Organic Chemistry Lesson 2 Functional Groups Functional Groups






















- Slides: 30

Organic Chemistry Lesson # 2: Functional Groups

Functional Groups A functional group is an attachment on a hydrocarbon which completely changes its physical and chemical properties, and determines a new homologous series. A functional group can be as simple as a single atom or a much more complex structure. Note that double or triple bonds are NOT functional groups. Functional groups usually contain oxygen, nitrogen, or halogens, and can be single, double, or triply bonded to carbon.

1. The Hydroxyls There are two types of hydroxyl groups: the alcohols and the ethers. These groups contain an - O - group in the place of a hydrogen.

Alcohols Methanol is highly toxic, and drinking even small quantities can lead to blindness and death. Ethanol is the most important alcohol economically. Apart from its uses as an industrial solvent, it can be used as a fuel. Many gasolines contain up to 15% ethanol and in South America, up to 50%. 2 -Propanol is also known as rubbing alcohol.

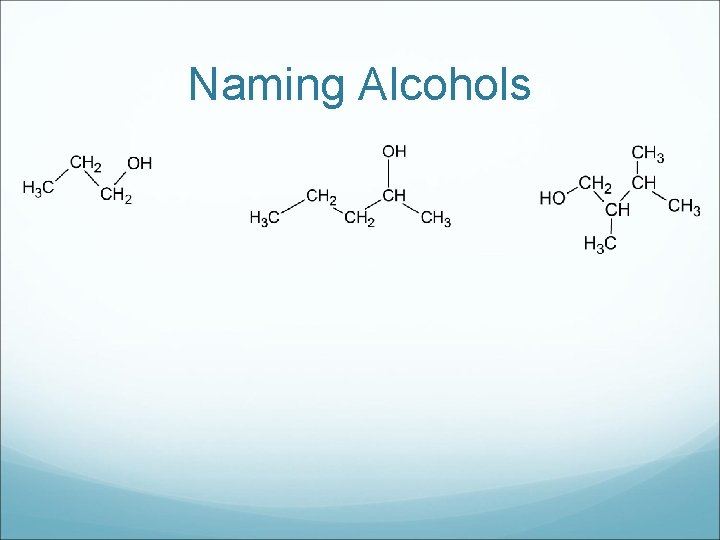
Drawing Alcohols a) ethanol b) 2 -butanol c) 2 -methyl-3 -hexanol

Naming Alcohols

Ethers are compounds in which two alkyl groups are joined to oxygen. Ethers are highly flammable, polar solvents. Ethoxyethane has a boiling point of 34°C and its vapours are extremely flammable. If it is poured into one corner of a room with an open flame in the opposite corner, it could ignite. It was actually used as an anaesthetic for dentistry when laughing gas was proved unreliable. Note that when name ethers, the shortest hydrocarbon alkyl group is named first, no matter to which side of oxygen it lies.

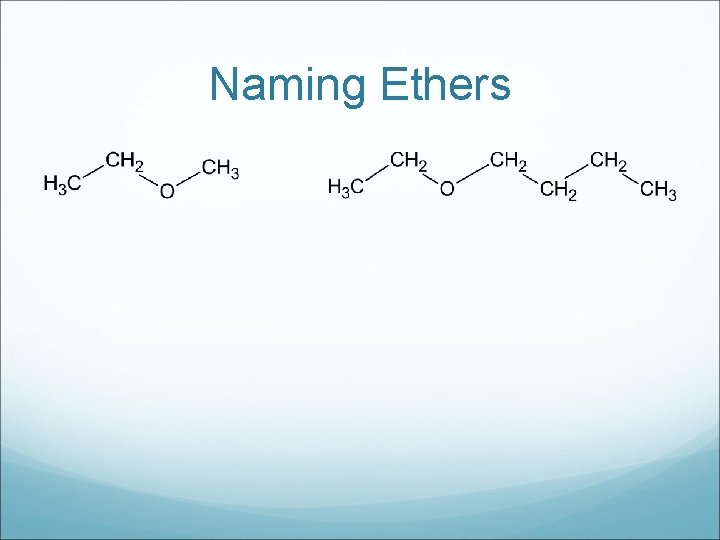
Drawing Ethers a) Methoxybutane b) Ethoxypentane c) Propoxypropane

Naming Ethers

2. The Carbonyls There are two types of carbonyl groups: the aldehydes and the ketones.

Aldehydes are compounds in which the carbonyl group is attached to a terminal (end) carbon. Methanal is used as a preservative for biological specimens, however it is thought to be carcinogenic. Its old name is formaldehyde. Aldehydes with higher molecular mass tend to have pleasant and flowery odour, and are found in essential oils of plants.

Ketones are compounds in which the carbonyl is on a central or non-terminal carbon (in the middle). Ketones are used as industrial solvents. Propanone (old name – acetone) is a widely used household solvent.

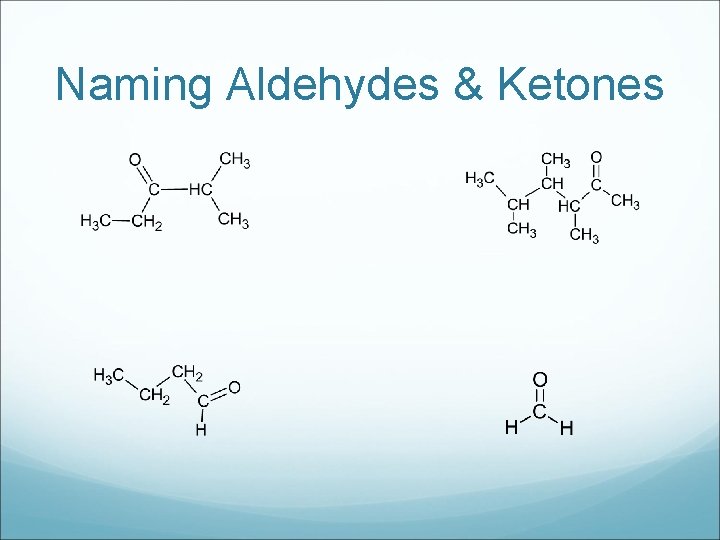
Drawing Aldehydes & Ketones a) Propanal b) 3 -hexanone c) Propanone d) 2 -methyl butanal e) Pentanal f) 3, 4 -dimethyl-2 -heptanone

Naming Aldehydes & Ketones

3. The Carboxyls There are two types of carboxyl groups: the carboxylic acids and the esters.

Carboxylic Acids Carboxylic acids are a combination of aldehyde and alcohol. Carboxylic acids are weak acids, which means they do not completely dissolve in water. Another name for ethanoic acid is acetic acid, which is in vinegar.

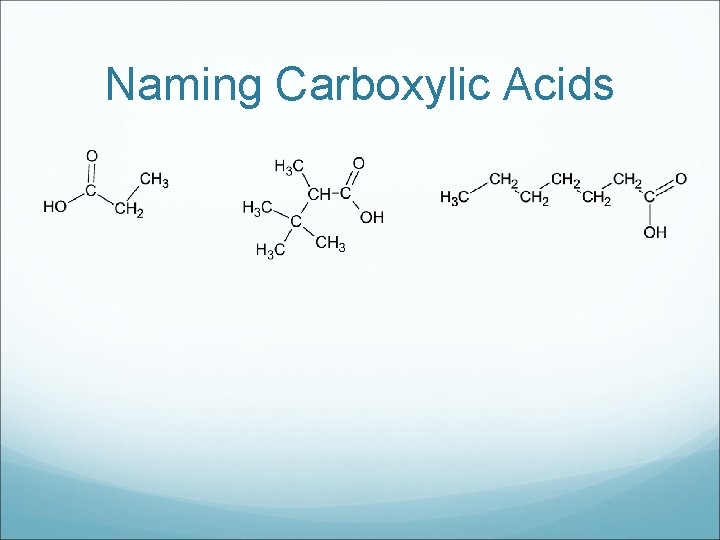
Drawing Carboxylic Acids a) Ethanoic Acid c) 3 -methyl pentanoic acid b) Butanoic Acid

Naming Carboxylic Acids

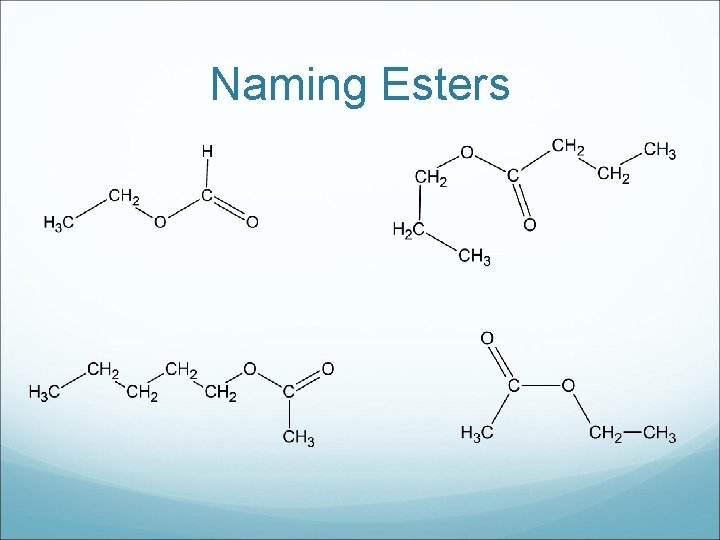
Esters are sweet smelling and many esters occurred naturally in fruit and flowers. They are widely used as flavour and aroma additives in the food industry. To name, the carbon chain attached directly to the single bond oxygen is named as an alkyl group, and the other chain becomes the ending name.

Drawing Esters a) methyl butanoate c) pentyl propanoate b) octyl ethanoate

Naming Esters

4. The Nitrogen Compounds There are two functional groups involving nitrogen: the amines and the amides.

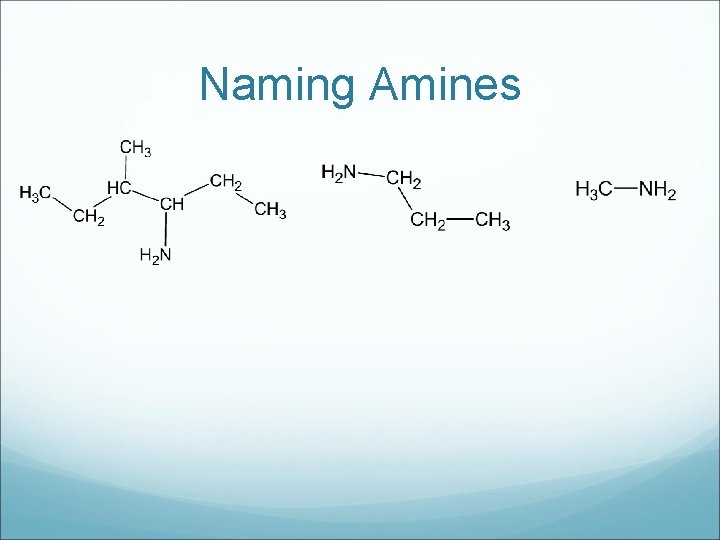
Amines are sometimes called organic bases. The lone pair of electrons on the nitrogen is responsible for most of the properties. The word “amino” starts the hydrocarbon naming, instead of using a suffix.

Drawing Amines a) 2 -aminobutane c) 3 -methyl 2 -aminopropane b) aminoethane

Naming Amines

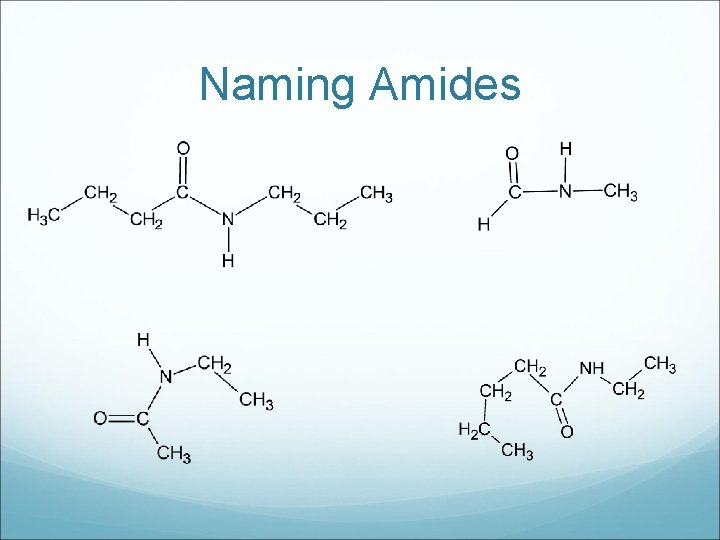
Amides are important because they are the linkages contained in proteins and some synthetic polymers. To name, the carbon chain attached directly to the single bond nitrogen is named as an alkyl group, and the other chain becomes the ending name, similar to esters.

Drawing Amides a) propyl ethanamide c) pentyl methanamide b) methyl butanamide d) butyl ethanamide

Naming Amides

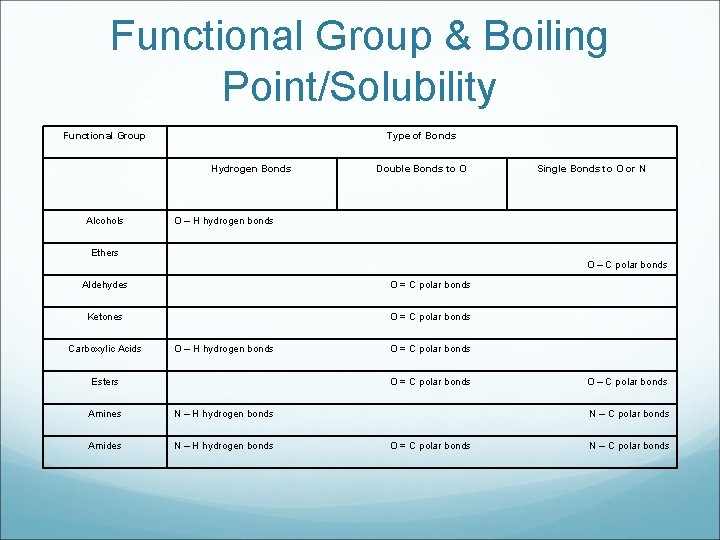
Functional Group & Boiling Point/Solubility Functional Group Type of Bonds Hydrogen Bonds Alcohols Double Bonds to O Single Bonds to O or N O – H hydrogen bonds Ethers O – C polar bonds Aldehydes O = C polar bonds Ketones O = C polar bonds Carboxylic Acids O – H hydrogen bonds Esters O = C polar bonds Amines N – H hydrogen bonds Amides N – H hydrogen bonds O – C polar bonds N – C polar bonds O = C polar bonds N – C polar bonds

Polarity & Boiling Points and Solubility The functional groups with hydrogen bonds have the highest boiling points and can dissolve the most substances, and hydrogen bonding is the strongest intermolecular force. *intermolecular means “between molecules”* The functional groups with polar bonds have slightly lower boiling points and have a limited ability to dissolve substances, both polar and nonpolar. The hydrocarbons have by far the lowest boiling points, and can only dissolve non-polar substances, as they do not contain any polar bonds. “polar” means they do not share electrons equally – stronger bonds “non-polar” means they share electrons equally – weaker bonds