Organic Chemistry Nomenclature Alkanes Alkanes Hydrocarbon chains where
Organic Chemistry Nomenclature: Alkanes
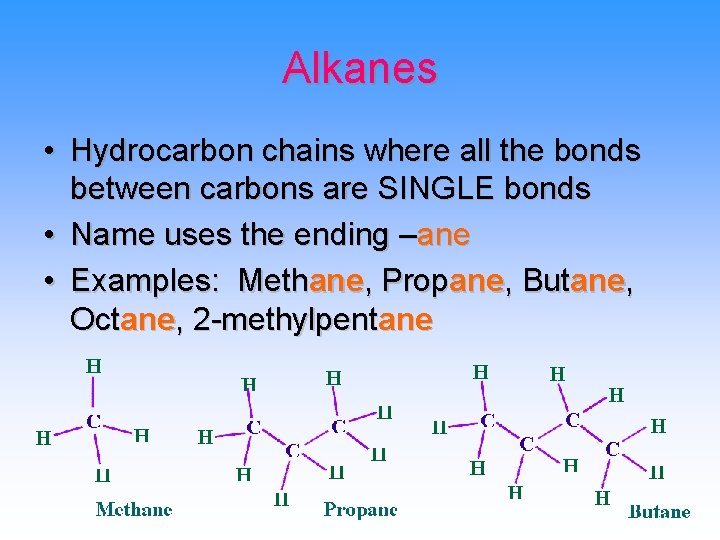
Alkanes • Hydrocarbon chains where all the bonds between carbons are SINGLE bonds • Name uses the ending –ane • Examples: Methane, Propane, Butane, Octane, 2 -methylpentane

Summary: IUPAC Rules for Alkane Nomenclature 1. Find and name the longest continuous carbon chain. This is called the parent chain. (Examples: methane, propane, etc. ) 2. Number the chain consecutively, starting at the end nearest an attached group (substituent). 3. Identify and name groups attached to this chain. (Examples: methyl-, bromo-, etc. ) 4. Designate the location of each substituent group with the number of the carbon parent chain on which the group is attached. Place a dash between numbers and letters. (Example: 3 -chloropentane) 5. Assemble the name, listing groups in alphabetical order. The prefixes di, tri, tetra etc. , used to designate several groups of the same kind, are not considered when alphabetizing. Place a comma between multiple numbers. (Example: 2, 3 -dichloropropane)

Step 1. Find the parent chain. • Where is the longest continuous chain of carbons?
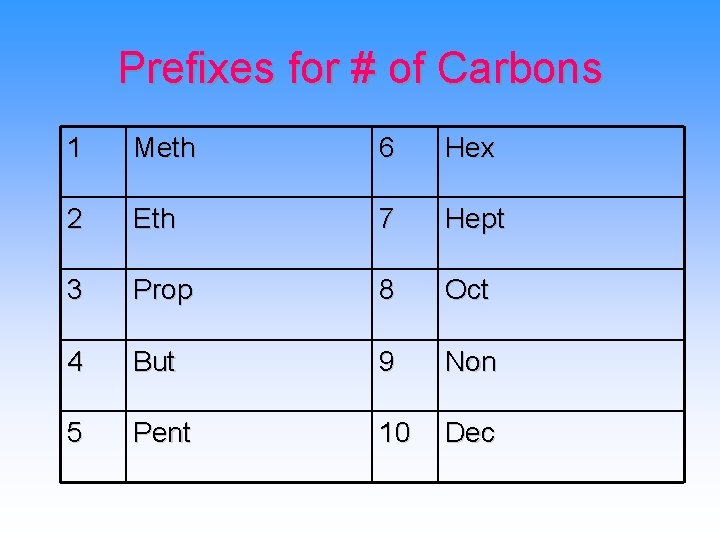
Prefixes for # of Carbons 1 Meth 6 Hex 2 Eth 7 Hept 3 Prop 8 Oct 4 But 9 Non 5 Pent 10 Dec
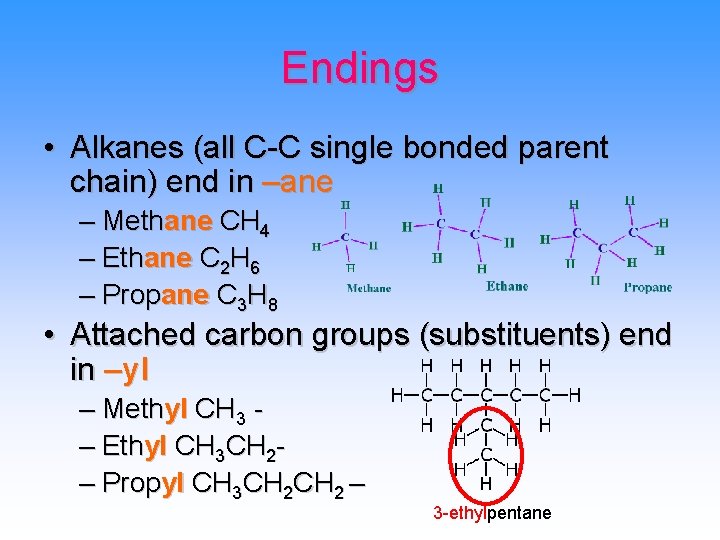
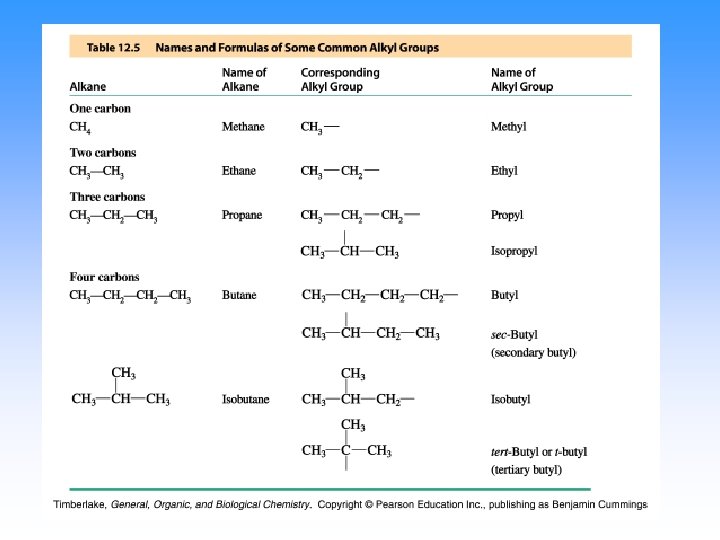
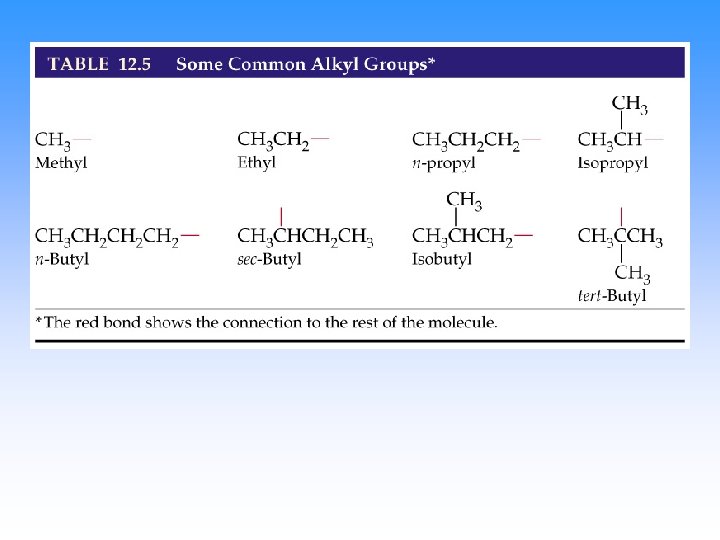
Endings • Alkanes (all C-C single bonded parent chain) end in –ane – Methane CH 4 – Ethane C 2 H 6 – Propane C 3 H 8 • Attached carbon groups (substituents) end in –yl – Methyl CH 3 – Ethyl CH 3 CH 2– Propyl CH 3 CH 2 – 3 -ethylpentane
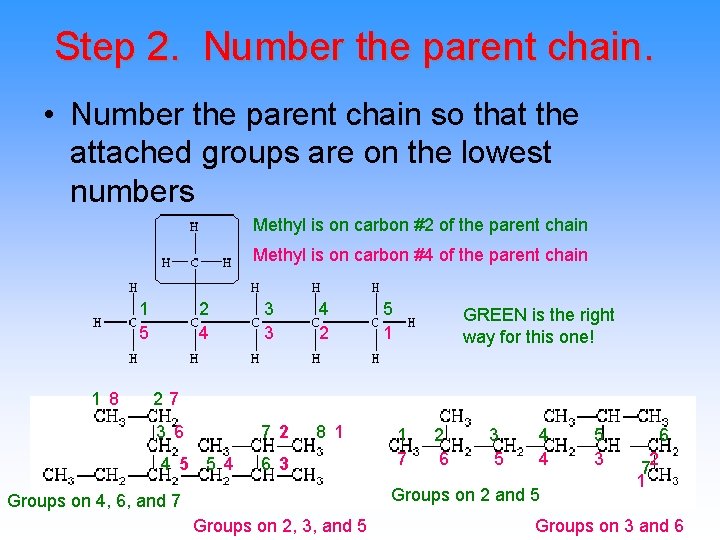
Step 2. Number the parent chain. • Number the parent chain so that the attached groups are on the lowest numbers Methyl is on carbon #2 of the parent chain Methyl is on carbon #4 of the parent chain 1 5 1 8 2 4 3 3 4 2 3 6 7 2 8 1 4 5 5 4 6 3 5 1 GREEN is the right way for this one! 27 1 7 2 6 3 5 4 4 Groups on 2 and 5 Groups on 4, 6, and 7 Groups on 2, 3, and 5 5 3 6 72 1 Groups on 3 and 6

Step 3. Name the attached groups. • Carbon (alkyl) groups – Methyl CH 3 – Ethyl CH 3 CH 2– Propyl CH 3 CH 2 – • Halogens – Fluoro (F-) – Chloro (Cl-) – Bromo (Br-) – Iodo (I-)
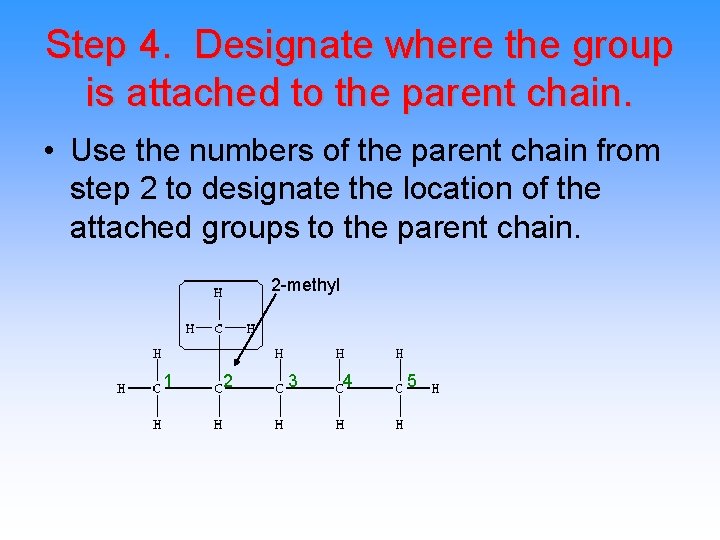
Step 4. Designate where the group is attached to the parent chain. • Use the numbers of the parent chain from step 2 to designate the location of the attached groups to the parent chain. 2 -methyl 1 2 3 4 5
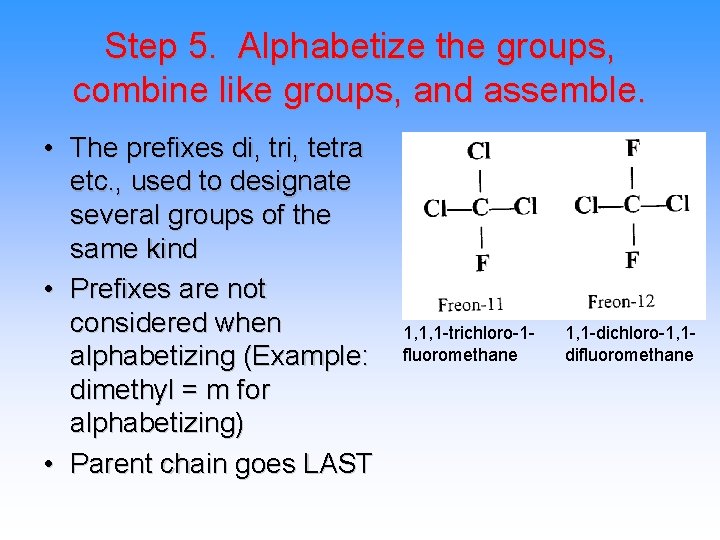
Step 5. Alphabetize the groups, combine like groups, and assemble. • The prefixes di, tri, tetra etc. , used to designate several groups of the same kind • Prefixes are not considered when alphabetizing (Example: dimethyl = m for alphabetizing) • Parent chain goes LAST 1, 1, 1 -trichloro-1 fluoromethane 1, 1 -dichloro-1, 1 difluoromethane

Draw Some Simple Alkanes • 2 -methylpentane • 3 -ethylhexane • 2, 2 -dimethylbutane • 2, 3 -dimethylbutane

Structural Formulas • “Lazy” way to write the Hydrogens • Instead of drawing the bonds, just state how many hydrogens are attached • NOTE: The bonds are between CARBONS in a parent chain, and not hydrogens! Structural Formula Lewis Structure
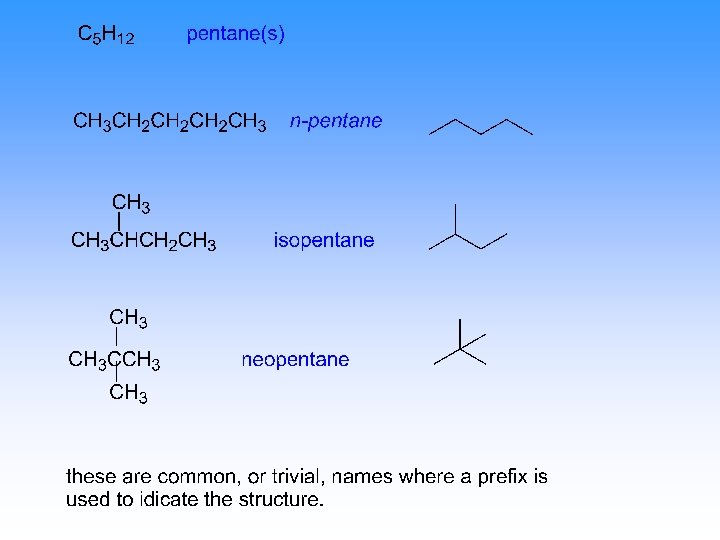

hexanes C 6 H 14 common names CH 3 CH 2 CH 2 CH 3 n-hexane CH 3 CH 2 CH 3 ? ? ? CH 3 CHCHCH 3 ? ? CH 3 CHCH 2 CH 3 isohexane CH 3 CCH 2 CH 3 neohexane

IUPAC nomenclature (Geneva, 1920) names of radicals (alkyl groups): CH 3 - “methyl” CH 3 Cl methyl chloride CH 3 OH methyl alcohol, etc. CH 3 CH 2 - “ethyl” CH 3 CH 2 - “n-propyl” CH 3 CHCH 3 | “isopropyl”

CH 3 CH 2 CH 2 - CH 3 CH 2 CHCH 3 | CH 3 CHCH 2 CH 3 CCH 3 | “n-butyl” or CH 3 CH 2 CH- “isobutyl” “tert-butyl” “sec-butyl”
Alkanes
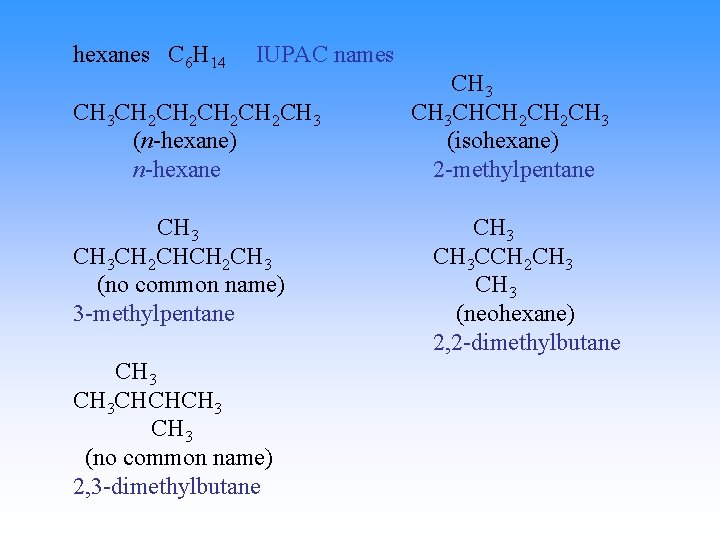
hexanes C 6 H 14 IUPAC names CH 3 CH 2 CH 2 CH 3 (n-hexane) n-hexane CH 3 CH 2 CH 3 (no common name) 3 -methylpentane CH 3 CHCHCH 3 (no common name) 2, 3 -dimethylbutane CH 3 CHCH 2 CH 3 (isohexane) 2 -methylpentane CH 3 CCH 2 CH 3 (neohexane) 2, 2 -dimethylbutane
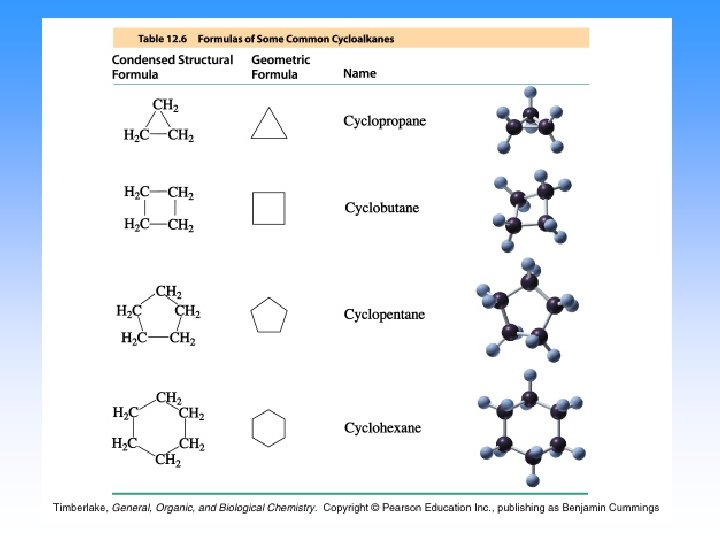
Alkanes

Physical Properties

Boiling points
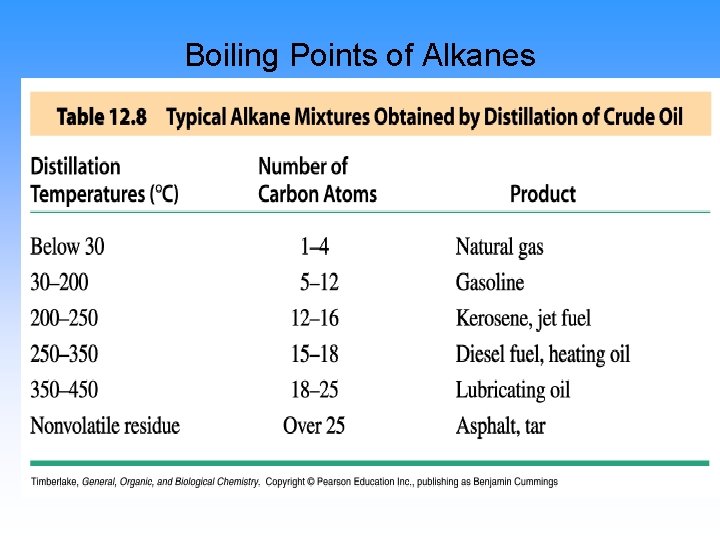
Boiling Points of Alkanes

Isomers • Straight chain alkanes: An alkane that has all its carbons connected in a row. • Branched chain alkanes: An alkane that has a branching connection of carbons. • Isomers: Compounds with same molecular formula but different structures.

Alkane name isomers CH 4 methane 1 C 2 H 6 ethane 1 C 3 H 8 propane 1 C 4 H 10 butanes 2 C 5 H 12 pentanes 3 C 6 H 14 hexanes 5 C 7 H 16 heptanes 9 C 8 H 18 octanes 18 C 9 H 20 nonanes 35 C 10 H 22 decanes 75 ……. C 20 H 42 eicosanes 366, 319 each new common name requires a new prefix…
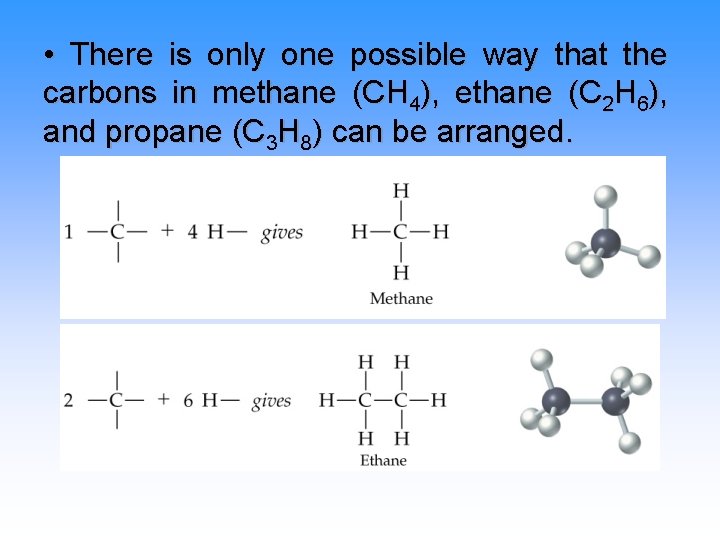
• There is only one possible way that the carbons in methane (CH 4), ethane (C 2 H 6), and propane (C 3 H 8) can be arranged.
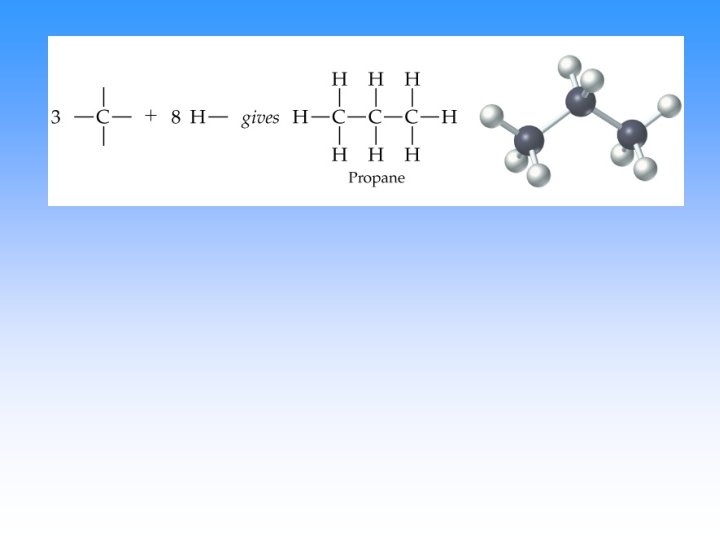
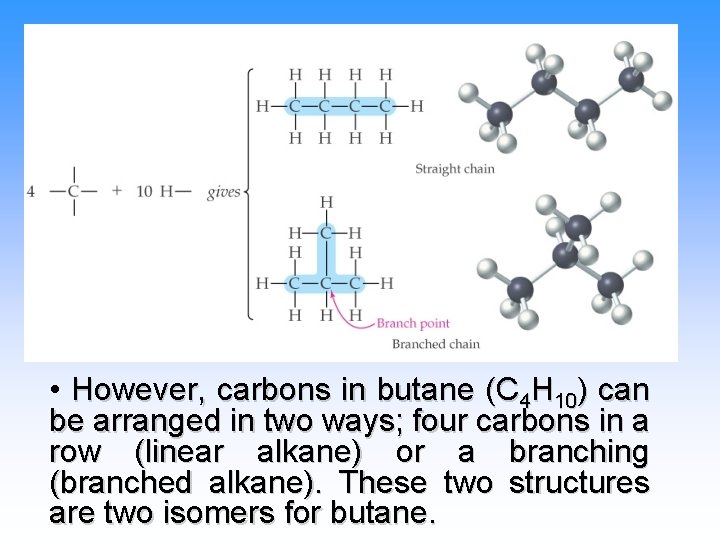
• However, carbons in butane (C 4 H 10) can be arranged in two ways; four carbons in a row (linear alkane) or a branching (branched alkane). These two structures are two isomers for butane.
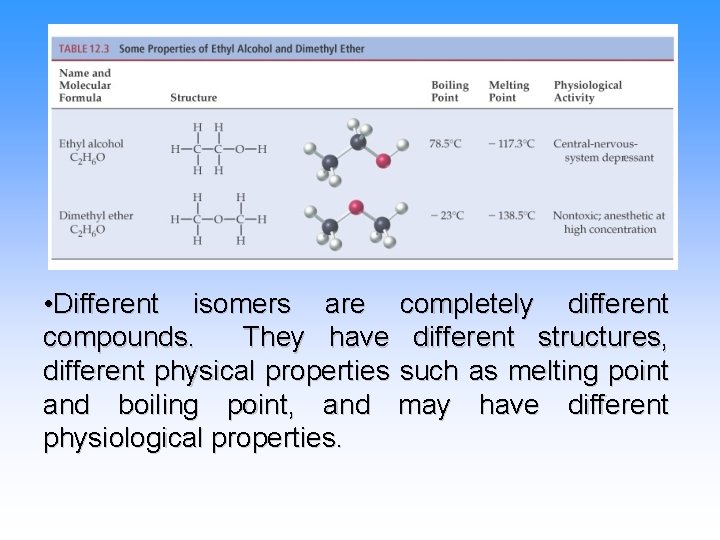
• Different isomers are completely different compounds. They have different structures, different physical properties such as melting point and boiling point, and may have different physiological properties.

Learning Check • Draw all possible structural isomers of C 5 H 12

Types of Carbon Atoms • Primary carbon (1 o) – a carbon bonded to one other carbon • Secondary carbon (2 o) – a carbon bonded to two other carbons • Tertiary carbon (3 o) – a carbon bonded to three other carbons

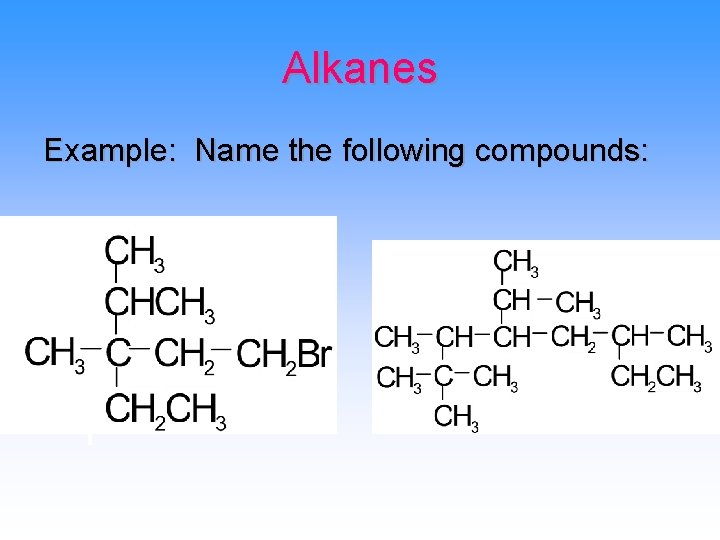
Alkanes Example: Name the following compounds:

Alkanes Example: Write the condensed structure for the following compounds: 3, 3 -dimethylpentane 2 -methyl-4 -sec-butyloctane 1, 2 -dichloro-3 -methylheptane

Environmental transformations • When released in the environment, alkanes don't undergo rapid biodegradation, because they have no functional groups (like hydroxyl or carbonyl) that are needed by most organisms in order to metabolize the compound. • However, some bacteria can metabolize some alkanes (especially those linear and short), by oxidizing the terminal carbon atom. The product is an alcohol, that could be next oxidized to an aldehyde, and finally to a carboxylic acid. The resulting fatty acid could be metabolized through the fatty acid degradation pathway.

Hazards • Methane is explosive when mixed with air (1 – 8% CH 4). Other lower alkanes can also form explosive mixtures with air. The lighter liquid alkanes are highly flammable, although this risk decreases with the length of the carbon chain. Pentane, hexane, heptane, and octane are classed as dangerous for the environment and harmful. The straight-chain isomer of hexane is a neurotoxin. • Considerations for detection /risk control: – – Methane is lighter than air (possibility of accumulation on roofs) Ethane is slightly Heavier than air (possibility of pooling at ground levels / pits) Propane is Heavier than air (possibility of pooling at ground levels / pits) Butane is Heavier than air (possibility of pooling at ground levels / pits)

Assignment • Reaction of Alkene. – Reactions with oxygen (combustion reaction) – Reactions with halogens
- Slides: 37