Organic Chemistry Dr Walker What is Organic Chemistry










- Slides: 21

Organic Chemistry Dr. Walker

What is Organic Chemistry? • Organic chemistry is the study of carbon compounds. – Organic compounds have carbon AND hydrogen – Carbon Dioxide is NOT organic • The versatility and stability of carbon’s molecular structures provides the enormous range of properties of its compounds. • Carbon can bond to other carbons – Reason for the degree of structural complexity

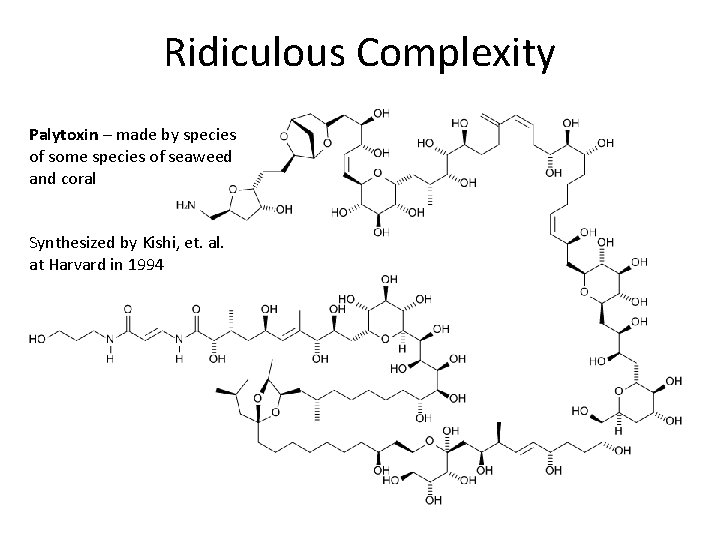
Ridiculous Complexity Palytoxin – made by species of some species of seaweed and coral Synthesized by Kishi, et. al. at Harvard in 1994

Organic Chemistry • Carbon – Has 4 valence electrons – Makes 4 covalent bonds to fill its octet • Can include double and triple bonds – Hydrocarbon • Compound with only carbon and hydrogen

Organic Chemistry in Everyday Life • • Smells & tastes: fruits, fish, mint Medications: aspirin, Tylenol, decongestants, sedatives, insulin Addictive substances: caffeine, nicotine, alcohol, narcotics Hormones/Neurotransmitters: adrenaline, dopamine, serotonin Food: carbohydrates, protein, fat, vitamins Genetics: DNA, RNA Consumer products: plastics, nylon, rayon, polyester

Organic Materials • Addition to previous notes – Before, it was stated that covalent compounds were mostly liquid/gas at room temp – This is primarily for binary compounds (2 elements) – More complicated structures can be solid at room temp

General Formulas • Alkanes – Contains only single bonds – Cx H 2 x+2 • Alkenes – Contains at least one double bond • Alkynes – Contains at least one triple bond • Alkenes and Alkynes are more reactive than alkanes

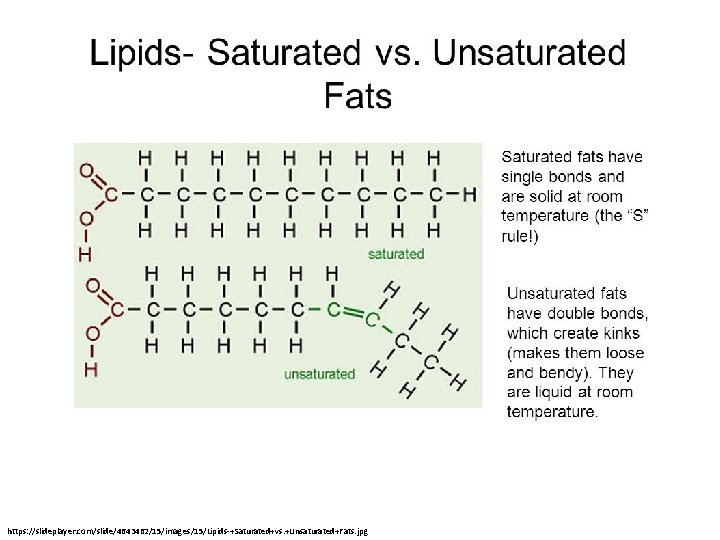
Saturation • Saturated: a carbon chain contains as many hydrogens as possible. • Saturated compounds contain only single C-H bonds. Alkanes are saturated. • Unsaturated means that a carbon chain contains at least one multiple (double or triple) bond. – Alkene = double bond – Alkyne = triple bond

https: //slideplayer. com/slide/4643462/15/images/15/Lipids-+Saturated+vs. +Unsaturated+Fats. jpg

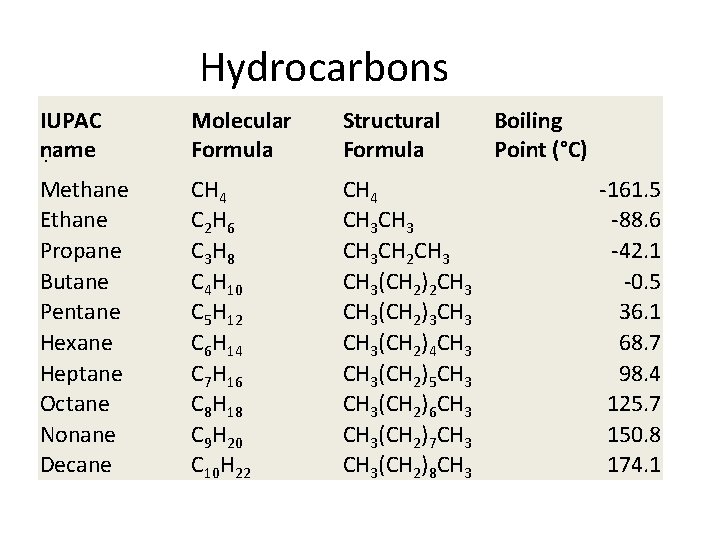
Hydrocarbons IUPAC name. Molecular Formula Structural Formula Methane Ethane Propane Butane Pentane Hexane Heptane Octane Nonane Decane CH 4 C 2 H 6 C 3 H 8 C 4 H 10 C 5 H 12 C 6 H 14 C 7 H 16 C 8 H 18 C 9 H 20 C 10 H 22 CH 4 CH 3 CH 2 CH 3(CH 2)3 CH 3(CH 2)4 CH 3(CH 2)5 CH 3(CH 2)6 CH 3(CH 2)7 CH 3(CH 2)8 CH 3 Boiling Point (°C) -161. 5 -88. 6 -42. 1 -0. 5 36. 1 68. 7 98. 4 125. 7 150. 8 174. 1

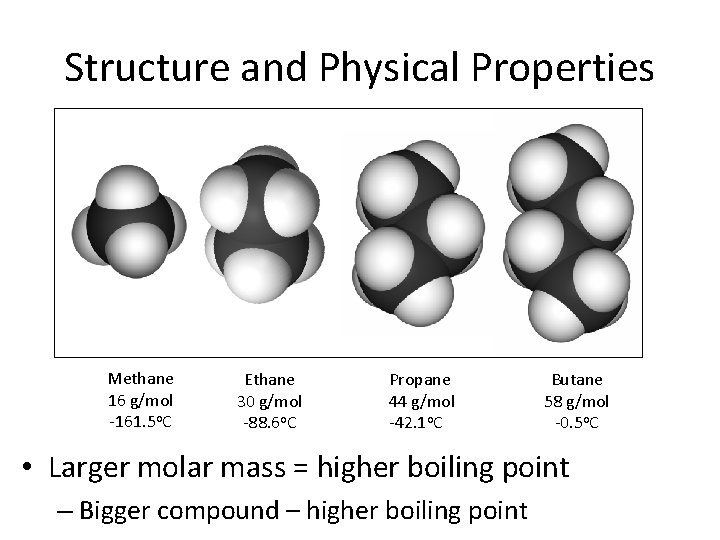
Structure and Physical Properties Methane 16 g/mol -161. 5 o. C Ethane 30 g/mol -88. 6 o. C Propane 44 g/mol -42. 1 o. C Butane 58 g/mol -0. 5 o. C • Larger molar mass = higher boiling point – Bigger compound – higher boiling point

Petrochemicals • Simple, small hydrocarbons are petrochemicals – Chemicals isolated from crude oil (petroleum) • Octane – used in gasoline • Propane – used in grills, heaters, etc. • Butane – lighter fluid

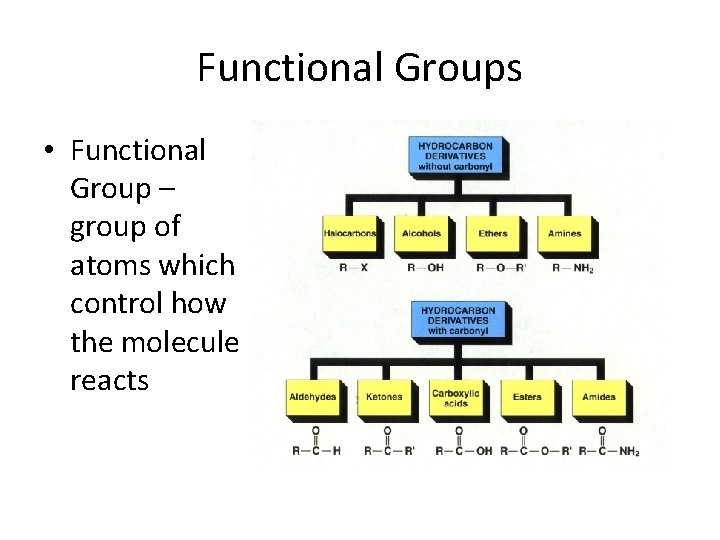
Functional Groups • Functional Group – group of atoms which control how the molecule reacts

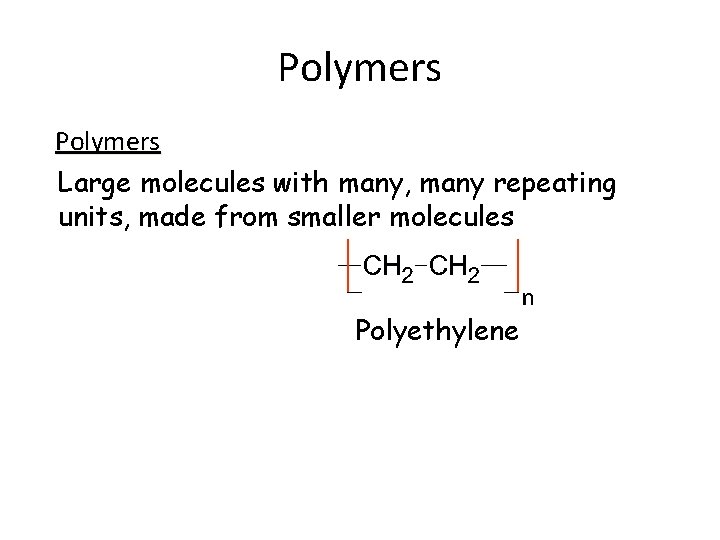
Polymers Large molecules with many, many repeating units, made from smaller molecules Polyethylene

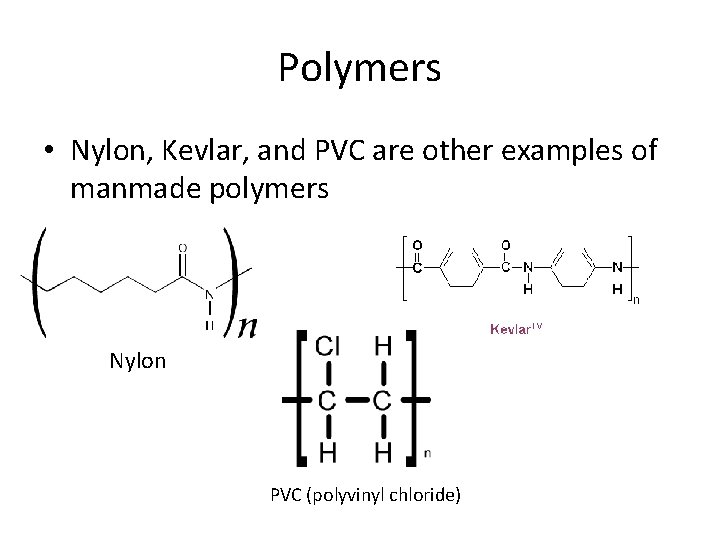
Polymers • Nylon, Kevlar, and PVC are other examples of manmade polymers Nylon PVC (polyvinyl chloride)

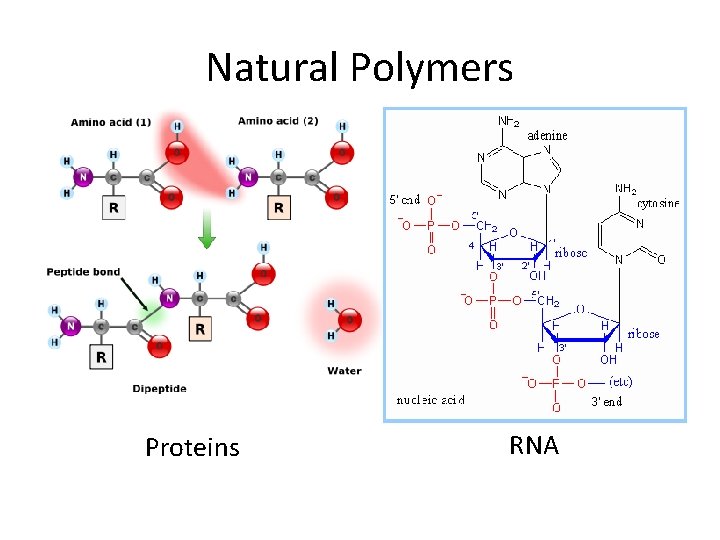
Natural Polymers Proteins RNA

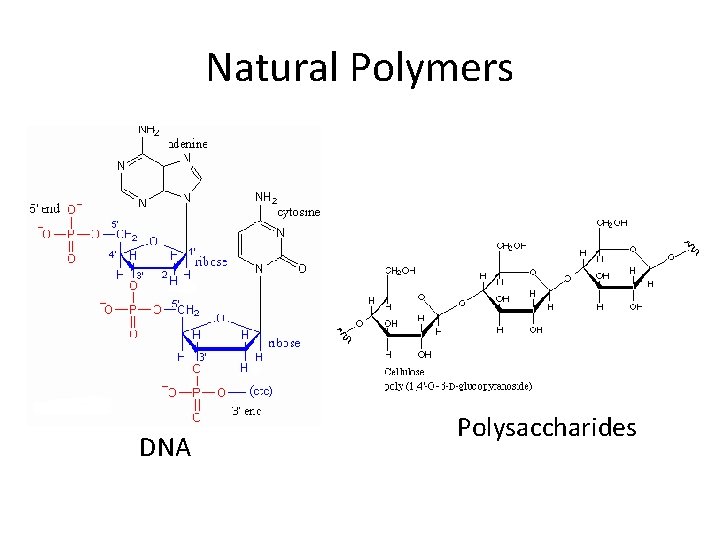
Natural Polymers DNA Polysaccharides

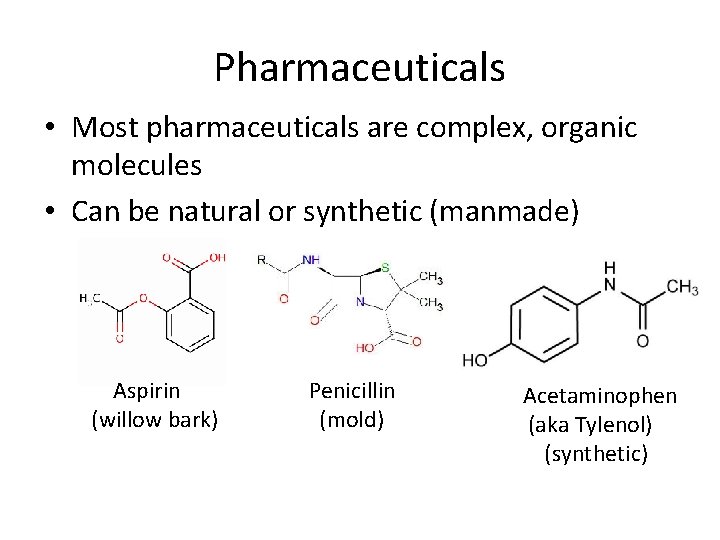
Pharmaceuticals • Most pharmaceuticals are complex, organic molecules • Can be natural or synthetic (manmade) Aspirin (willow bark) Penicillin (mold) Acetaminophen (aka Tylenol) (synthetic)

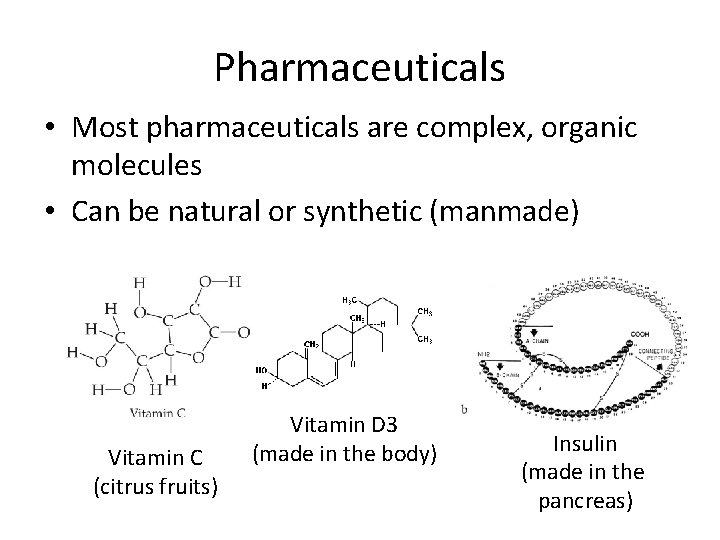
Pharmaceuticals • Most pharmaceuticals are complex, organic molecules • Can be natural or synthetic (manmade) Vitamin C (citrus fruits) Vitamin D 3 (made in the body) Insulin (made in the pancreas)

Terms To Know • • • Hydrocarbon Alkane Alkene Alkyne Saturated Unsaturated Petrochemicals Functional Group Polymers

Skills To Master • Differentiating alkanes, alkenes, and alkynes • Differentiating saturated and unsaturated compounds • Differentiating natural polymers and manmade polymers • Recognizing common pharmaceuticals