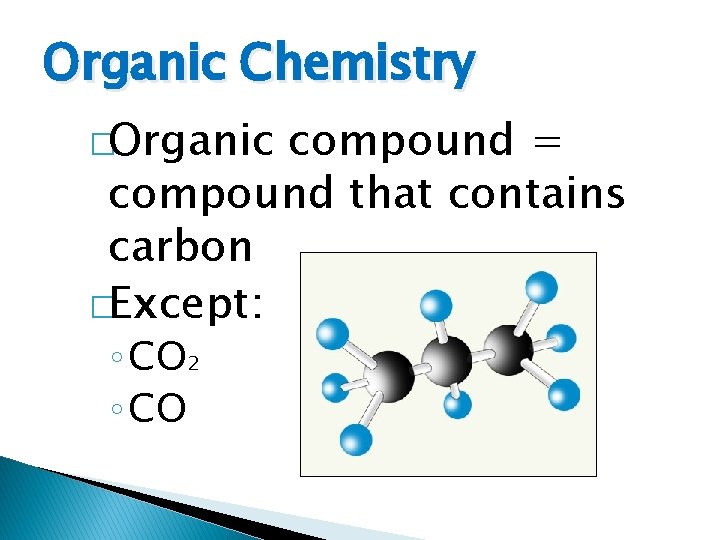
Organic Chemistry �Organic compound = compound that contains carbon �Except: ◦ CO 2 ◦ CO
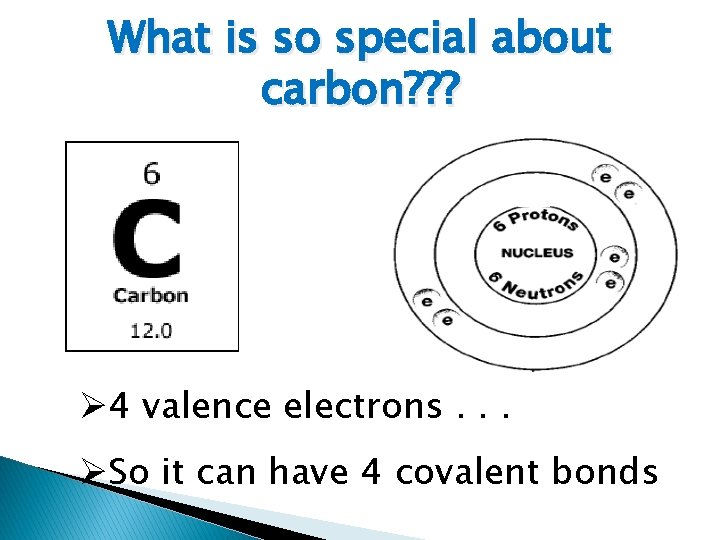
What is so special about carbon? ? ? Ø 4 valence electrons. . . ØSo it can have 4 covalent bonds
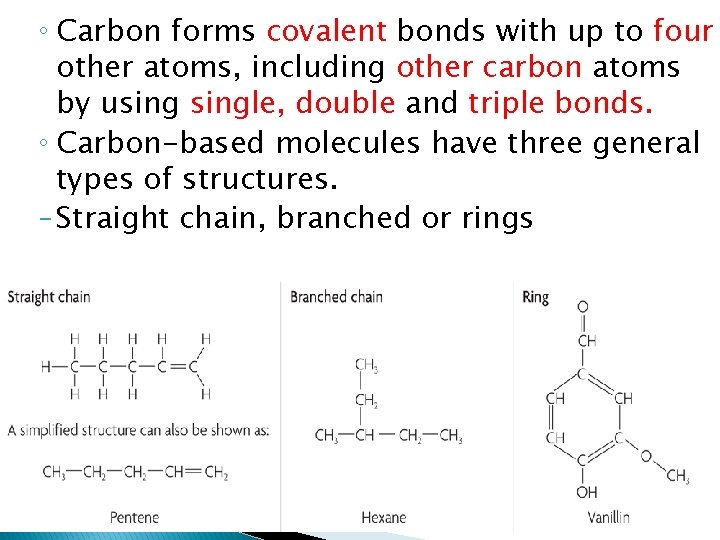
◦ Carbon forms covalent bonds with up to four other atoms, including other carbon atoms by usingle, double and triple bonds. ◦ Carbon-based molecules have three general types of structures. – Straight chain, branched or rings

Remember: Chemical Formula vs. Structural Formula Chemical Formula: CH 4 CO 2 H 20 NH 3 CH 3 CH 2 CH 3 Structural Formula:

Terms • Macromolecules: The 4 molecules of life are called macromolecules (large) – Carbohydrates, Proteins, Lipids, Nucleic acids • Monomer = building blocks • Polymer = large compound made up of smaller monomers

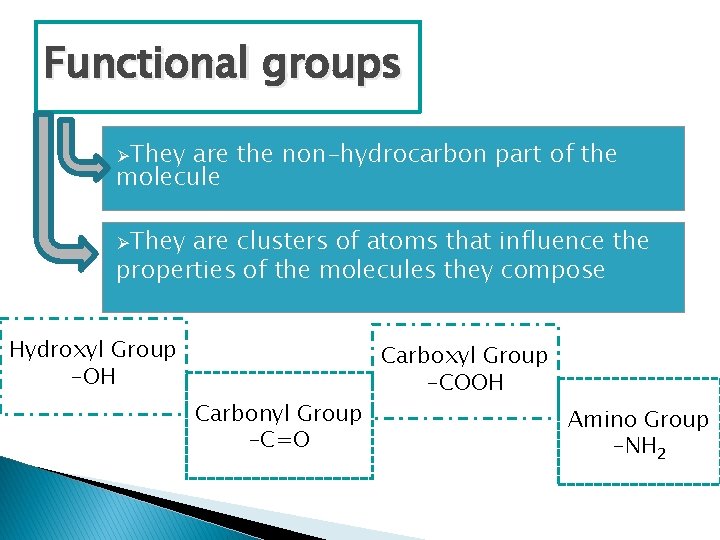
Functional groups ØThey are the non-hydrocarbon part of the molecule ØThey are clusters of atoms that influence the properties of the molecules they compose Hydroxyl Group -OH Carbonyl Group -C=O Carboxyl Group -COOH Amino Group -NH 2
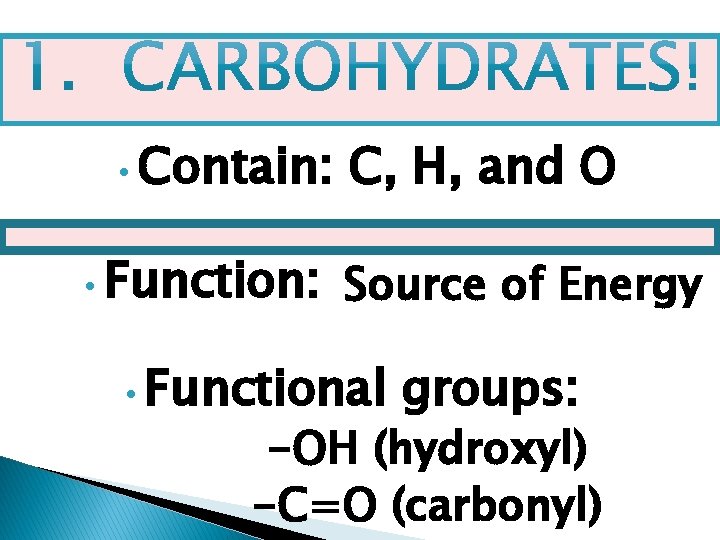
• Contain: • Function: C, H, and O Source of Energy • Functional groups: -OH (hydroxyl) -C=O (carbonyl)

• Monomer • Polymer = Monosaccharide (simple sugar) = Disaccharides and polysaccharides
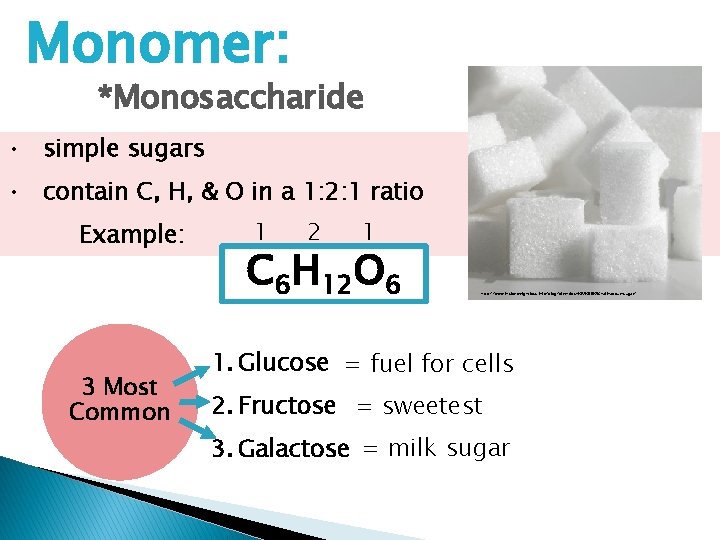
Monomer: *Monosaccharide • simple sugars • contain C, H, & O in a 1: 2: 1 ratio Example: 3 Most Common 1 2 1 C 6 H 12 O 6 http: //www. insideweightloss. info/blog/diet-tips-%E 2%80%93 -all-about-sugar/ 1. Glucose = fuel for cells 2. Fructose = sweetest 3. Galactose = milk sugar

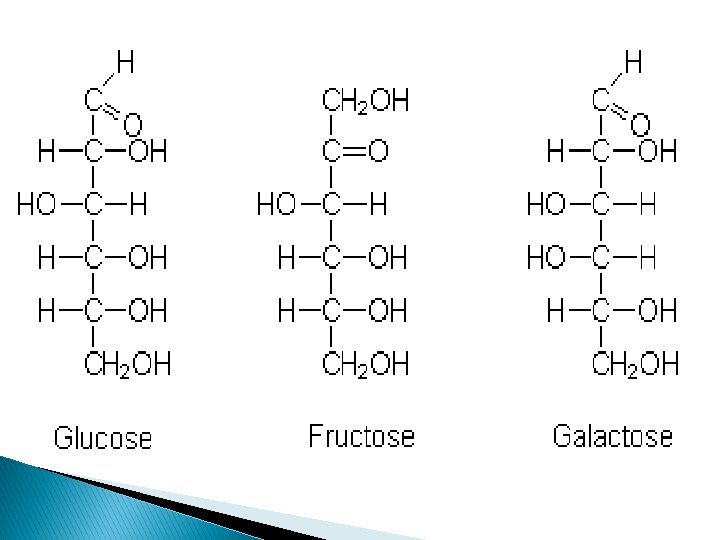

Isomer= compounds with the SAME chemical formula, but different structural formulas Example: glucose, fructose, and galactose are all isomers of each other. . . C 6 H 12 O 6

Polymer: ØIs *Disaccharide a double sugar formed when 2 monosaccharides combine in a dehydration synthesis rxn 3 Type s 1. Maltose = glucose + glucose 2. Sucrose = glucose + fructose 3. Lactose = glucose + galactose
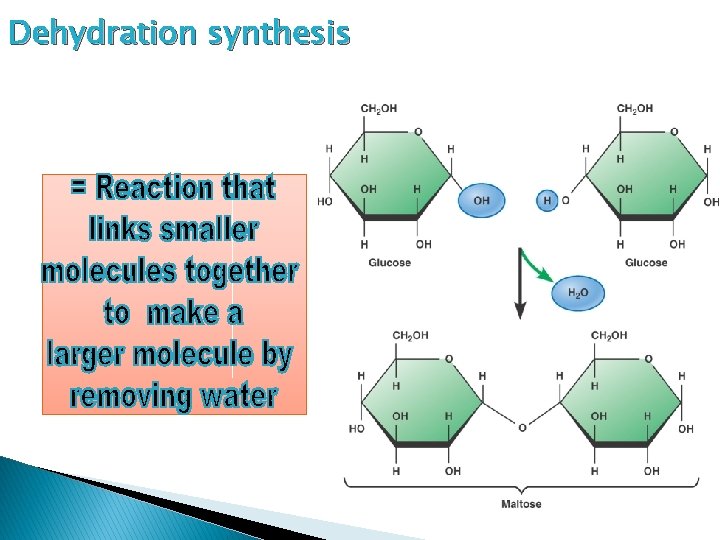
Dehydration synthesis
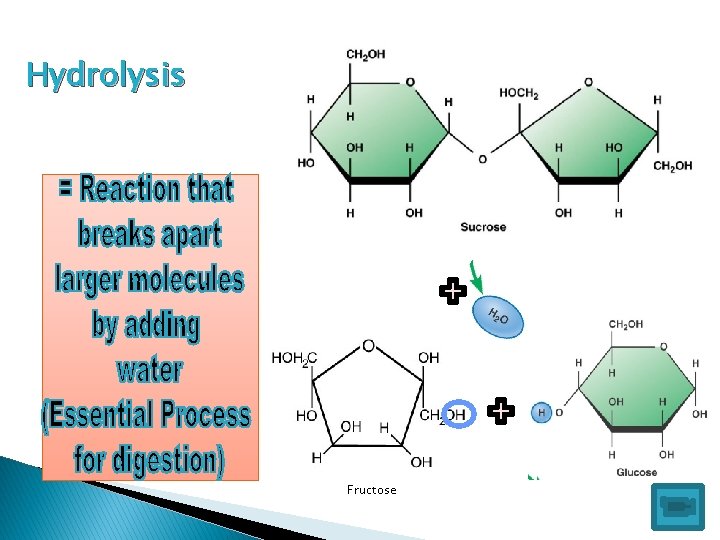
Hydrolysis Fructose

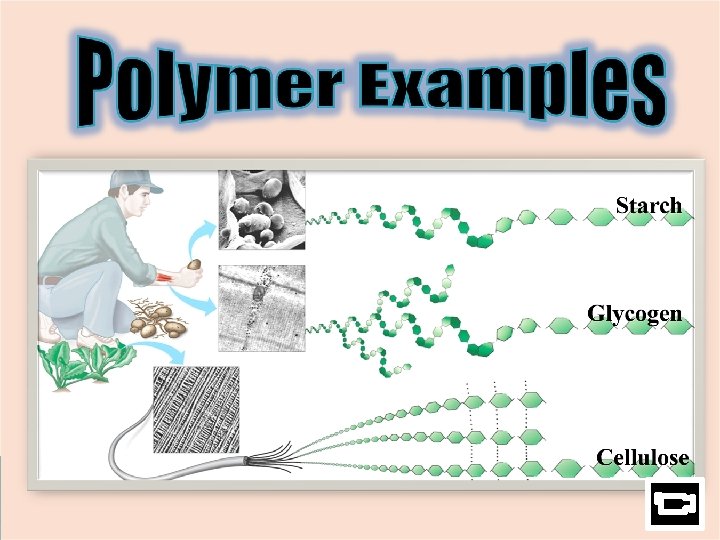
Polymer: ØIs *Polysaccharide a complex molecule made of 3 or more monosaccharides 3 Type s 1. starch = sugar storage in plants 2. glycogen = sugar storage in animals (in muscles) 3. cellulose = structural component to plant cells (cell wall) ! ! r e Fib
• Contain: C, H, O, and N • Function: 1. 2. Structural component to living things (helps build muscle mass) Control cell processes and reactions (enzymes)
• Monomer • Polymer = amino acid = protein (folded polypeptide chain)
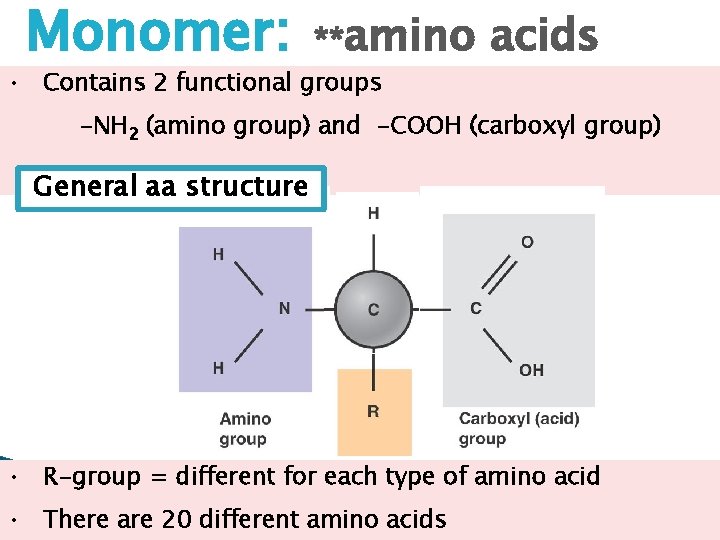
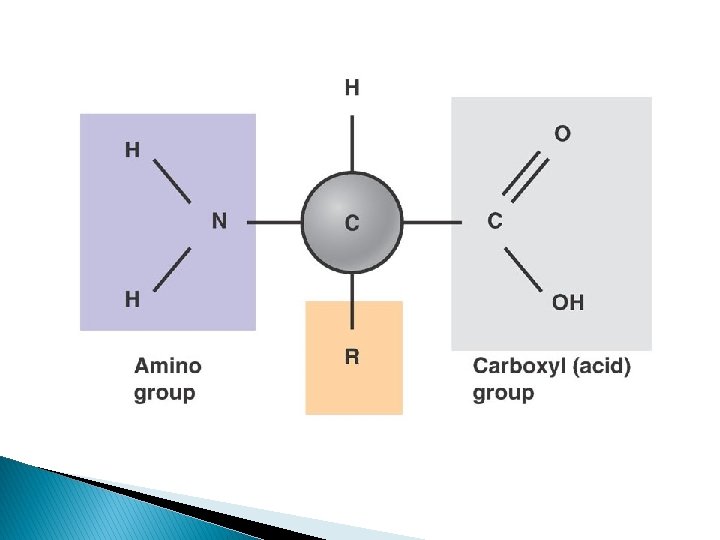
Monomer: **amino acids • Contains 2 functional groups -NH 2 (amino group) and -COOH (carboxyl group) General aa structure • R-group = different for each type of amino acid • There are 20 different amino acids
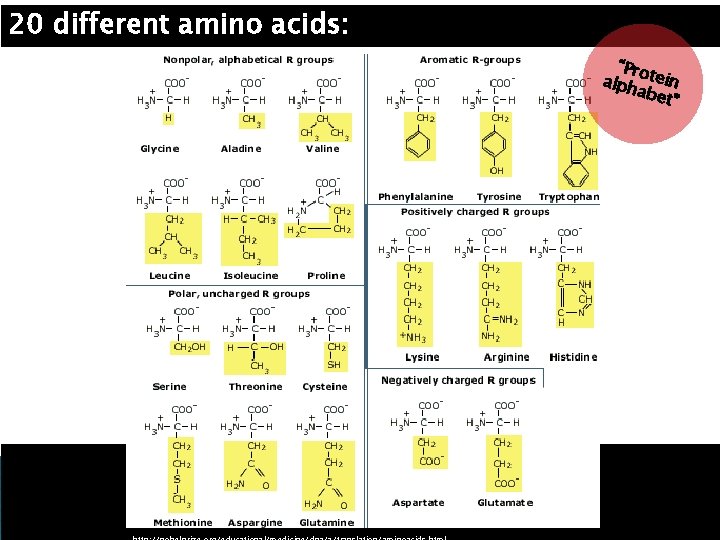
20 different amino acids: “Pro alph tein abet ”
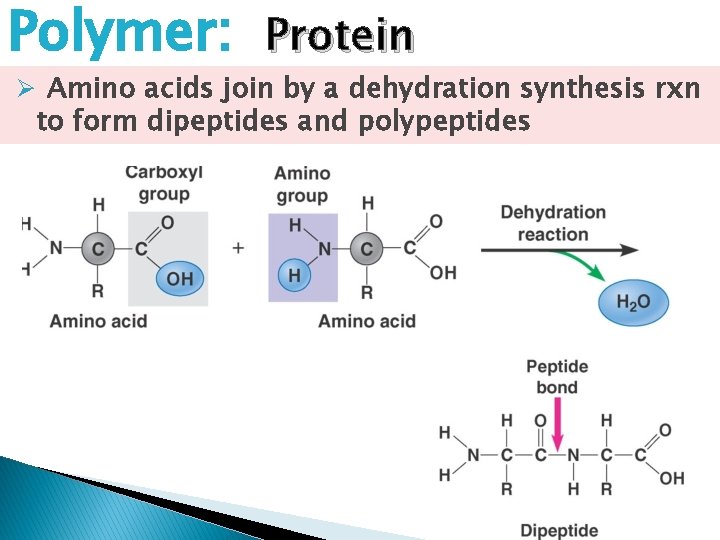
Polymer: Protein Ø Amino acids join by a dehydration synthesis rxn to form dipeptides and polypeptides

Primary Structure 1. Form a chain of aa Secondary Structure 2. Twist or fold the chain of aa (alpha helix or pleated sheet) Tertiary structure 3. Fold the chain onto itself (globular subunit) Quaternary Structure 4. Bring 2 to 4 aa subunits together
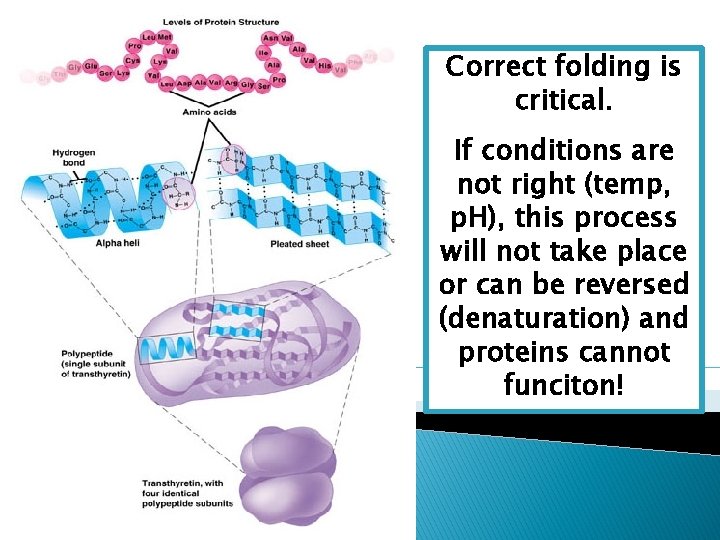
Correct folding is critical. If conditions are not right (temp, p. H), this process will not take place or can be reversed (denaturation) and proteins cannot funciton!

Enzymes are made of proteins. Ø They help speed up reactions and are UNCHANGED by the reaction. Ø Image from: http: //www. cas. muohio. edu/~wilsonkg/old/gene 2005/syllabus_F 03_23. jpg

• Contain: C, H, and O • Function: Depends on type http: //www. red-spirit-energy-healing. com/essential-fats. html

• Lipids are not typical monomers and polymers

• Most contain (“monomer”): Glycerol and fatty acid
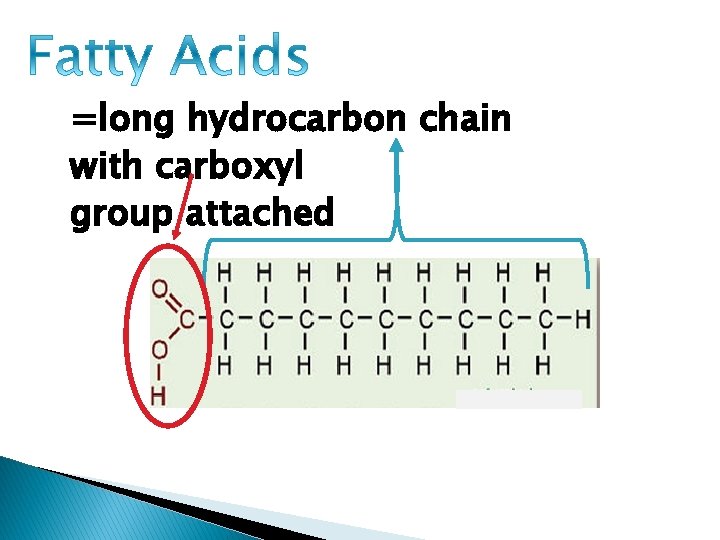
=long hydrocarbon chain with carboxyl group attached
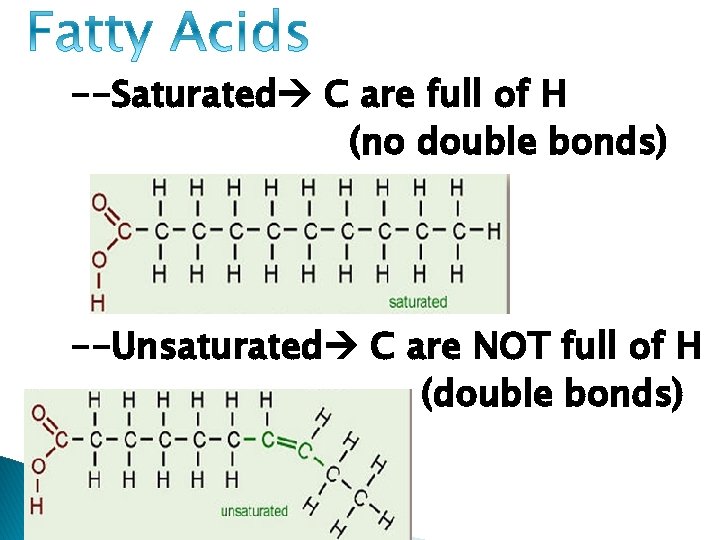
--Saturated C are full of H (no double bonds) --Unsaturated C are NOT full of H (double bonds)
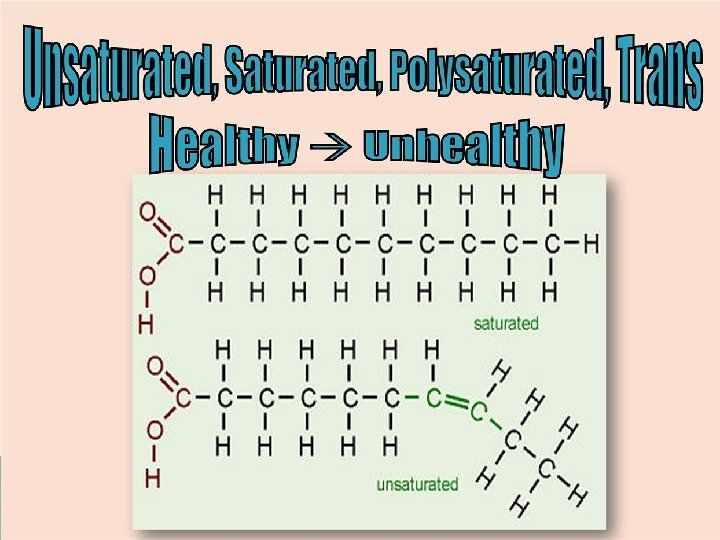
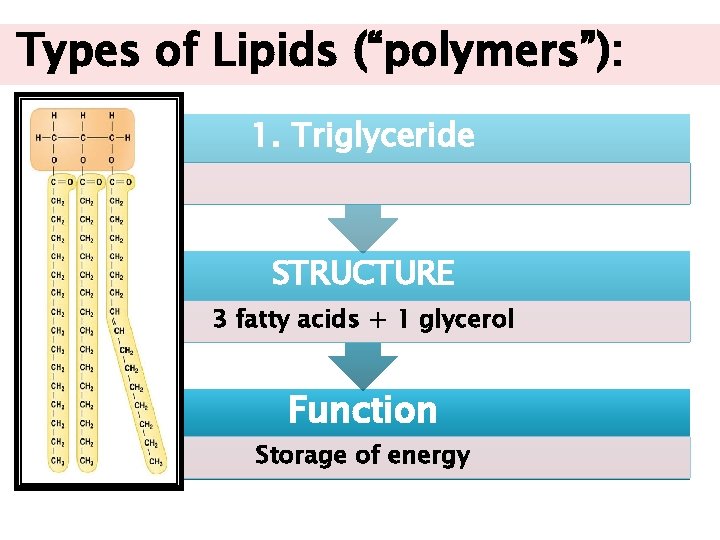
Types of Lipids (“polymers”): 1. Triglyceride STRUCTURE 3 fatty acids + 1 glycerol Function Storage of energy
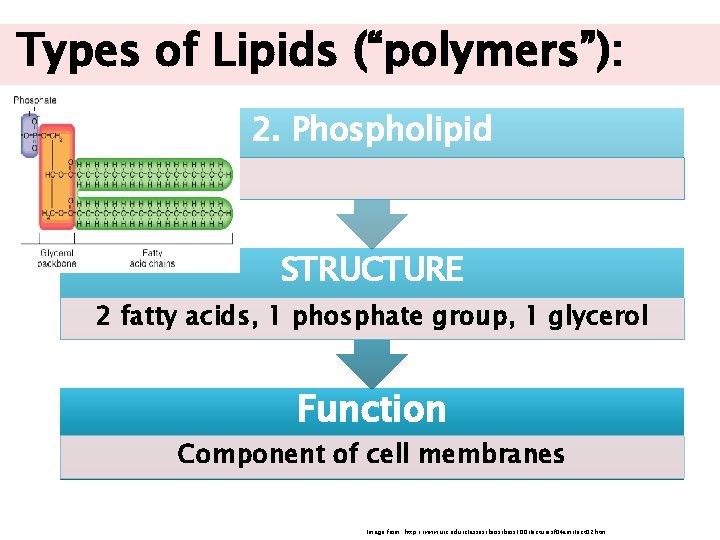
Types of Lipids (“polymers”): 2. Phospholipid STRUCTURE 2 fatty acids, 1 phosphate group, 1 glycerol Function Component of cell membranes Image from: http: //www. uic. edu/classes/bios 100/lecturesf 04 am/lect 02. htm
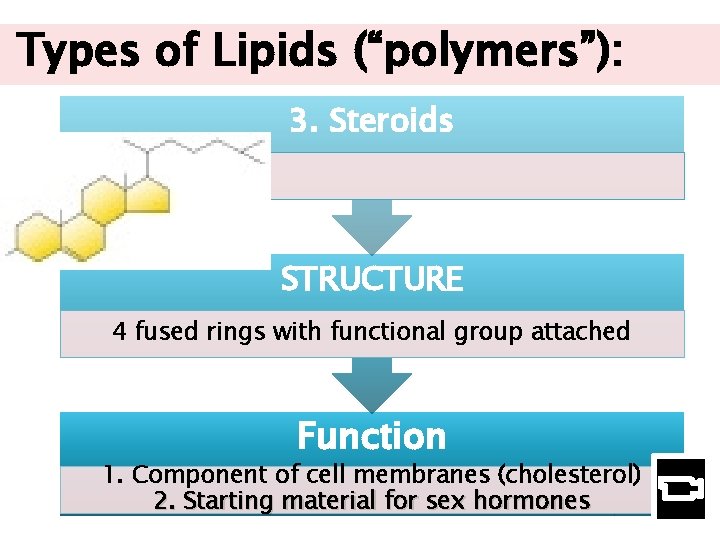
Types of Lipids (“polymers”): 3. Steroids STRUCTURE 4 fused rings with functional group attached Function http: //www. uic. edu/classes/bios 100/lectures f 04 am/lect 02. htm 1. Component of cell membranes (cholesterol) 2. Starting material for sex hormones

• Contain: C, H, O, N, P • Function: 1. Store and transmit info to carry out cell processes and make protein 2. Transmit genetic info from one generation to the next • Functional groups: None that we discussed
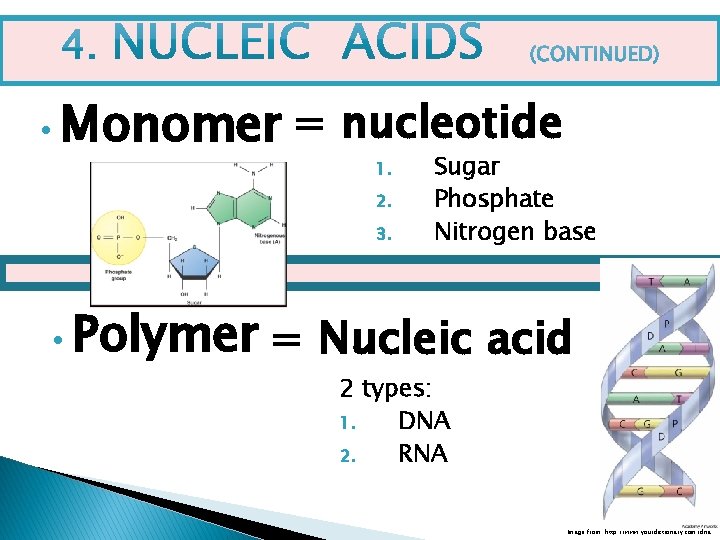
• Monomer = nucleotide 1. 2. 3. • Polymer Sugar Phosphate Nitrogen base = Nucleic acid 2 types: 1. DNA 2. RNA Image from: http: //www. yourdictionary. com/dna
- Slides: 39