Organic Chemistry Chapter 3 Part II Addition to










- Slides: 14

Organic Chemistry Chapter 3 Part II Addition to Alkenes - Markovnikov’s rule. Nanoplasmonic Research Group

Reactions of Alkenes: Addition • Electrophile – Literally ‘ELECTRON’ lovers: electron-POOR reagents – Positive ions or electron-deficient species • Nucleophile – Literally ‘NUCLEUS’ lovers: electron-RICH reagents – Donate electrons to an electrophile E+ + Nu- E Nu

Addition of Unsymmetric Reagents to Unsymmetric Alkenes: Markovnikov’s Rule

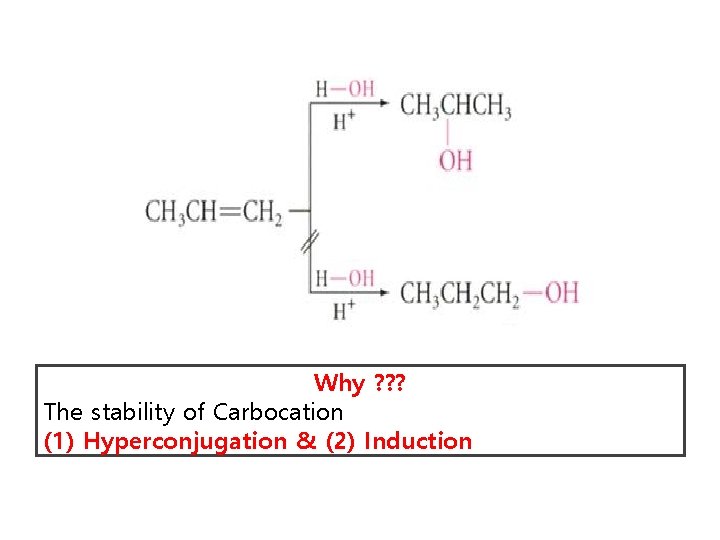
Markovnikov’s Rule The RULE says “The addition of a protic acid such as H-X to an alkene, the acid hydrogen (H) becomes attached to the carbon atom with the greatest number of hydrogens, and the halide (X) group becomes attached to the carbon with the fewest hydrogens”

Why ? ? ? The stability of Carbocation (1) Hyperconjugation & (2) Induction

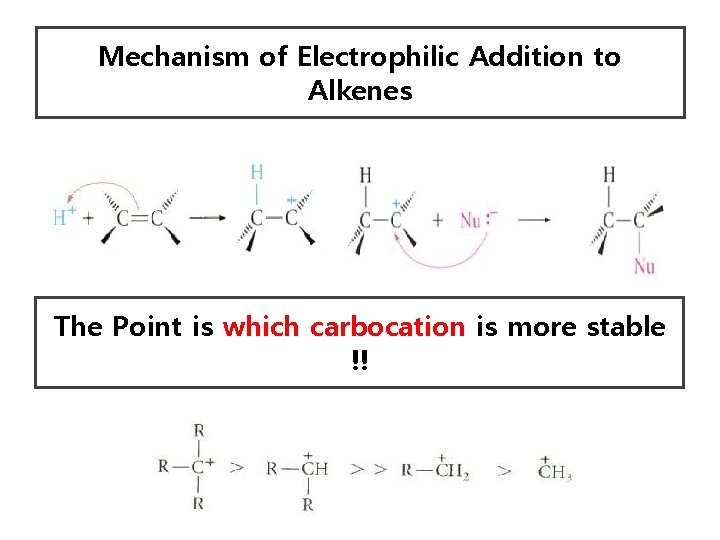
Mechanism of Electrophilic Addition to Alkenes The Point is which carbocation is more stable !!

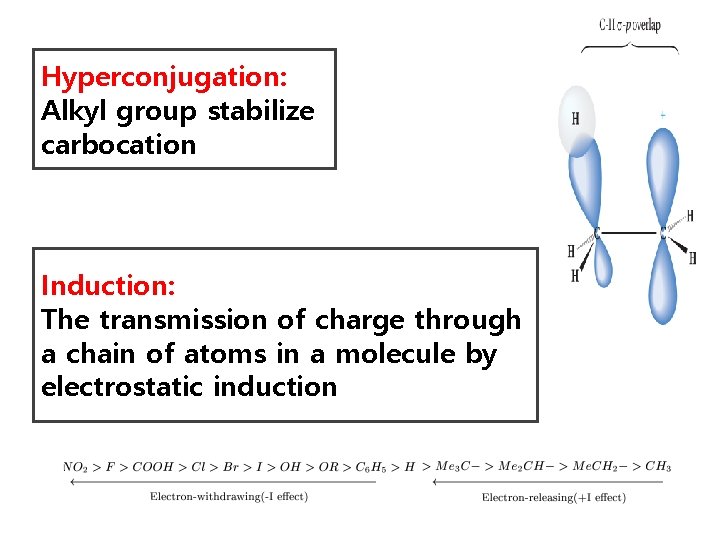
Hyperconjugation: Alkyl group stabilize carbocation Induction: The transmission of charge through a chain of atoms in a molecule by electrostatic induction

Types of Addition Rxns • Addition of Halogens • Addition of Water (hydration) • Addition of Acids

Hydroboration • Addition of a hydrogen-boron bond to an alkene • The Point is that ‘hydrogen’ is more electronegative than ‘boron’

Quick Reminder • Alkene Reaction goes through the pathway of the most stable intermediate • The more substituents on an electron deficient carbon (carbocation or radical), the greater its stability

Additions to Conjugated Systems 1, 2 -addition 1, 4 -addition How to Predict ? 1. Electrophile adds to alkene according to Markovnikov’s rule 2. Draw possible resonance structures and see how many structures come out 3. Selectivity may differ depending on the stability of each resonance structure

Cycloaddition to Conjugated Dienes - The Diels-Alder Reaction - Features 1. Concerted as with hydroboration 2. Easy to synthesize a cyclic compound 3. Converts three pi bonds to two sigma bonds & one new pi bond 4. HOMO of diene and LUMO of dienophile

Free-Radical Additions Features 1. Initiation by a radical initiator: A radical is created from a non-radical precursor 2. Chain propagation: A radical reacts with a nonradical to produce a new radical species 3. Chain termination: Two radicals react with each other to create a non-radical species 4. Chain branching: see page 102 middle

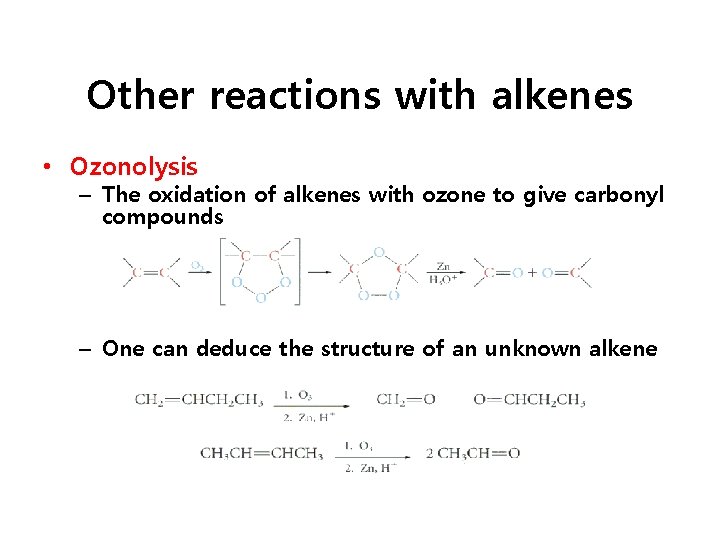
Other reactions with alkenes • Ozonolysis – The oxidation of alkenes with ozone to give carbonyl compounds – One can deduce the structure of an unknown alkene