Organic Chemistry Mr Calmer Lawndale High School Hydrocarbons

Organic Chemistry Mr. Calmer Lawndale High School

Hydrocarbons • Hydrocarbons is the simplest organic compounds that contain only hydrogens and carbons • The 2 simplest hydrocarbons are methane and ethane • Carbon has 4 valence electrons, so it always forms 4 covalent bonds

Methane • The major component of natural gas • Formed by the action of bacteria on decaying plants in swamps • Livestock and termites emit substantial quantities of methane as a product of digestion

Alkanes • Alkane are hydrocarbons with only single covalent bonds • The carbon atoms in an alkane can be arranged in a straight chain or in a chain that has branches
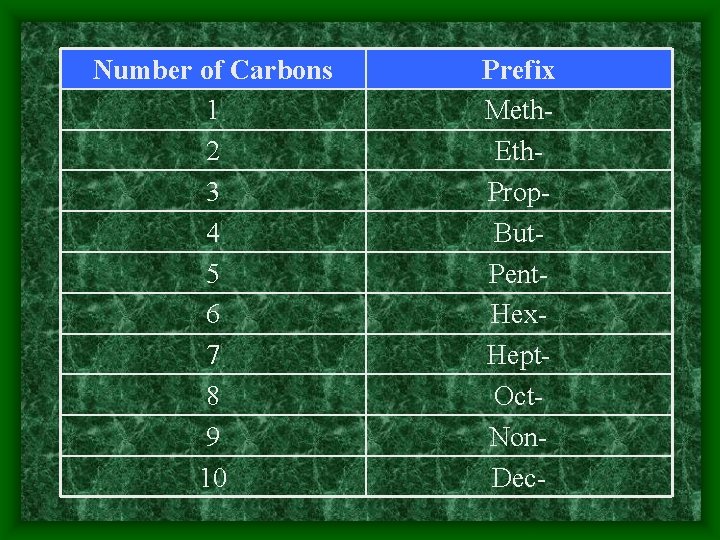
Number of Carbons 1 2 3 4 5 6 7 8 9 10 Prefix Meth. Eth. Prop. But. Pent. Hex. Hept. Oct. Non. Dec-

Branched-Chain Alkane • Parent Chain – the longest chain of carbons (main chain) • Branched-chain alkane – an alkane with one or more side group • Side Group – an atom or group of atoms that is attached to the side of a parent chain

Rules for Naming Alkane 1. Name the longest continuous carbon chain (parent chain) 2. Name the groups attached to this chain in alpha order (change ending to -yl) 3. Designate the location of each side group starting from the nearest side group • Use di-, tri-, tetra- for the same substituents • Use fluoro-, chloro-, bromo-, iodo for halogens

Saturation • Saturated compounds contain only single bonds • Unsaturated compound contain at least one double or triple bond

Alkene • Alkenes are hydrocarbons that contain one or more double covalent bonds Alkyne • Alkyne are hydrocarbons that contain one or more triple covalent bonds

Rules for naming Alkenes and Alkynes 1. Name the parent chain with the corresponding ending (-ene or –yne) 2. Name the side groups in alpha. order 3. The chain is numbered nearest to the double bond 4. If there are more than one double bond in the chain, use di- and tri-
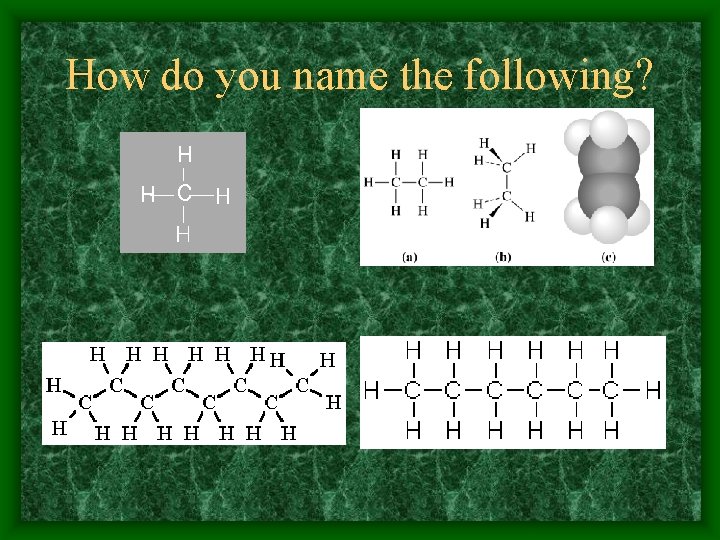
How do you name the following?
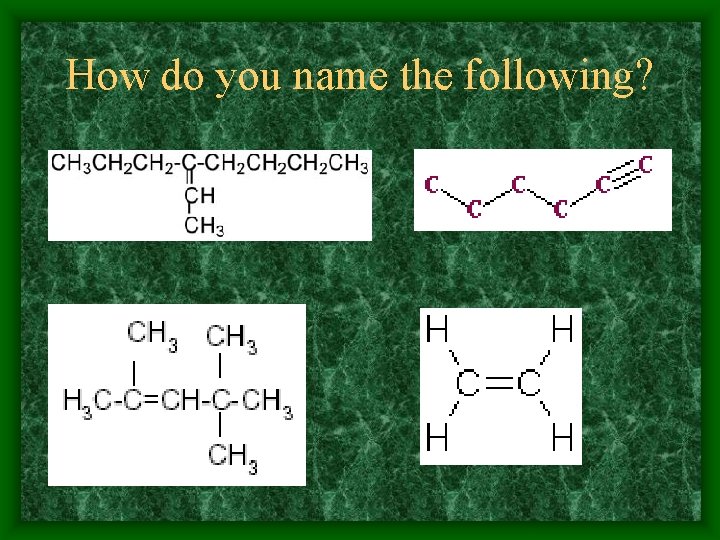
How do you name the following?
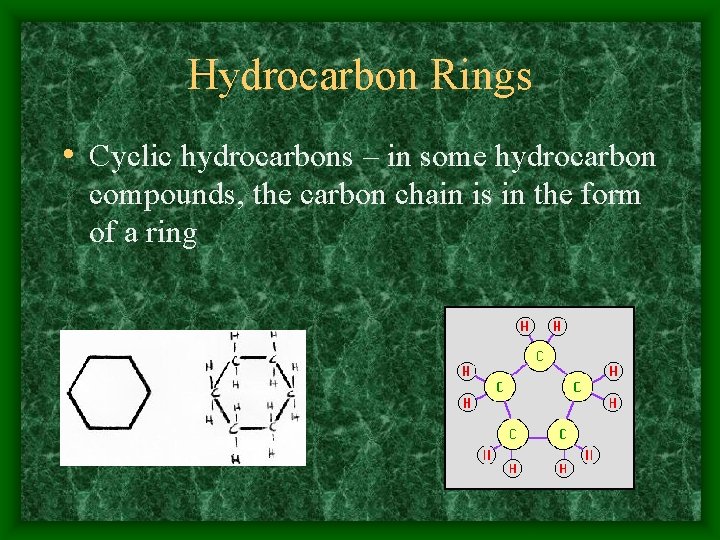
Hydrocarbon Rings • Cyclic hydrocarbons – in some hydrocarbon compounds, the carbon chain is in the form of a ring

Aromatic Hydrocarbons • An organic compound that contains a benzene ring or other ring

Rules for Aromatic Hydrocarbons 1. Name the cycloalkane 2. Number all carbons starting with the first alphabetical substituent. 3. Name each substituent in alphabetical order.
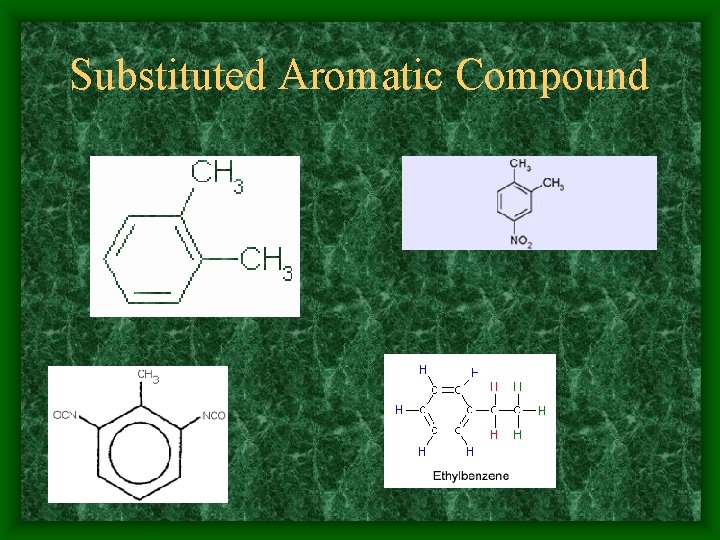
Substituted Aromatic Compound
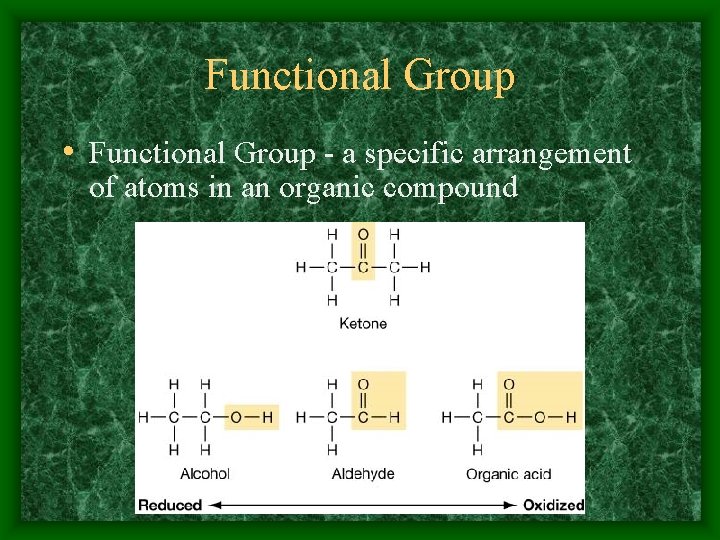
Functional Group • Functional Group - a specific arrangement of atoms in an organic compound
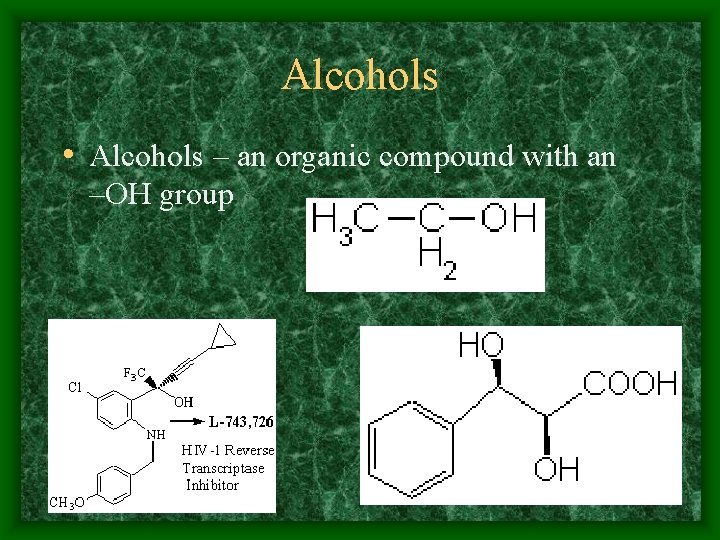
Alcohols • Alcohols – an organic compound with an –OH group

Ethers • Ether – a compound in which oxygen is bonded to two carbon groups
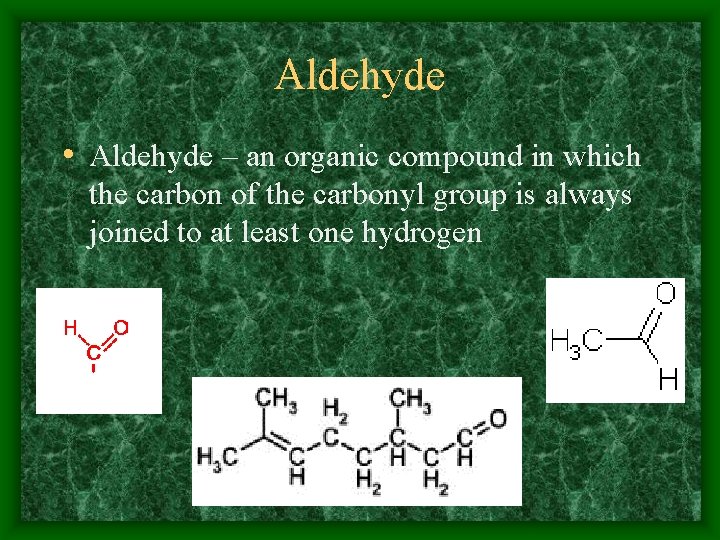
Aldehyde • Aldehyde – an organic compound in which the carbon of the carbonyl group is always joined to at least one hydrogen

Ketone • Ketone – an organic compound in which the carbon of the carbonyl group is joined to two other carbons
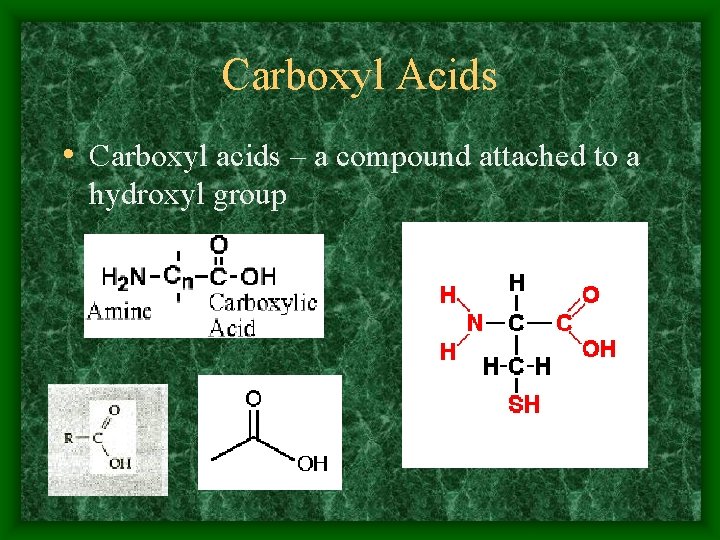
Carboxyl Acids • Carboxyl acids – a compound attached to a hydroxyl group
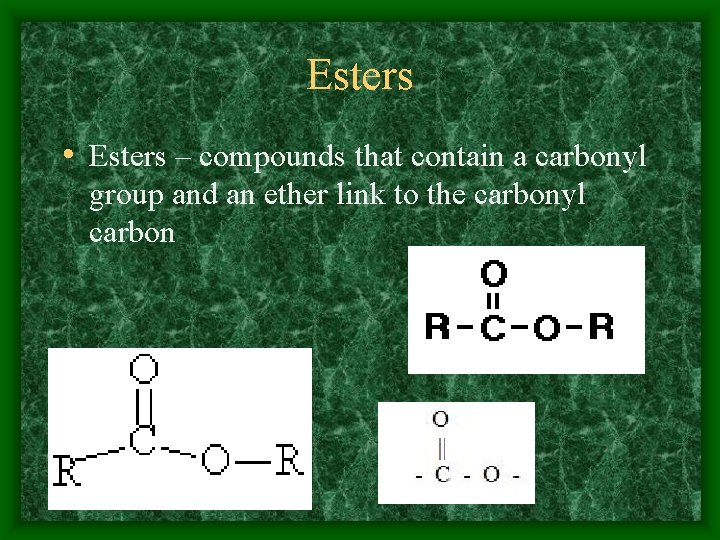
Esters • Esters – compounds that contain a carbonyl group and an ether link to the carbonyl carbon

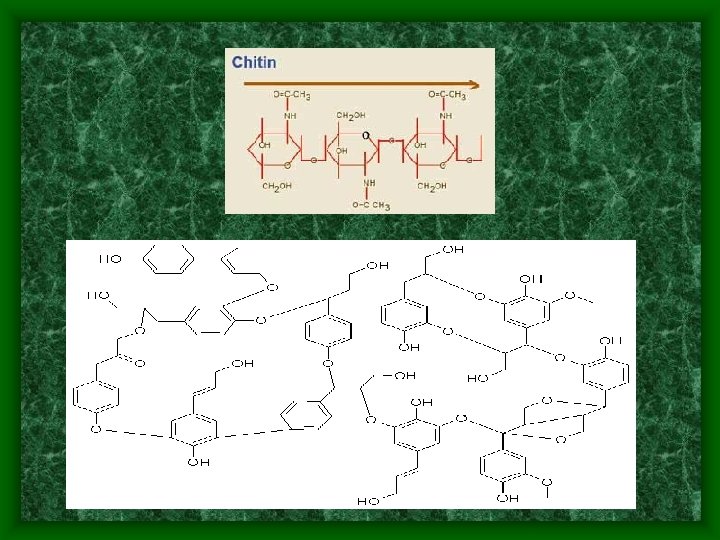
Polymerization • Polymer – a large molecule formed by the covalent bonding of repeating smaller molecules • Monomer – smaller molecules that combine to form a polymer • An addition polymer forms when unsaturated monomers react to form polymers
- Slides: 25