Organic chemistry Alkenes The alkenes were described as

Organic chemistry

Alkenes: The alkenes were described as being obtained from alkanes by loss of hydrogen in the cracking process. The general formula for this family is Cn. H 2 n. Since alkenes evidently contain less than the maximum quantity of hydrogen, they are referred to as unsaturated hydrocarbons. The simplest member of the alkene family is ethylene, C 2 H 4.

The carbon-carbon double bond is the distinguishing feature of the alkene structure. In forming the sp 2 orbitals, each carbon atom has used only two of its three p orbitals. The remaining p orbital consists of two equal lobes, one lying above and the other lying below the plane of the three sp 2 orbitals; it is occupied by a single electron. If the p orbital of one carbon atom overlaps the p orbital of the other carbon atom, the electrons pair up and an additional bond is formed. Because it is formed by the overlap of p orbitals, and to distinguish it from the differently shaped σ bonds, this bond is called a π bond (pi bond).
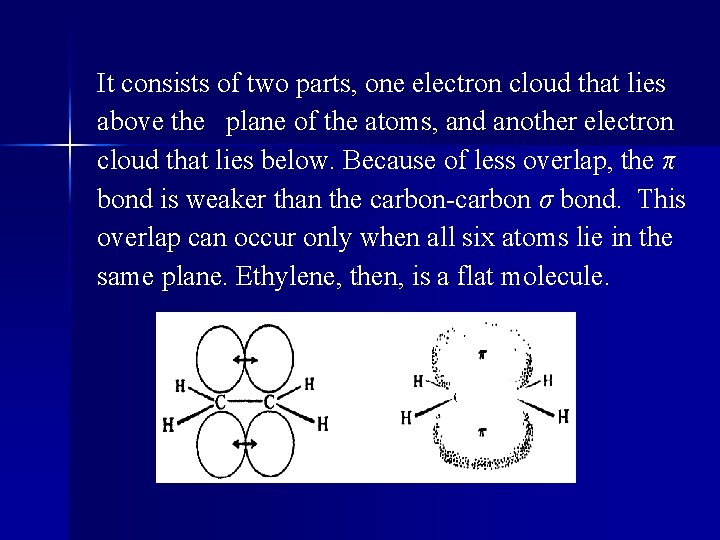
It consists of two parts, one electron cloud that lies above the plane of the atoms, and another electron cloud that lies below. Because of less overlap, the π bond is weaker than the carbon-carbon σ bond. This overlap can occur only when all six atoms lie in the same plane. Ethylene, then, is a flat molecule.

l The carbon-carbon "double bond" is thus made up of a strong σ bond a weak π bond. Since the carbon atoms are held more tightly together, the C─C distance in ethylene is less than the C─C distance in ethane; that is to say, the carbon-carbon double bond is shorter than the carbon-carbon single bond.

l Names of alkenes: Common names are seldom used except for three simple alkenes: ethylene, propylene, and isobutylene. The various alkenes of a given carbon number are sometimes referred to collectively as the pentylenes (amylenes), hexylenes, heptylenes, and so on. (One sometimes encounters the naming of alkenes as derivatives of ethylene: as, for example, tetramethylene for

l Most alkenes are named by the IUPAC system. The rules of the IUPAC system are: 1 - Select as the parent structure the longest continuous chain that contains the carbon-carbon double bond; then consider the compound to have been derived from this structure by replacement of hydrogen by various alkyl groups. The parent structure is known as ethene, propene, butene, pentene, and so on, depending upon the number of carbon atoms; each name is derived by changing the ending -ane of the corresponding alkane name to -ene:
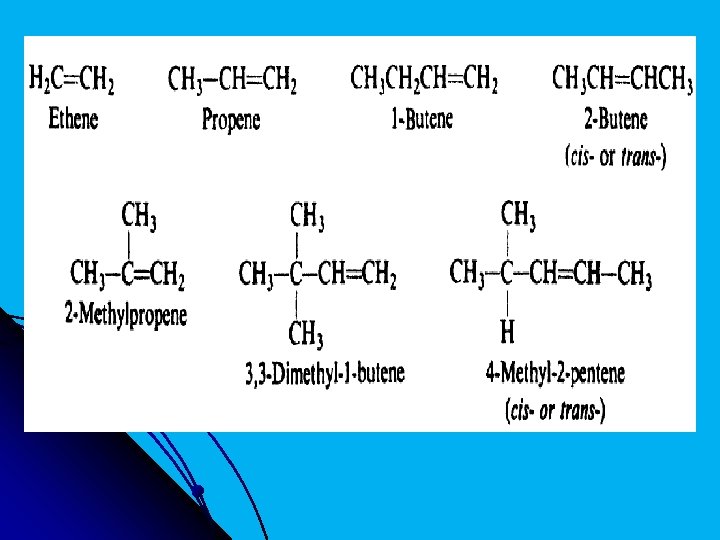

l 2. Indicate by a number the position of the double bond in the parent chain. Although the double bond involves two carbon atoms, designate its position by the number of the first doublybonded carbon encountered when numbering from the end of the chain nearest the double bond; thus 1 -butene and 2 -butene. 3 - Indicate by numbers the positions of the alkyl groups attached to the parent chain.

Physical properties: The alkenes possess physical properties that are essentially the same as those of the alkanes. They are insoluble in water, but quite soluble in nonpolar solvents like benzene, ether, and chloroform, the boiling point rises with increasing carbon number; as with the alkanes.

l Preparation of alkenes: Alkyl halides are converted into alkenes by dehydrohalogenation: elimination of the elements of hydrogen halide. Dehydrohalogenation involves removal of the halogen atom together with a hydrogen atom from a carbon adjacent to the one bearing the halogen. As we can see, in some cases this reaction yields a single alkene and in other cases yields a mixture.

For example:

n-Butyl chloride can eliminate hydrogen only from C-2 and hence yields only 1 -butene. sec-Butyl chloride, on the other hand, can eliminate hydrogen from either C-l or C-3 and hence yields both 1 -butene and 2 -butene. Where the two alkenes can be formed, 2 -butene is the chief product.

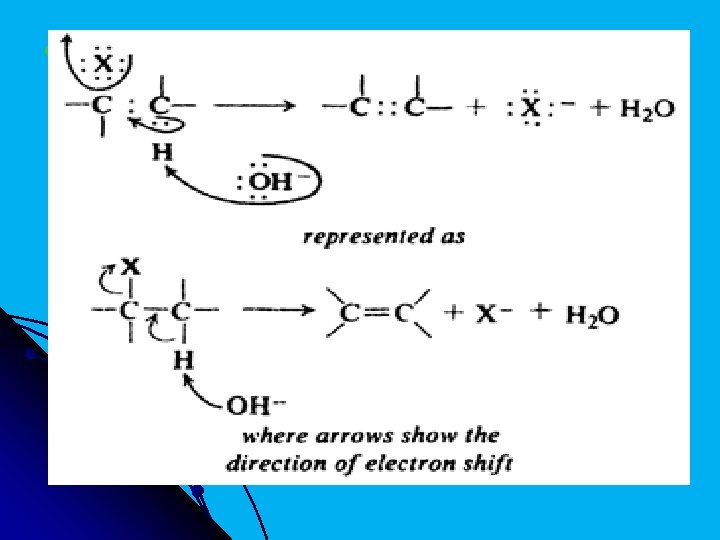
l Mechanism of dehydrohalogenation: The function of hydroxide ion is to pull a hydrogen ion away from carbon; simultaneously a halide ion separates and the double bond forms. We should notice that, in contrast to free radical reactions, the breaking of the C─H and C ─X bonds occurs in an unsymmetrical fashion: hydrogen relinquishes both electrons to carbon, and halogen retains both electrons. The electrons left behind by hydrogen are now available formation of the second bond (the π bond) between the carbon atoms.
l

l

l Alcohols are compounds of the general formula, ROH, where R is any alkyl group and the hydroxyl group OH, is characteristic of alcohols.

l Dehydration requires the presence of an acid and the application of heat. It is generally carried out in either of two ways: (a) heating the alcohol with sulfuric or phosphoric acid to temperatures as high as 200, or (b) passing the alcohol vapor over alumina, A 12 O 3, at 350400, alumina here serving as a Lewis acid.
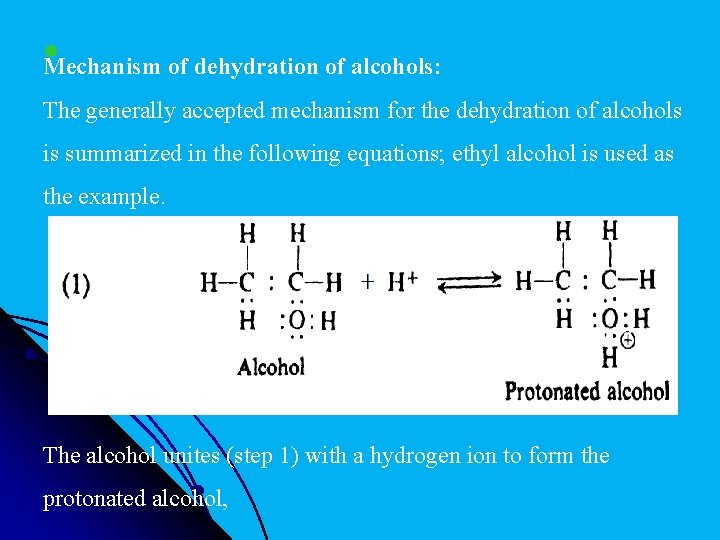
l Mechanism of dehydration of alcohols: The generally accepted mechanism for the dehydration of alcohols is summarized in the following equations; ethyl alcohol is used as the example. The alcohol unites (step 1) with a hydrogen ion to form the protonated alcohol,

lwhich dissociates (step 2) into water and a carbonium ion; + the carbonium ion then loses (step 3) a hydrogen ion to form the alkene.

l The double bond is thus formed in two stages, OH being lost (as H 2 O) in step (2) and H being lost in step (3).
- Slides: 21