Organic Chemistry of Carbon Compounds except oxides carbides

Organic Chemistry of Carbon Compounds (except oxides, carbides, & carbonates)

Bonding Capacity • • H can form only 1 bond O and S like to form 2 bonds The halogens (F, Cl, Br, I) form only 1 bond N and P form 3 bonds

Why so many C compounds? • Carbon atoms can bond with other carbon atoms in chains, rings, and networks • Bonds are covalent

Lewis Diagram of C • • Carbon has 4 unpaired electrons so it can form 4 covalent bonds

Properties of Covalent Substances • • • (also called molecular substances) Low melting & boiling points Poor conductors of heat & electricity May be soft or brittle Generally nonpolar – van der Waals forces Tend to dissolve in nonpolar solvents React more slowly than ionic compounds

Chemical Formulas • show kind & number of atoms CH 3 OH CH 4 CH 2 Cl 2 CH 3 Cl

Structural Formulas • Show kind & number of atoms • Also show bonding patterns and approximate shapes of molecules • 2 -D rep of 3 -D object so structural formulas aren’t totally realistic H H–C–H H

Structural Formulas • – A single line represents one pair of electrons (a single bond) • = A double line represents two pairs of electrons (a double bond) ● A triple line represents three pairs of electrons (a triple bond)

Shape • 4 unpaired electrons around C atom located at the corners of a tetrahedron • 109. 5 apart

Vocabulary • Hydrocarbons: Hydrocarbons organic compounds containing only C and H • Homologous Series: Series – group of compounds with related structures and properties – each member of series differs from the one before it by adding one more unit

Vocabulary Interlude • Saturated: Saturated organic compounds containing only Single bonds • Unsaturated: Unsaturated organic compounds containing one or more double or triple bonds

Homologous Series • The molecules have a fixed, numerical relationship among the numbers of atoms

Condensed Structural Formula • Shows kind & number of atoms • Shows some structural information, but not all the details structural formula: H H–C–C–C–H H becomes: CH 3 CH 2 CH 3

ALKANES Cn. H 2 n+2

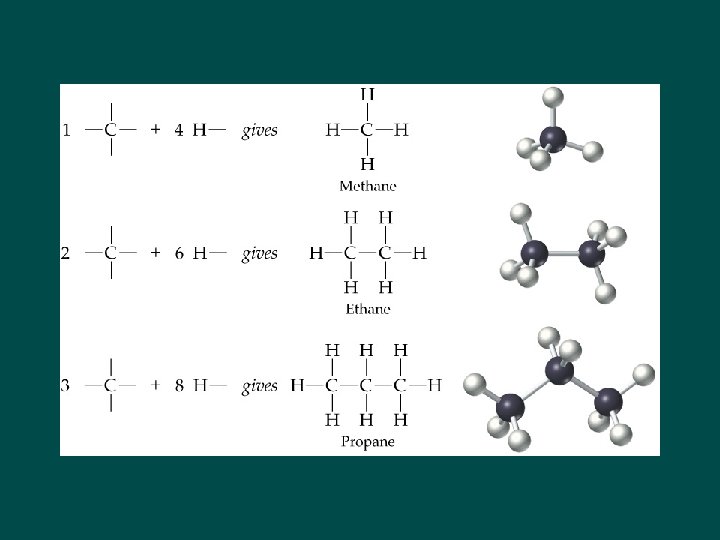
Alkanes • Homologous series of saturated hydrocarbons • Release energy when burned CH 4 C 2 H 6 H H–C–H H H–C–C–H H H
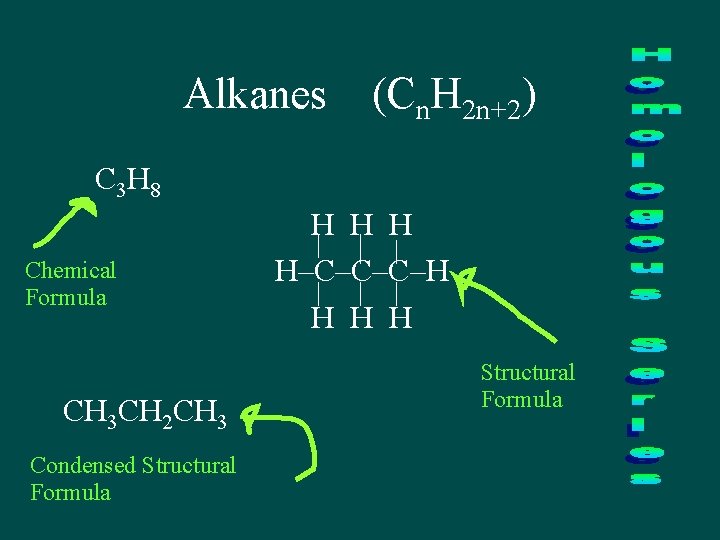
Alkanes (Cn. H 2 n+2) C 3 H 8 Chemical Formula CH 3 CH 2 CH 3 Condensed Structural Formula H H–C–C–C–H H Structural Formula
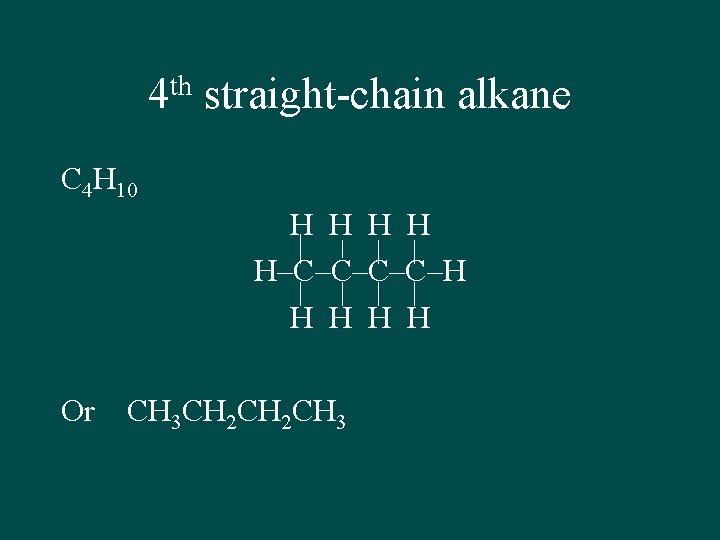
th 4 straight-chain alkane C 4 H 10 H H H–C–C–H H H Or CH 3 CH 2 CH 3

Alkanes: base unit CH 4 CH 3 or C 2 H 6 CH 3 CH 2 CH 3 or C 3 H 8 CH 3 CH 2 CH 3 or C 4 H 10 CH 3 CH 2 CH 2 CH 3 or C 5 H 12 Difference between each is CH 2

Naming straight-chain Alkanes • Name describes molecule so you can draw it • All alkanes ane have the suffix –ane • The prefix depends on the number of C’s
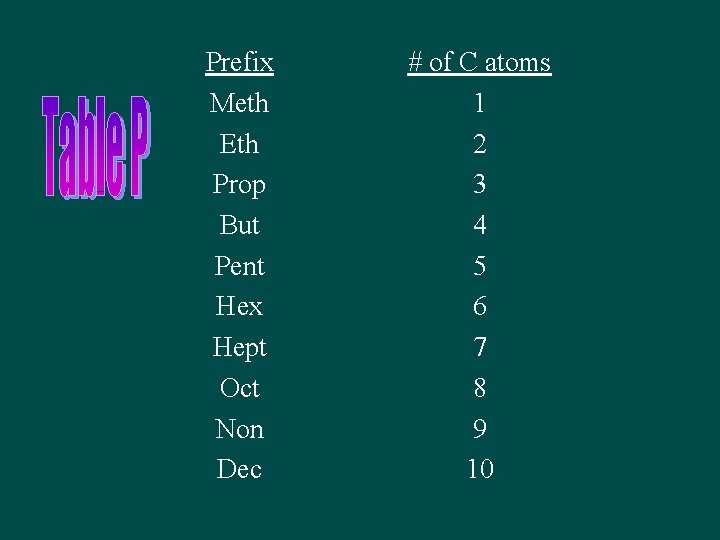
Prefix Meth Eth Prop But Pent Hex Hept Oct Non Dec # of C atoms 1 2 3 4 5 6 7 8 9 10
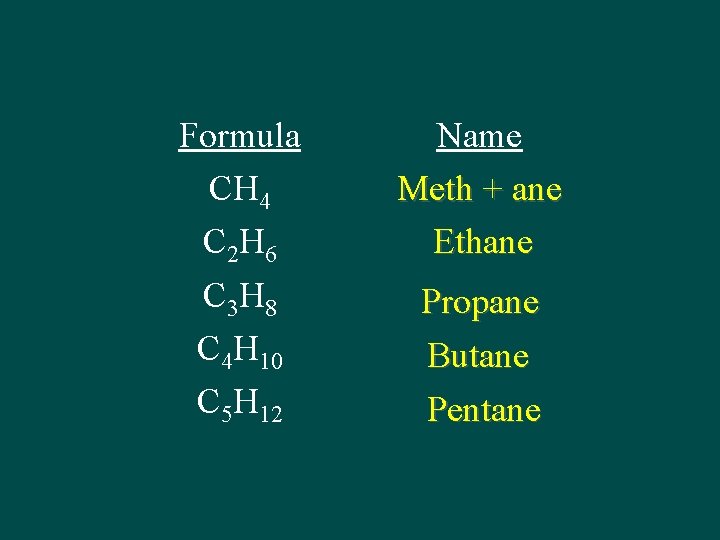
Formula CH 4 C 2 H 6 C 3 H 8 C 4 H 10 C 5 H 12 Name Meth + ane Ethane Propane Butane Pentane

Properties of Alkanes • Change systematically with number of C’s • As the number of C’s increases, the boiling point increases – The molecules get heavier & harder to change into the gas phase

Properties of Alkanes • • • Low Reactivity Except readily undergo combustion – fuels Nonpolar – Don’t dissolve well in water Low melting pts & boiling pts. (M. P. & B. P. increase with molecular mass) High vapor pressures

Which property is generally characteristic of an organic compound? A. B. C. D. Low melting point High melting point Soluble in polar solvents Insoluble in nonpolar solvents Correct response = A

Which of the following compounds has the highest boiling point? A. B. C. D. CH 4 C 2 H 6 C 3 H 8 C 4 H 10 Correct answer = D nonpolar coval cmpd: Bp depends on the strength of the van der Waals interactions (van der Waals forces ↑ as size of molecule ↑ - bigger molecule = bigger electron cloud)

Branched-chain alkanes • Beginning with butane, C 4 H 10, there is more than 1 way to arrange the atoms H H–C–H H H H–C–C–C–H H

Branched alkane: can’t link all the C’s without lifting pencil off paper Methyl propane

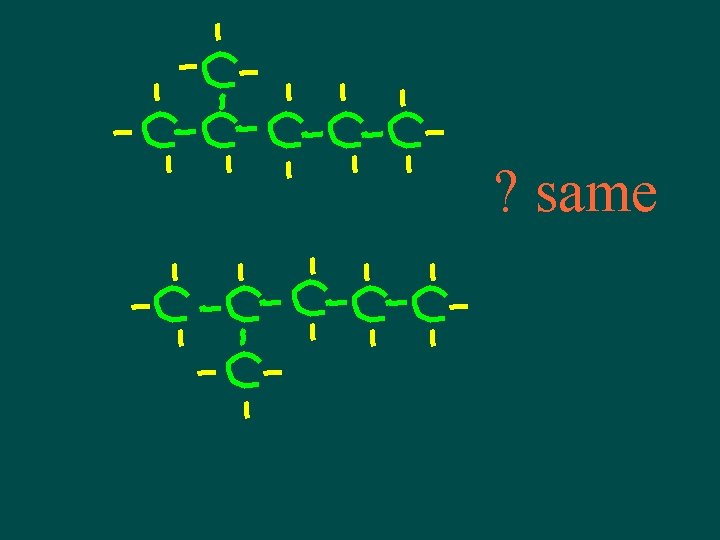
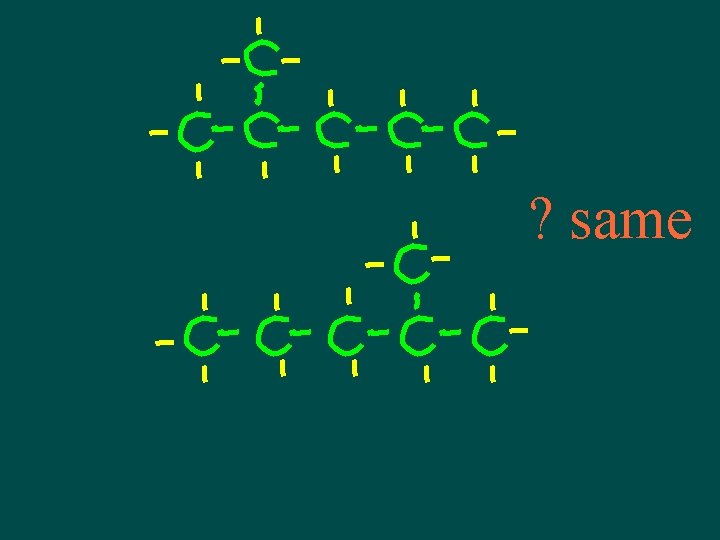
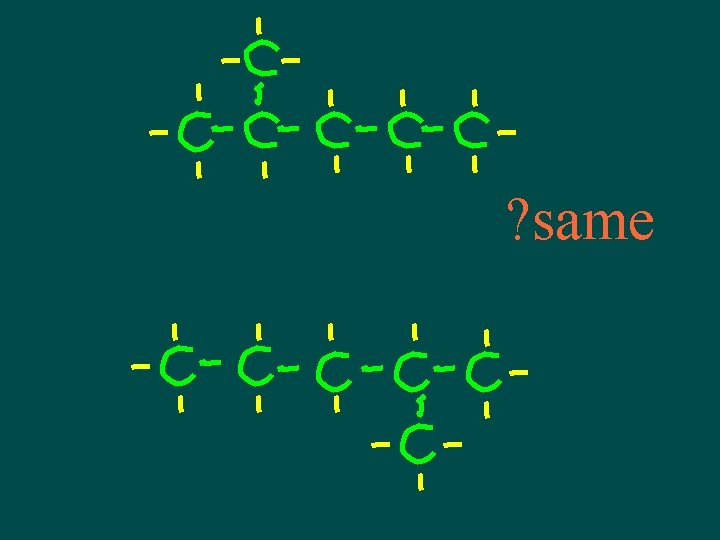
Isomers • Compounds with same molecular formula but different structural arrangement • The more C atoms there are the more isomers can have more possible ways to arrange them

Isomers • Different structures, different properties • Isomers have different chemical and physical properties

A note about isomers -- • If comparing 2 structural formulas & can superimpose them, they are not isomers – they are the same molecule! • If can rotate or flip one of the structural formulas & then superimpose it on the other one, they are not isomers – they are the same molecule!
? same
? same
? same
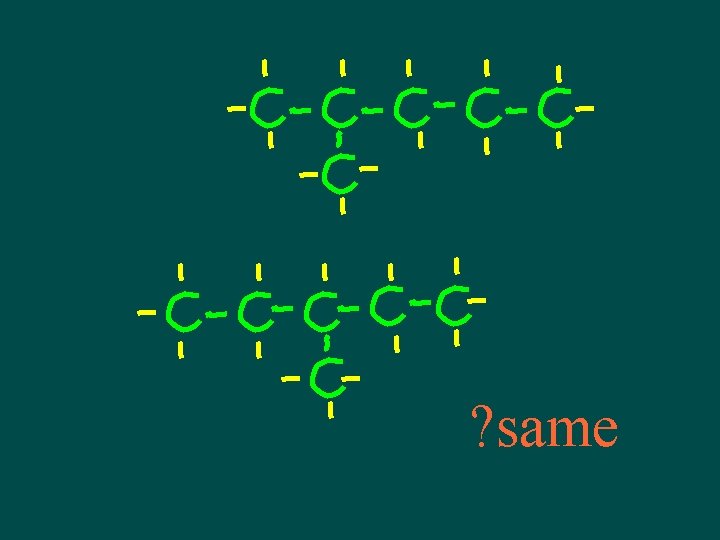
? same

Naming Branched-Chain Alkanes • The goal of the name is to describe the molecule so you can draw it

Naming branched-chain alkanes • Find longest continuous chain of C atoms – Bends don’t count! • base name derived from # of C’s in continuous chain • Branches named first – count # C atoms & add “yl” at end • # location of branch by assigning # to C branch is attached to in backbone – # C's so get lowest number for branch • more than one of same type of branch: Use di, tri, etc.
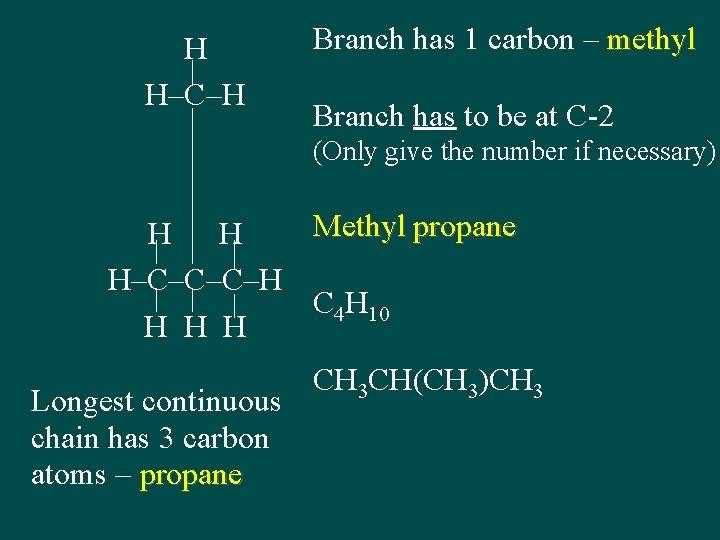
H H–C–H Branch has 1 carbon – methyl Branch has to be at C-2 (Only give the number if necessary) Methyl propane H H H–C–C–C–H C 4 H 10 H H H Longest continuous chain has 3 carbon atoms – propane CH 3 CH(CH 3)CH 3
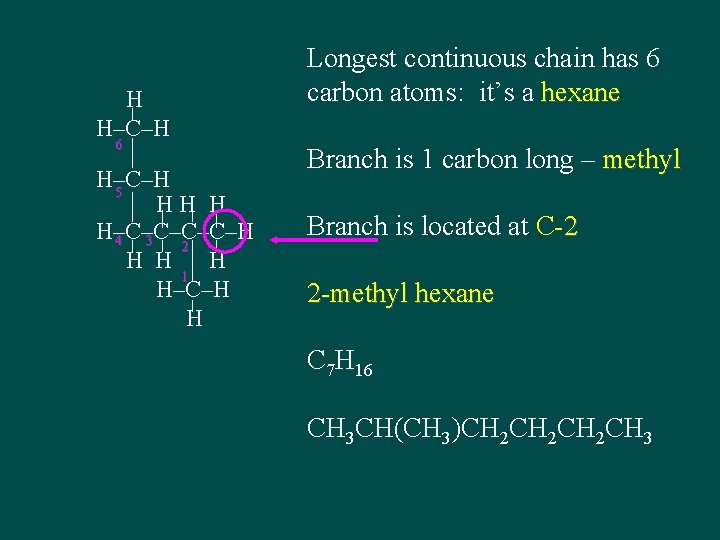
H H–C–H 6 H–C–H 5 HH H H–C–C–H 4 3 2 H H H 1 H–C–H H Longest continuous chain has 6 carbon atoms: it’s a hexane Branch is 1 carbon long – methyl Branch is located at C-2 2 -methyl hexane C 7 H 16 CH 3 CH(CH 3)CH 2 CH 2 CH 3

A note about branches • If you have several branches with the same # of C atoms, you can condense the name a little • 2 -methyl 3 -methyl pentane becomes: – 2, 3 -dimethyl pentane
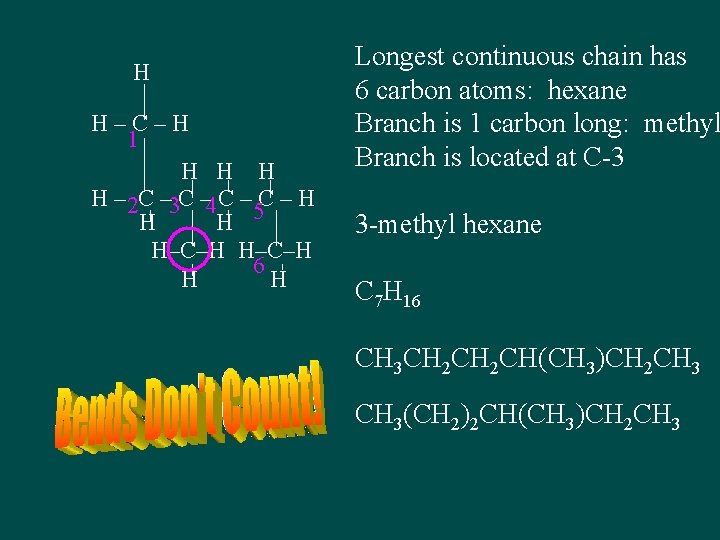
H H–C–H 1 H H – 2 C – 3 C – 4 C – H H H 5 H–C–H 6 H H Longest continuous chain has 6 carbon atoms: hexane Branch is 1 carbon long: methyl Branch is located at C-3 3 -methyl hexane C 7 H 16 CH 3 CH 2 CH(CH 3)CH 2 CH 3(CH 2)2 CH(CH 3)CH 2 CH 3

Alkenes • Another homologous series of hydrocarbons • Each member contains at least one double covalent bond between C atoms So alkenes are unsaturated • General formula = Cn. H 2 n
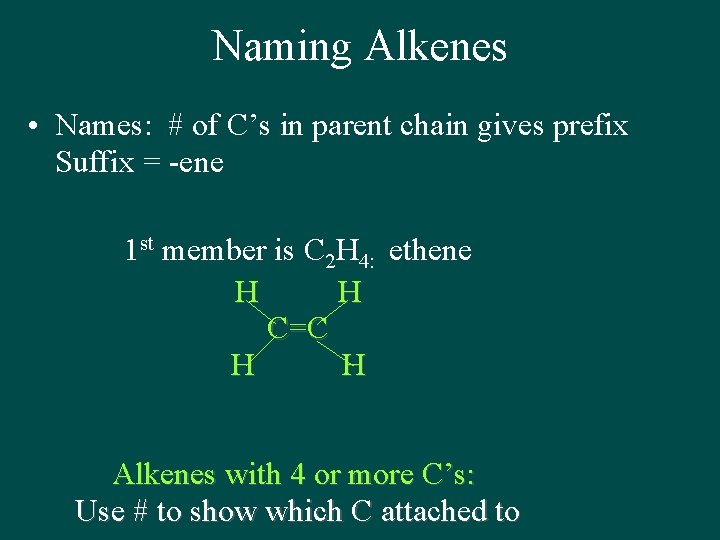
Naming Alkenes • Names: # of C’s in parent chain gives prefix Suffix = -ene 1 st member is C 2 H 4: ethene H H C=C H H Alkenes with 4 or more C’s: Use # to show which C attached to

Naming Alkenes • Number C atoms in backbone & give double bond the lowest possible number 1 -butene H HH C=C–C–C–H H H 2 -butene H H HH H–C–C=C–C–H H H C 4 H 8 CH 2 CH 3 CHCHCH 3

Naming Branched-Chain Alkenes • parent chain = longest chain that contains the double bond • position of double bond, not branches, determines numbering of backbone • Give 1 st C in bond lowest possible #

Properties of Alkenes • Nonpolar – low solubility in H 2 O • Fairly low M. P. & B. P. • More reactive than alkanes : – double bond = site of reactivity

Alkynes • Homologous series of unsaturated hydrocarbons that contain one triple bond • Each member contains one C≡C bond – Alkynes are unsaturated • General formula = Cn. H 2 n-2

Naming Alkynes • Name: Prefix depends on # of C’s – Suffix = yne • If necessary, number 1 st carbon atom at which the triple bond occurs
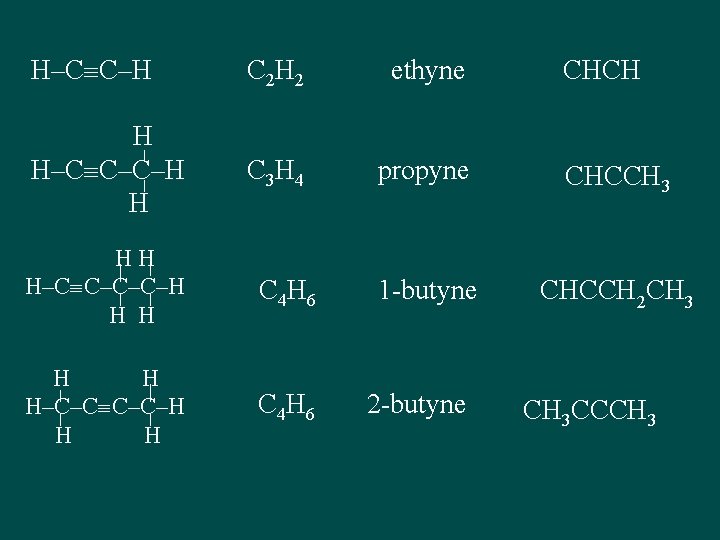
H–C C–H C 2 H 2 ethyne H H–C C–C–H H C 3 H 4 propyne CHCCH 3 1 -butyne CHCCH 2 CH 3 HH H–C C–C–C–H H H C 4 H 6 H H H–C–C C–C–H H H C 4 H 6 2 -butyne CHCH CH 3 CCCH 3

3 Homologous Series of HC’s Name of Series General Formula Ending Alkanes Cn. H 2 n+2 -ane Alkenes Cn. H 2 n -ene Alkynes Cn. H 2 n-2 -yne

Which compound belongs to the alkene series? A. B. C. D. C 2 H 2 C 2 H 4 C 6 H 6 C 6 H 14 Correct answer = B. Alkenes follow the format Cn. H 2 n A & C are Cn. Hn , D is Cn. H 2 n+2

In which group could the hydrocarbons all belong to the same homologous series? A. B. C. D. C 2 H 2, C 2 H 4, C 2 H 6 C 2 H 4, C 3 H 4, C 4 H 8 C 2 H 4, C 2 H 6, C 3 H 6 C 2 H 4, C 3 H 6, C 4 H 8 Correct answer = D Members of homologous series all have same relationship between atoms Every compound in set D fits the formula Cn. H 2 n

Which of the following is a saturated hydrocarbon? A. B. C. D. Ethene Ethyne Propene Propane Correct answer = D The alkanes are saturated
- Slides: 54