Organic Chemistry A Carbon Compounds organic compounds are

Organic Chemistry A. Carbon Compounds ¬ organic compounds are those in which carbon atoms are almost always bonded to each other, to hydrogen atoms and a few other atoms (O, N, S, P) ¬inorganic exceptions are the oxides of carbon, carbonates, cyanides and carbides (no C-C bonds or C-H bonds) eg) CO 2, Ca. CO 3, Na. CN, Si. C ¬ there are millions of organic compounds and all covalent bonds contain

¬carbon is unique for two reasons: a) it can bond with other carbon atoms to form long chains, rings, spheres, tubes, sheets etc. b) it can form combinations of single, double and triple bonds (no other element does this!!!!)

¬ recall polarity and intermolecular forces from the chemical bonding unit ¬ polar bonds are formed when there is an uneven pull on e ¬ polar compounds are formed when the polar bonds within a molecule do not cancel each other out ¬ the presence of dipole-dipole forces and hydrogen bonding will allow polar compounds to dissolve in water, since it is also polar ¬ non-polar compounds only have LD forces between molecules and will not dissolve in water

B. Structural Isomers ¬isomers are compounds with the same molecular formula but a different structure
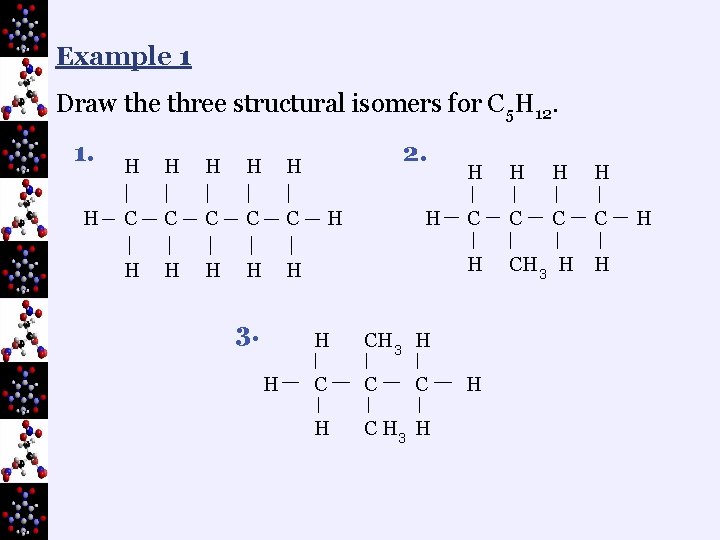
Example 1 Draw the three structural isomers for C 5 H 12. 1. H H H C C C H H H 3. H 2. H H H CH 3 H C C H 3 H C H H C C H CH 3 H H
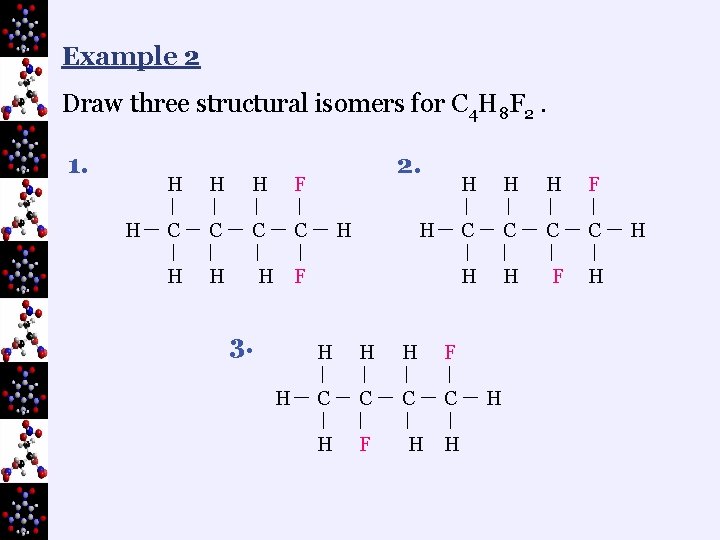
Example 2 Draw three structural isomers for C 4 H 8 F 2. 1. H H F C C H H 2. H H H F 3. H H F C C H F H H H F C C H H F H H H

¬different structures result in different properties ¬ the arrangement of the atoms determines the types of intermolecular attractions which then determines properties such as boiling point and solubility in water

Example Draw two isomers of C 3 H 8 O. Which one would have a boiling point of 7. 4 C and which would have a boiling point of 82. 5 C? Explain why the boiling points are so different. H H C C C H OH H 82. 5 C – has HB between molecules which makes the boiling point quite high H C H O H H C C H H H 7. 4 C – does not have HB between molecules therefore the boiling point is significantly lower

C. Formulas and Structural Diagrams ¬organic molecules can be drawn in three different ways: 1. complete structural diagram – shows all bonds eg) C 3 H 8 H H eg) C C C H H H C 3 H 7 F H H H C C C H H H F

2. condensed structural diagrams – shows carbon to carbon bonds but includes the hydrogens etc. attached to each carbon eg) C 3 H 8 eg) C 3 H 7 F CH 3 CH 2 F
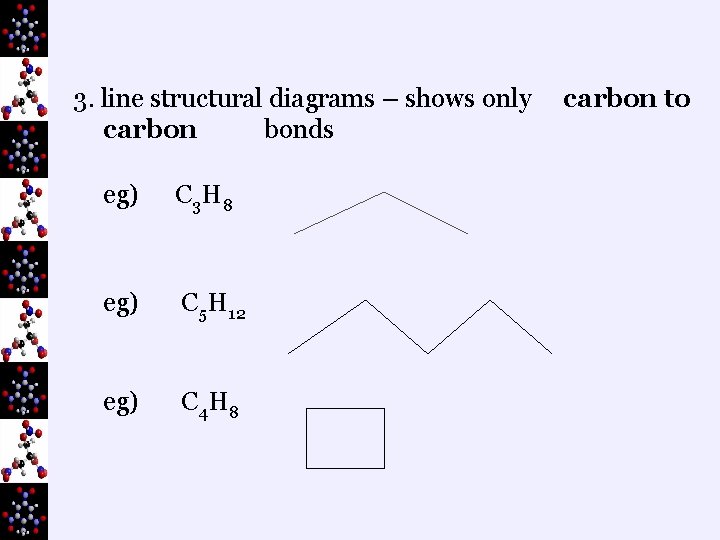
3. line structural diagrams – shows only carbon bonds eg) C 3 H 8 eg) C 5 H 12 eg) C 4 H 8 carbon to

D. Prefixes 1. Number of Functional Groups 6 = hexa 2 = di 7 = hepta 3 = tri 8 = octa 4 = tetra 9 = nona 5 = penta 10 = deca

2. Number of Carbons 1 = meth 6 = hex 2 = eth 7 = hept 3 = prop 8 = oct 4 = but 9 = non 5 = pent 10 = dec

E. Alkanes Cn. H 2 n+2 eg) C 5 H 12, C 20 H 42, etc. ¬ hydrocarbons containing only single bonds ie) they are SATURATED ¬ can be long continuous chains, branched chains, ring structures (cycloalkanes)

1. Properties ¬ nonpolar not soluble in water ¬ can be solid, liquid or gas number of carbon atoms depending on ¬ relatively unreactive because the single bonds are very stable

2. Uses ¬ natural gas, BBQ’s, lighter fluid, gasoline etc ¬ good for making plastics, lubricants 3. Naming ¬IUPAC = International Union of Pure and Applied Chemistry
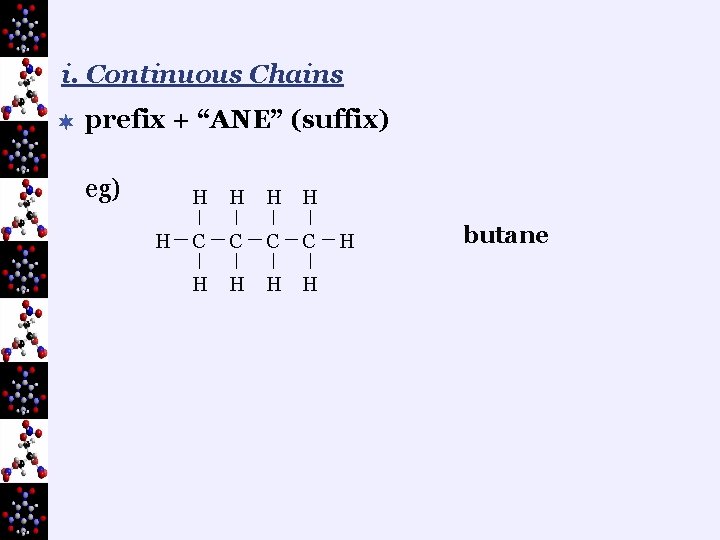
i. Continuous Chains ¬ prefix + “ANE” (suffix) eg) H H H C C H H H butane

ii. Branched Chains ¬ branches are called alkyl functional group ¬ 1 C = methyl ; 2 C = ethyl ; 3 C = propyl etc ¬find the longest carbon chain and number it so the branches get the lowest possible numbers ¬to name: name the groups first (in alphabetical order), including the number of the carbon where each group is found, then name the longest chain (parent name)
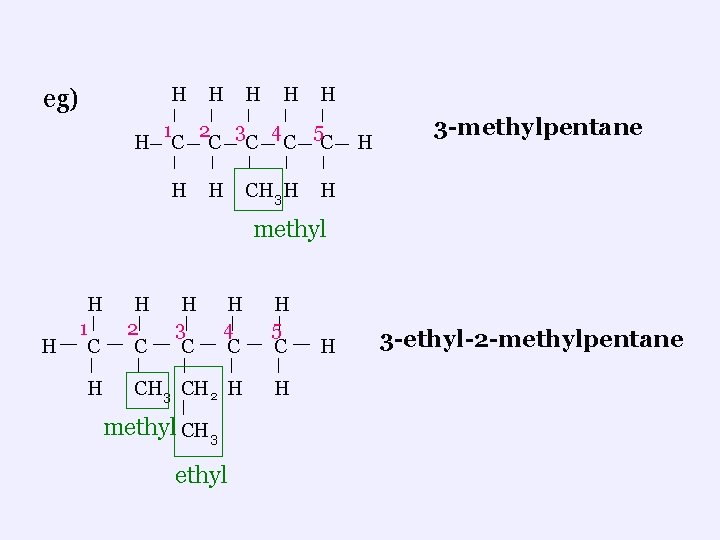
H eg) H H 1 2 3 4 5 H C C C H H CH 3 H H 3 -methylpentane H methyl H H 1 C H H 2 C H 3 C H 4 C H 5 C CH 3 CH 2 H H methyl CH 3 ethyl H 3 -ethyl-2 -methylpentane
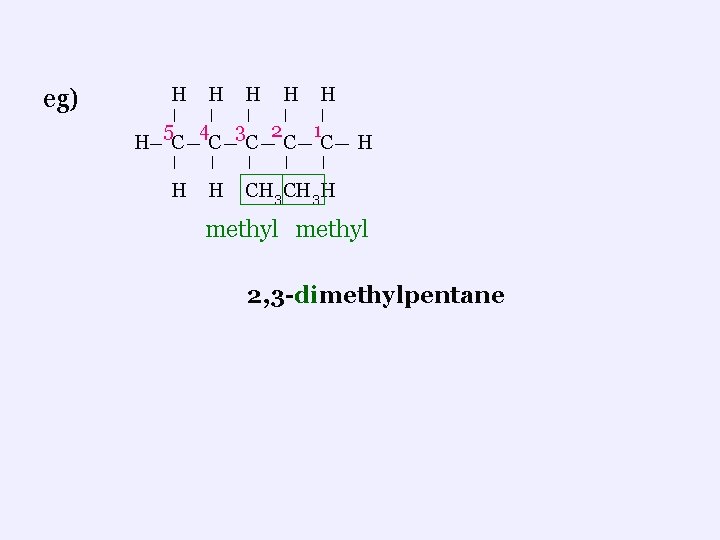
eg) H H H 5 4 3 2 1 H C C C H H H CH 3 H methyl 2, 3 -dimethylpentane
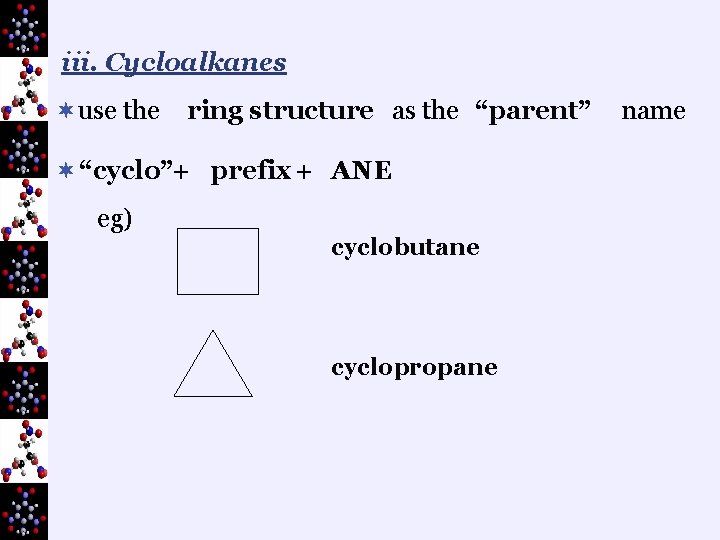
iii. Cycloalkanes ¬use the ring structure as the “parent” ¬ “cyclo”+ prefix + ANE eg) cyclobutane cyclopropane name
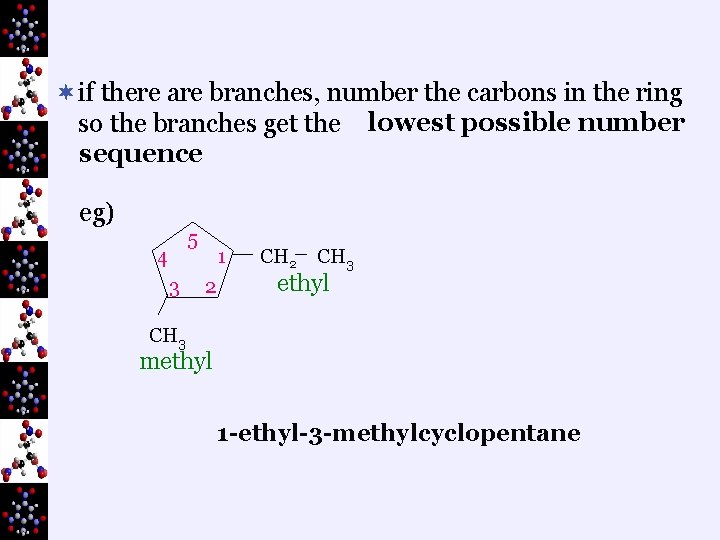
¬if there are branches, number the carbons in the ring so the branches get the lowest possible number sequence eg) 5 4 3 1 2 CH 3 ethyl CH 3 methyl 1 -ethyl-3 -methylcyclopentane

F. Alkenes Cn. H 2 n eg) C 5 H 10, C 20 H 40, etc. ¬ hydrocarbons containing one or more double bonds ie) they are UNSATURATED ¬ can be long continuous chains, branched chains, ring structures (cycloalkenes)

1. Properties ¬ nonpolar not soluble in water ¬ lower boiling point than corresponding alkane LD because they have fewer e which makes the forces of attraction weaker eg) ethane BP = 88. 6 C ¬ more reactive ethene BP = 103. 8 C than alkanes ¬ double bond has more e- in the same area greater repulsion and bond less stable
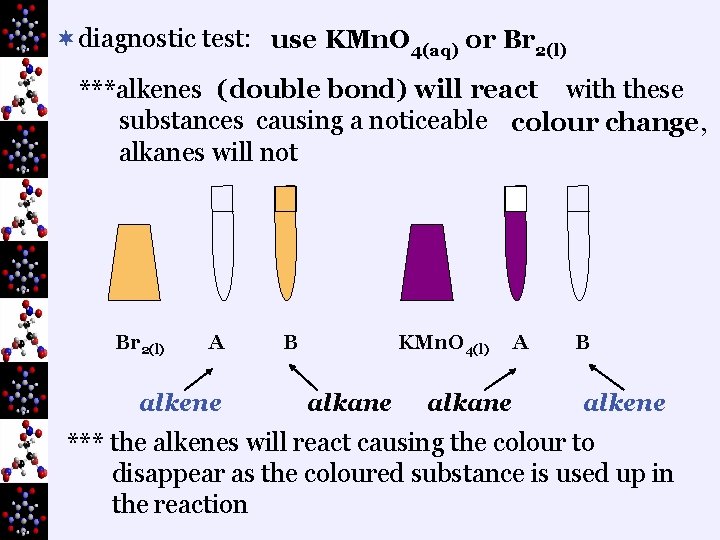
¬diagnostic test: use KMn. O 4(aq) or Br 2(l) ***alkenes (double bond) will react with these substances causing a noticeable colour change, alkanes will not Br 2(l) A alkene B KMn. O 4(l) alkane A alkane B alkene *** the alkenes will react causing the colour to disappear as the coloured substance is used up in the reaction

2. Uses ¬ plastics (PVC) ¬ steroids ¬ welding torches 3. Naming i. Continuous Chains ¬ prefix + “ENE” (suffix) ¬number carbons to give the double bond the lowest number ¬the number where the double bond starts is to be given as a “ # ” between the prefix and the suffix
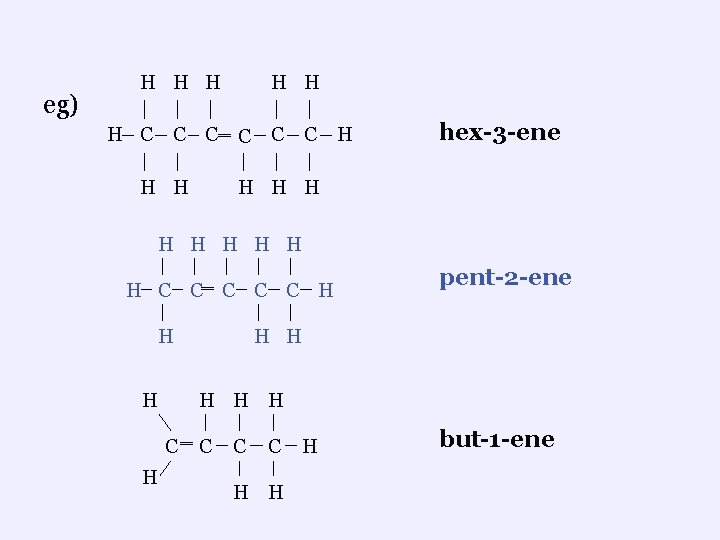
eg) H H C C H H C‗ C C H H C H hex-3 -ene H H H H ‗ H C C C H H H H C H pent-2 -ene ‗ C C C H H H but-1 -ene

ii. Branched Chains ¬find the longest carbon chain and number it so the double bond gets the lowest possible number ¬to name: name the groups first (in alphabetical order), including the number of the carbon where each is found, then the parent name including the number of the carbon where the double bond starts
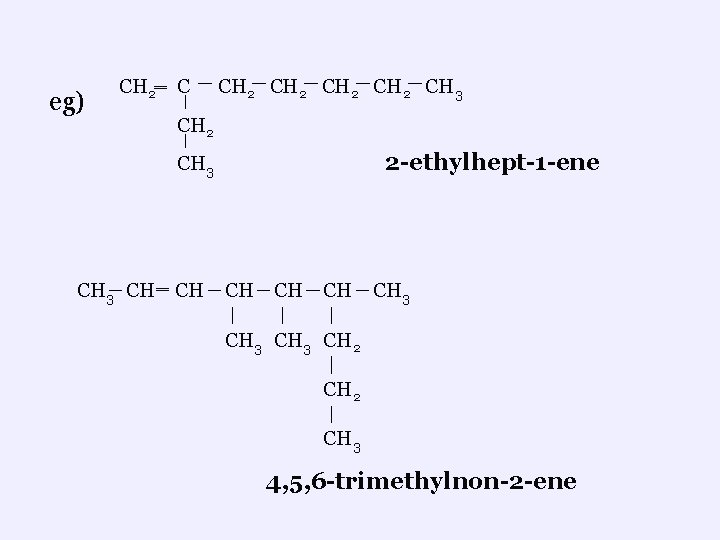
eg) CH 2‗ C CH 2 CH 3 CH 2 2 -ethylhept-1 -ene CH 3 CH ‗ CH CH CH 3 CH 2 CH 3 4, 5, 6 -trimethylnon-2 -ene

iii. Cycloalkenes ¬double bond is always numbered 1, 2 ¬ branches get the lowest numbering sequence after the double bond ¬ “cyclo”+ prefix + ENE ¬list branches in alphabetical order with the number of the carbon they are on
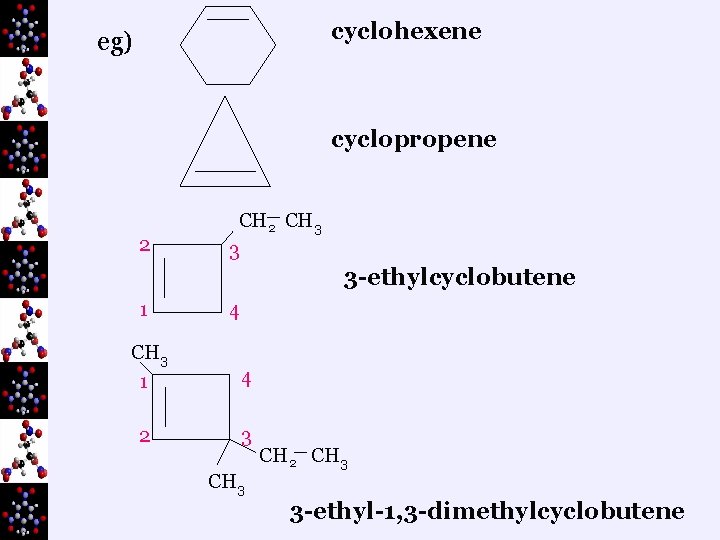
cyclohexene eg) cyclopropene 2 CH 3 3 3 -ethylcyclobutene 1 4 CH 3 1 4 2 3 CH 2 CH 3 3 -ethyl-1, 3 -dimethylcyclobutene

G. Alkynes Cn. H 2 n-2 eg) C 5 H 8, C 20 H 38, etc. ¬ hydrocarbons containing one or more triple bonds ie) they are also UNSATURATED ¬ can be long continuous chains, branched chains ¬ not plentiful in nature

1. Properties ¬ nonpolar ¬ very reactive (more than alkanes and alkenes) ¬ triple bond has 6 e- in the same area high force of repulsion ¬ boiling points are higher than corresponding alkanes and alkenes because of their linear structure and the nature of triple bonds 2. Uses ¬ welding torches

3. Naming i. Continuous Chains ¬ prefix + “YNE” ¬number carbons to give the triple bond lowest number the ¬the number where the triple bond starts is to be given as a “ # ” between the prefix and the suffix
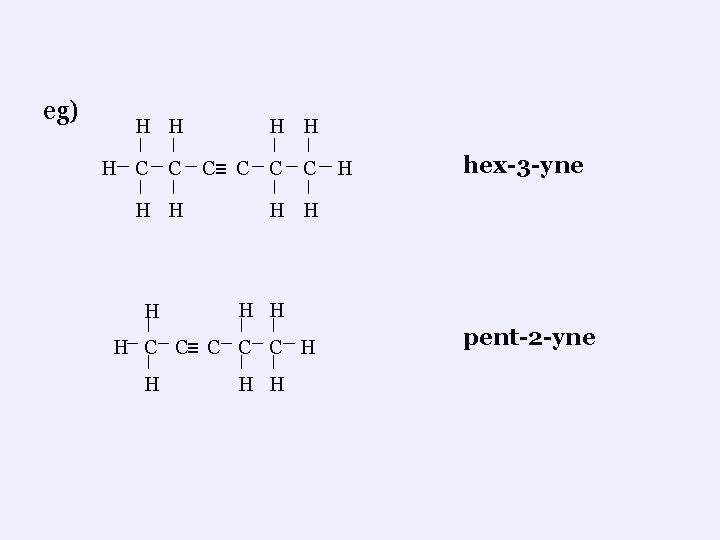
eg) H H H C C H H H C≡ C C C hex-3 -yne H H H C C≡ C C C H H H pent-2 -yne

ii. Branched Chains ¬find the longest carbon chain and number it so the triple bond gets the lowest number ¬to name: name the groups first (in alphabetical order), including the number of the carbon where each is found, then the parent name including the number of the carbon where the triple bond starts
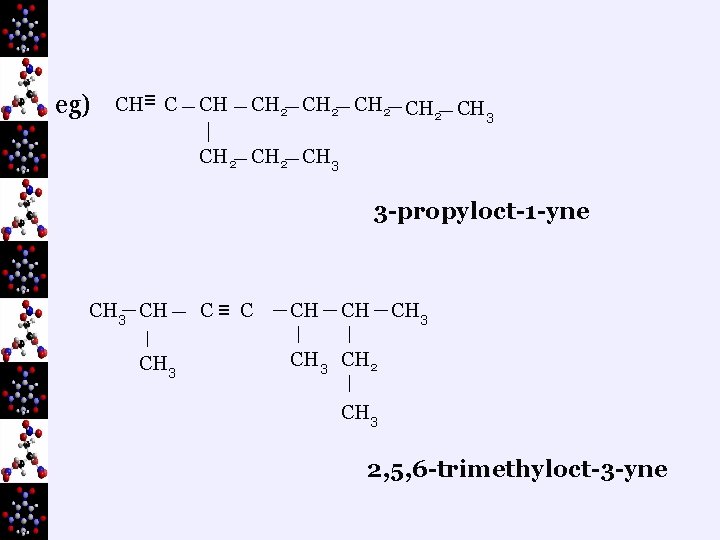
eg) CH≡ C CH CH 2 CH 3 CH 2 CH 3 3 -propyloct-1 -yne CH 3 CH CH 3 C≡ C CH CH CH 3 CH 2 CH 3 2, 5, 6 -trimethyloct-3 -yne

Review ¬ alkanes – branches, rings – least reactive ¬ alkenes – branches, rings ¬ alkynes – branches – most reactive ¬ all called aliphatics ¬ all nonpolar and not soluble in water ¬ major intermolecular forces are LD boiling points are low
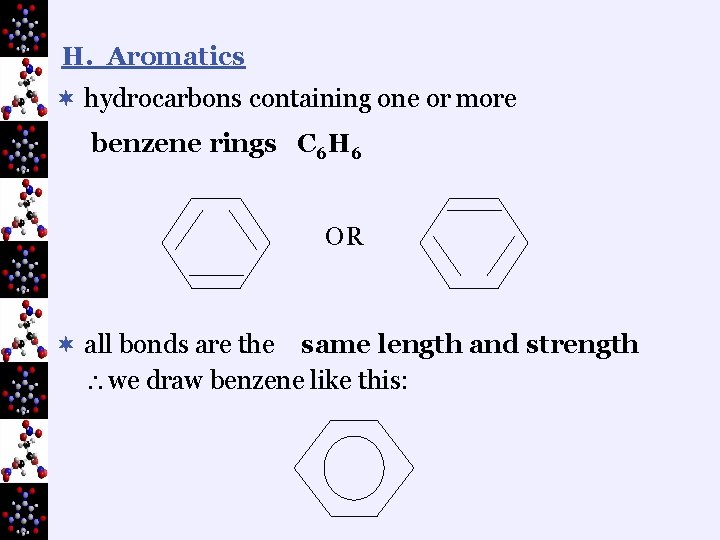
H. Aromatics ¬ hydrocarbons containing one or more benzene rings C 6 H 6 OR ¬ all bonds are the same length and strength we draw benzene like this:

1. Properties ¬ nonpolar ¬ the benzene ring structure is very stable ¬aromatics are characterized by strong aromas

2. Uses ¬ ASA, amphetamines, adrenaline, benzocaine (anesthetic) ¬ moth balls, TNT ¬ wintergreen, menthol, vanilla, cinnamon ¬ SPF in sunscreen
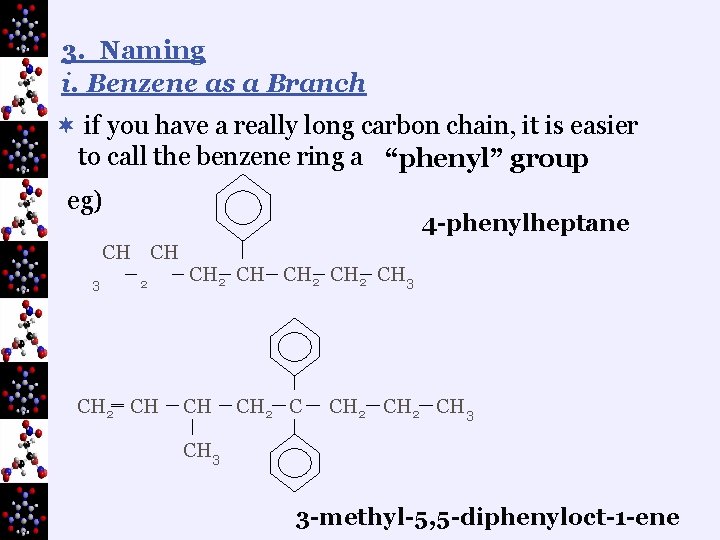
3. Naming i. Benzene as a Branch ¬ if you have a really long carbon chain, it is easier to call the benzene ring a “phenyl” group eg) 4 -phenylheptane CH CH 3 2 ‗ CH 2 CH 3 CH CH 2 CH 3 3 -methyl-5, 5 -diphenyloct-1 -ene
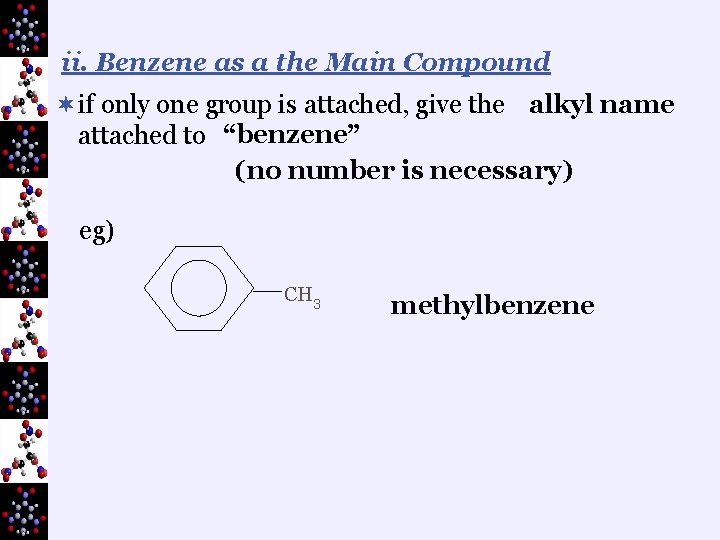
ii. Benzene as a the Main Compound ¬if only one group is attached, give the alkyl name attached to “benzene” (no number is necessary) eg) CH 3 methylbenzene
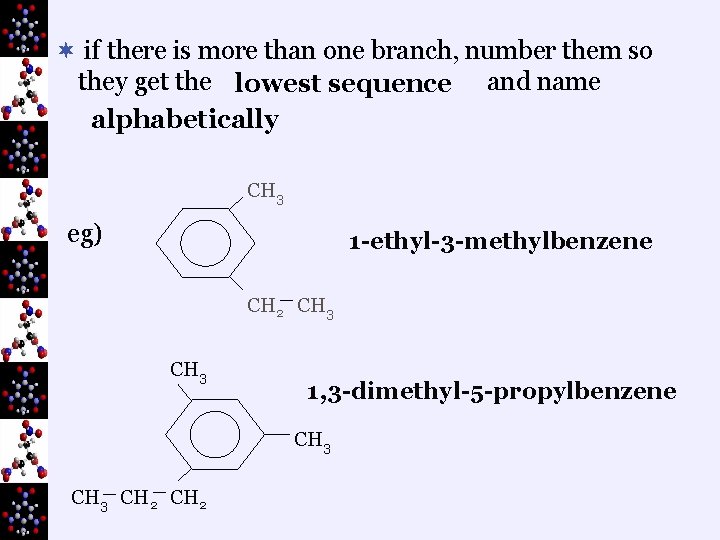
¬ if there is more than one branch, number them so they get the lowest sequence and name alphabetically CH 3 eg) 1 -ethyl-3 -methylbenzene CH 2 CH 3 1, 3 -dimethyl-5 -propylbenzene CH 3 CH 2
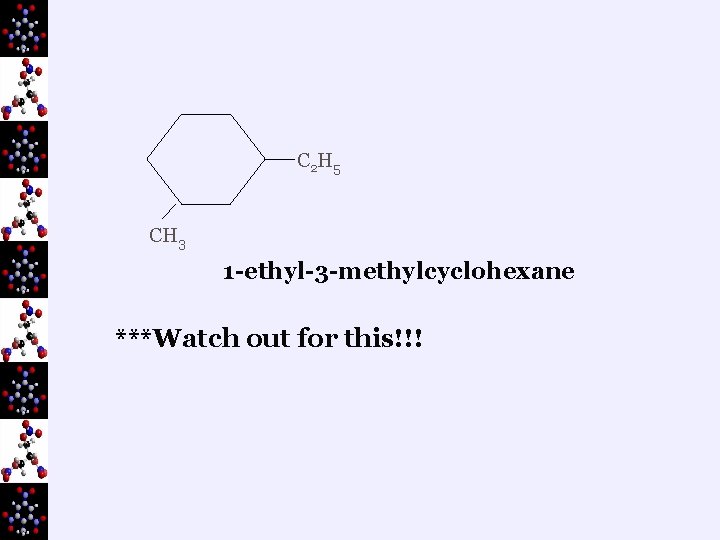
C 2 H 5 CH 3 1 -ethyl-3 -methylcyclohexane ***Watch out for this!!!

I. Alcohols R - OH ¬ organic compounds with one or more OH (hydroxyl) groups 1. Properties ¬ have much higher boiling points than corresponding aliphatics because of hydrogen bonding! eg) methane (CH 4) BP = -162 C methanol (CH 3 OH) BP = 65 C

¬ polar ¬ the –OH end of the alcohol is polar while the carbon chain end is not small alcohols are soluble in water and large alcohols are not 2. Uses ¬ antifreeze, rubbing alcohol, beverages, moistening agent

3. Naming ¬ number the longest carbon chain containing the hydroxyl group so the –OH group gets the lowest number ¬ aliphatic name (without “e” at end) + “OL” ¬ give the number for the carbon where the –OH group is found between the parent name and the suffix

¬if there is more than one hydroxyl group, use a prefix ( di , tri , tetra ) to indicate the number of OH groups and place the numbers between the parent name and the suffix ***Note, if the suffix starts with a vowel, drop the “e” on the parent name; if the suffix starts with a consonant, keep the “e” on the parent name
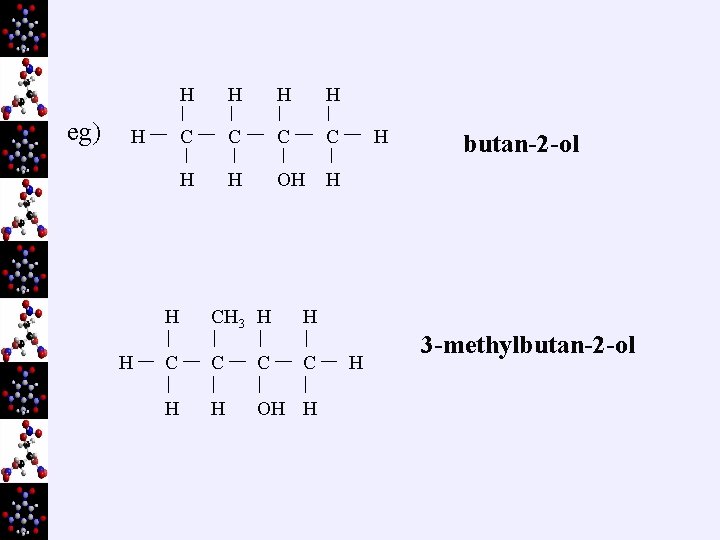
eg) H H C C H H OH H CH 3 H H C C H H OH H H H butan-2 -ol 3 -methylbutan-2 -ol
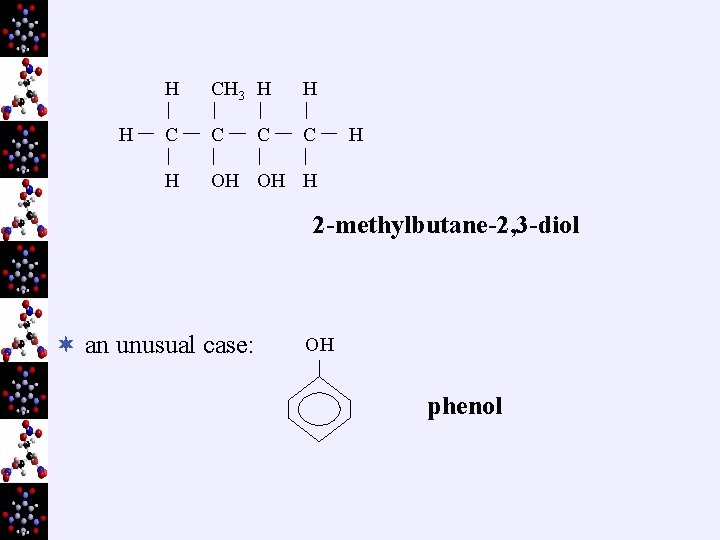
H H CH 3 H H C C C H OH OH H C H 2 -methylbutane-2, 3 -diol ¬ an unusual case: OH phenol

J. Organic Halides R-X ¬ where R is carbon chain or ring halogen and X is a ¬ organic compounds where hydrogen has been replaced by one or more halogens (F, Cl, Br, I) ¬ do not readily occur in nature

1. Properties ¬ can be polar or nonpolar , depending on the placement of the halogen groups ¬many are toxic and dangerous 2. Uses ¬ manufactured for human use eg) DDT, PCB, CFC ¬ anesthetics ¬ dry cleaning fluid ¬ plastics, polymers (Teflon)

3. Naming ¬ same rules as before… name branches ( halogens included now) alphabetically F = fluoro Cl = chloro Br = bromo I = iodo eg) H H Cl Cl H C C H H H 2, 3 -dichlorobutane
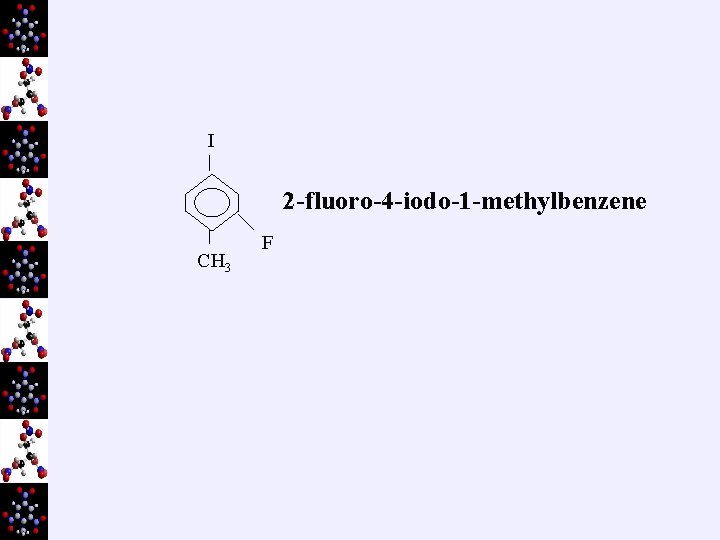
I 2 -fluoro-4 -iodo-1 -methylbenzene CH 3 F

K. Carboxylic Acids R O ║ C OH ¬ where R is carbon chain or ring ¬ organic compounds containing the carboxyl functional group (-COOH) 1. Properties ¬ polar dissolve in water ¬ high boiling points due to hydrogen bonding ¬ weakly acidic ¬ diagnostic test: use litmus paper (will turn red ), readily react with metals, neutralize bases

2. Uses ¬ recycling rubber – methanoic acid ¬ vinegar – ethanoic (acetic) acid ¬ rust remover – oxalic acid ¬ fruits – citric acid 3. Naming ¬ count the longest carbon chain including the carbon in the carboxyl group ¬the carbon in the carboxyl group is always number 1 ¬ drop “e” and add “OIC ACID”
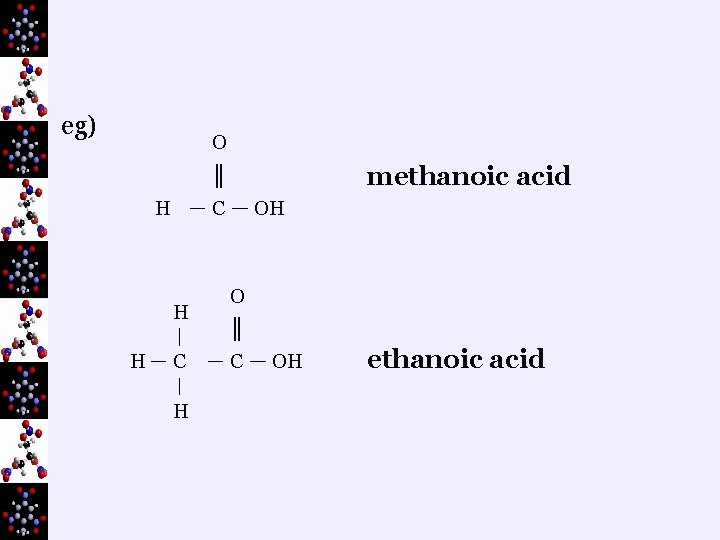
eg) O methanoic acid ║ H C H OH O ║ C OH ethanoic acid
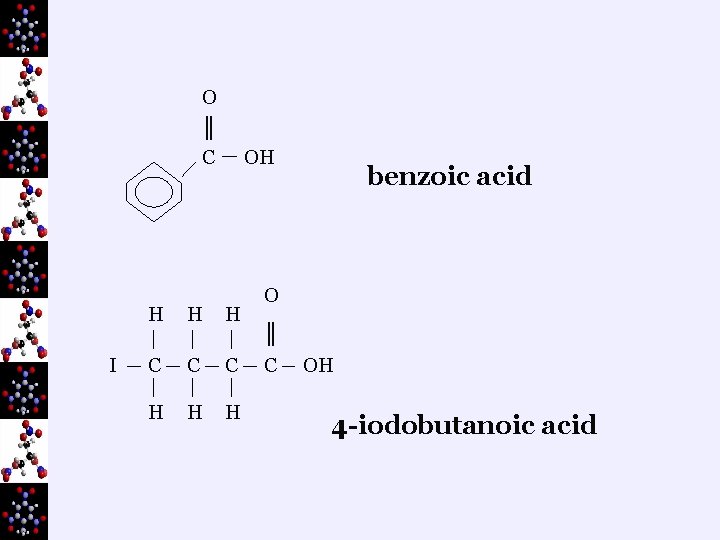
O ║ C I OH H C C C H H H benzoic acid O ║ C OH 4 -iodobutanoic acid

L. Esters R O ║ C O R’ ¬where R can be a carbon chain or hydrogen and R’ can be a carbon chain ¬combination of a carboxylic acid an alcohol 1. Properties ¬ polar ¬ small esters dissolve in water, large esters do not ¬ boiling points slightly lower than corresponding carboxylic acids and alcohols due to lack of hydrogen bonding

¬very volatile aromas which allows them to generate 2. Uses ¬ flavouring agents 3. Naming ¬identify the alcohol used to make the ester ¬change the alcohol name to the corresponding alkyl name eg) methanol would become methyl ¬ identify the made from carboxylic acid the ester was

¬drop the “oic acid” and replace with “oate” eg) butanoic acid would become “butanoate” ¬put the two names between together with a space in eg) methyl butanoate ¬you can have branches on esters…they follow the alphabetical rule, numbering begins at the O end of the alcohol and the C=O end of the carboxylic acid
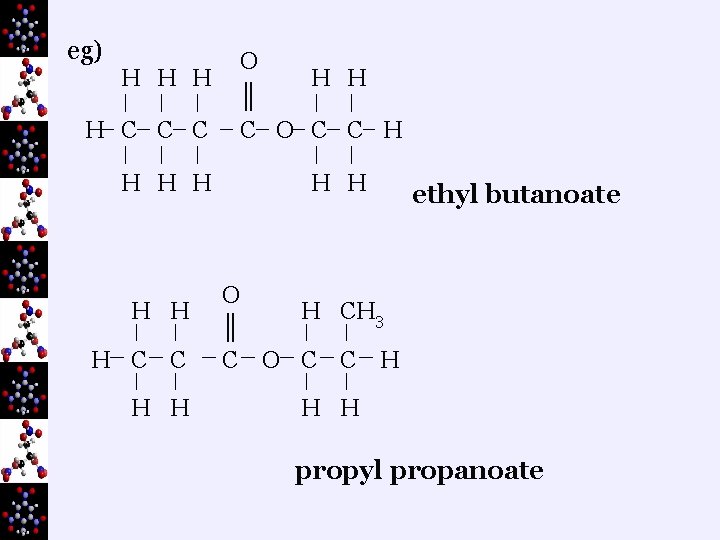
eg) H H C C C H H H C C H H O H H ║ C O C C H H H ethyl butanoate O H CH 3 ║ C O C C H H H propyl propanoate
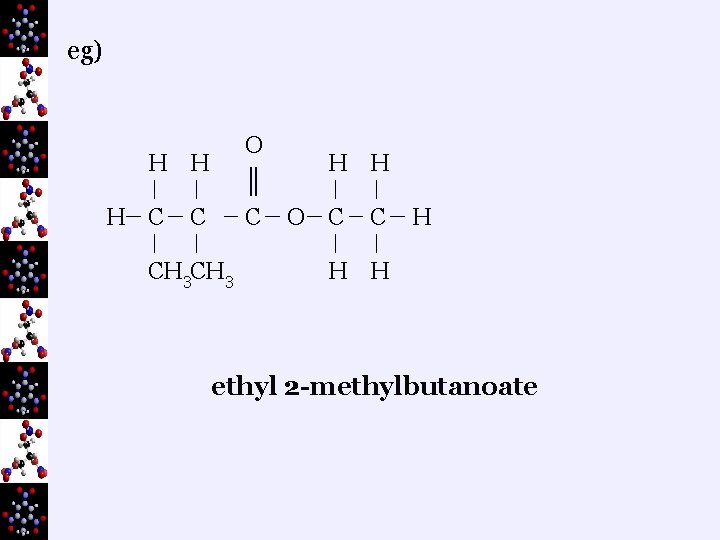
eg) O H H H C ║ C C CH 3 H H O C C H H H ethyl 2 -methylbutanoate


M. Boiling Points and Solubility ¬we can compare the boiling points of various organic compounds using their and polarity the intermolecular attractions between the molecules Example 1 Put the following organic compounds in order from highest boiling point to lowest boiling point. alcohol, alkane, alkene, aromatic, carboxylic acid alcohol aromatic alkane alkene highest lowest
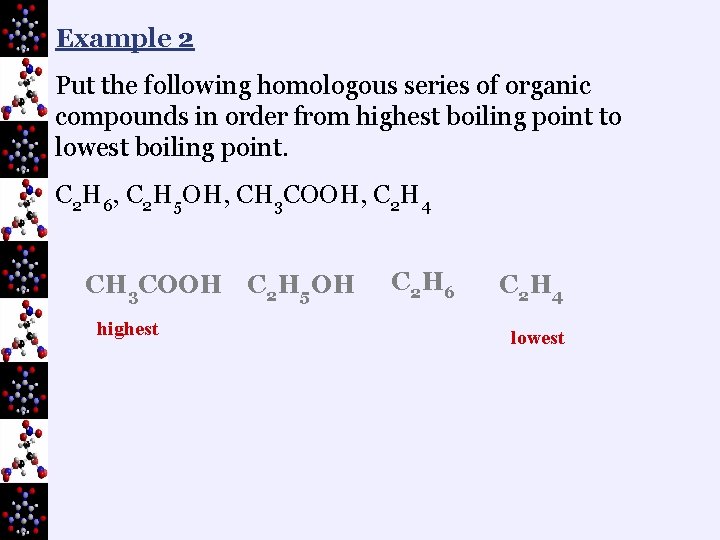
Example 2 Put the following homologous series of organic compounds in order from highest boiling point to lowest boiling point. C 2 H 6, C 2 H 5 OH, CH 3 COOH, C 2 H 4 CH 3 COOH C 2 H 5 OH highest C 2 H 6 C 2 H 4 lowest

¬we can also compare the solubility of various organic compounds using their polarity Insoluble Organic Compounds Soluble Organic Compounds aliphatics – alkanes, alkenes, alkynes carboxylic acids aromatics alcohols – large (7 or more carbons) alcohols – small (less than 7 carbons) esters – large esters – small organic halides

N. Organic Reactions 1. Combustion Reactions ¬ occurs when a hydrocarbon reacts with oxygen ¬ products are always carbon dioxide and water ¬these are economically important reactions for they are the major reactions that produce thermal energy required for fuelling our vehicles, heating our homes, and producing electricity eg) 1 C 5 H 12(l) + 8 O 2(g) 5 CO 2(g) + 6 H O 2 (g)
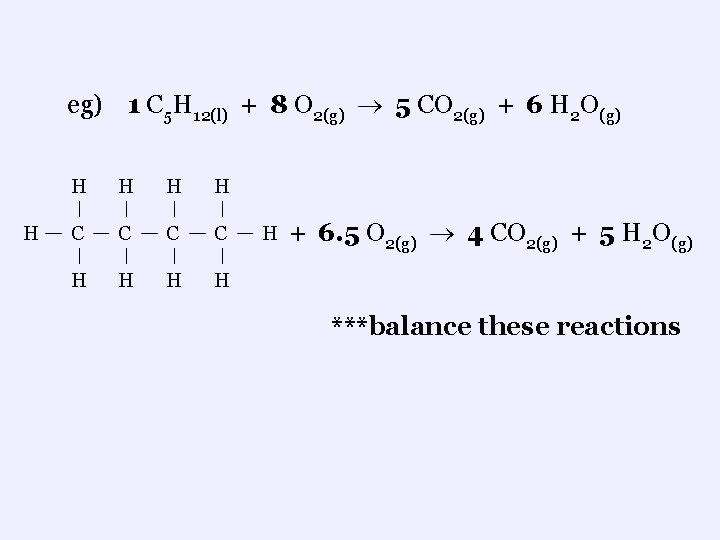
eg) 1 C 5 H 12(l) + 8 O 2(g) 5 CO 2(g) + 6 H 2 O(g) H H H C C H H H + 6. 5 O 2(g) 4 CO 2(g) + 5 H 2 O(g) ***balance these reactions
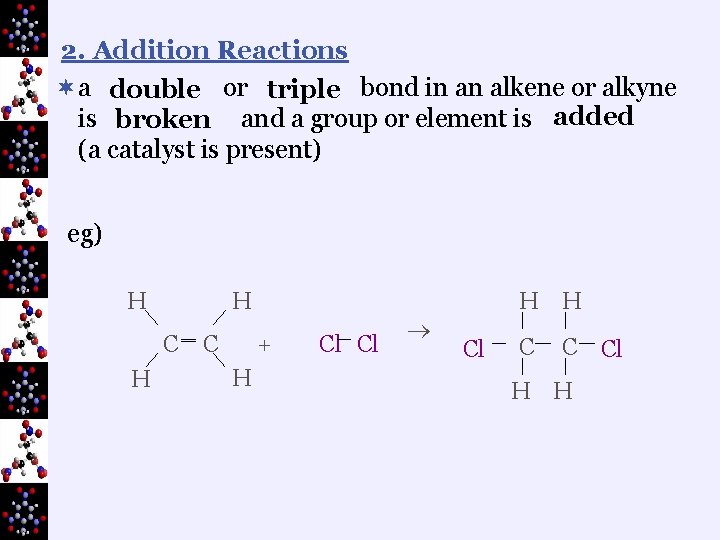
2. Addition Reactions ¬a double or triple bond in an alkene or alkyne is broken and a group or element is added (a catalyst is present) eg) H H C H ‗ C + H Cl Cl Cl H H C C Cl H H
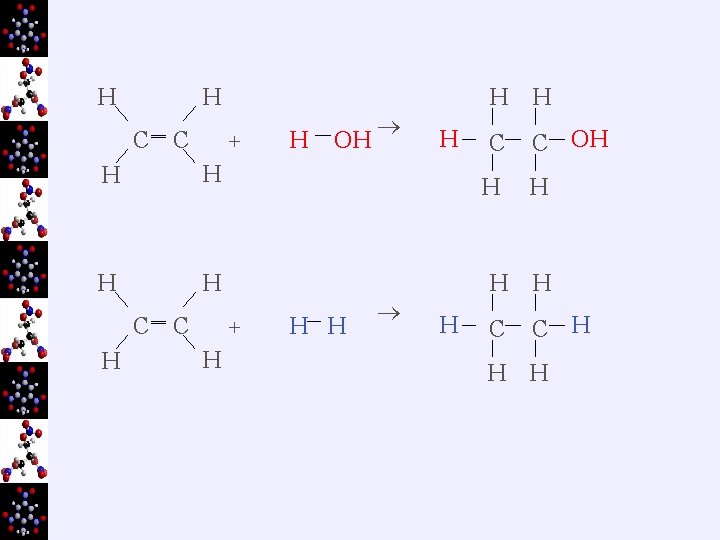
H H C ‗ C + H H C H ‗ C OH H H C C OH H + H H H H H C C H H H
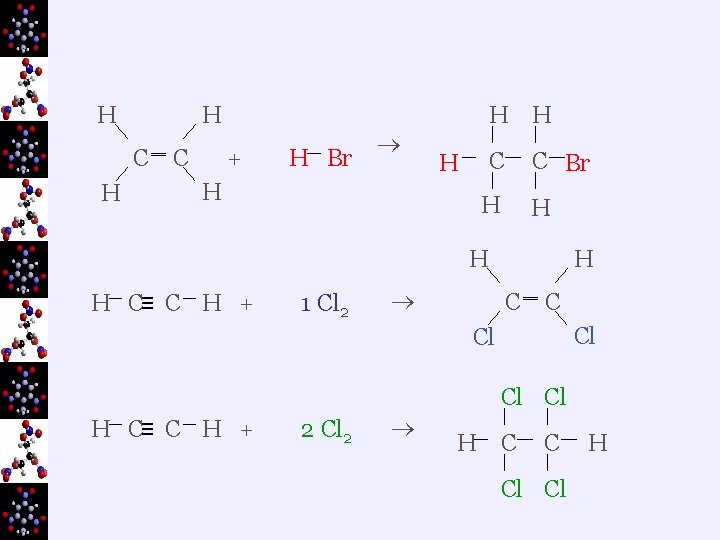
H C H ‗ C + H Br H H C C Br H H H C≡ C H + 1 Cl 2 H C ‗ C Cl Cl H C≡ C H + 2 Cl 2 H C C Cl Cl H

3. Substitution Reactions ¬the replacement ( substitution ) of a hydrogen on an alkane or aromatic with another atom (eg. F, Cl etc) ¬commonly used to make organic halides Examples 1. H H C H H H + cat Cl – Cl H C Cl H + H - Cl
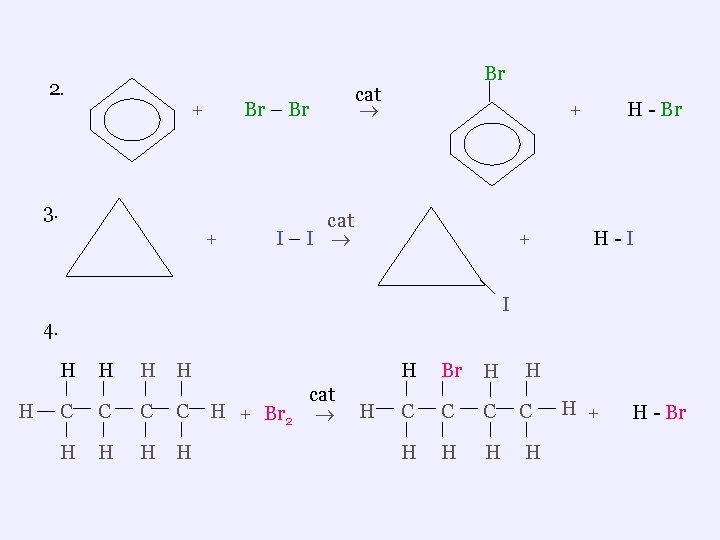
2. + Br – Br 3. + Br cat + cat I–I + H - Br H-I I 4. H H H C C H H H + Br 2 cat H H Br H C C C H H + H - Br
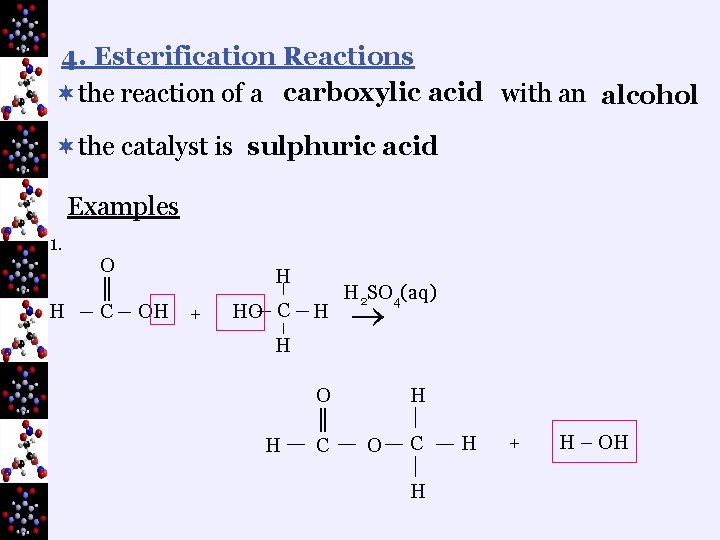
4. Esterification Reactions ¬the reaction of a carboxylic acid with an alcohol ¬the catalyst is sulphuric acid Examples 1. H O ║ C H OH + HO C H H 2 SO 4(aq) H H O ║ C H O C H H + H – OH
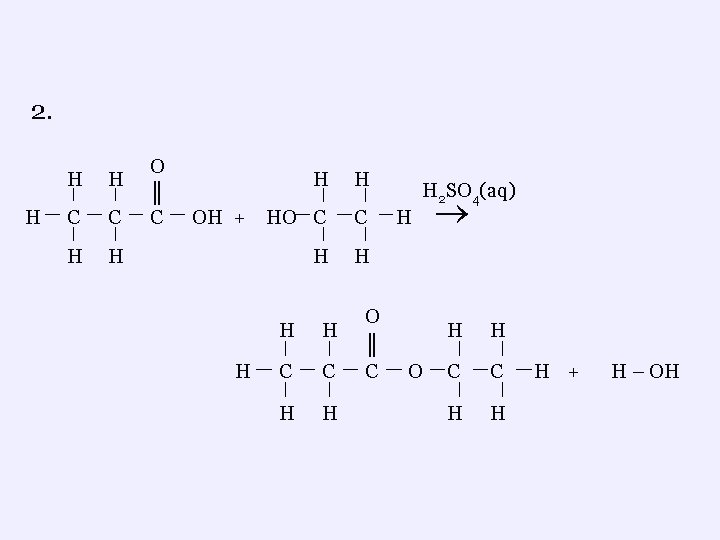
2. H H H C C H H O ║ C H H OH + HO C C H H H C C H H O ║ C H 2 SO 4(aq) H O H H C C H H H + H – OH

5. Elimination Reactions ¬an alcohol has water removed, forming an alkene plus the water ¬ organic halides can react with a base (hydroxide) to produce an alkene , a halide ion and water Examples 1. H H H C C H H OH cat H H H C‗ C H + H – OH
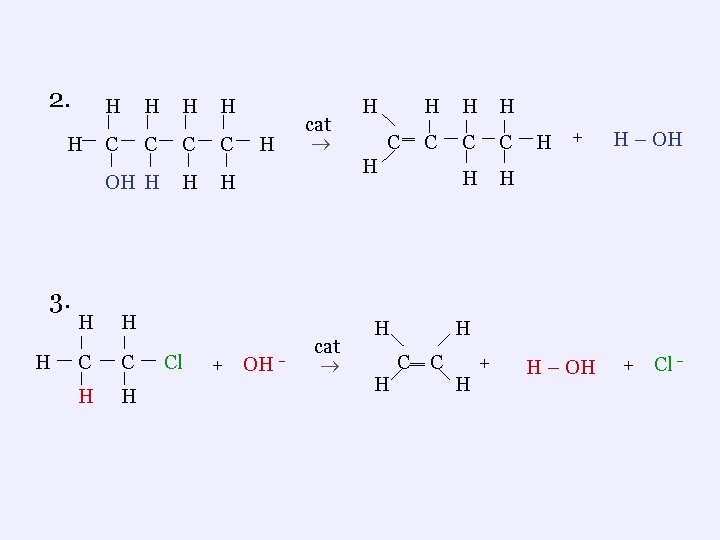
2. H 3. H H H C C OH H H C C H H Cl cat H + OH H C ‗ H H H C C C H H H cat H H H + H – OH + Cl H C‗ C H +

6. Polymerization Reactions ¬a monomer is a simple molecule the base unit for a polymer that forms ¬a polymer is a very, very long molecule formed by the covalent bonding of bazillions of monomers ¬depending on the polymer, the monomers that make it up can be the sameor different
¬polymers can be natural eg) carbohydrates, proteins, DNA

¬polymers can be synthetic eg) nylon, PVC, Teflon, polyester

¬polymers that can be heated and molded into specific shapes are commonly called plastics ¬plastics are one type of synthetic compound that has been of great benefit to society (although there also problems associated with then) ¬the names of polymers are the monomer name with “poly” in front ¬many have classical names instead of IUPAC names
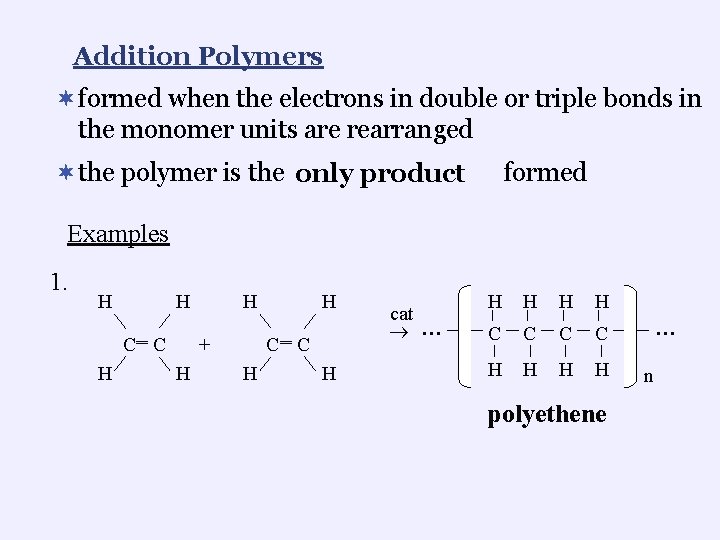
Addition Polymers ¬formed when the electrons in double or triple bonds in the monomer units are rearranged ¬the polymer is the only product formed Examples 1. H H ‗ C C H H ‗ + H H C C H H cat … H H C C H H polyethene … n
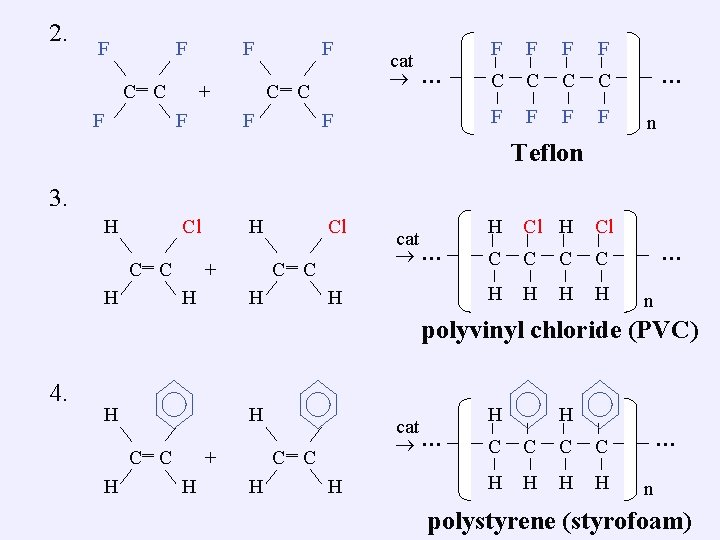
2. F F ‗ C C F F ‗ + F F C C F cat … F F F C C F F … n Teflon 3. H Cl ‗ C C H H ‗ C C + H Cl H cat … H H Cl C C H H … n polyvinyl chloride (PVC) 4. H H ‗ C C H ‗ + H cat … C C H H … n polystyrene (styrofoam)

Condensation Polymers ¬polymerization reactions that involve the formation of a small molecule (commonly water ) as well as the polymer ¬each monomer must have two functional groups ¬two common linkages formed: 1. ester linkage – between carboxyl group ( COOH) and hydroxyl group ( OH) 2. amide linkage – between amino group ( NH 2) and carboxyl group ( COOH)
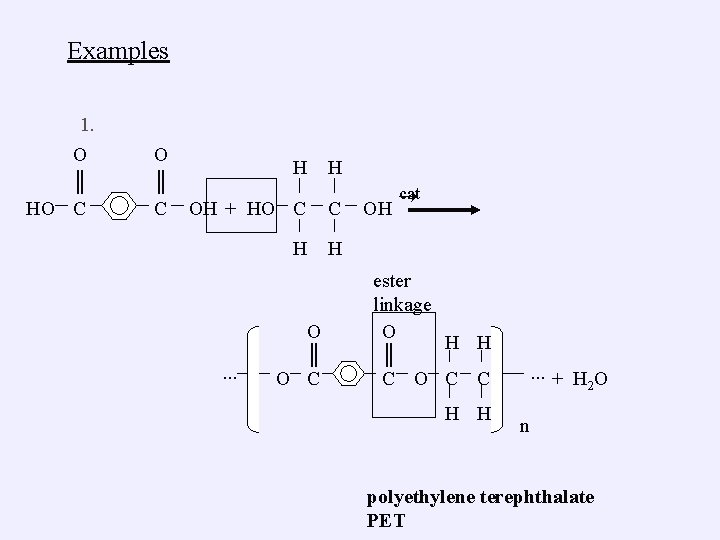
Examples 1. O ║ HO C O ║ C H H OH + HO C C H H ∙∙∙ O ║ O C OH cat ester linkage O H H ║ C O C C H H ∙∙∙ + H 2 O n polyethylene terephthalate PET
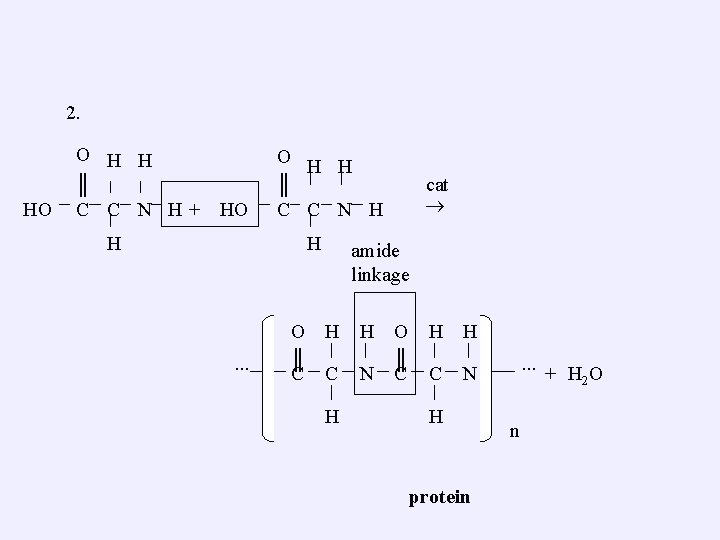
2. HO O H H ║ C C N H + HO O H H ║ C C N H H H ∙∙∙ cat amide linkage O H H ║ ║ C C N H H protein ∙∙∙ + H 2 O n

O. Petroleum Refining ¬Alberta has vast reserves of petroleum in the form of natural gas, crude oil and oil sand deposits ¬most of this petroleum is refined and then burned as fuel ¬petrochemicals are also used in the production of plastics

¬refining of petroleum separates the crude mixture into purified components ¬ fractional distillation is used to separate the components ¬distillation works because of the different boiling points of the components of crude oil ¬the lighter the molecule and the lower the boiling point, the it rises in the tower (asphalt, fuel higher oil, wax at bottom; gasoline at top) distillation tower: http: //home. att. net/~cat 6 a/fuels-IV. htm

¬ two types of reactions in petroleum refining: 1. Cracking ¬ breaks long chain hydrocarbons into smaller units ¬cracking requires heat and pressure ¬there are many different types of cracking reactions, forming different products (alkanes, alkenes) eg) catalytic cracking, steam cracking, hydrocracking ¬hydrocracking requires H 2(g)

Examples 1. C 17 H 36 + cat H 2(g) C 9 H 20 + C 8 H 18 2. CH 3 CH 2 cat CH 3 CH 2 + H 2(g) CH 3 CH 2 CH 3 + CH 3 CH 2 CH 3

2. Reforming ¬small hydrocarbons are joined to make larger molecules ¬requires heat and pressure ¬there are several types of reforming reactions eg) alkylation to produce “high octane” gasoline ¬all reforming reactions produce H 2(g) Example C 7 H 16 + C 12 H 26 C 19 H 40 + H 2

Examples 1. C 7 H 16 + C 12 H 26 C 19 H 40 + H 2 2. ethane + octane decane + hydrogen gas
- Slides: 94