Organic Chemistry Lab 318 Spring 2012 DUE DATES

Organic Chemistry Lab 318 Spring, 2012

DUE DATES • Today – At beginning of lab – Separation Scheme for Nitration of Methyl Benzoate – Spectroscopy Problem Set, Part II, 2. – at end of lab -- copy of laboratory notebook pages for today's experiment (do not turn in your personal notes) • Next Week – At beginning of lab - Nitration of Methyl Benzoate Report – Spectroscopy Problem Set, Part II, 3. • Two Weeks – Synthesis of 4, 4’-di-t-Butylbiphenyl Report – Spectroscopy Problem Set, Part II, 4.

Report from last week • Please review the errors you might have made on the Bromination Report. • Most likely, you will need to review significant digits; reaction mechanisms (not chemical equations) • Notice that the graded parts correspond to the explicit instructions on my web page and the Manual for writing this report. • If you have questions about how it was graded, please ask.

Report for next week • There are instructions for writing a Synthesis Report in the Manual and on my web site. Please follow the instructions so that you can earn the maximum points. • Pay attention to significant digits (0. 002 has only one sig. fig. ) • Note the example for calculation of moles of strong acids such as H 2 SO 4 and HNO 3. • Note the example for completing the table on the report form that is the calculation of theoretical yield. • Pay special attention to citing physical property data (M. W. , density, etc. ) as explained
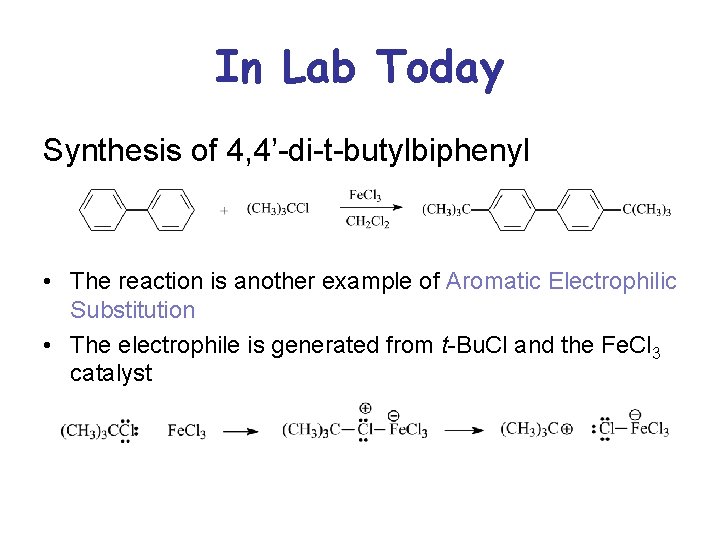
In Lab Today Synthesis of 4, 4’-di-t-butylbiphenyl • The reaction is another example of Aromatic Electrophilic Substitution • The electrophile is generated from t-Bu. Cl and the Fe. Cl 3 catalyst
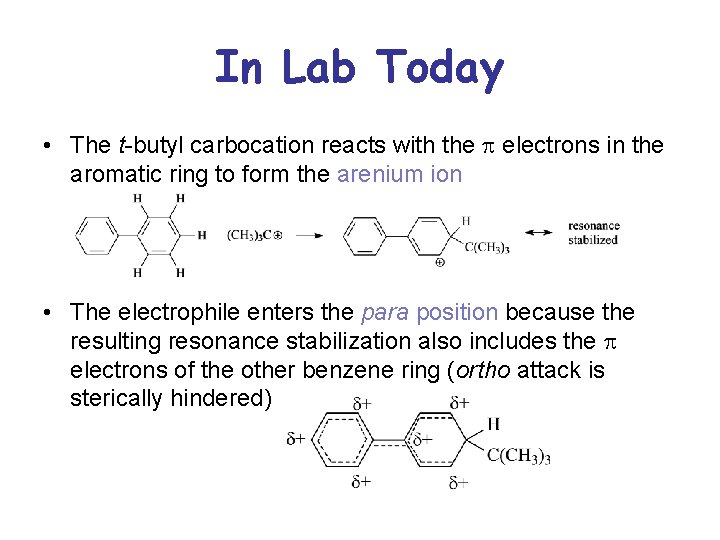
In Lab Today • The t-butyl carbocation reacts with the p electrons in the aromatic ring to form the arenium ion • The electrophile enters the para position because the resulting resonance stabilization also includes the p electrons of the other benzene ring (ortho attack is sterically hindered)
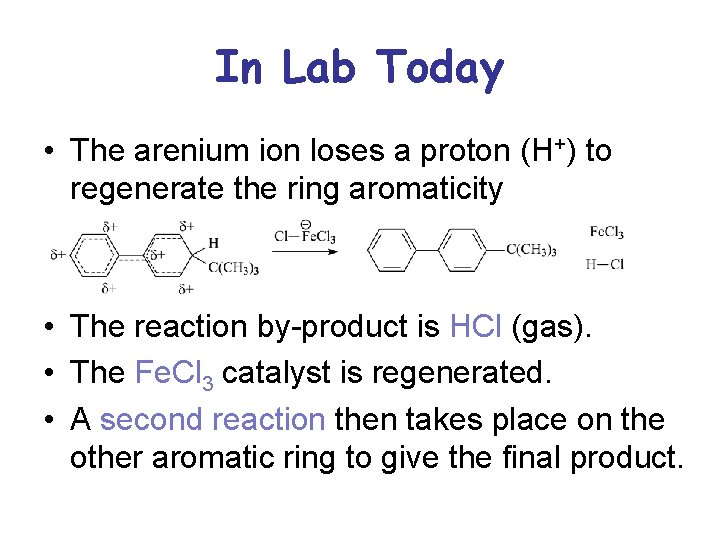
In Lab Today • The arenium ion loses a proton (H+) to regenerate the ring aromaticity • The reaction by-product is HCl (gas). • The Fe. Cl 3 catalyst is regenerated. • A second reaction then takes place on the other aromatic ring to give the final product.

Experiment Notes • Set up hot plate in hood; cool 10% HCl – all ready ahead of time • Weigh the vial containing biphenyl; empty contents into Erlenmeyer; reweigh vial (±. 001 g) • Fe. Cl 3 is hydroscopic and must be added quickly • Keep reaction in the hood (use the one near you). • This is where the Separation Scheme begins (the reaction is over and then 10% HCl is added to begin the separation of the product from the mixture – it removes Fe. Cl 3).

Experiment Notes • Use a funnel when pouring the acidified reaction mixture into the separatory funnel. • Review extraction with separatory funnel and drying agents (p. 681 -682; 686 -688) l NEVER throw anything out until you are sure you have your product isolated! Always keep a beaker under the funnel in case of leaks Keep the extract in an Erlenmeyer flask (not a beaker)

Experiment Notes • Review drying agents (p. 693 -697) – Solution should be in the small Erlenmeyer – Add “some” drying agent with a spatula. Swirl to mix. – Observe the result and consult Table 12. 3 p. 697 – Decant into a small beaker; evaporate solvent in Hood • See additional instructions for recrystallization from ethanol

In Lab Today • Finish the Nitration experiment – Weigh the final product: weigh empty vial, place product in vial, weigh again. – Take melting point of product. • Increase temperature slowly • Range: from first drop of liquid to complete melting – Properly label the vial containing the product. – Turn the vial in to me before the end of the period. – Cleanup – Chung and Reyes
- Slides: 11