Organic Chemistry Organic Chemistry Has Carbon 4 covalent

Organic Chemistry

Organic Chemistry Has Carbon 4 covalent bonds C has 4 valence e-’s Low M. P. tetrahedral Poor conductors Slow reactors Inorganic Organic Contains only small amount of Carbon (I. e. , carbides, oxides) Contains Carbon!!!! Ionic or Covalent bonds High MP, strong intermolecular forces EXCEPT CO 2 it’s inorganic (CHON) Hydrocarbons contain Carbon & Hydrogen ONLY!!!!!!
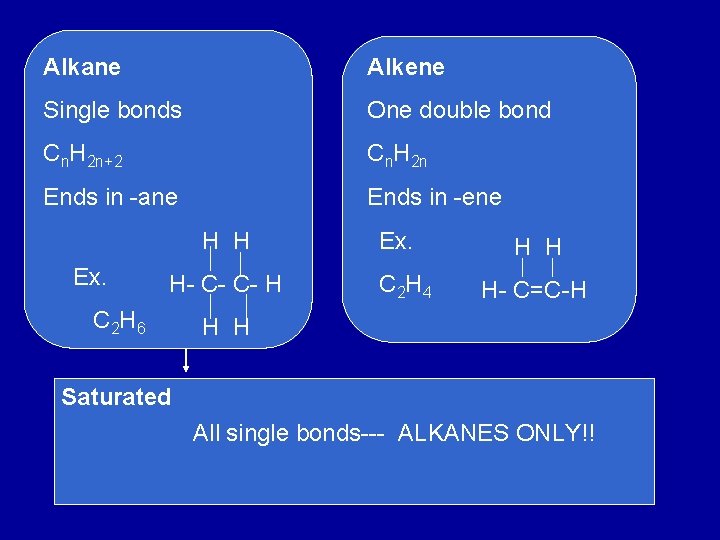
Alkane Alkene Single bonds One double bond Cn. H 2 n+2 Cn. H 2 n Ends in -ane Ends in -ene H H Ex. H- C- C- H C 2 H 6 Ex. C 2 H 4 H H H- C=C-H H H Saturated All single bonds--- ALKANES ONLY!!
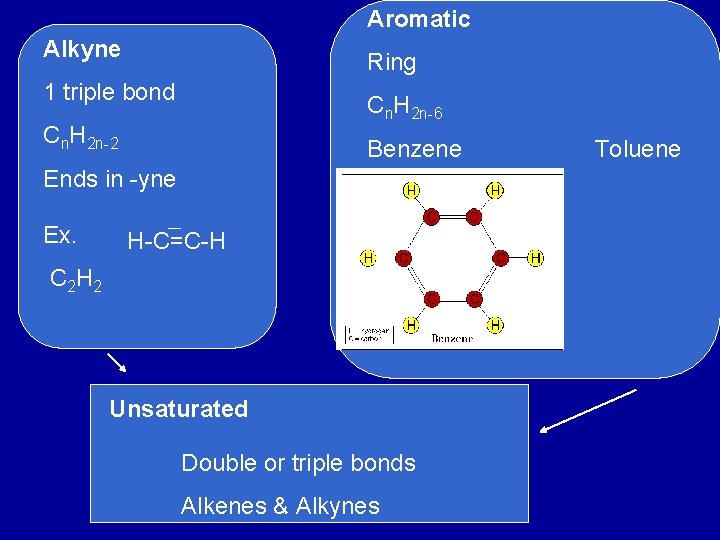
Aromatic Alkyne Ring 1 triple bond Cn. H 2 n-6 Cn. H 2 n-2 Benzene Ends in -yne Ex. H-C=C-H C 2 H 2 Unsaturated Double or triple bonds Alkenes & Alkynes Toluene

Naming Hydrocarbons Prefix 1 -meth 2 -eth Branches 6 -hex 7 -hept 3 -prop 8 -oct 4 -but 9 -non 5 -pent 10 -dec F-fluoro. Cl-chloro. Br-bromo. CH 3 - methyl CH 3 CH 2 - ethyl
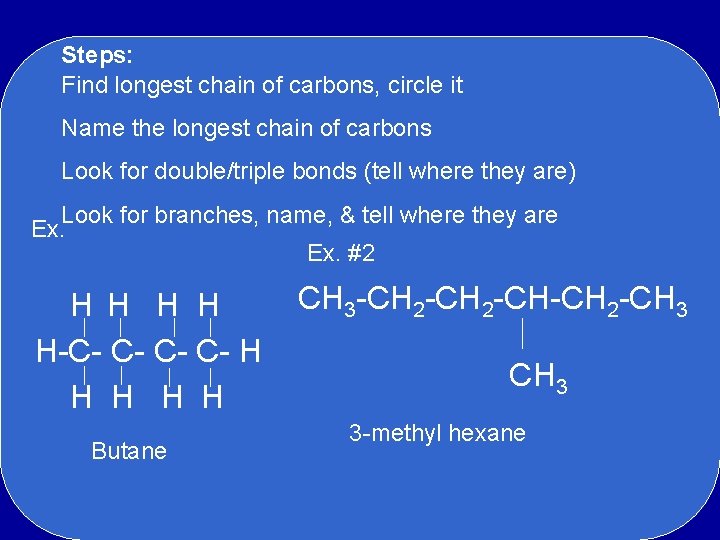
Steps: Find longest chain of carbons, circle it Name the longest chain of carbons Look for double/triple bonds (tell where they are) Look for branches, name, & tell where they are Ex. #2 H H H-C- C- H H H Butane CH 3 -CH 2 -CH-CH 2 -CH 3 3 -methyl hexane

Characteristics of Hydrocarbons: Contains only Carbon & Hydrogen Can be found in nature Can be straight chained or branched Soluble in nonpolar solvents Non polar Low M. P. Non electrolytes Carbon has 4 valence e-’s, so it makes 4 covalent bonds

Isomers Same molecular formula Different structural formula Count up the atoms, put in different order C 5 H 12 -C-C-C- C 5 H 12 -C-CC
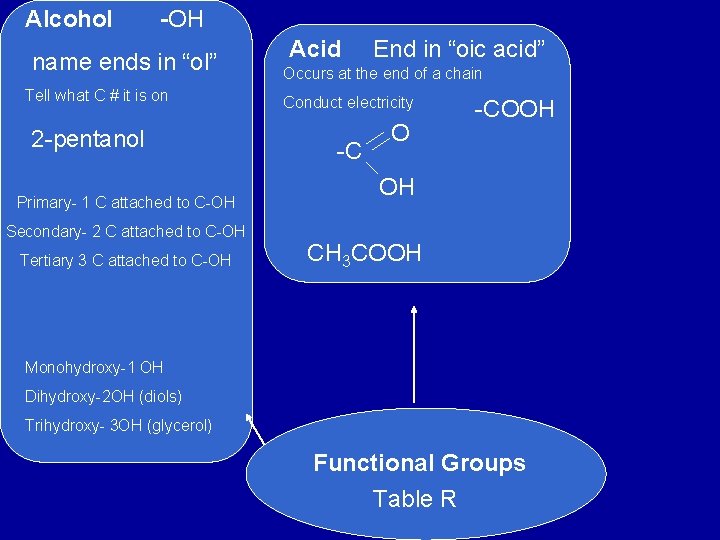
Alcohol -OH name ends in “ol” Tell what C # it is on 2 -pentanol Primary- 1 C attached to C-OH Secondary- 2 C attached to C-OH Tertiary 3 C attached to C-OH Acid End in “oic acid” Occurs at the end of a chain Conduct electricity -C O -COOH OH CH 3 COOH Monohydroxy-1 OH Dihydroxy-2 OH (diols) Trihydroxy- 3 OH (glycerol) Functional Groups Table R
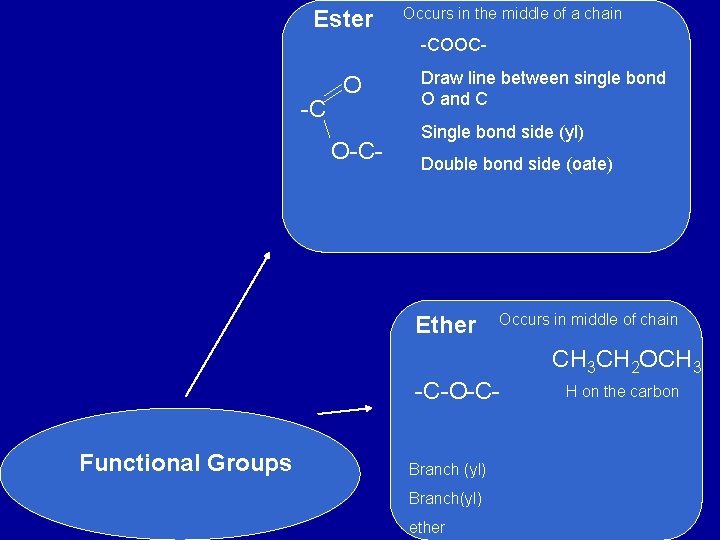
Ester Occurs in the middle of a chain -COOC- -C O O-C- Draw line between single bond O and C Single bond side (yl) Double bond side (oate) Ether Occurs in middle of chain CH 3 CH 2 OCH 3 -C-O-CFunctional Groups Branch (yl) Branch(yl) ether H on the carbon
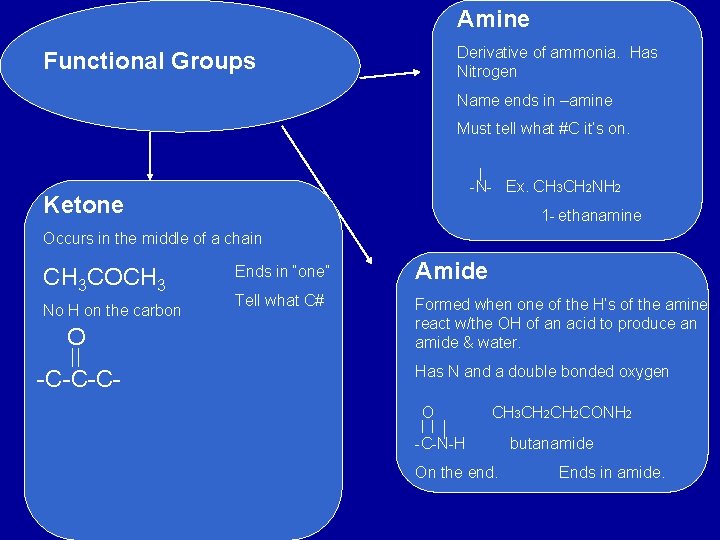
Amine Derivative of ammonia. Has Nitrogen Functional Groups Name ends in –amine Must tell what #C it’s on. -N- Ex. CH 3 CH 2 NH 2 Ketone 1 - ethanamine Occurs in the middle of a chain CH 3 COCH 3 No H on the carbon O -C-C-C- Ends in “one” Amide Tell what C# Formed when one of the H’s of the amine react w/the OH of an acid to produce an amide & water. Has N and a double bonded oxygen O CH 3 CH 2 CONH 2 -C-N-H On the end. butanamide Ends in amide.
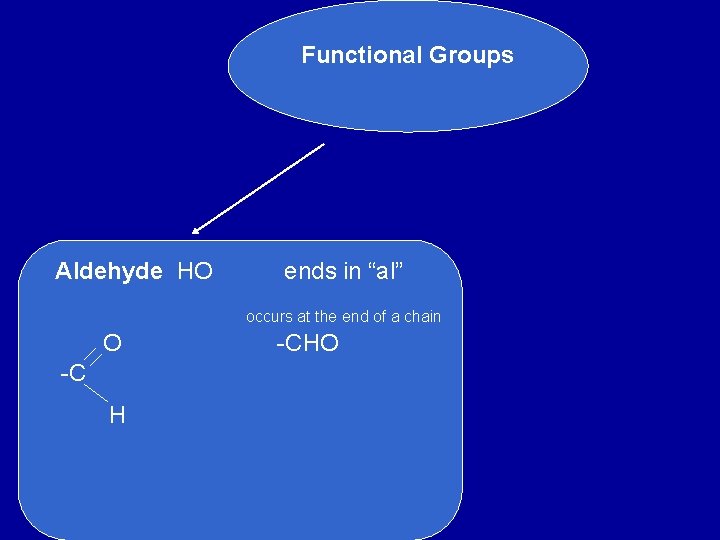
Functional Groups Aldehyde HO ends in “al” occurs at the end of a chain O -C H -CHO
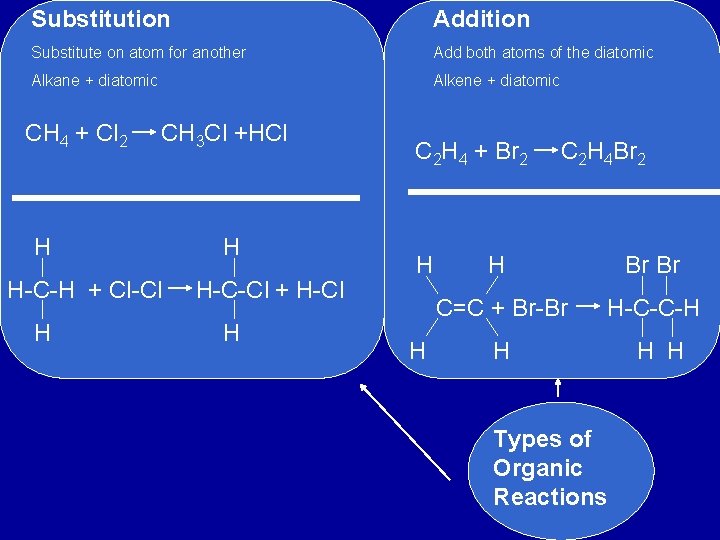
Substitution Addition Substitute on atom for another Add both atoms of the diatomic Alkane + diatomic Alkene + diatomic CH 4 + Cl 2 CH 3 Cl +HCl H H-C-H + Cl-Cl H H H-C-Cl + H-Cl H C 2 H 4 + Br 2 H H C 2 H 4 Br 2 H Br Br C=C + Br-Br H-C-C-H H Types of Organic Reactions
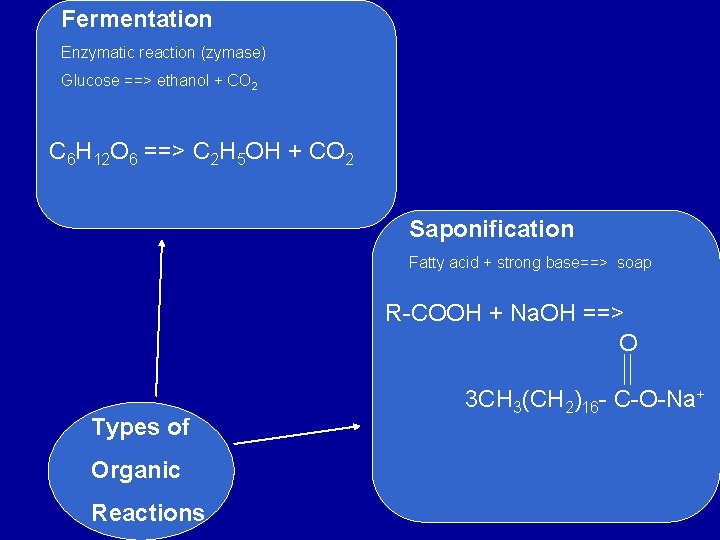
Fermentation Enzymatic reaction (zymase) Glucose ==> ethanol + CO 2 C 6 H 12 O 6 ==> C 2 H 5 OH + CO 2 Saponification Fatty acid + strong base==> soap R-COOH + Na. OH ==> O Types of Organic Reactions 3 CH 3(CH 2)16 - C-O-Na+
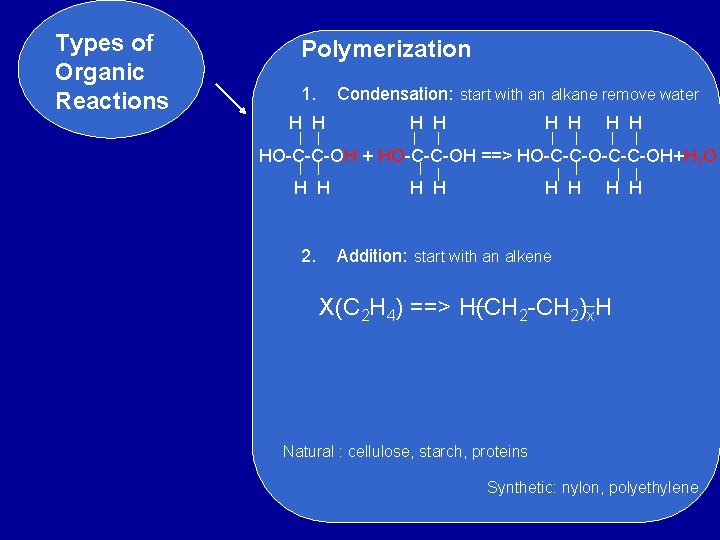
Types of Organic Reactions Polymerization 1. Condensation: start with an alkane remove water H H H H HO-C-C-OH + HO-C-C-OH ==> HO-C-C-OH+H 2 O H H 2. H H H Addition: start with an alkene X(C 2 H 4) ==> H(CH 2 -CH 2)x. H Natural : cellulose, starch, proteins Synthetic: nylon, polyethylene
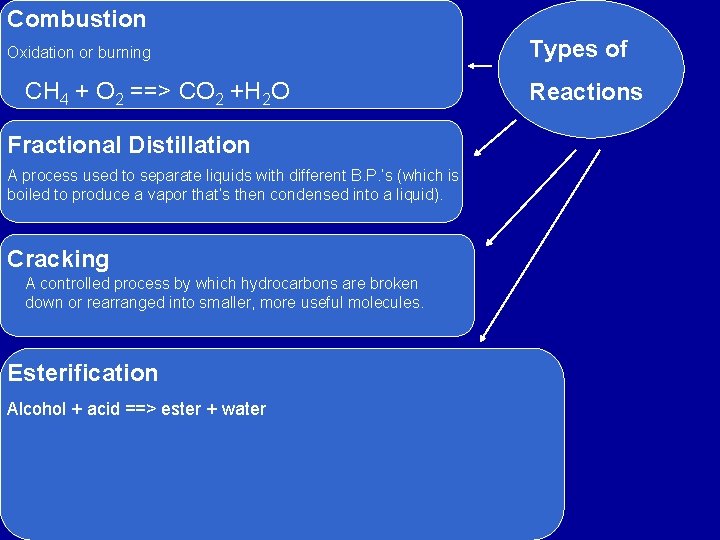
Combustion Oxidation or burning CH 4 + O 2 ==> CO 2 +H 2 O Fractional Distillation A process used to separate liquids with different B. P. ’s (which is boiled to produce a vapor that’s then condensed into a liquid). Cracking A controlled process by which hydrocarbons are broken down or rearranged into smaller, more useful molecules. Esterification Alcohol + acid ==> ester + water Types of Reactions
- Slides: 16