Organic Chemistry Ch 19 w Organic chemistry is






























































- Slides: 65

Organic Chemistry : Ch. 19 w Organic chemistry is the branch of chemistry which deals with “carbon based” compounds. w Carbon is an element which has the unique ability to bond long chains, rings, and react with other elements to form a variety of compounds.

Carbon Compounds w Carbon compounds are found in many forms : gases, aqueous, and solids. w Carbon is recycled in nature by the carbon cycle. w Carbon chains can be modified by reactions to form different type of carbon based (organic) compounds.

Story of Carbon w Carbon is a major component of living things. w Lipids (fats and oils), sugars, starches, proteins, DNA, etc. are composed of carbon, hydrogen, oxygen, nitrogen, and other atoms. w Lipids, sugars, and starches are composed of carbon, hydrogen, and oxygen. w Proteins are composed of carbon, hydrogen, oxygen, nitrogen, and sometimes sulfur.

Story of Carbon w When organisms die and decompose in a low oxygen environment, these carbon compounds(with the aide of anaerobic bacteria) will break off the oxygen, nitrogen, and other atoms bonded to the compounds. Animal Fat (one molecule)

Story of Carbon w When the oxygen, nitrogen, sulfur, and other atoms are removed by bacteria, what is left is a hydrocarbon ( Carbon and hydrogen only) w These hydrocarbons are of mixed sizes, and are known as “crude oil” when recovered from the earth. Hydrocarbons : C & H atoms

Story of Carbon w Carbon atoms have four electrons in their outer energy level, and will want to covalently bond (share electrons) with itself and other atoms to form more stable chain and ring compounds. w The crude oil contains a mixture of hydrocarbons including : butane(lighter fluid), gasoline, diesel, candle wax, etc.

Story of Carbon w The hydrocarbons found in crude oil can be separated based upon their boiling points and refined or distilled. w By adding substances to them a host of other organic compounds can be produced : (alcohols, esters, ethers, plastic, styrofoam, etc. )

Hydrocarbons w Hydrocarbons are a class of organic compounds which contain only carbon and hydrogen. w Hydrocarbons are characterized as carbon chains, branched chains, and rings.

Hydrocarbons w Carbon has 4 valence electrons and will form 4 covalent bonds with hydrogen. A line between carbon and another atom represents the sharing of 2 electrons. Therefore every carbon in an organic compound needs 4 lines around it. w Hydrocarbons with all single carbon bonds are described as the Alkanes. w http: //www. nyu. edu/pages/mathmol/library/hydrocarbons/

Journal Activity #1 w In your journal prepare a data table which shows the following : Fill in the information for methane through decane. Use the website given to find the uses for. Alkane Name Structural Formula Molecular Formula Uses For

Hydrocarbons : Alkenes w Carbon atoms have the ability to bond with other atoms with single, double, or even triple bonds. w When carbon bonds to another carbon with a double bond, and only carbon and hydrogen atoms are in a compound it’s termed an alkene. w Ethylene(ethene) is the simplest alkene. http: //users. rcn. com/jkimball. ma. ultranet/Biology. Pages/E/ Ethylene. html

Journal Activity #2 • In your journal fill in the data table below for the alkenes ( ethene – octene) Name Structural Formula Molecular Formula Uses

Alkynes w Alkynes are hydrocarbons which contain a triple bond between carbons. w Alkynes end in “yne”. w Ethyne, Propyne, Butyne, etc.

Journal Activity #3 w Diagram the following hydrocarbons: a) Propene b) Butyne c) 2 hexene d) 2 - octyne

Journal Activity #4 w http: //www. ausetute. com. au/ethene. html • Using the website above to answer the questions below in regards to ethene or ethylene. Explain how ethene can be made into ethanol. Explain how ethene can be made into ethylene glycol (antifreeze). What is ethene extracted from? Describe one other chemical which can be produced from ethyene. a) b) c) d)

Branched Hydrocarbons w Hydrocarbons come in a variety of forms. w Not all hydrocarbons are linear, some are branched and some are cyclic. w Branched hydrocarbons have a carbon “backbone” with “groups” attached to this backbone. w To name these compounds you need to follow the basic rules below: (IUPAC) w Find the longest carbon chain (backbone) and write its corresponding name. w Identify the groups attached to this chain. w Assign the carbons in the chain numbers, which make the “group” have the lowest number. w Identify any double or triple bonds with the corresponding carbon number.

Journal Activity #5 w Diagram the following branched hydrocarbons: a) 2 methyl, hexane b) 2, 2, dimethyl pentane c) 2, 3 dimethyl octane d) 2, 4 dimethyl 3 -decene

Branched Hydrocarbons w Branched hydrocarbons are found in crude oil and can also by synthesized by chemical reactions. w Hydrocarbons with only single bonds are termed “saturated” hydrocarbons. w Hydrocarbons with a double or triple bond are called “unsaturated” hydrocarbons. Saturated hydrocarbon : Pentane

Isomers w Diagram the following hydrocarbons : a) Butane • • • b) 2 -methyl propane The molecular formula for both of these is C 4 H 10 Even though the formula is the same the properties of these compounds is very different. This is an example of isomers. Isomers are compounds with the same formula but different chemical and physical properties. (Boiling point, etc…)

Journal Activity #6 w Diagram a pentane molecule, and two isomers of this molecule. Name each isomer.

Aromatic Hydrocarbons w Hydrocarbons can also form ring structures. w The simplest would be cyclopropane, cyclobutane, and cyclopentane. w When six carbons form a ring we enter a new class of hydrocarbons called “aromatic” hydrocarbons. w Benzene is the first member of aromatic hydrocarbons.

Journal Activity #7 w Diagram the following cyclic hydrocarbons and give the molecular formula of each. a) Cyclopentane b) cyclobutene c) Benzene d) cyclohexane

Benzene w Benzene is a member of the aromatic (ring) hydrocarbons. w These hydrocarbons have a variety of uses as pesticides, flavorings, and a host of other uses.

Journal Activity #8 w http: //www. uyseg. org/greener_industry/pages/benz ene/2 Benzene. Uses. htm * Use the website above to describe how benzene is used to produce : styrene, nylon rope, dynamite (TNT), and 2, 4 D( weed killer).

Hydrocarbon Summary w At this point we’ve concluded our study of hydrocarbons. Below is a summary of the hydrocarbon compounds we’ve studied: w w w Alkanes Alkenes Alkynes Branched alkanes, alkenes, alkynes Cyclic hydrocarbons Aromatic hydrocarbons

Hydrocarbon Tutorial Websites w http: //chemed. chem. purdue. edu/genchem/topicrevie w/bp/1 organic/organic. html • Provides a good overview of hydrocarbons with naming practice. • http: //www. ausetute. com. au/namsanes. html * Great web-site for all concepts of chemistry. Choose a tutorial from the drop down on alkanes, alkenes, etc.

Organic Compounds w Hydrocarbons are used to synthesize a large amount of different organic compounds. w In addition some of these organic compounds are naturally produced in living things. w By adding substances and breaking bonds the hydrocarbon can become a new “class” of organic compound.

Organic Compounds w The organic compounds we will study include: n n n n Alcohols * sugars Aldehydes * starches Amine’s * lipids Carboxylic Acids (Organic Acids) Esters * proteins Ethers Ketones

Alcohols w Alcohols can be produced 3 different ways. Alcohols are compounds with an OH functional group (Hydroxyl) on a carbon skeleton. C- C– OH Hydroxyl Group (Alcohol)

Alcohol Properties w Alcohols with up to 4 carbon atoms are soluble in water as a result of the OH group on the carbon chain. w As more carbon atoms are added to the chain the solubility decreases. w Alcohols have higher boiling points than do the hydrocarbons from which they are formed from.

Types of Alcohols w There are three types of alcohols: Primary, Secondary, and tertiary. These types of alcohols are determined by “where” the OH is on the carbon chain. w Primary : OH is at the end of a carbon chain. w Secondary: OH is in the middle, with 2 C atoms attached to the C with the OH. w Tertiary : OH is in the middle with 3 C atoms attached to the C with the OH.

Alcohol Types w Diagram a primary alcohol below : ( ethanol) • Diagram a secondary alcohol below: (2 – butanol) * Diagram a tertiary alcohol below: 2 -methyl-2 -propanol

Naming Alcohols w Alcohols are named by finding their hydrocarbon skeleton and ending this with “ol”. w The OH group is identified to which carbon in the chain it is attached to. Name the following alcohol and identify it as primary, secondary, or tertiary.

Diols/Triols w Alcohols can have more than one OH group on a hydrocarbon chain. w Alcohols with two OH’s are called diols, and the prefix di is added before the “ol” in naming. w Alcohols with three OH’s are called triols, and the prefix “tri” is added before the “ol” in naming. Write the structural formula for 1, 2 -ethanediol. This is also known as ethylene glycol or antifreeze.

Naming and Classifying Alcohols 1) Draw the structural formula for the following alcohols and classify them: a) 3 -hexanol b) methanol c) 1, 2, 3 -propanetriol (glycerol)

Alcohol Formation w Alcohols can be produced three ways : 1) From using bacteria or yeast in the process of fermentation. (ethanol and carbon dioxide are the waste products of yeast as they eat sugar) 12% 2) Addition of water to an alkene. (with an acid catalyst) 3) Hydrolysis of a halocarbons.

Alcohol Formation • Diagram the formation of ethanol from ethene + water ( with an acid catalyst H 2 SO 4) * Diagram the formation of ethanol from the hydrolysis (splitting by water) of chloroethane.

Helpful Websites for Alcohols w http: //dl. clackamas. cc. or. us/ch 106 -03/nomencla. htm http: //jchemed. chem. wisc. edu/JCESoft/CCA 5/M AIN/HOME. HTM

Halocarbons w Halocarbons are organic compounds in which halogens have replaced hydrogen. w Halocarbons are named as to where the halogens are attached, and how many halogens there are. w The halogen prefix is used : fluoro, chloro, bromo, etc.

Halocarbon Uses w Halocarbons are used extensively as pesticides, insecticides, dry cleaning, and other areas. w Many of the most toxic chemicals have been halocarbons. w PCB’s : poly chlorinated biphenols, are toxic synthesized halocarbons. DDT: 1, 1, 1 -trichloro-2, 2 -bis(pchlorophenyl) ethane DDT : a halocarbon which was banned in 1972

PCB’s w PCB’s are halocarbons which contain two phenols with chlorine on them. w PCB’s have been shown to be accumulative in fatty tissue, and bioaccumulate. w The biphenols typically can persist in soil for 15 -25 years.

Naming Halocarbons w Draw the structure for the following : a) Trichloromethane (chloroform) b) Tetraiodomethane: c) 2 -fluoro pentane

Ethers w Ethers are organic compounds which have an oxygen atom bonded to two different carbons. w Ethers are named by naming the compound on each side and ending in “ether”. Or by writing the short version of each with “oxy” in the middle. w Ethers were used anesthetics, but safer gases have been found. w Small ethers are soluble in water, but larger ethers are insoluble. w Applications of ethers are solvents, gasoline additive, etc.

Naming Ethers • Draw the structural diagram for the following : a) Dimethyl ether b) methoxypentane c) Diethyl ether d) Diphenyl ether

Aldehydes w Aldehydes are organic compounds which have a carbonyl group(C=O) at the end of a carbon chain. w Aldehydes are named by ending the molecule with an “al”. Diagram ethanal, and propanal below:

Uses of Aldehydes w Aldehydes are used as preservatives, flavorings, perfumes, etc. w Vanilla flavoring and cinnamon are natural aldehyde flavors. w Ethanol is converted to acetaldehyde (propanal) in the human body.

Ketones w Ketones are a class of organic compounds with a carbonyl group (C=O) in the middle of a carbon chain. (Ketones are never alone) w Propanone is the simplest ketone and is known as acetone. Acetone is a top 50 industrial chemical. w Acetone is a common solvent for nonpolar substances. (fingernail polish remover) w To name ketones end the carbon chain in “one”.

Organic Acids (Carboxylic Acids) w Carboxylic acids are a group of organic compounds which have a (COOH) functional group. w Carboxylic or organic acids are weak acids. w Examples are : acetic acid, citric acid, benzoic acid, etc. w To name these acids use the carbon chain root : and add the suffix “oic” and the word acid.

Naming Organic Acids w Draw the structure for the following organic acids: a) Methanoic acid b) Ethanoic c) Butanoic acid d) Propanoic acid

Properties of Organic Acids w w w Methanoic acid (formic acid) is the acid in bee and ant stings. Ethanoic acid is acetic acid, found in vinegar Small chain organic acids are soluble in water. Lactic acid is an organic acid which builds up in body cells. Ascorbic acid (Vitamin C) is found in citrus fruits and tomatoes. w Bacteria reacts butter into butanoic acid. (rancid butter)

Esters w Esters are organic compounds formed when an alcohol is added to an organic acid. w Esters have distinctive odors. w Esters have the functional group : (COOC) w Esters are named by naming the alcohol name first ending in “yl”, then the acid name is given ending in “oate”. w When an ester is synthesized from an alcohol and organic acid, the alcohol gives off the (O) atom , and the acid gives two (H) atoms to form water.

Esters w Diagram the reaction of ethanoic acid with methanol. Name the ester which forms.

Esters w Draw the structural formula for the following esters: a) Ethyl butanoate b) pentyl ethanoate

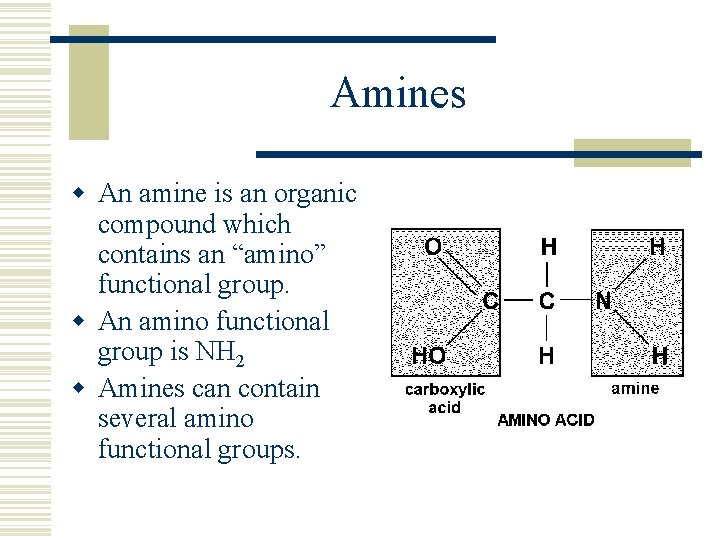
Amines w An amine is an organic compound which contains an “amino” functional group. w An amino functional group is NH 2 w Amines can contain several amino functional groups.

Amines w Amines are named by adding the suffix “amine” to the root name of the hydrocarbon. w Amines are also named by naming the hydrocarbon ending in “yl” with the suffix “amine”.

Amines w Amines are important biological compounds. w Amino acids which make up proteins contain an amino group along with an organic acid group. w Amines are found in DNA. w Amines are the basis of several addictive drugs : nicotine, cocaine, and amphetamines. Amino Group Methamphetamine

Naming Amines w Draw the structure for the following amines. a) Butanamine b) pentylamine

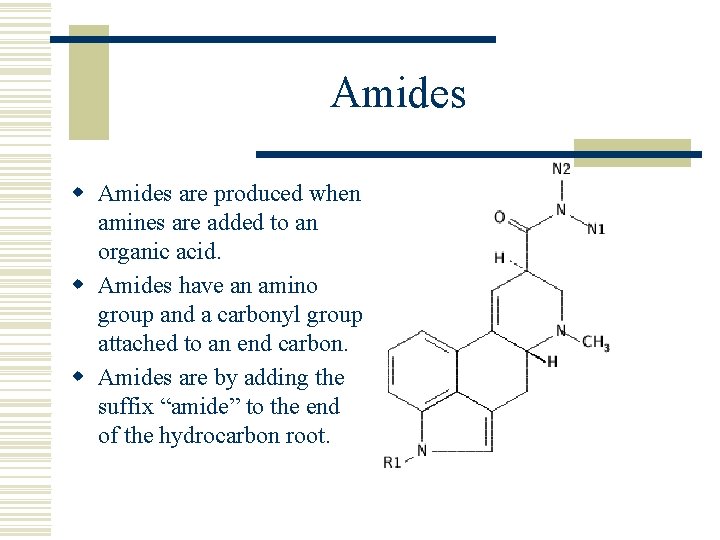
Amides w Amides are produced when amines are added to an organic acid. w Amides have an amino group and a carbonyl group attached to an end carbon. w Amides are by adding the suffix “amide” to the end of the hydrocarbon root.

Amides w Amides are found in biological compounds and in some synthetic polymers such as nylon. Nylon : a polymer with an amide group in the chain.

Naming Amides w Draw the structure of ethanamide. What is the name of the amide above?

Organic Compound Summary • The following are the organic compounds we’ve studied. They are identified by their “functional group”. • Hydrocarbons * Esters • Alcohols * Organic Acids • Ethers * Amines • Ketones * Amides • Aldehydes * Halocarbons

Polymers w Small organic molecules (monomers) can be reacted to form long organic molecules (polymers) w Example of polymers are : polyethylene (plastic), polyvinylchloride (PVC), nylon, polyester, etc. w Polymers can be formed by a condensation reaction (water is produced)

Polymers w Polymers can be formed by an elimination reaction, where a simple molecule (water, etc. ) is removed and a new compound is formed. PVC : polyvinyl chloride polymer

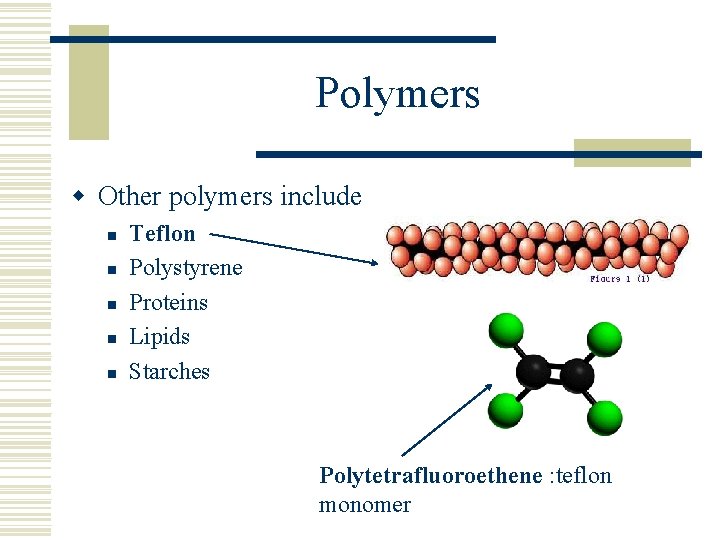
Polymers w Other polymers include n n n Teflon Polystyrene Proteins Lipids Starches Polytetrafluoroethene : teflon monomer

Polymers w Nylon is a polymer involving an amide monomer. w Nylon was synthesiszed in the 1930’s and named after the cities of New York and London. w Nylon is produced by adding an organic acid to an amine. w Nylon has many uses.