Organic Chemistry Chapter 22 Vocabulary Organic Chemistry Hydrocarbons
Organic Chemistry Chapter 22

Vocabulary • • Organic Chemistry Hydrocarbons Saturated Unsaturated Alkanes Alkenes Alkynes Cis-trans isomerism • • Carbonyl group Ketones Aldehydes Carboxylic acids Carboxyl group Ester Ether Amine

Saturated vs. Unsaturated Hydrocarbons • Hydrocarbons are molecules composed of carbon & hydrogen – Each carbon atom forms 4 chemical bonds – A saturated hydrocarbon is one where all C - C bonds are “single” bonds & the molecule contains the maximum number of H-atoms – An unsaturated hydrocarbon is one where at least 1 C=C bond is double.

Straight-Chain Alkanes • Straight-chain alkanes contain any number of carbon atoms, one after the other, in a chain pattern - meaning one linked to the next (not always straight) C-C-C-C etc.
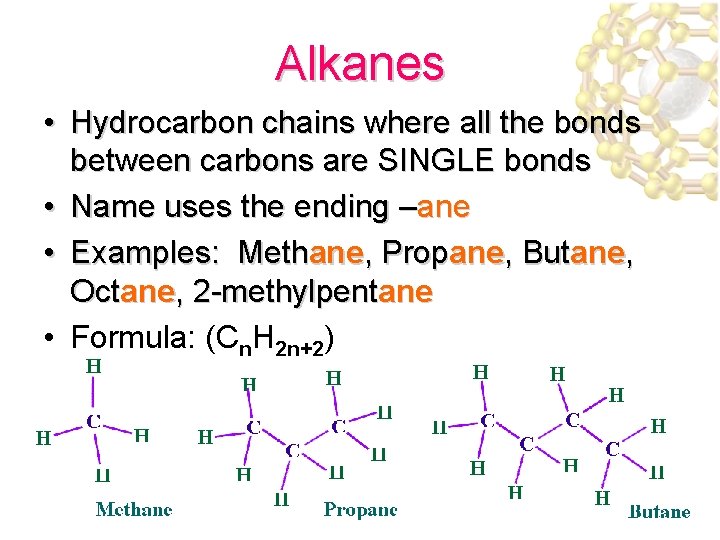
Alkanes • Hydrocarbon chains where all the bonds between carbons are SINGLE bonds • Name uses the ending –ane • Examples: Methane, Propane, Butane, Octane, 2 -methylpentane • Formula: (Cn. H 2 n+2)

Prefixes for # of Carbons 1 Meth 6 Hex 2 Eth 7 Hept 3 Prop 8 Oct 4 But 9 Non 5 Pent 10 Dec
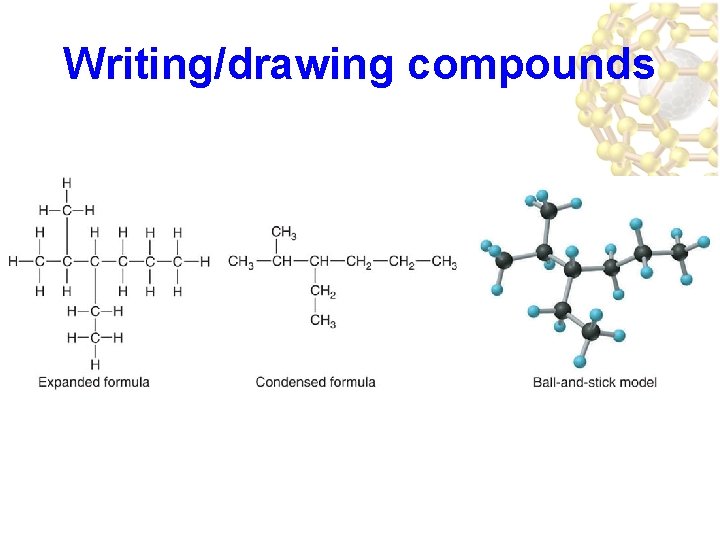
Writing/drawing compounds
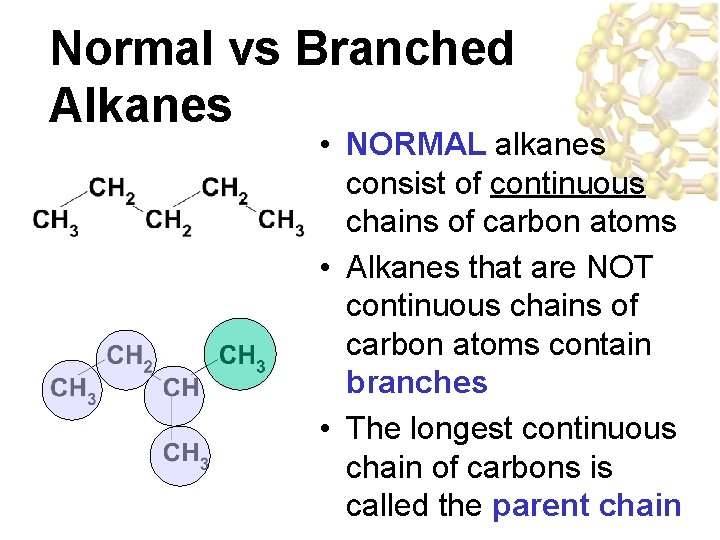
Normal vs Branched Alkanes • NORMAL alkanes consist of continuous chains of carbon atoms • Alkanes that are NOT continuous chains of carbon atoms contain branches • The longest continuous chain of carbons is called the parent chain

Endings • Attached carbon groups (substituents) end in –yl – Methyl CH 3 – Ethyl CH 3 CH 2– Propyl CH 3 CH 2 – – 3 ethylpentane

Names of branches • Carbon (alkyl) groups – Methyl CH 3 – Ethyl CH 3 CH 2– Propyl CH 3 CH 2 – • Halogens – Fluoro (F-) – Chloro (Cl-) – Bromo (Br-) – Iodo (I-)

Branched-Chain Alkanes • Rules for naming – 1. Longest C-C chain is parent 2. Number so branches have lowest # 3. Give position number to branch 4. Prefix (di, tri) more than one branch 5. Alphabetize branches (not prefix) 6. Use proper punctuation ( - and , )

Some Simple Alkanes • 2 -methylpentane • 3 -ethylhexane • 2, 2 -dimethylbutane • 2, 3 -dimethylbutane

Branched-Chain Alkanes • From the name, draw the structure, in a right-to-left manner: 1. Find the parent, with the -ane 2. Number carbons on parent 3. Identify substituent groups (give lowest number); attach 4. Add remaining hydrogens
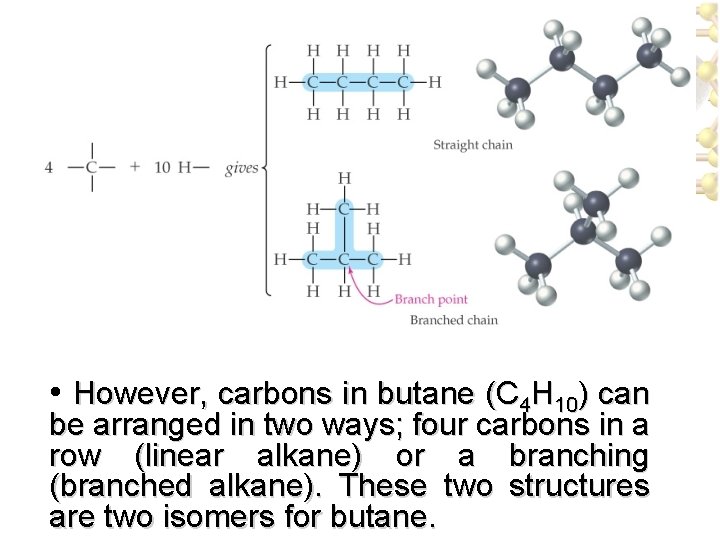
• However, carbons in butane (C 4 H 10) can be arranged in two ways; four carbons in a row (linear alkane) or a branching (branched alkane). These two structures are two isomers for butane.
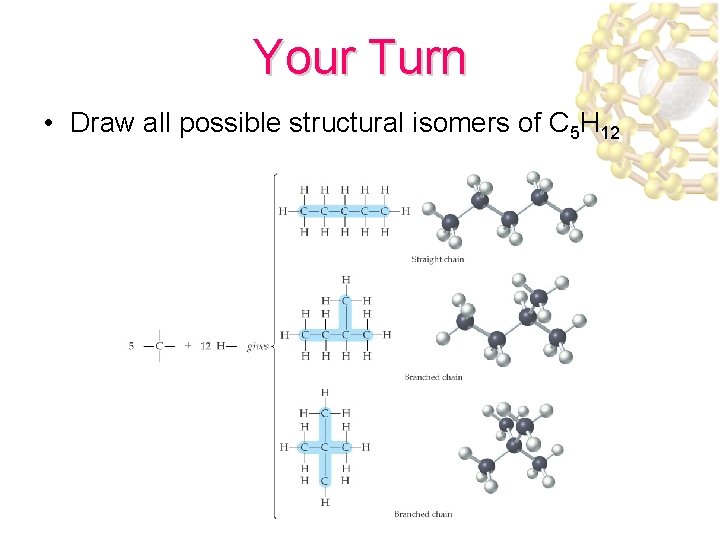
Your Turn • Draw all possible structural isomers of C 5 H 12

Structural Isomerism • Structural isomers are molecules with the same chemical formulas but different molecular structures n-pentane, C 5 H 12 2 -methlbutane, C 5 H

IUPAC Rules for Naming Branched Alkanes – Find and name the parent chain in the Find and name the parent chain hydrocarbon - this forms the root of the hydrocarbon name – Number the carbon atoms in the parent chain starting at the end closest to the branching – Name alkane branches by dropping the “ane” Name alkane branches from the names and adding “yl”. A one-carbon branch is called “methyl”, a two-carbon branch is “ethyl”, etc… – When there are more than one type of branch (ethyl and methyl, for example), they are named
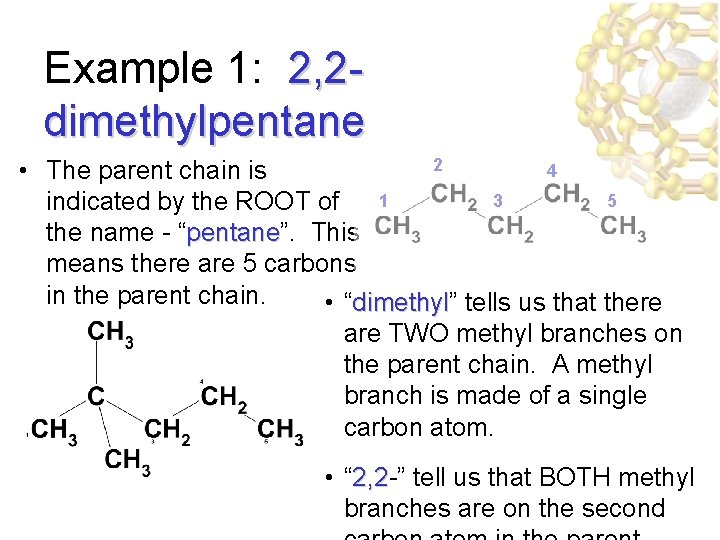
Example 1: 2, 2 dimethylpentane 2 4 • The parent chain is 3 5 indicated by the ROOT of 1 the name - “pentane”. This pentane means there are 5 carbons in the parent chain. • “dimethyl” tells us that there dimethyl are TWO methyl branches on the parent chain. A methyl branch is made of a single carbon atom. • “ 2, 2 -” tell us that BOTH methyl 2, 2 branches are on the second

Example 2: 3 -ethyl-2, 4 dimethylheptane 2 • The parent chain is 1 indicated by the ROOT of the name - “heptane”. This heptane means there are 7 carbons in the parent chain. 3 4 5 6 7 • “ 2, 4 -dimethyl” tells us there 2, 4 -dimethyl are TWO methyl branches on the parent chain, at carbons #2 and #4. • “ 3 -ethyl-” tell us there is an 3 -ethyl branch (2 -carbon branch) on carbon #3 of the

Example 3: 2, 3, 3 -trimethyl-4 propyloctane • The parent chain is indicated by the ROOT of the name - “octane”. This octane means there are 8 carbons in the parent chain. 2 1 3 5 4 6 7 8 3 2 1 5 4 6 7 8 • “ 2, 3, 3 -trimethyl” tells us there 2, 3, 3 -trimethyl are THREE methyl branches - one on carbon #2 and two on carbon #3. • “ 4 -propyl-” tell us there is a 4 -propyl branch (3 -carbon branch) on carbon #4 of the

Example 4: Name the molecules shown! • parent chain has 5 carbons - “pentane” • two methyl branches - start counting from the right - #2 and #3 • 2, 3 -dimethylpentane • parent chain has 8 carbons - 3 4 5 “octane” • two methyl branches - start counting from the left - #3 and #4 • one ethyl branch - #5 3, 4 -dimethyl octane 5 -ethyl • name branches alphabetically

Alkenes & Alkynes • Alkenes are • The suffix for the hydrocarbons that parent chains are contain at least one changed from “ane” carbon-carbon to “ene” and “yne” double bond – e. g. ethene, propyne • Alkynes are • the BONDS are hydrocarbons that numbered like contain at least one branches so that the carbon-carbon triple location of the bond multiple bond may be indicated
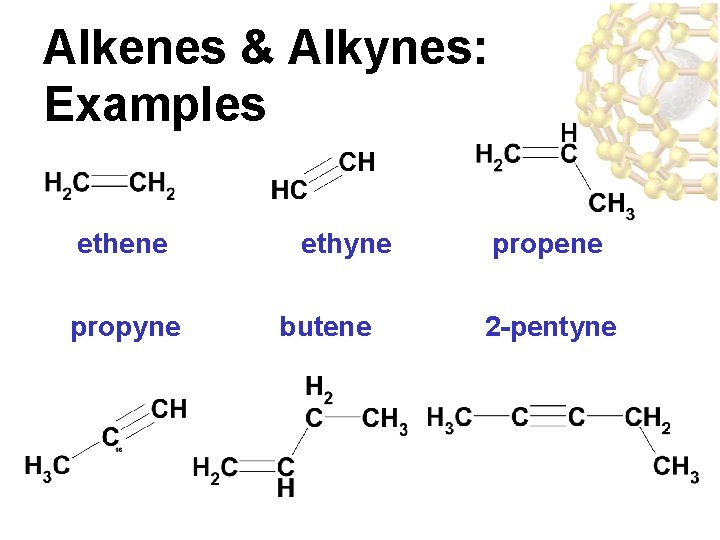
Alkenes & Alkynes: Examples ethene propyne ethyne butene propene 2 -pentyne

Aromatic Hydrocarbons Cycloalkanes • A cycloalkane is made of a hydrocarbon chain that has been joined to make a “ring”. • Note that two hydrogen atoms were lost in forming the ring!
Aromatic Hydrocarbons Cycloalkanes • The two ends of the carbon chain are attached in a ring in a cyclic hydrocarbon – named as “cyclo- ____”

Aromatic Hydrocarbons • Benzene is a sixcarbon ring, with alternating double and single bonds

Aromatic Compounds and Benzene Aromatic compounds contain benzene. Benzene, C 6 H 6 , is represented as a six carbon ring with 3 double bonds. Two possible resonance structures can be drawn to show benzene in this form.

Aromatic Hydrocarbons • Benzene derivatives can have two C or more substituents: – 1, 2 -dimethylbenzene – 1, 3 -dimethylbenzene – 1, 4 -dimethylbenzene C C C • Can use ortho for 1, 2; meta for 1, 3; and para for 1, 4 (page 711)

Isomers With organic compounds

Isomers • There is a lack of rotation around a carbon to carbon multiple bond – Two possible arrangements: 1. trans configuration - substituted groups on opposite sides of double bond 2. cis configuration - same side
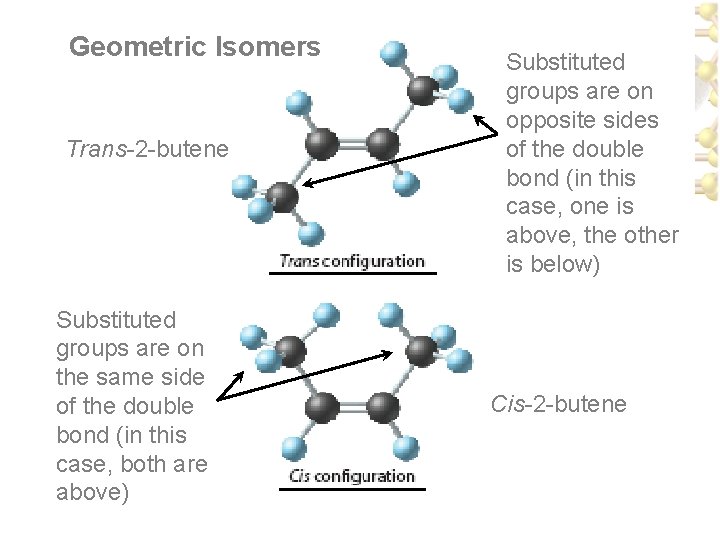
Geometric Isomers Trans-2 -butene Substituted groups are on the same side of the double bond (in this case, both are above) Substituted groups are on opposite sides of the double bond (in this case, one is above, the other is below) Cis-2 -butene

Cis-Trans Isomers Examples cis-1, 3 -dimethylcyclobutane cis-1, 2 -dichlorocyclohexane trans-1 -ethyl-2 -methylcyclopro

Reactions With organic compounds
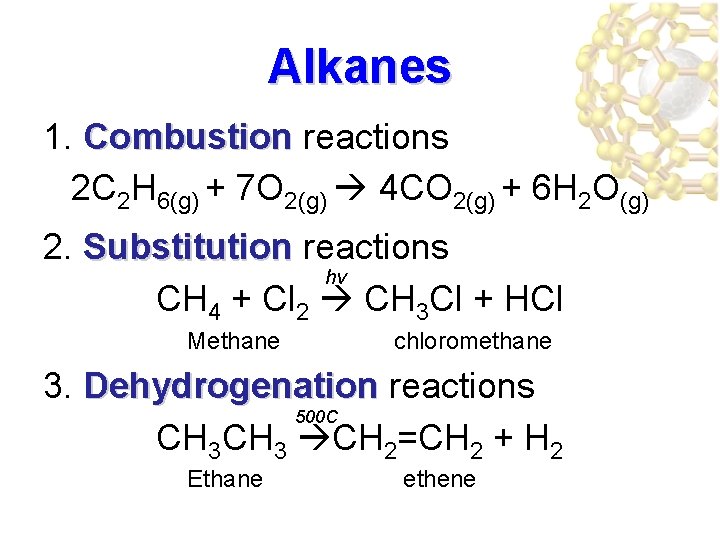
Alkanes 1. Combustion reactions 2 C 2 H 6(g) + 7 O 2(g) 4 CO 2(g) + 6 H 2 O(g) 2. Substitution reactions Substitution hv CH 4 + Cl 2 CH 3 Cl + HCl Methane chloromethane 3. Dehydrogenation reactions Dehydrogenation 500 C CH 3 CH 2=CH 2 + H 2 Ethane ethene
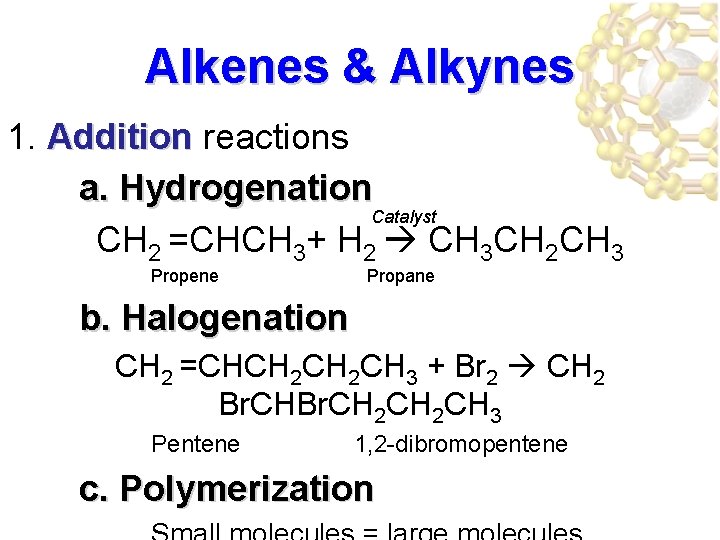
Alkenes & Alkynes 1. Addition reactions a. Hydrogenation Catalyst CH 2 =CHCH 3+ H 2 CH 3 CH 2 CH 3 Propene Propane b. Halogenation CH 2 =CHCH 2 CH 3 + Br 2 CH 2 Br. CH 2 CH 3 Pentene 1, 2 -dibromopentene c. Polymerization

Aromatic 1. Substitution reactions Catalyst = Fe. Cl + Cl 2 3 + HCl

Functional Groups With organic compounds

Functional Groups u. Functional group: an Functional group: atom or group of atoms within a molecule
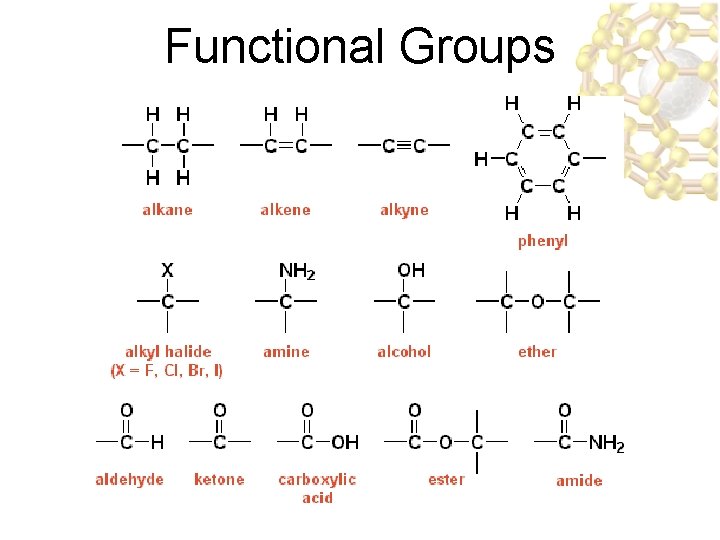
Functional Groups
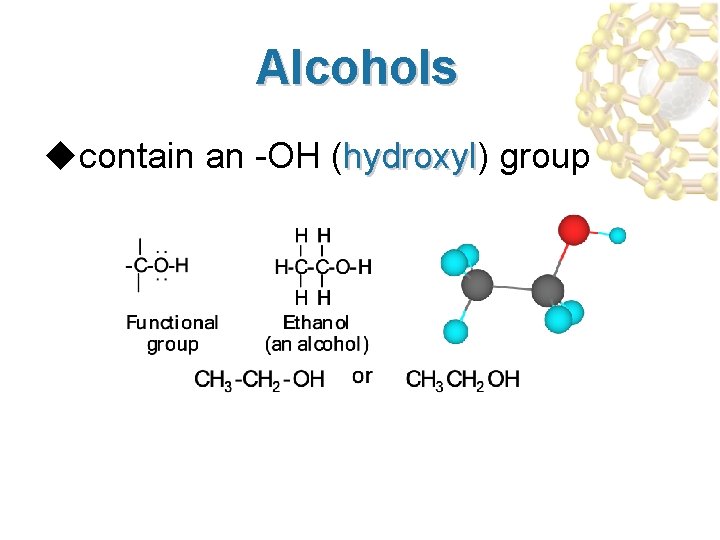
Alcohols ucontain an -OH (hydroxyl) group hydroxyl

Halides & Carboxylic Acids ucontain an -X (Halogen) group Halogen u. F, Cl, Br, I, At ucontain a carboxyl (-COOH) group
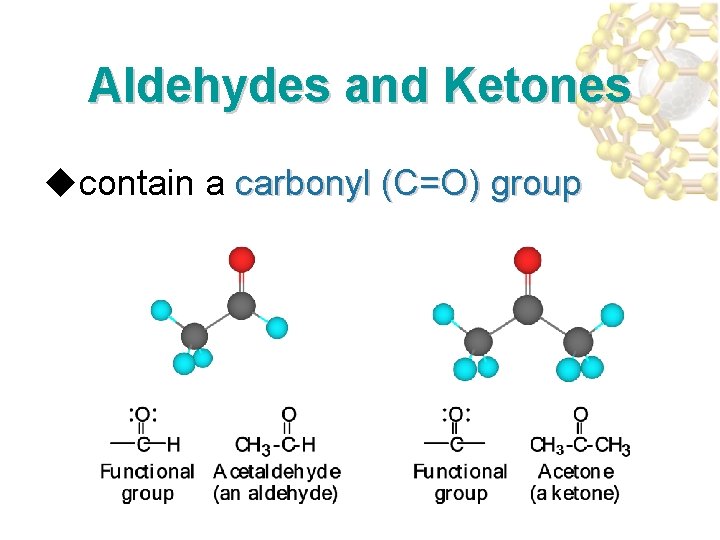
Aldehydes and Ketones ucontain a carbonyl (C=O) group
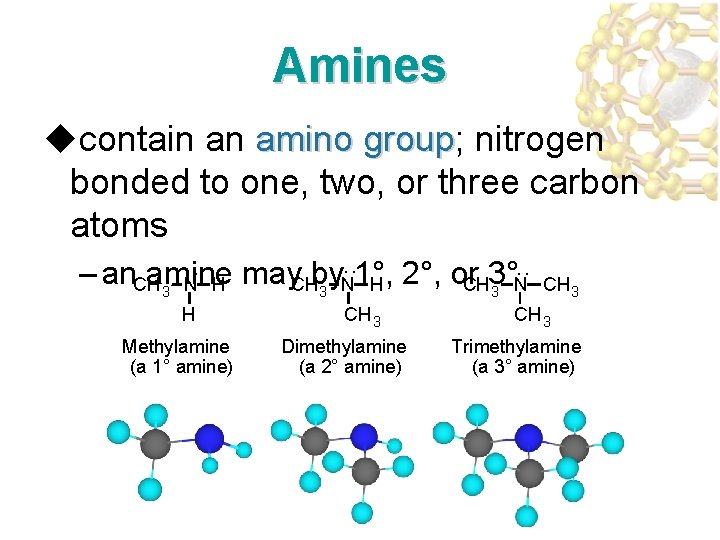
Amines ucontain an amino group; nitrogen amino group bonded to one, two, or three carbon atoms : : : – an amine may by 1°, 2°, or 3° CH 3 N H CH 3 Methylamine (a 1° amine) Dimethylamine (a 2° amine) CH 3 Trimethylamine (a 3° amine)
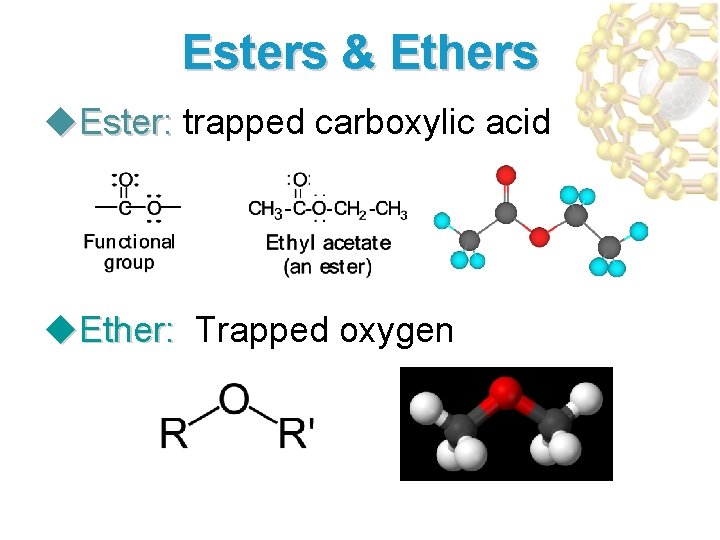
Esters & Ethers u. Ester: trapped carboxylic acid Ester: u. Ether: Trapped oxygen Ether:
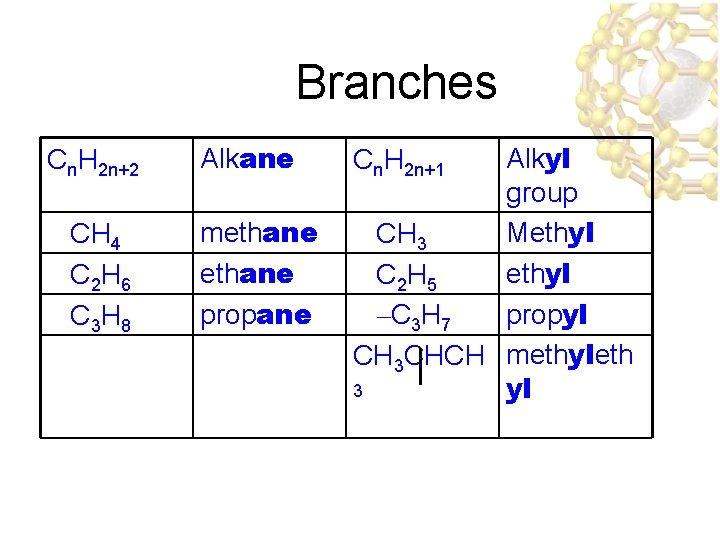
Branches Cn. H 2 n+2 Alkane CH 4 C 2 H 6 C 3 H 8 methane propane Alkyl group Methyl CH 3 ethyl C 2 H 5 propyl –C 3 H 7 CH 3 CHCH methyleth 3 yl Cn. H 2 n+1
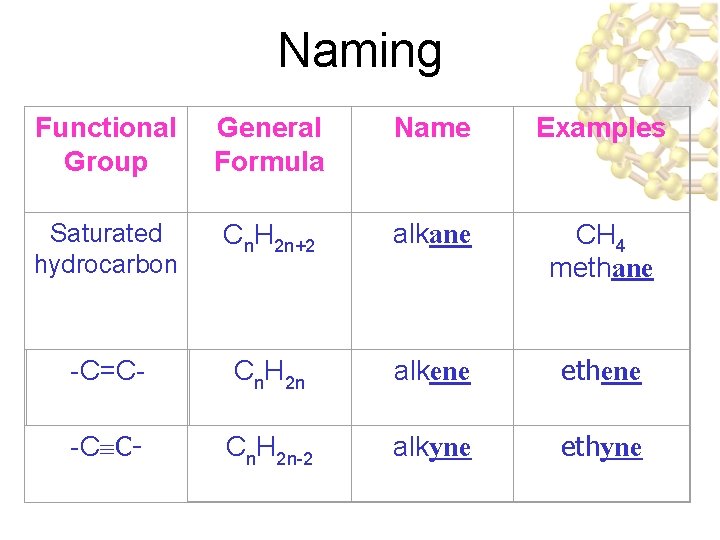
Naming Functional Group General Formula Name Examples Saturated hydrocarbon Cn. H 2 n+2 alkane CH 4 methane -C=C- Cn. H 2 n alkene ethene -C C- Cn. H 2 n-2 alkyne ethyne
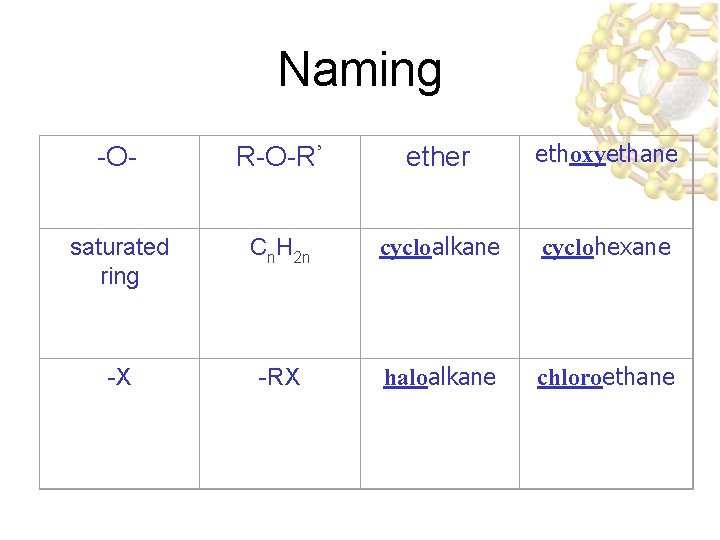
Naming -O- R-O-R’ ether ethoxyethane saturated ring Cn. H 2 n cycloalkane cyclohexane -X -RX haloalkane chloroethane
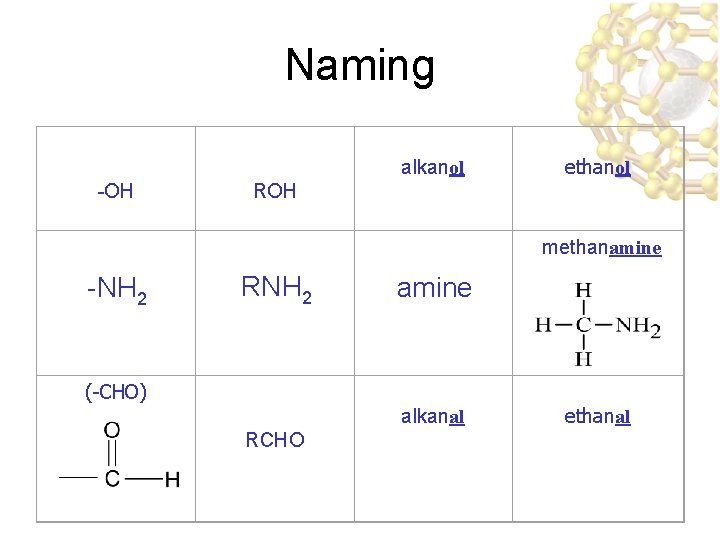
Naming alkanol -OH ethanol ROH methanamine -NH 2 RNH 2 amine (-CHO) alkanal RCHO ethanal
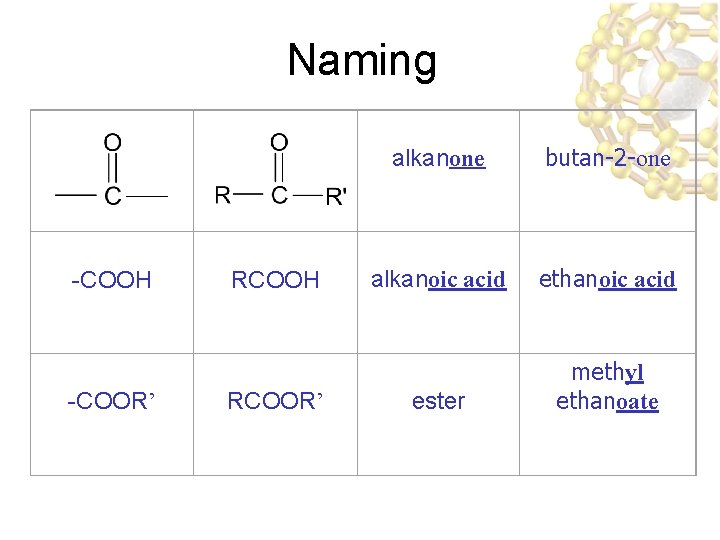
Naming -COOH -COOR’ RCOOH RCOOR’ alkanone butan-2 -one alkanoic acid ethanoic acid ester methyl ethanoate
- Slides: 49