Organic Chemistry Alkanes end with ane simplest hydrocarbons
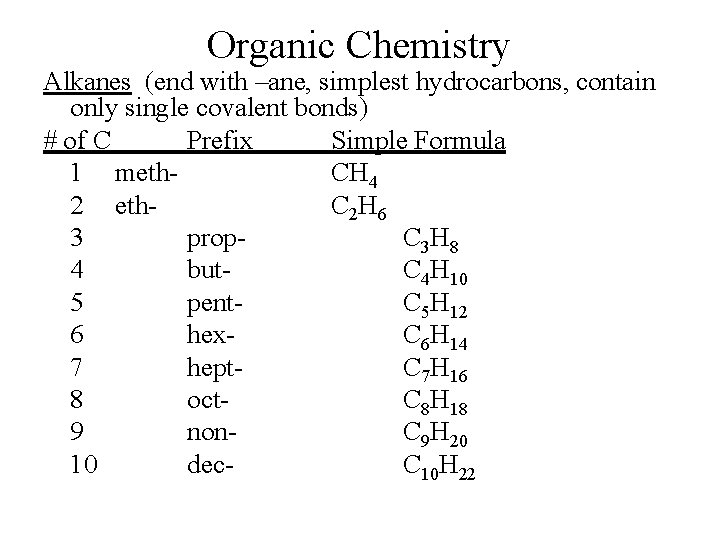
Organic Chemistry Alkanes (end with –ane, simplest hydrocarbons, contain only single covalent bonds) # of C Prefix Simple Formula 1 meth. CH 4 2 eth. C 2 H 6 3 prop. C 3 H 8 4 but. C 4 H 10 5 pent. C 5 H 12 6 hex. C 6 H 14 7 hept. C 7 H 16 8 oct. C 8 H 18 9 non. C 9 H 20 10 dec. C 10 H 22

-all alkanes that are straight chains are named n-alkane (means normal) C 4 H 10 = n-butane -look at pg 1010 *iso-, and neo-

Branched Chain Alkanes -alkane group takes the place of a H atom -called a substituent alkyl group- hydrocarbon substituent -ends with –yl (take away -ane) -contains one less H -look at # of C to name ex- CH 3 = methyl C 2 H 5 = ethyl

Naming Branched Chain Alkanes 1) Find the longest chain = parent molecule ex- heptane 2) # the C’s in the chain, making sure alkyl groups have lower numbers, can count backwards ex- 2, 3, 4 instead of 4, 5, 6 3) Add #’s to names of alkyl groups, with a dash in between ex: 2 -methyl 3 -methyl 4 -ethyl

4) Use prefixes to denote multiples of the same alkyl groups, commas between #’s ex- 2, 3 -dimethyl 5) Put alkyl groups in alphabetical order ignoring prefixes ex- 4 -ethyl-2, 3 -dimethyl 6) **Use proper punctuation -commas separate #’s -hyphens separate #’s and letters -no spaces

7) Add parent chain name ex- 4 -ethyl-2, 3 -dimethylheptane *Remember prefixes* 2 = di 3 = tri 4 = tetra 5 = penta 6 = hexa 7 = hepta 8 = octa 9 = nona 10 = deca-

Drawing Structural Formula From Name 1) Find parent chain and draw 2) # C’s on parent chain 3) Identify substituents and attach to proper C 4) Add hydrogen as needed Try These!! 1) 3 -ethylhexane 2) 2, 2, 4 -trimethylpentane 3) 3 -ethyl-3, 4 -dimethyloctane

saturated compounds- contain max # of H -all alkanes, all single C-C bonds unsaturated compounds- do not contain max # of H -contain double or triple C-C bonds -alkenes and alkynes Alkenes -contain at least one double C═C bond

Naming Alkenes 1) Find longest chain containing double bond -this is parent chain -gets –ene ending ex- butene 2) # so that C atoms of the double bond get the lowest #’s, then look at alkyl groups to get lowest # -double bond gets numbered in the name ex- 2 -butene

3) Substituents get named the same as alkanes ex- 2 -methyl 4) Put all together ex: 2 -methyl-2 -butene Alkynes -named same way except end in –yne http: //www. chembio. uoguelph. ca/educmat/chm 1 9104/organic_nomenclature_quizzes. htm

Structural Isomers -compounds that have the same simple formula, but different structural formulas Ex- C 4 H 10 n-butane 2 -methylpropane Try This!! Write the structural formulas/skeleton formulas and names for the nine isomers of heptane

Cyclic Hydrocarbons-contain a hydrocarbon ring -all single bonded cyclic hydrocarbons are named with the prefix cycloexcyclopropane -if have subgroups, name just like before
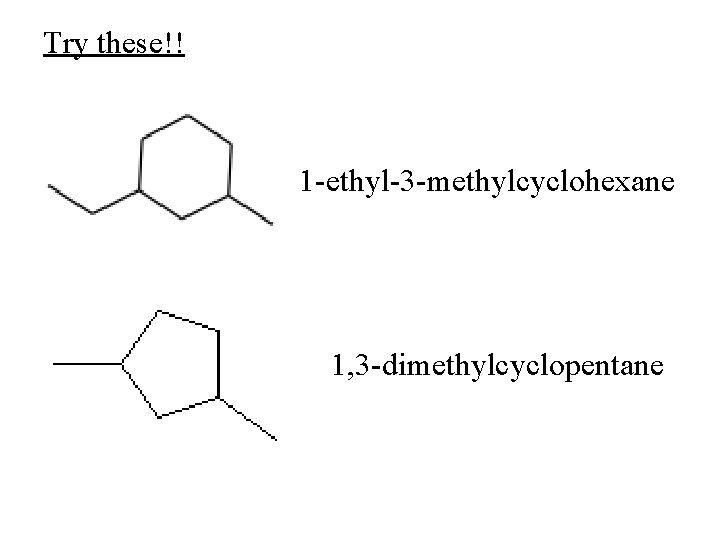
Try these!! 1 -ethyl-3 -methylcyclohexane 1, 3 -dimethylcyclopentane
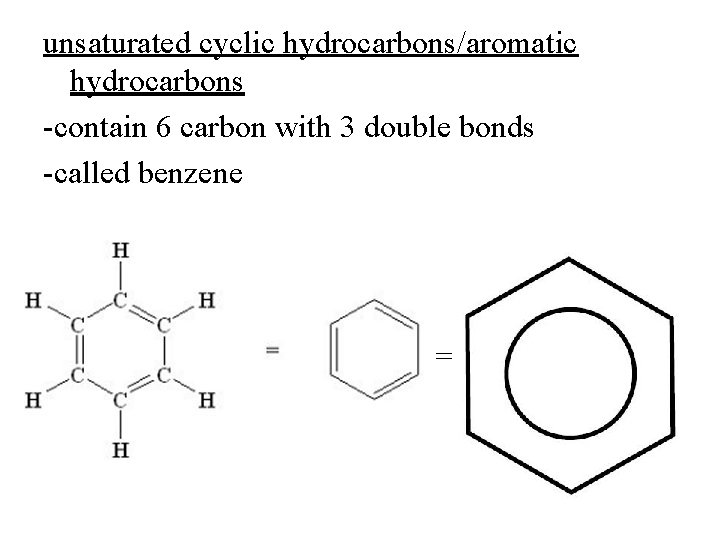
unsaturated cyclic hydrocarbons/aromatic hydrocarbons -contain 6 carbon with 3 double bonds -called benzene =
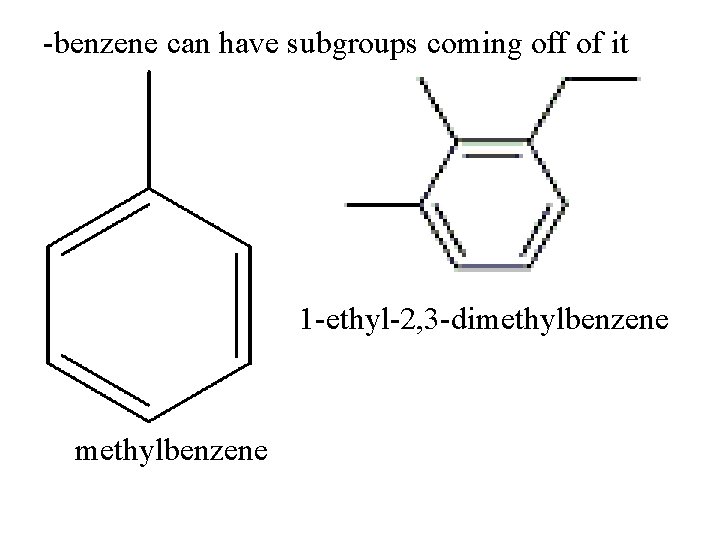
-benzene can have subgroups coming off of it 1 -ethyl-2, 3 -dimethylbenzene
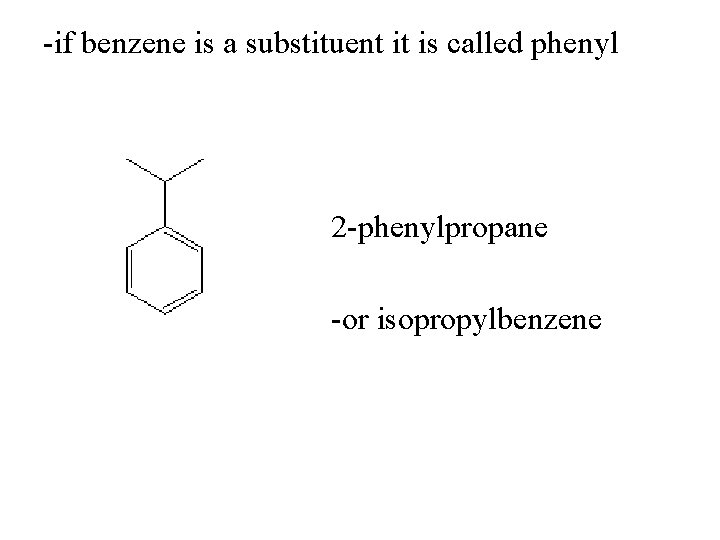
-if benzene is a substituent it is called phenyl 2 -phenylpropane -or isopropylbenzene

Functional Groups **R represents C chain attached to functional group Halocarbons R-X X= a halogen functional group = halogen -use root of halogen to name
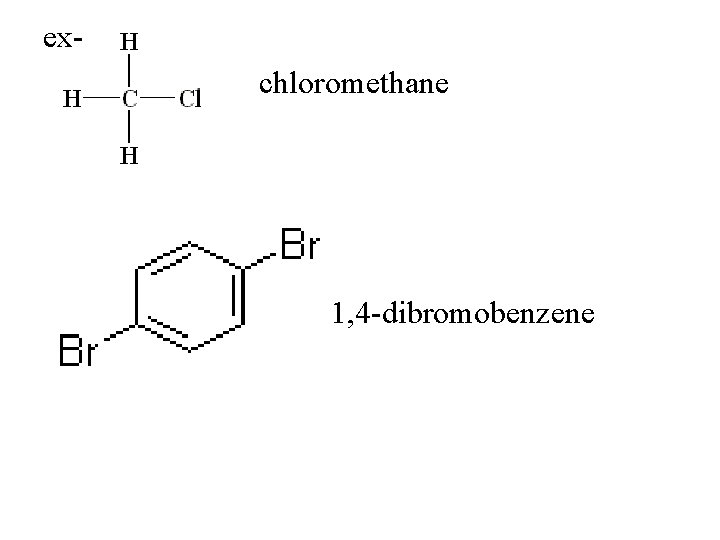
exchloromethane 1, 4 -dibromobenzene
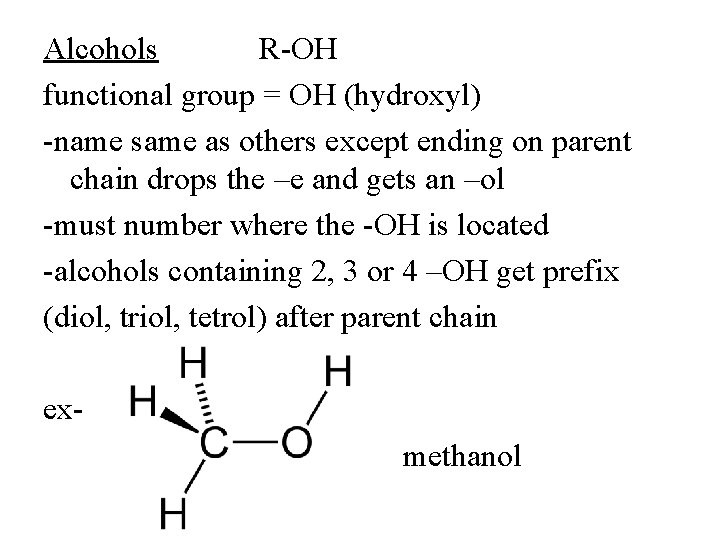
Alcohols R-OH functional group = OH (hydroxyl) -name same as others except ending on parent chain drops the –e and gets an –ol -must number where the -OH is located -alcohols containing 2, 3 or 4 –OH get prefix (diol, triol, tetrol) after parent chain exmethanol
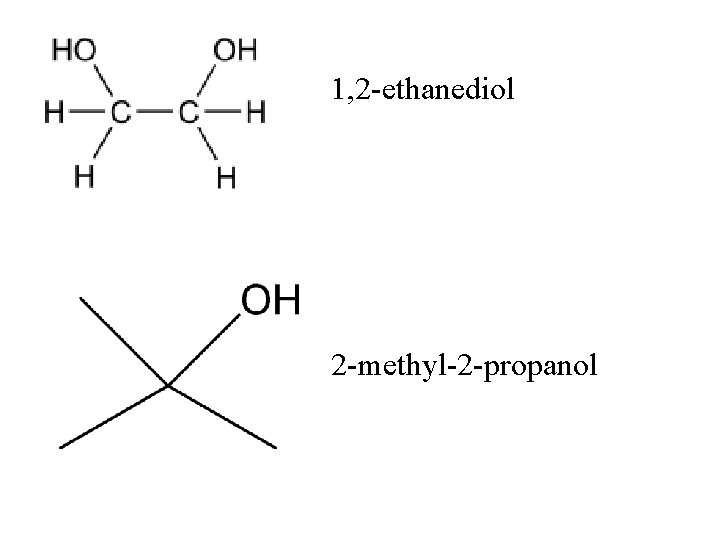
1, 2 -ethanediol 2 -methyl-2 -propanol
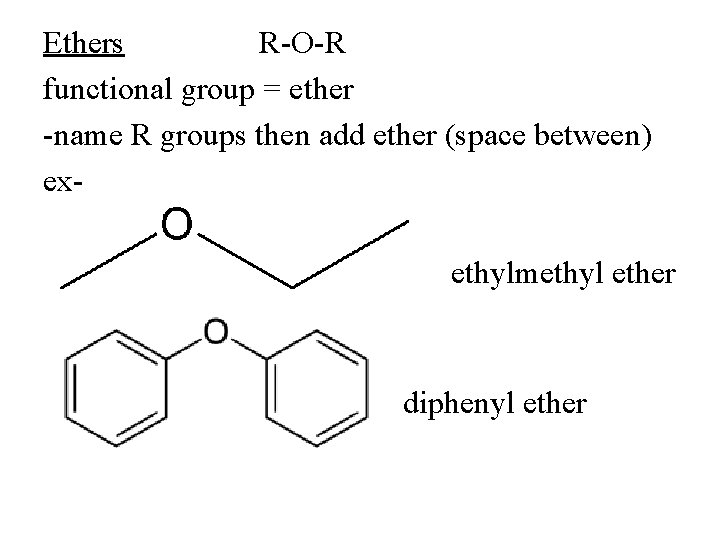
Ethers R-O-R functional group = ether -name R groups then add ether (space between) exethylmethyl ether diphenyl ether
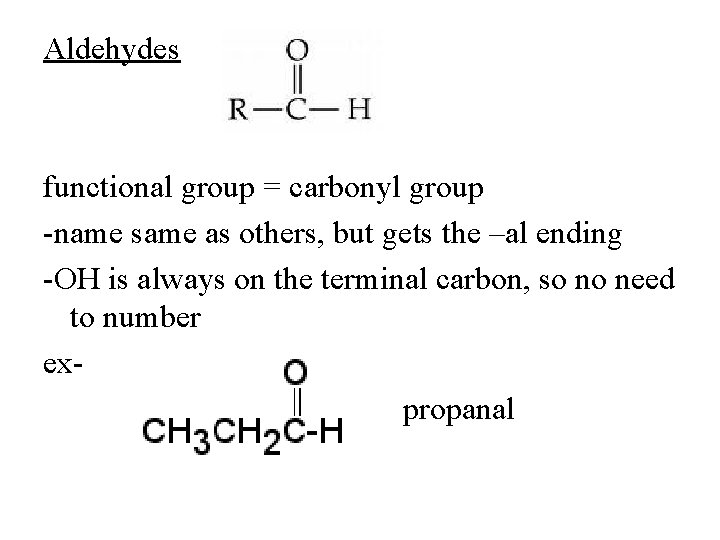
Aldehydes functional group = carbonyl group -name same as others, but gets the –al ending -OH is always on the terminal carbon, so no need to number expropanal
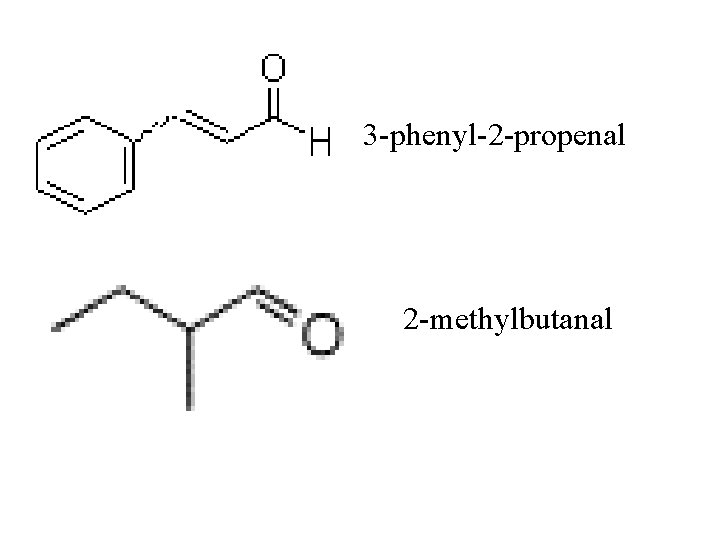
3 -phenyl-2 -propenal 2 -methylbutanal
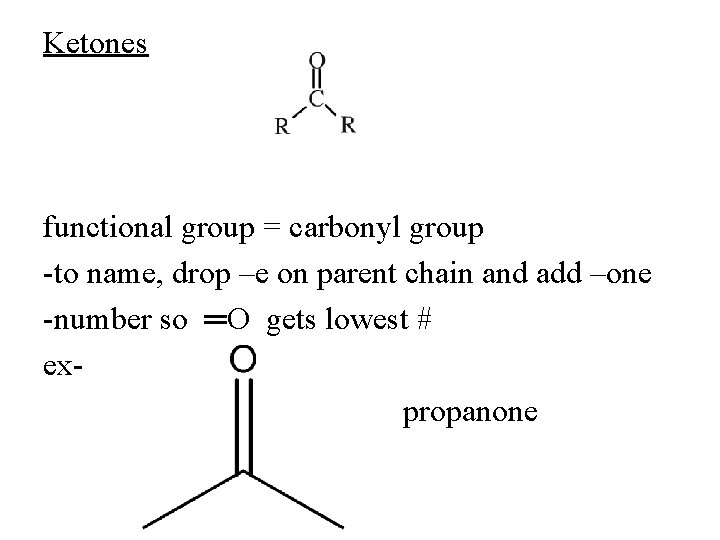
Ketones functional group = carbonyl group -to name, drop –e on parent chain and add –one -number so ═O gets lowest # expropanone
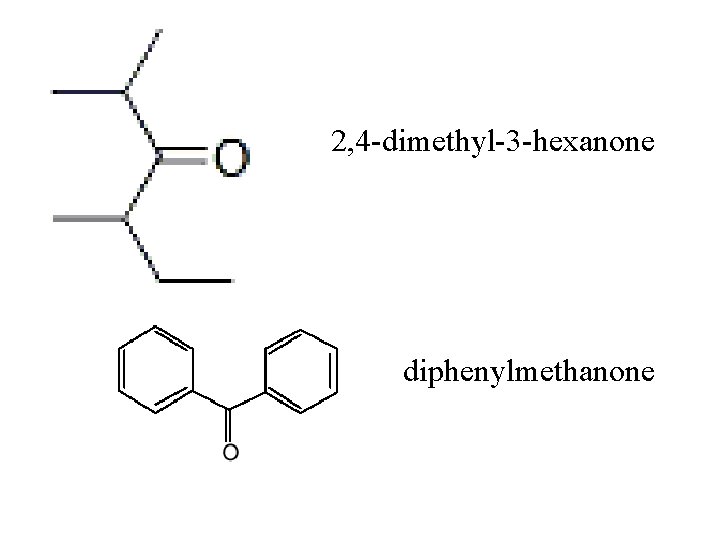
2, 4 -dimethyl-3 -hexanone diphenylmethanone
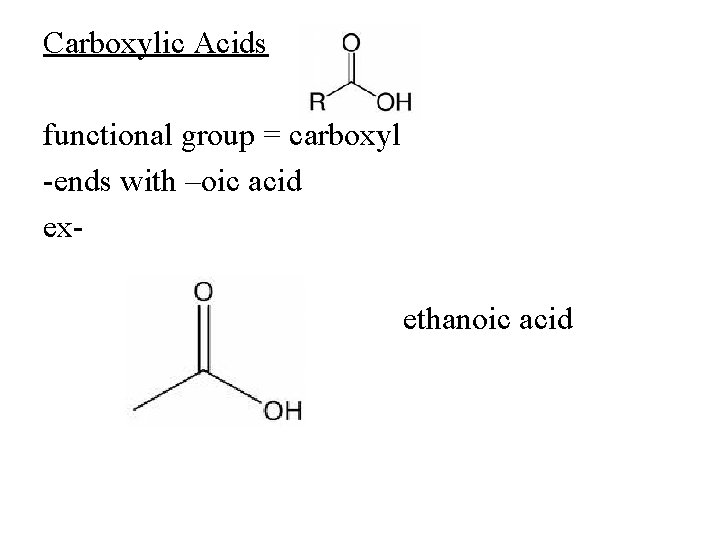
Carboxylic Acids functional group = carboxyl -ends with –oic acid exethanoic acid
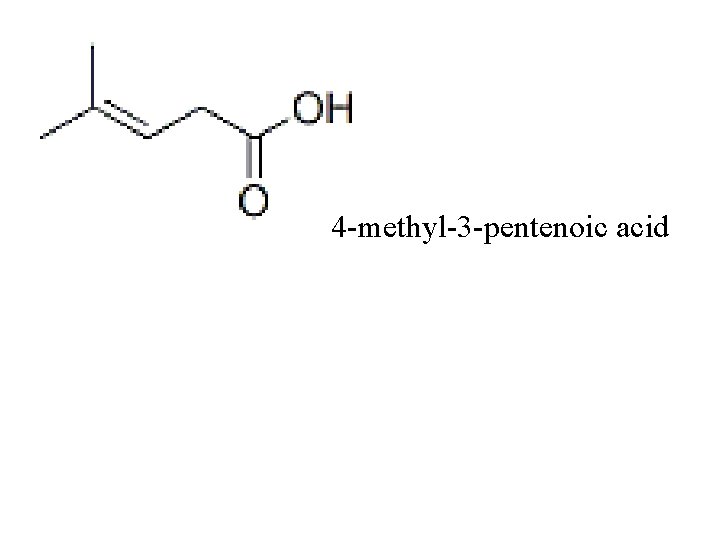
4 -methyl-3 -pentenoic acid
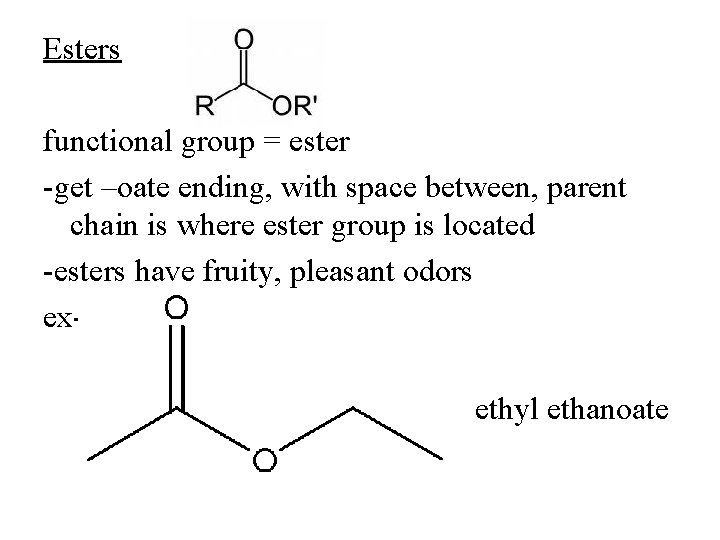
Esters functional group = ester -get –oate ending, with space between, parent chain is where ester group is located -esters have fruity, pleasant odors exethyl ethanoate
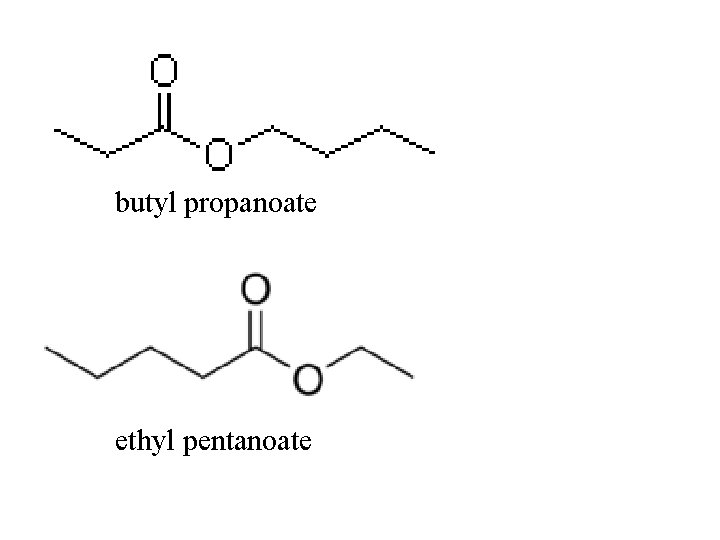
butyl propanoate ethyl pentanoate
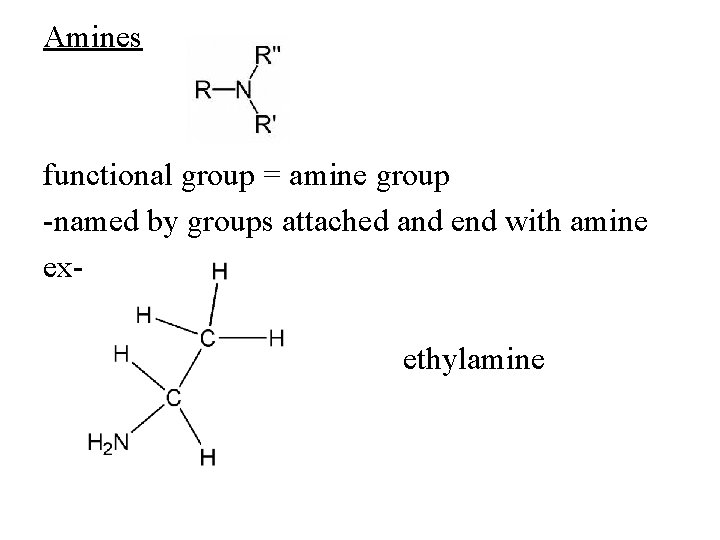
Amines functional group = amine group -named by groups attached and end with amine exethylamine
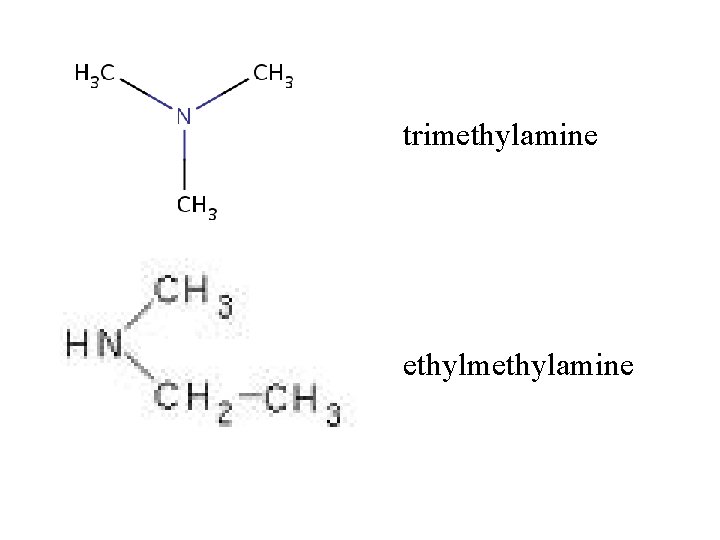
trimethylamine ethylmethylamine
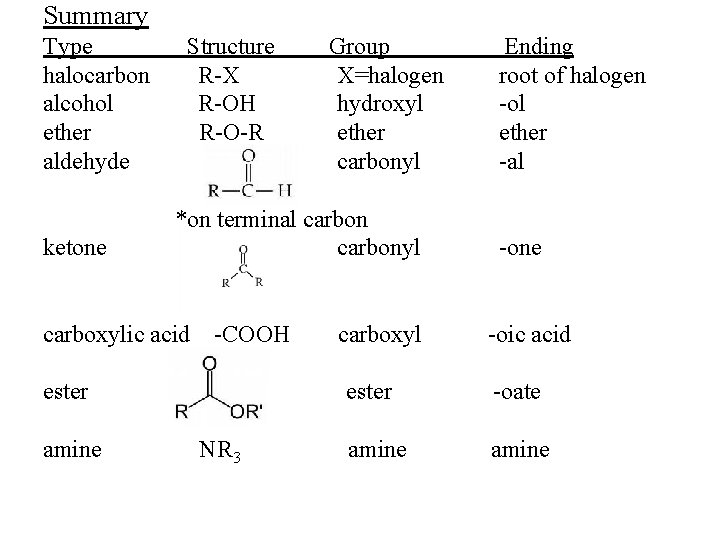
Summary Type halocarbon alcohol ether aldehyde ketone Structure R-X R-OH R-O-R *on terminal carbonyl carboxylic acid -COOH ester amine Group X=halogen hydroxyl ether carbonyl NR 3 carboxyl Ending root of halogen -ol ether -al -one -oic acid ester -oate amine
- Slides: 32