Organic Chemistry Introduction 1 2 3 4 bonds

Organic Chemistry Introduction 1 2 3 4 bonds HONC 3/3/2021 Chem-100 1
What is organic chemistry? • study of carbon, the compounds it makes, and the reactions it undergoes • over 16 million carbon-containing compounds are known • because the C-C single bond (348 k. J mol-1) and the C-H bond (412 k. J mol-1) are strong, carbon compounds are stable • carbon can form chains and rings 3/3/2021 Chem-100 2
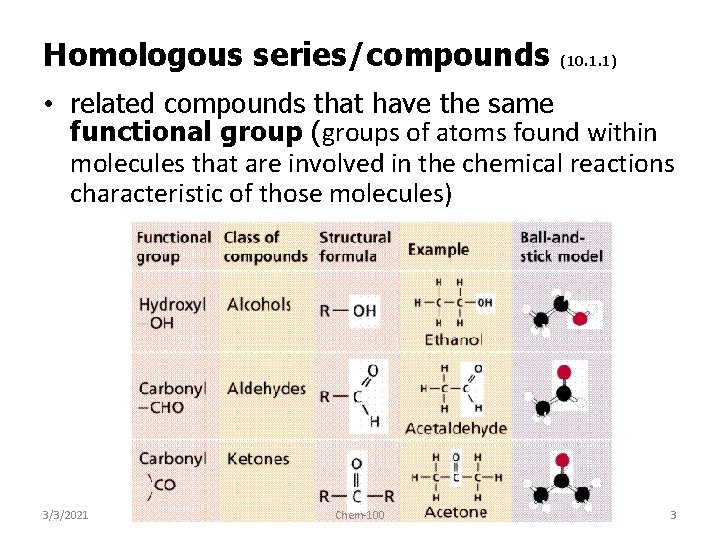
Homologous series/compounds (10. 1. 1) • related compounds that have the same functional group (groups of atoms found within molecules that are involved in the chemical reactions characteristic of those molecules) 3/3/2021 Chem-100 3
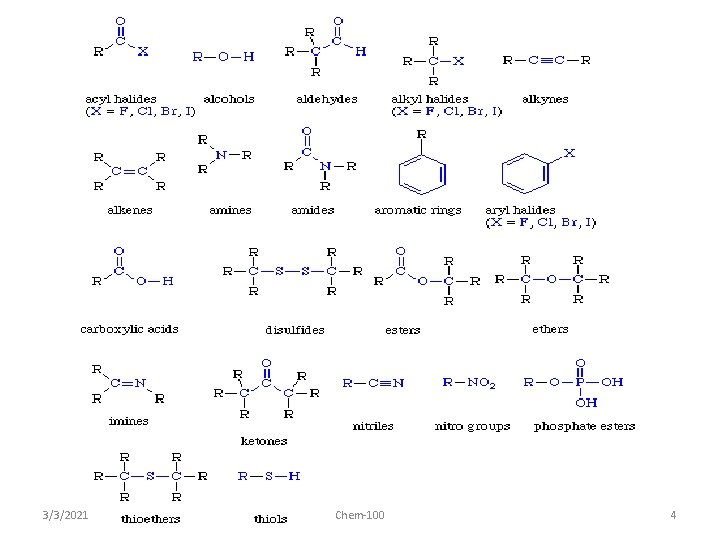
3/3/2021 Chem-100 4

• differ from each other by a CH 2 unit • can be represented by a general formula – examples: • Cn. H 2 n+2 (alkanes) or Cn. H 2 n (alkenes) or… 3/3/2021 Chem-100 5
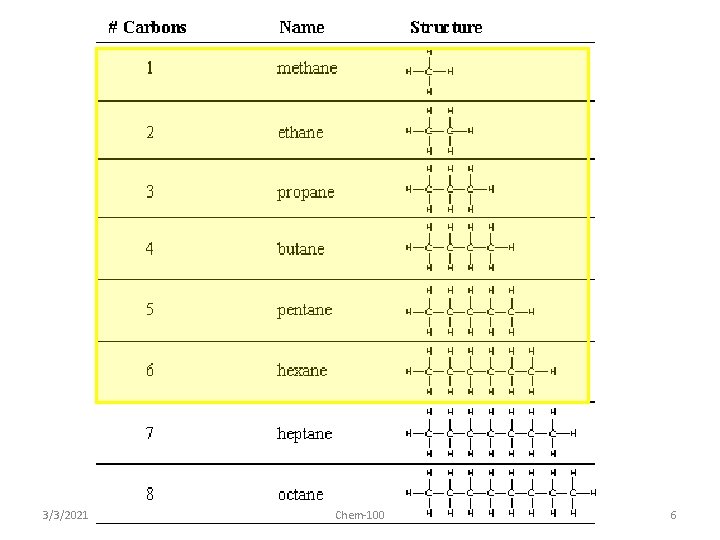
3/3/2021 Chem-100 6
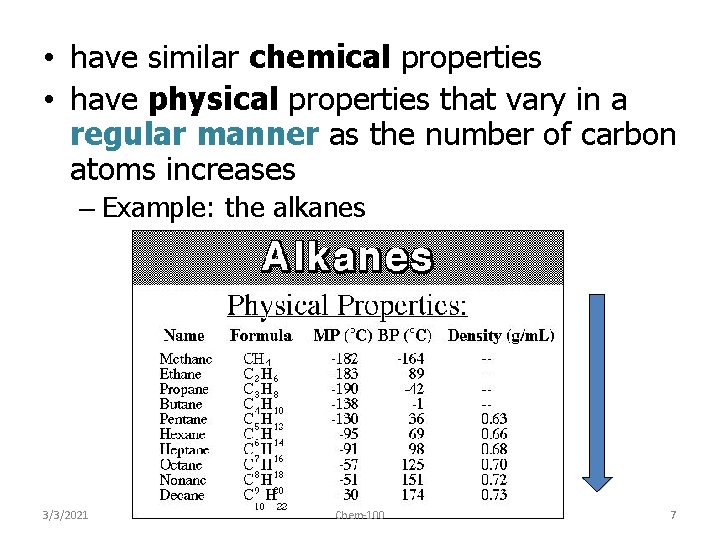
• have similar chemical properties • have physical properties that vary in a regular manner as the number of carbon atoms increases – Example: the alkanes 3/3/2021 Chem-100 7
3/3/2021 Chem-100 8

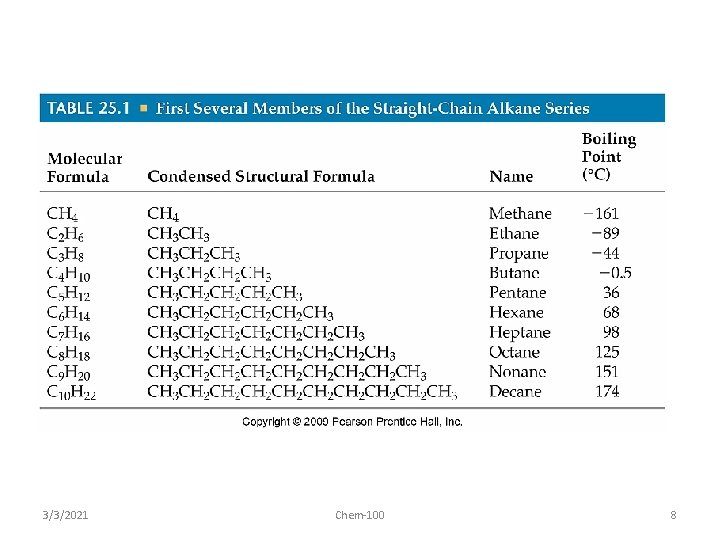
Trends in boiling points of members of a homologous series (10. 1. 2) • melting point and boiling point increase Alkane Formula Boiling Pt. /o. C with more carbon atoms methane CH 4 -162. 0 • Why? – – intermolecular forces increase adding a CH 2 adds more electrons • 3/3/2021 ethane this increases the Van der Waal’s forces C 2 H 6 -88. 6 propane C 3 H 8 -42. 2 butane Chem-100 C 4 H 10 -0. 5 9
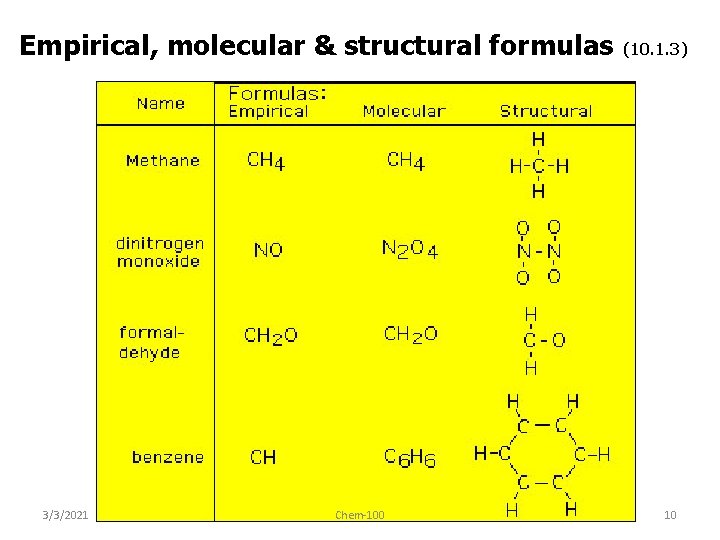
Empirical, molecular & structural formulas 3/3/2021 Chem-100 (10. 1. 3) 10

• empirical formula – simplest ratio of atoms in a molecule • molecular formula – actual numbers of atoms in a molecule 3/3/2021 Empirical Formula Molecular Formula CH 4 CH 3 C 2 H 6 CH 2 O C 6 H 12 O 6 CH 2 C 4 H 8 CH 2 C 8 H 16 Chem-100 11
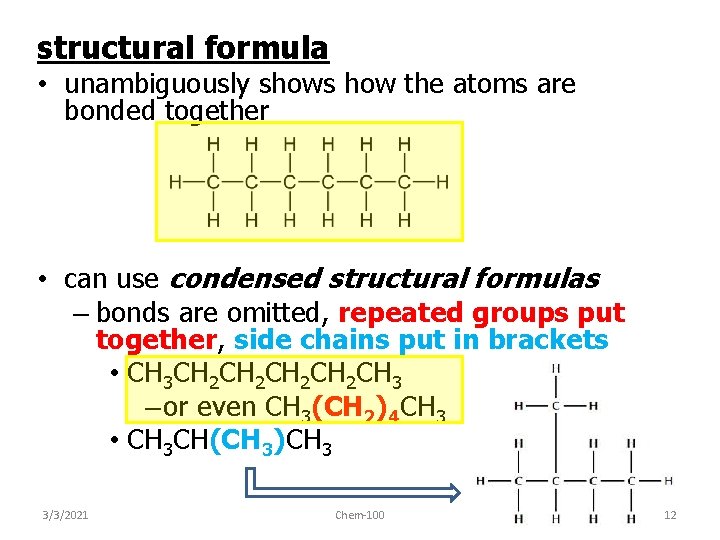
structural formula • unambiguously shows how the atoms are bonded together • can use condensed structural formulas – bonds are omitted, repeated groups put together, side chains put in brackets • CH 3 CH 2 CH 2 CH 3 – or even CH 3(CH 2)4 CH 3 • CH 3 CH(CH 3)CH 3 3/3/2021 Chem-100 12
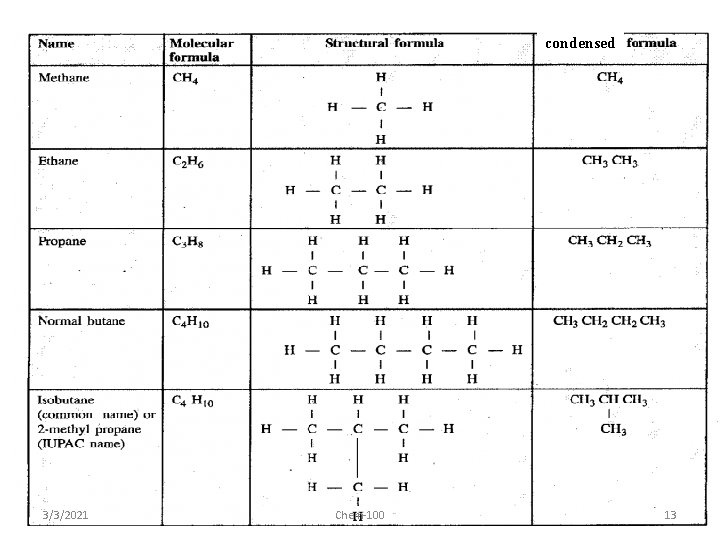
condensed 3/3/2021 Chem-100 13
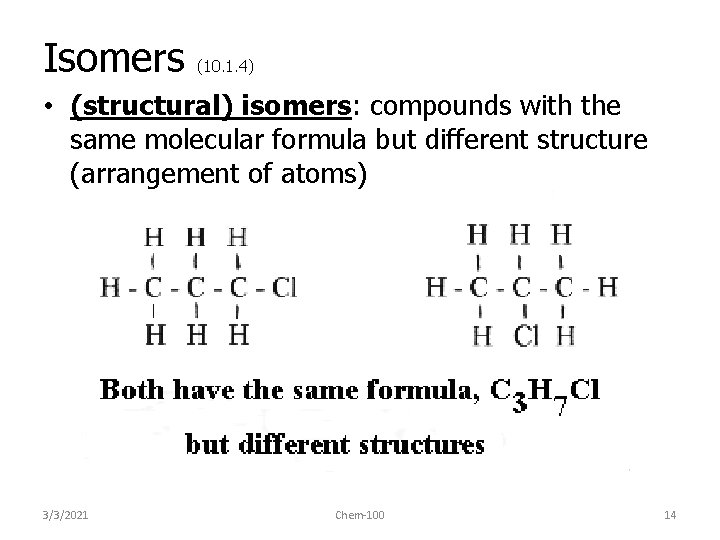
Isomers (10. 1. 4) • (structural) isomers: compounds with the same molecular formula but different structure (arrangement of atoms) 3/3/2021 Chem-100 14
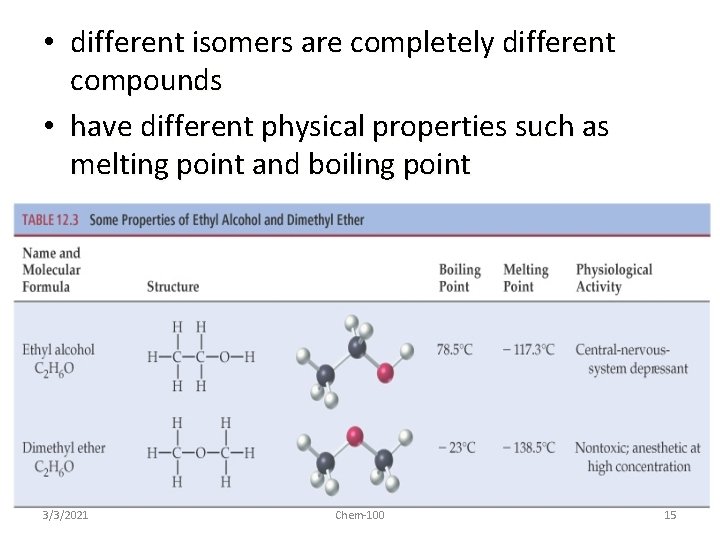
• different isomers are completely different compounds • have different physical properties such as melting point and boiling point 3/3/2021 Chem-100 15
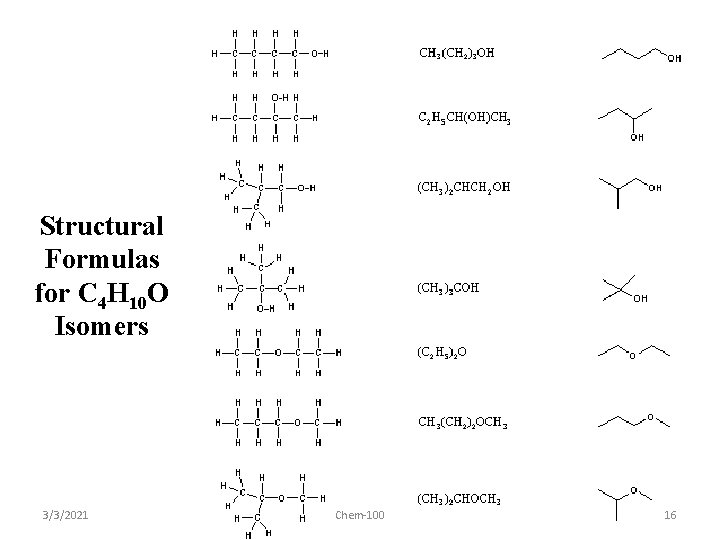
Structural Formulas for C 4 H 10 O Isomers 3/3/2021 Chem-100 16

Alkanes Structural formulas for the isomers of noncyclic alkanes up to C 6 (10. 1. 5) • hydrocarbon chains where all the bonds between carbons are SINGLE bonds • Cn. H 2 n+2 • draw out and write the structural formulas for all isomers that can be formed by: – CH 4 – C 2 H 6 – C 3 H 8 – C 4 H 10 – C 5 H 12 – C 6 H 14 3/3/2021 Richard Thornley 10. 1. 5 2: 54 Chem-100 17

Naming the isomers (IUPAC) of non-cyclic alkanes up to C 6 (10. 1. 6) 1. Richard Thornley 3: 35 2. Determine the longest carbon chain – 1 2 3 4 5 6 3/3/2021 Use the prefix to denote the number carbons Meth. Eth. Prop. But. Pent. Hex- Monkeys Eat Peeled Bananas Chem-100 18

3. use the suffix “-ane” to indicate that the substance is an alkane 4. number the carbons in the chain consecutively, starting at the end closest to a substituent (groups attached to the main chain/most busy end) 3/3/2021 Chem-100 19

5. name and number the location of each substituent – the name of the substituent will be written before the main chain and will end with “–yl” (or just memorize the below) • • • CH 3 is methyl C 2 H 5 is ethyl C 3 H 7 is propyl And with 2 or more side chains: 5. use prefixes di-, tri-, tetra-, to indicate when there are multiple side chains of the same type 6. use commas to separate numbers and hyphens to separate numbers or letters. 7. name the side chains in alphabetical order 3/3/2021 Chem-100 20
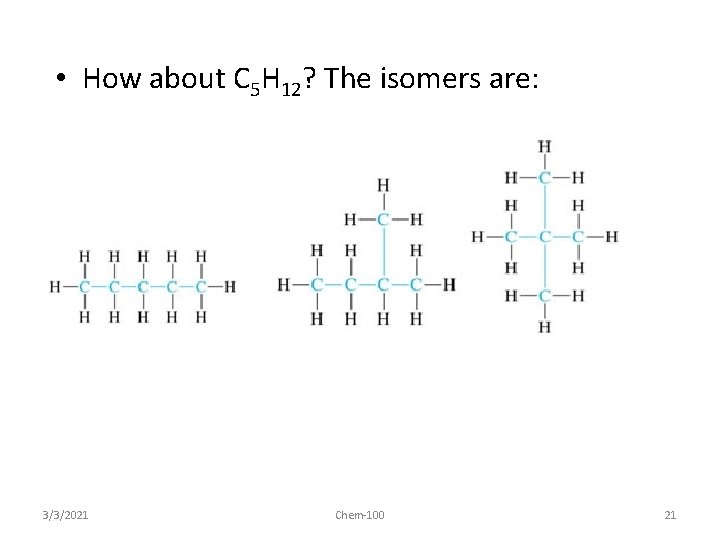
• How about C 5 H 12? The isomers are: Pentane 3/3/2021 2 -methyl-butane 2, 2 -dimethyl propane Chem-100 21
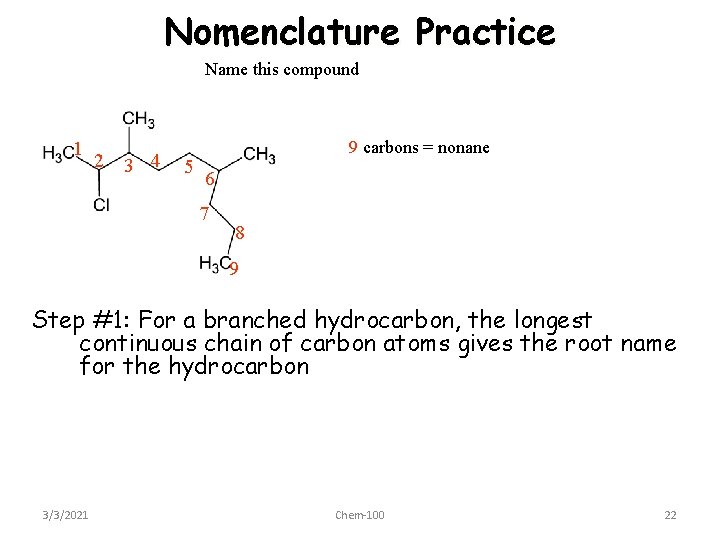
Nomenclature Practice Name this compound 1 2 3 4 5 9 carbons = nonane 6 7 8 9 Step #1: For a branched hydrocarbon, the longest continuous chain of carbon atoms gives the root name for the hydrocarbon 3/3/2021 Chem-100 22
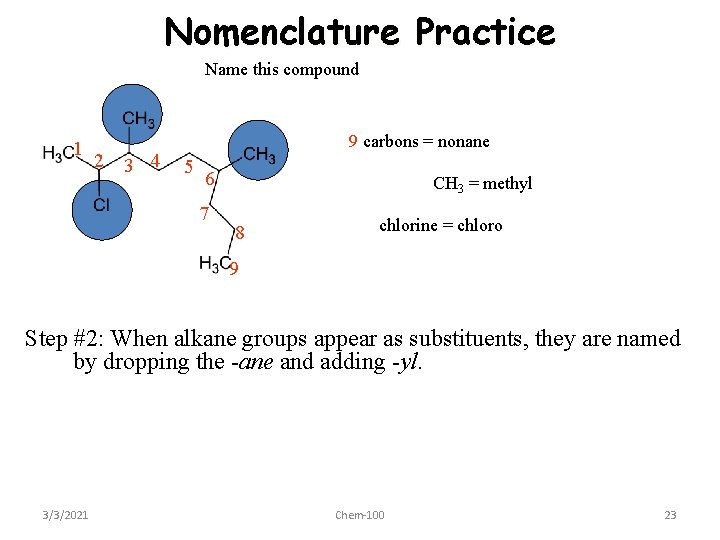
Nomenclature Practice Name this compound 1 2 3 4 9 carbons = nonane 5 6 7 CH 3 = methyl 8 chlorine = chloro 9 Step #2: When alkane groups appear as substituents, they are named by dropping the -ane and adding -yl. 3/3/2021 Chem-100 23
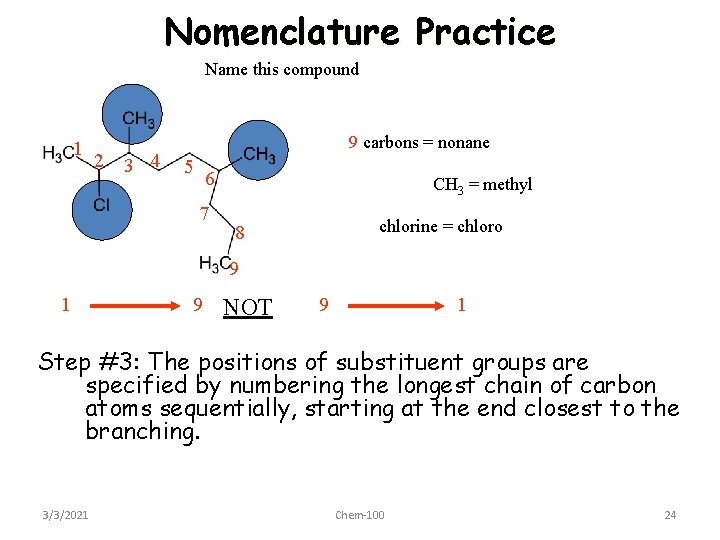
Nomenclature Practice Name this compound 1 2 3 4 9 carbons = nonane 5 6 7 CH 3 = methyl chlorine = chloro 8 9 1 9 NOT 9 1 Step #3: The positions of substituent groups are specified by numbering the longest chain of carbon atoms sequentially, starting at the end closest to the branching. 3/3/2021 Chem-100 24
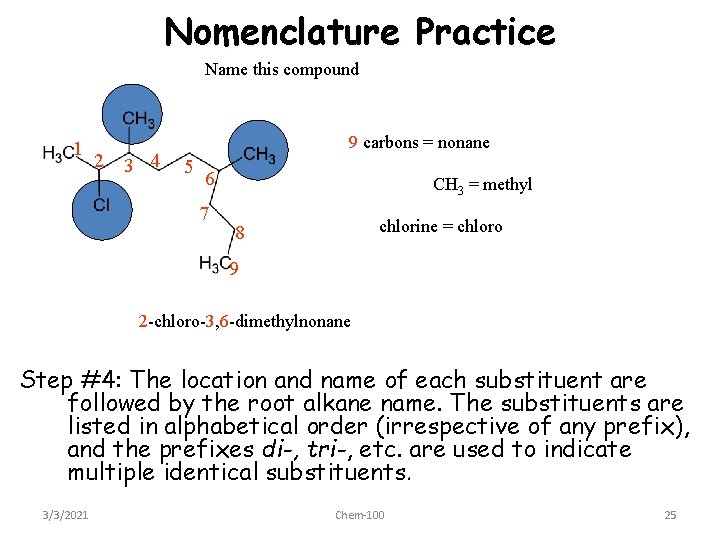
Nomenclature Practice Name this compound 1 2 3 4 9 carbons = nonane 5 6 7 CH 3 = methyl chlorine = chloro 8 9 2 -chloro-3, 6 -dimethylnonane Step #4: The location and name of each substituent are followed by the root alkane name. The substituents are listed in alphabetical order (irrespective of any prefix), and the prefixes di-, tri-, etc. are used to indicate multiple identical substituents. 3/3/2021 Chem-100 25

Alkenes Structural formulas for the isomers of the straight chain alkenes up to C 6 (10. 1. 7) • alkenes have a double bond between two or more of the carbons • Cn. H 2 n • draw out and write the structural formulas for all isomers that can be formed by each – C 2 H 4 – C 3 H 6 Richard Thornley 10. 1. 7 (1: 37) – C 4 H 8 – C 5 H 10 – C 6 H 12 3/3/2021 Chem-100 26

Naming the isomers (IUPAC) of straight chain alkenes up to C 6 (10. 1. 8) 1. suffix changes to “-ene” 2. when there are 4 or more carbon atoms in a chain, the location of the double bond is indicated by a number 3. begin counting the carbons closest to the end with the C=C bond – numbering the location of the double bond(s) takes precedence over the location of any substituents 3/3/2021 1 -butene but-1 -ene 2 -butene but-2 -ene Chem-100 27
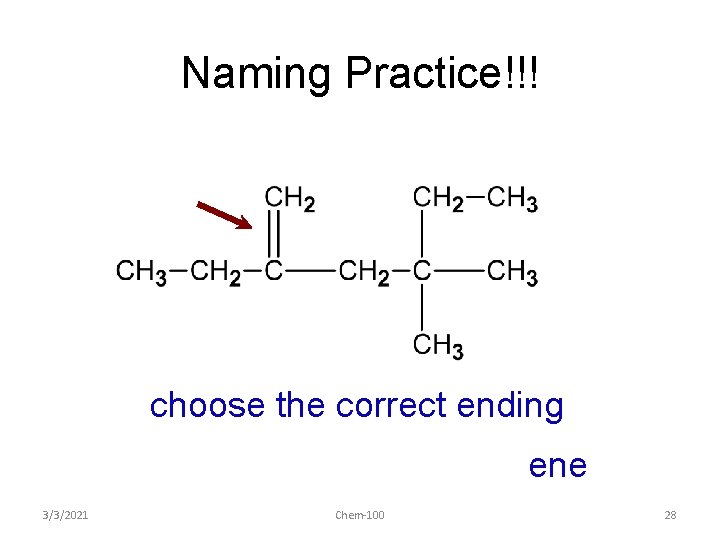
Naming Practice!!! choose the correct ending ene 3/3/2021 Chem-100 28
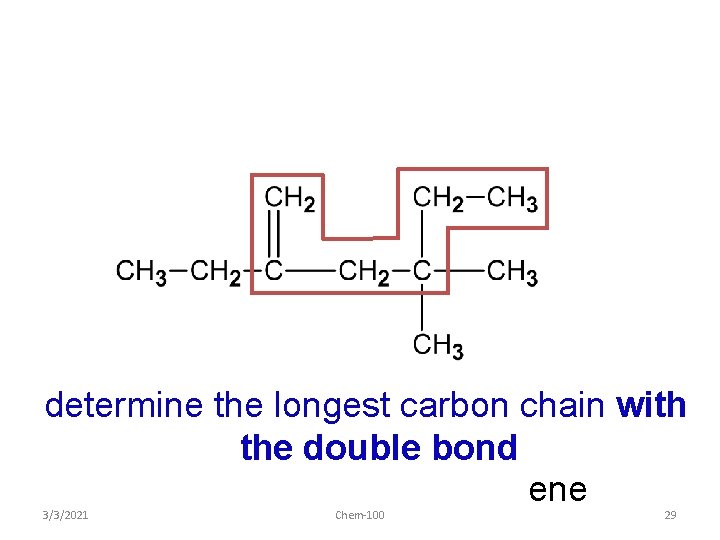
determine the longest carbon chain with the double bond ene 3/3/2021 Chem-100 29
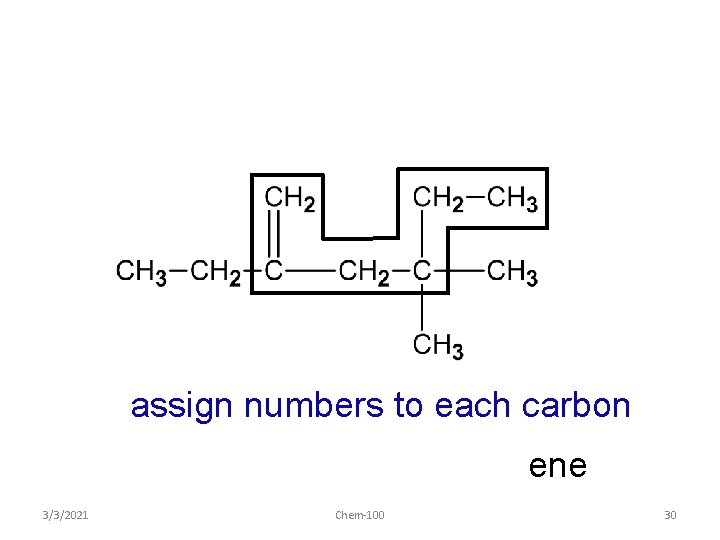
assign numbers to each carbon ene 3/3/2021 Chem-100 30
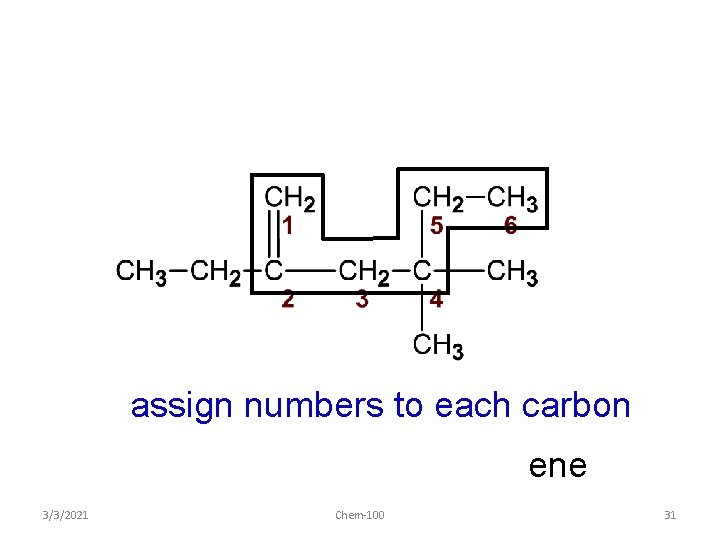
assign numbers to each carbon ene 3/3/2021 Chem-100 31
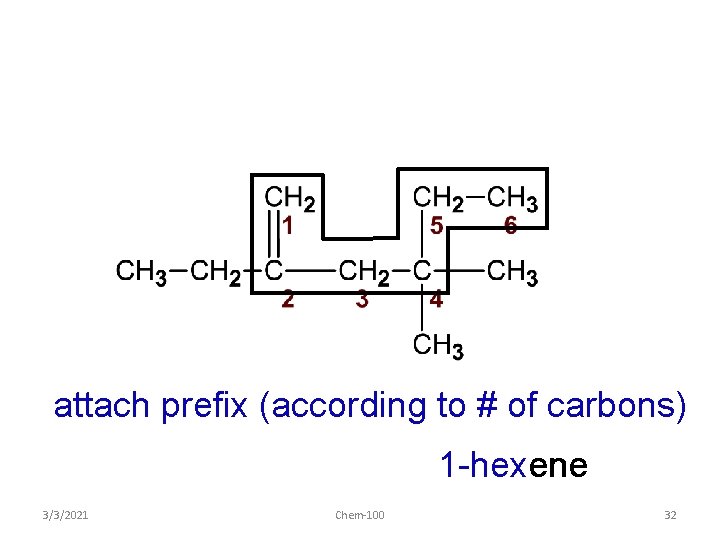
attach prefix (according to # of carbons) 1 -hexene 3/3/2021 Chem-100 32
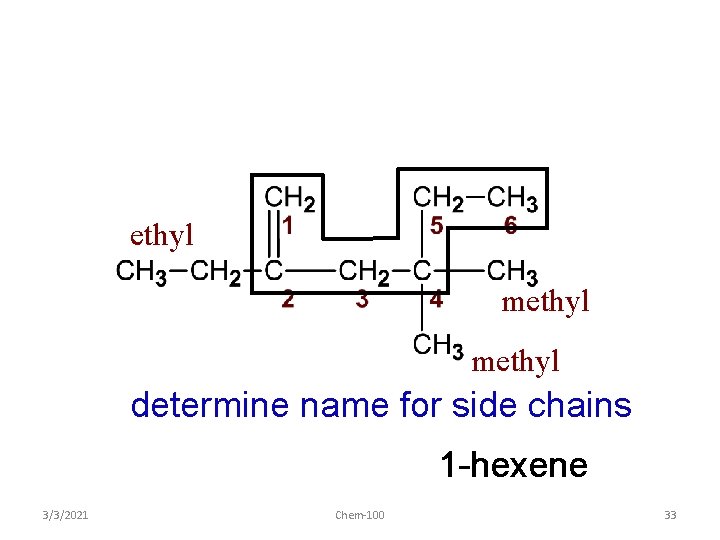
ethyl methyl determine name for side chains 1 -hexene 3/3/2021 Chem-100 33
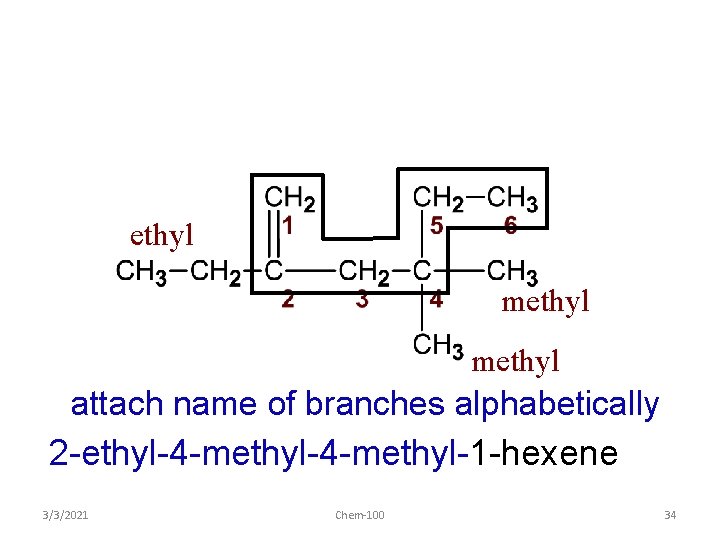
ethyl methyl attach name of branches alphabetically 2 -ethyl-4 -methyl-1 -hexene 3/3/2021 Chem-100 34
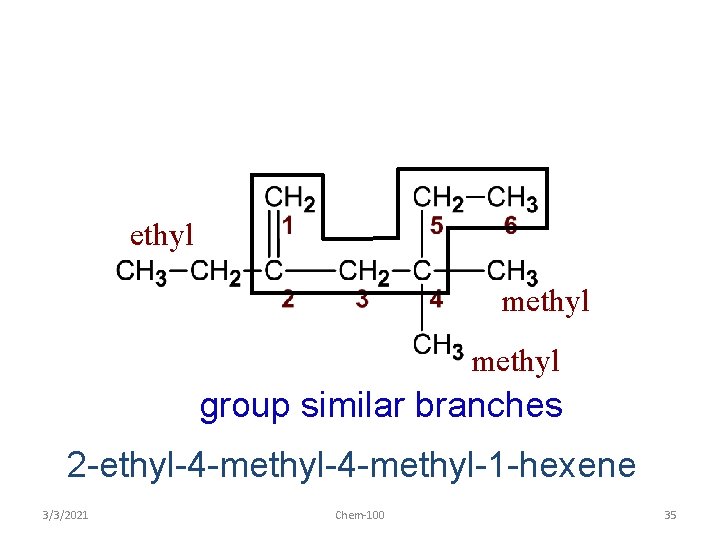
ethyl methyl group similar branches 2 -ethyl-4 -methyl-1 -hexene 3/3/2021 Chem-100 35
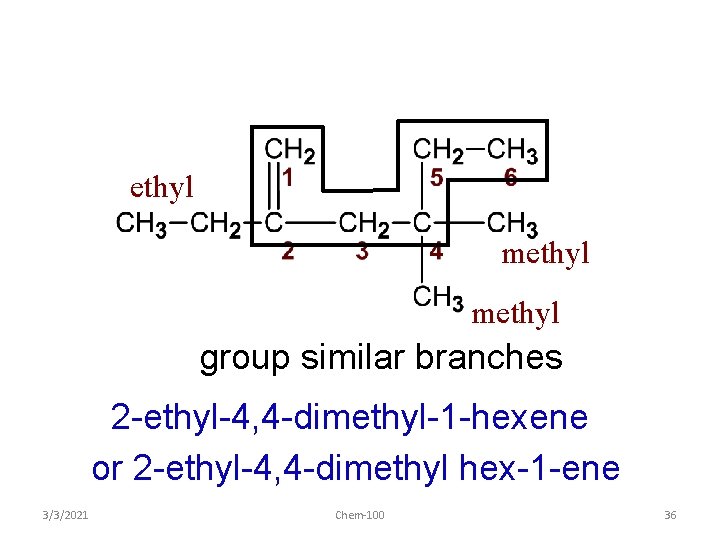
ethyl methyl group similar branches 2 -ethyl-4, 4 -dimethyl-1 -hexene or 2 -ethyl-4, 4 -dimethyl hex-1 -ene 3/3/2021 Chem-100 36
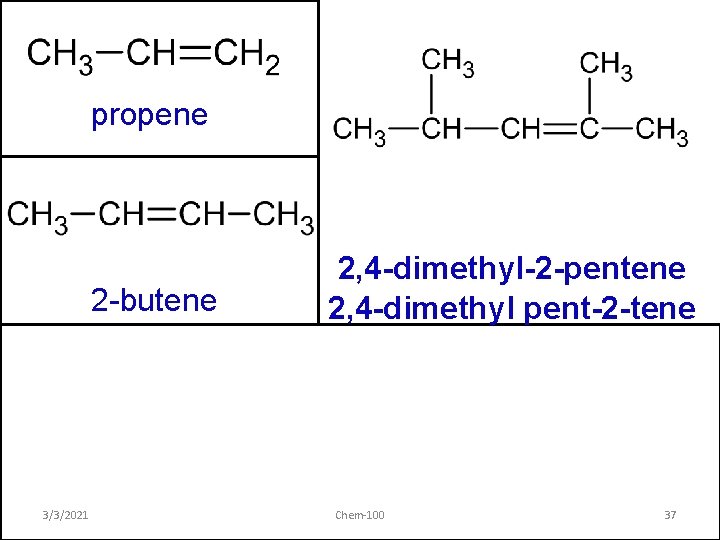
propene 2 -butene 3/3/2021 2, 4 -dimethyl-2 -pentene 2, 4 -dimethyl pent-2 -tene Chem-100 37
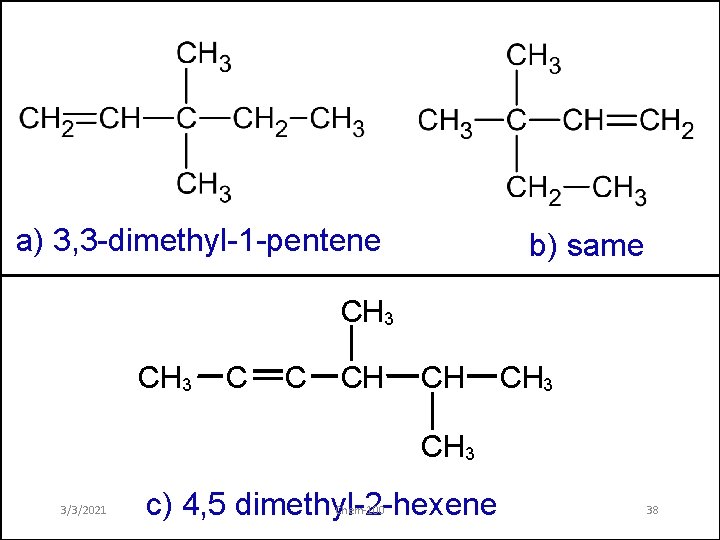
a) 3, 3 -dimethyl-1 -pentene b) same CH 3 C C CH CH CH 3 3/3/2021 c) 4, 5 dimethyl-2 -hexene Chem-100 38
- Slides: 38