Organic Chemistry for Hydrocarbon 1 Alkanes Hydrocarbon chains
Organic Chemistry for Hydrocarbon 1
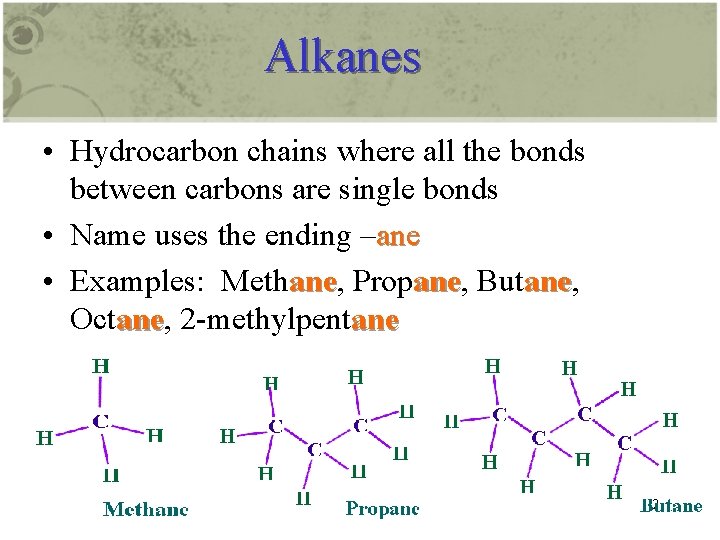
Alkanes • Hydrocarbon chains where all the bonds between carbons are single bonds • Name uses the ending –ane • Examples: Methane, Propane, Butane, Octane, 2 -methylpentane 2
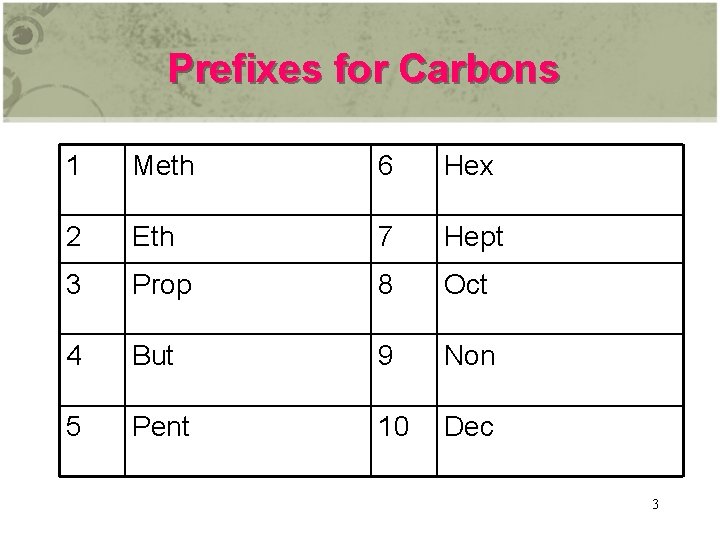
Prefixes for Carbons 1 Meth 6 Hex 2 Eth 7 Hept 3 Prop 8 Oct 4 But 9 Non 5 Pent 10 Dec 3

Physical & Chemical Properties • Alkanes • Alkenes • Alkynes • Aromatics 4

Physical & Chemical Properties Alkanes & Cycloalkanes 5
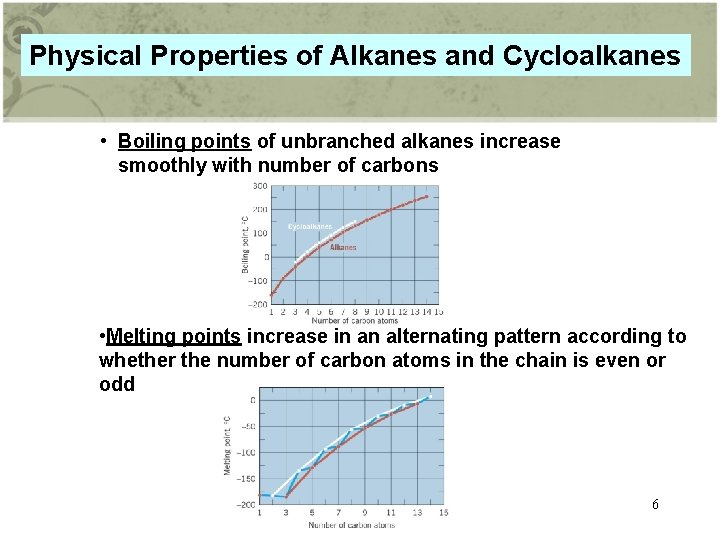
Physical Properties of Alkanes and Cycloalkanes • Boiling points of unbranched alkanes increase smoothly with number of carbons • Melting points increase in an alternating pattern according to whether the number of carbon atoms in the chain is even or odd 6

Physical Properties of Alkanes and Cycloalkanes • Branched chain has higher boiling point than strength chain (in case of number of carbon is equal) • Alkanes are non-polar molecules, this indicates that alkanes are water-insoluble compounds. • Alkanes are non-conductive compounds. 7

Chemical Properties of Alkanes and Cycloalkanes 1. Alkanes & cycloalkanes • are well flammable. • no soot from combustion. 8

Chemical Properties of Alkanes and Cycloalkanes Alkanes & cycloalkanes are saturated compounds. (having only single bonds) Chemical reactions: Substitution reactions 9
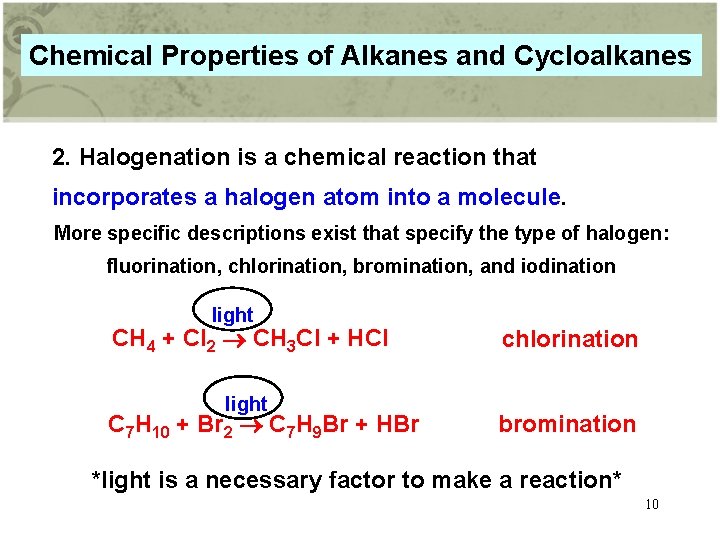
Chemical Properties of Alkanes and Cycloalkanes 2. Halogenation is a chemical reaction that incorporates a halogen atom into a molecule. More specific descriptions exist that specify the type of halogen: fluorination, chlorination, bromination, and iodination light CH 4 + Cl 2 CH 3 Cl + HCl light C 7 H 10 + Br 2 C 7 H 9 Br + HBr chlorination bromination *light is a necessary factor to make a reaction* 10
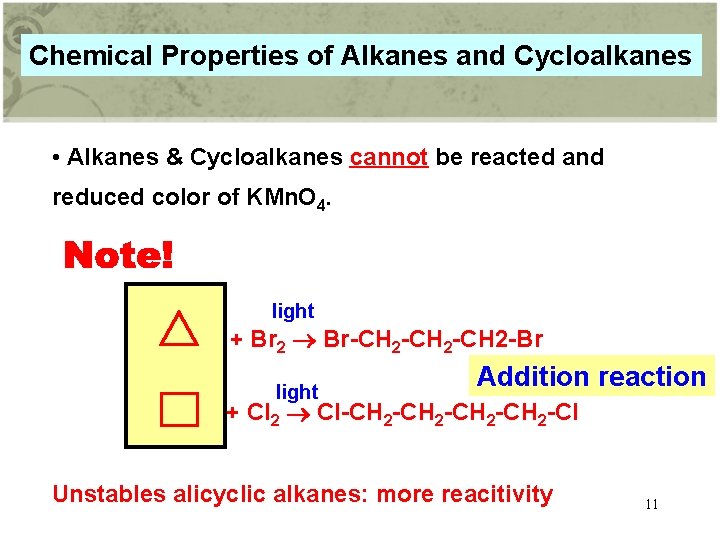
Chemical Properties of Alkanes and Cycloalkanes • Alkanes & Cycloalkanes cannot be reacted and reduced color of KMn. O 4. light + Br 2 Br-CH 2 -CH 2 -Br light Addition reaction + Cl 2 Cl-CH 2 -CH 2 -Cl Unstables alicyclic alkanes: more reacitivity 11

Physical & Chemical Properties Alkenes & Cycloalkenes 12

Physical Properties of Alkenes and Cycloalkenes • Boiling points and melting point of alkenes increase with number of carbons. If no. of carbon is same, alkenes have less boiling point and melting point than alkanes, i. e. , b. p. of C 4 H 10 >C 4 H 8 • Alkenes are non-water soluble compounds, but can soluble in non-polar solvent i. e. ether, hexane • Alkenes are non-conductive compounds. 13

Chemical Properties of Alkenes and Cycloalkenes 1. Combustion reaction 2 C 5 H 10 +15 O 2 10 CO 2 + 10 H 2 O +heat 2. Addition reaction addition at double bond of carbon atom 2. 1 Halogenation – halide addition, not required light CH 2=CH 2 + Cl 2 Cl-CH 2 -Cl 14

Chemical Properties of Alkenes and Cycloalkenes 2. 2 Hydrogenation – hydrogen addition with Ni or Pt acted as catalyst, alkanes will be obtained. Ni CH 3 -CH=CH 2 + H 2 CH 3 -CH-CH 2 H H 2. 3 Hydrohalogenation – acid halide addition and alkyl halide will be obtained CH 2=CH 2 + HCl H-CH 2 -Cl 15

Chemical Properties of Alkenes and Cycloalkenes 2. 4 Hydration – water addition with H 2 SO 4 acted as catalyst, alcohols will be obtained. H 2 SO 4 CH 2=CH 2 +H 2 O HO-CH 2 -OH 3. Oxidation react with KMn. O 4 (Oxidizing agent) in water, glycol will be obtained. 2 KMn. O 4 + 4 H 2 O + CH 3 -CH=CH-CH 3 3 CH 3 -CH-CH-CH 3 + 2 Mn. O 2 + 2 KOH OH OH 16

Physical & Chemical Properties Alkynes 17

Physical Properties of Alkynes • Boiling points and melting point of alkenes increase with number of carbons. If no. of carbon is same: boiling point of alkynes > alkanes > alkenes • Alkynes are non-water soluble compounds, but can soluble in non-polar solvent i. e. ether, hexane 18

Chemical Properties of Alkynes 1. Combustion reaction 2 C 2 H 2 +5 O 2 4 CO 2 + 2 H 2 O +heat 2. Addition reaction addition at triple bond of carbon atom 2. 1 Halogenation – halide addition, not required light CH CH + Cl 2 Cl-CH=CH-Cl + Cl 2 Cl-CH-CH-Cl Cl Cl 19
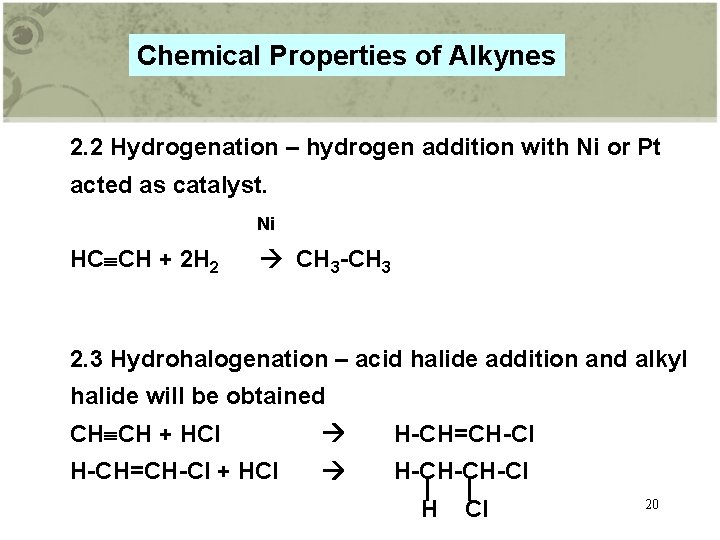
Chemical Properties of Alkynes 2. 2 Hydrogenation – hydrogen addition with Ni or Pt acted as catalyst. Ni HC CH + 2 H 2 CH 3 -CH 3 2. 3 Hydrohalogenation – acid halide addition and alkyl halide will be obtained CH CH + HCl H-CH=CH-Cl + HCl H-CH-CH-Cl H Cl 20
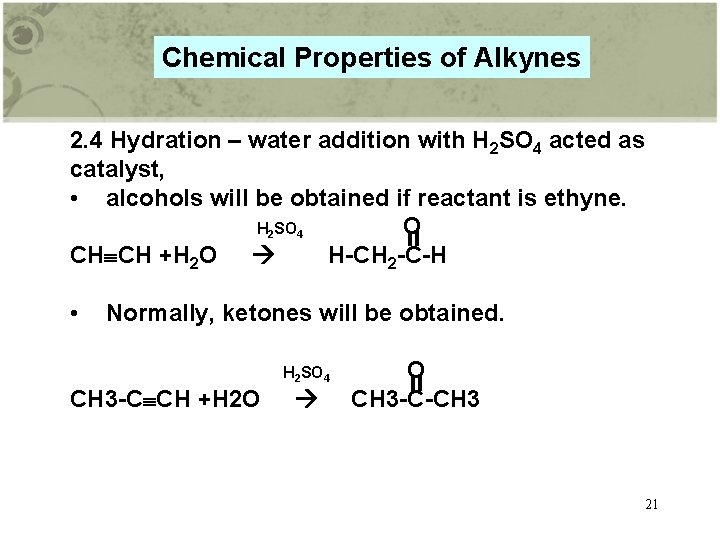
Chemical Properties of Alkynes 2. 4 Hydration – water addition with H 2 SO 4 acted as catalyst, • alcohols will be obtained if reactant is ethyne. H 2 SO 4 O CH CH +H 2 O H-CH 2 -C-H • Normally, ketones will be obtained. H 2 SO 4 CH 3 -C CH +H 2 O O CH 3 -C-CH 3 21

Chemical Properties of Alkynes 3. Oxidation react with KMn. O 4 (Oxidizing agent) in water • Carboxylic acid and CO 2 will be obtained for alkyne with primary carbon 3 CH CH + 8 KMn. O 4 + 4 H 2 O 3 HCOOH + 3 CO 2 + 8 Mn. O 2 + 8 KOH • Ketone will be obtained for alkyne with secondary carbon OO 3 RC CR + 4 KMn. O 4 +2 H 2 O 3 R-C-C-R + 4 Mn. O 2 + 4 KOH 22

Physical & Chemical Properties Aromatic compounds 23

Physical Properties of Aromatics • Non-water soluble compounds • Soluble in non-polar organic solvent i. e. ether • Low boiling point and melting point • Individual odor compounds 24
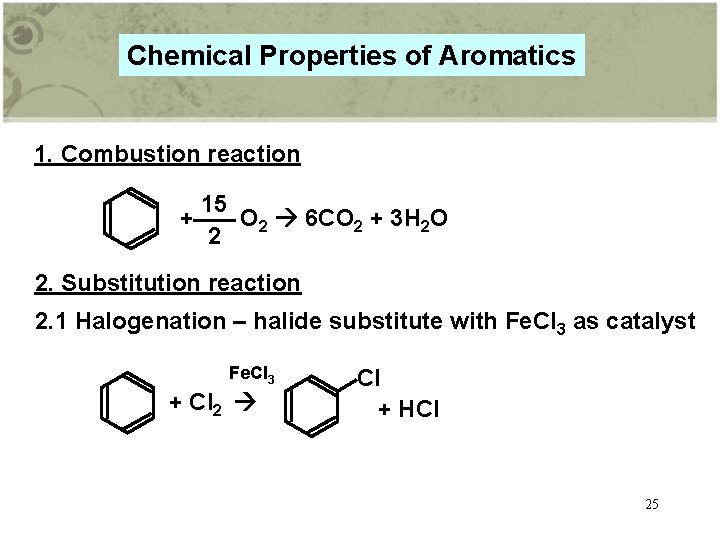
Chemical Properties of Aromatics 1. Combustion reaction 15 + O 2 6 CO 2 + 3 H 2 O 2 2. Substitution reaction 2. 1 Halogenation – halide substitute with Fe. Cl 3 as catalyst Fe. Cl 3 + Cl 2 Cl + HCl 25
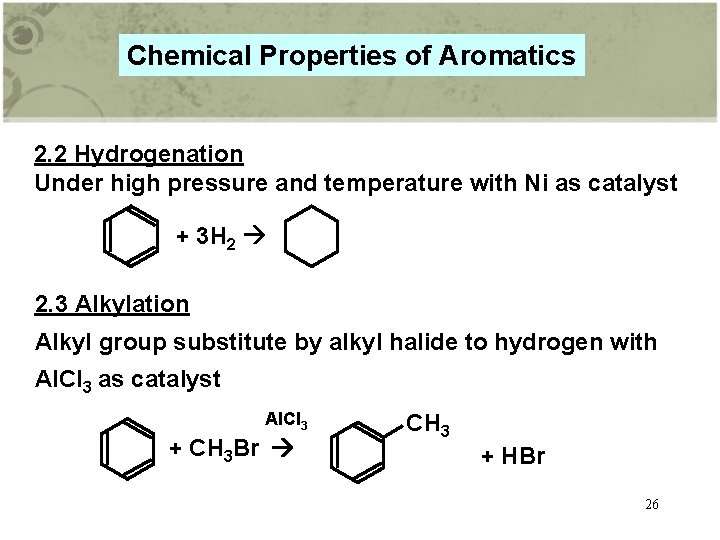
Chemical Properties of Aromatics 2. 2 Hydrogenation Under high pressure and temperature with Ni as catalyst + 3 H 2 2. 3 Alkylation Alkyl group substitute by alkyl halide to hydrogen with Al. Cl 3 as catalyst Al. Cl 3 + CH 3 Br CH 3 + HBr 26
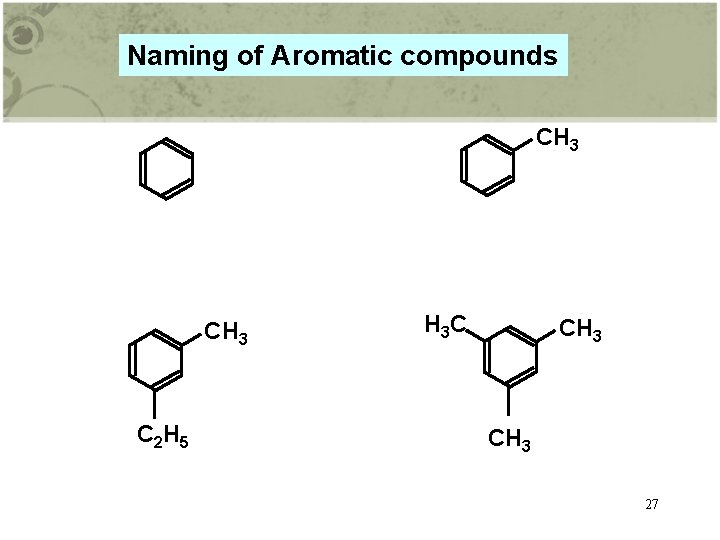
Naming of Aromatic compounds CH 3 C 2 H 5 H 3 C CH 3 27
Naming of Aromatic compounds 28
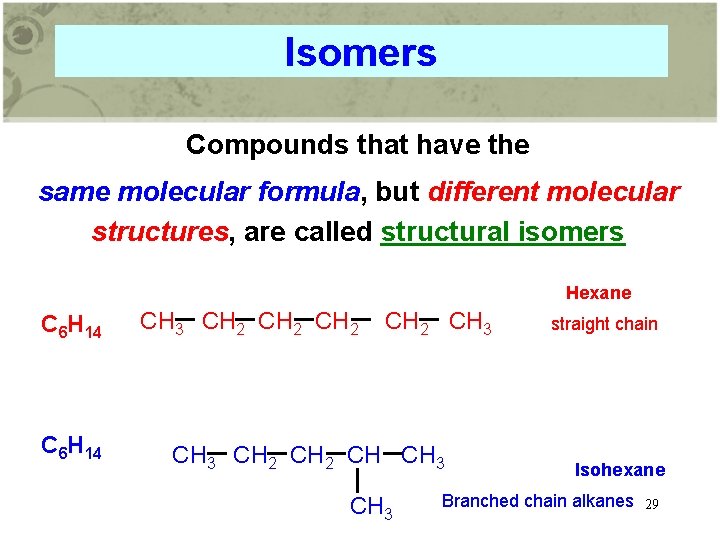
Isomers Compounds that have the same molecular formula, but different molecular structures, are called structural isomers Hexane C 6 H 14 CH 3 CH 2 CH 3 CH 2 CH CH 3 straight chain Isohexane Branched chain alkanes 29
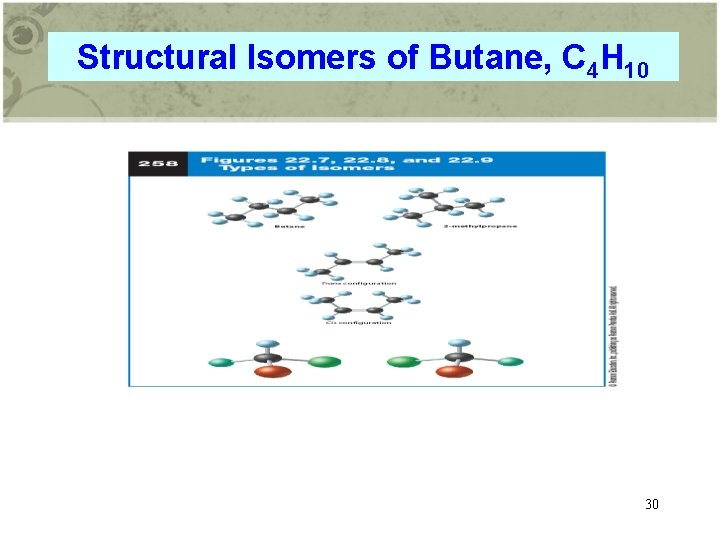
Structural Isomers of Butane, C 4 H 10 30
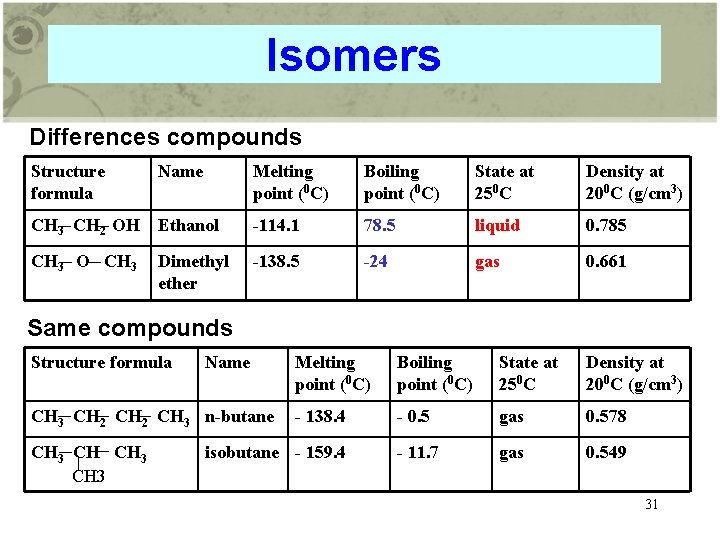
Isomers Differences compounds Structure formula Name Melting point (0 C) Boiling point (0 C) State at 250 C Density at 200 C (g/cm 3) CH 3 CH 2 OH Ethanol -114. 1 78. 5 liquid 0. 785 CH 3 O CH 3 -138. 5 -24 gas 0. 661 Dimethyl ether Same compounds Structure formula Name CH 3 CH 2 CH 3 n-butane CH 3 Melting point (0 C) Boiling point (0 C) State at 250 C Density at 200 C (g/cm 3) - 138. 4 - 0. 5 gas 0. 578 - 11. 7 gas 0. 549 isobutane - 159. 4 31

Isomers C 5 H 12 C 4 H 8 C 3 H 8 O Dichlorocyclomethane Trichlorocyclomethane 32
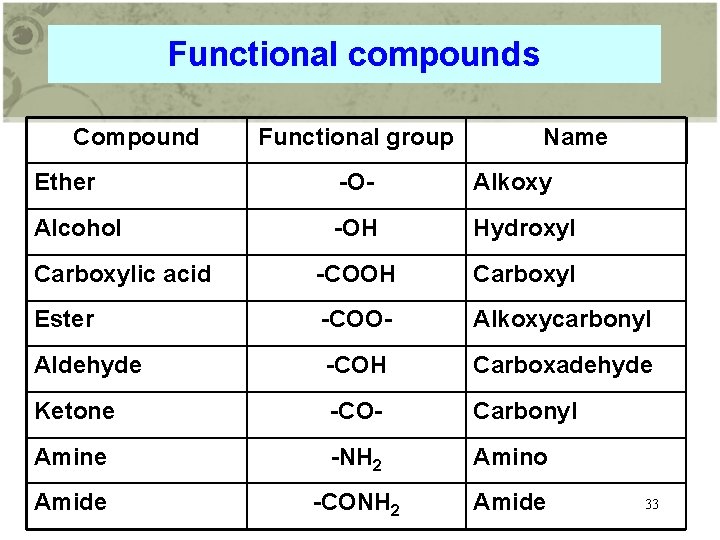
Functional compounds Compound Functional group Name Ether -O- Alkoxy Alcohol -OH Hydroxyl Carboxylic acid -COOH Carboxyl Ester -COO- Alkoxycarbonyl Aldehyde -COH Carboxadehyde Ketone -CO- Carbonyl Amine -NH 2 Amino Amide -CONH 2 Amide 33

Functional compounds Identify the functional group of following compounds CH 3 CH 2 COOH C 3 H 7 OH HCOH CH 3 COOCH 2 CH 3 34
- Slides: 34