Organic Chemistry 6 th Edition Paula Yurkanis Bruice
Organic Chemistry 6 th Edition Paula Yurkanis Bruice Chapter 22 The Organic Chemistry of Carbohydrates Introduction, Basic structural features and types of carbohydrates, Reactions and conversions, role in biological systems. 1 © 2011 Pearson Education, Inc.

Carbohydrates • General molecular formula is Cn(H 2 O)n…. ’hydrates of carbon’ • Structurally, carbohydrades are polyhydroxy aldehydes or ketones. • The most abundant carbohydrate in nature is glucose. The carbohydrates may be defined as optically active polyhydroxy aldehydes or ketones or the compounds which produce such units on hydrolysis Exam. Sucrose: used in our home Lactose: present in milk 2 © 2011 Pearson Education, Inc.

Carbohydrates are also called saccharides (Greek: sakcharon means sugar) Simple carbohydrates are monosaccharides: Cannot be hydrohyzed further; About 20 monosaccharides are known in nature…glucose, fructose, ribose etc Complex carbohydrates contain two or more sugar units linked together: Sucrose on hydrolysis gives glucose and fructose but maltose gives only two • Disaccharides molecules of glucose • Oligosaccharides Polysaccharides are not sweet and are • Polysaccharides called non-sugar 3 © 2011 Pearson Education, Inc.

Polyhydroxy aldehydes are aldoses Polyhydroxy ketones are ketoses Position of carbonyl group at C 1, carbonyl is an aldehyde: aldose at any other carbon, carbonyl is a ketone: ketose Number of carbons three carbons: triose four carbons: tetrose five carbons: pentose six carbons: hexose seven carbons: heptose etc. 4 © 2011 Pearson Education, Inc.
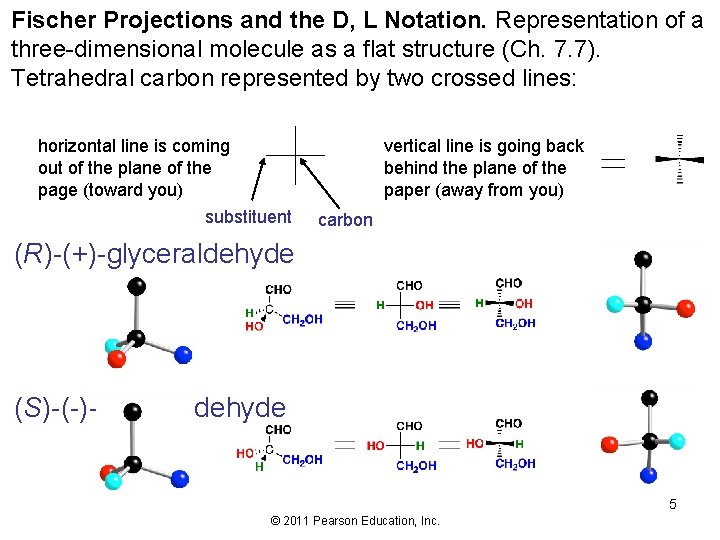
Fischer Projections and the D, L Notation. Representation of a three-dimensional molecule as a flat structure (Ch. 7. 7). Tetrahedral carbon represented by two crossed lines: horizontal line is coming out of the plane of the page (toward you) vertical line is going back behind the plane of the paper (away from you) substituent carbon (R)-(+)-glyceraldehyde (S)-(-)-glyceraldehyde 5 © 2011 Pearson Education, Inc.
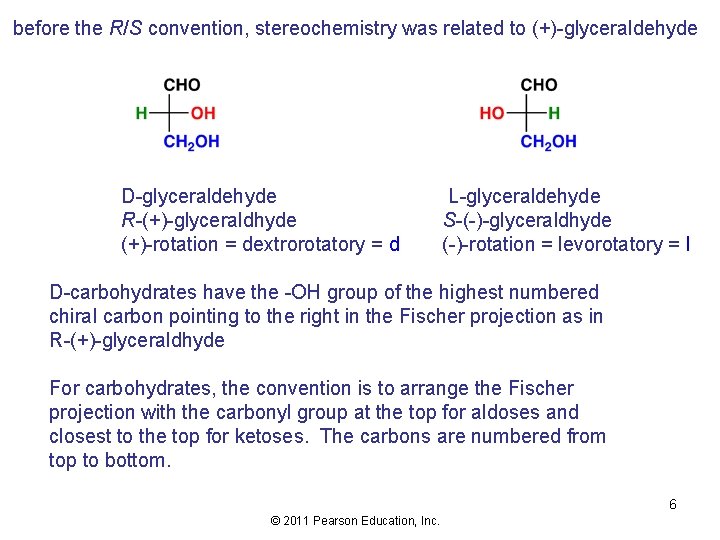
before the R/S convention, stereochemistry was related to (+)-glyceraldehyde D-glyceraldehyde R-(+)-glyceraldhyde (+)-rotation = dextrorotatory = d L-glyceraldehyde S-(-)-glyceraldhyde (-)-rotation = levorotatory = l D-carbohydrates have the -OH group of the highest numbered chiral carbon pointing to the right in the Fischer projection as in R-(+)-glyceraldhyde For carbohydrates, the convention is to arrange the Fischer projection with the carbonyl group at the top for aldoses and closest to the top for ketoses. The carbons are numbered from top to bottom. 6 © 2011 Pearson Education, Inc.

Carbohydrates are designated as D- or L- according to the stereochemistry of the highest numbered chiral carbon of the Fischer projection. If the hydroxyl group of the highest numbered chiral carbon is pointing to the right, the sugar is designated as D (Dextro: Latin for on the right side). If the hydroxyl group is pointing to the left, the sugar is designated as L (Levo: Latin for on the left side). Most naturally occurring carbohydrates are of the D-configuration. 7 © 2011 Pearson Education, Inc.
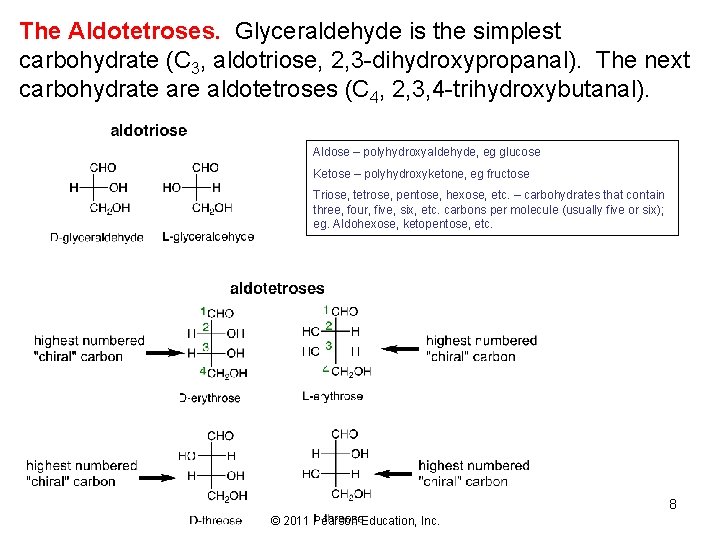
The Aldotetroses. Glyceraldehyde is the simplest carbohydrate (C 3, aldotriose, 2, 3 -dihydroxypropanal). The next carbohydrate are aldotetroses (C 4, 2, 3, 4 -trihydroxybutanal). Aldose – polyhydroxyaldehyde, eg glucose Ketose – polyhydroxyketone, eg fructose Triose, tetrose, pentose, hexose, etc. – carbohydrates that contain three, four, five, six, etc. carbons per molecule (usually five or six); eg. Aldohexose, ketopentose, etc. 8 © 2011 Pearson Education, Inc.

Aldopentoses and Aldohexoses. Aldopentoses: C 5, three chiral carbons, eight stereoisomers Aldohexoses: C 6, four chiral carbons, sixteen stereoisomers 9 © 2011 Pearson Education, Inc.

• A ketose has one fewer asymmetric center than an aldose. • Therefore, ketoses have fewer stereoisomers than aldoses. • Naturally occurring ketoses have the ketone group in the 2 -position. 10 © 2011 Pearson Education, Inc.
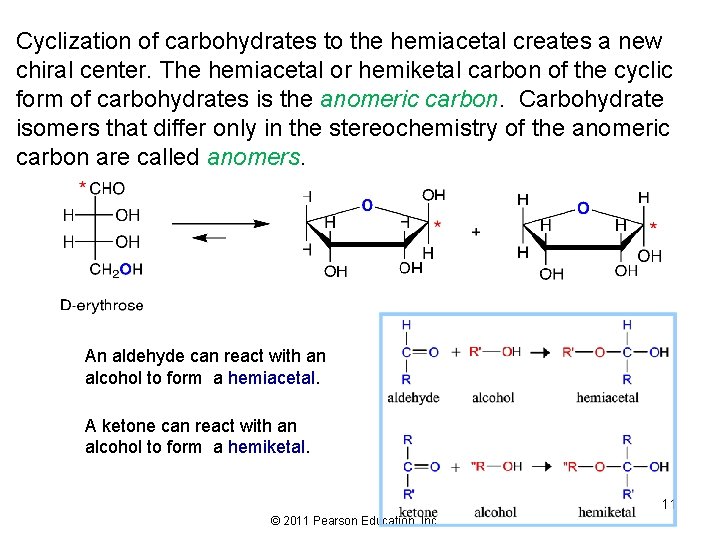
Cyclization of carbohydrates to the hemiacetal creates a new chiral center. The hemiacetal or hemiketal carbon of the cyclic form of carbohydrates is the anomeric carbon. Carbohydrate isomers that differ only in the stereochemistry of the anomeric carbon are called anomers. An aldehyde can react with an alcohol to form a hemiacetal. A ketone can react with an alcohol to form a hemiketal. 11 © 2011 Pearson Education, Inc.
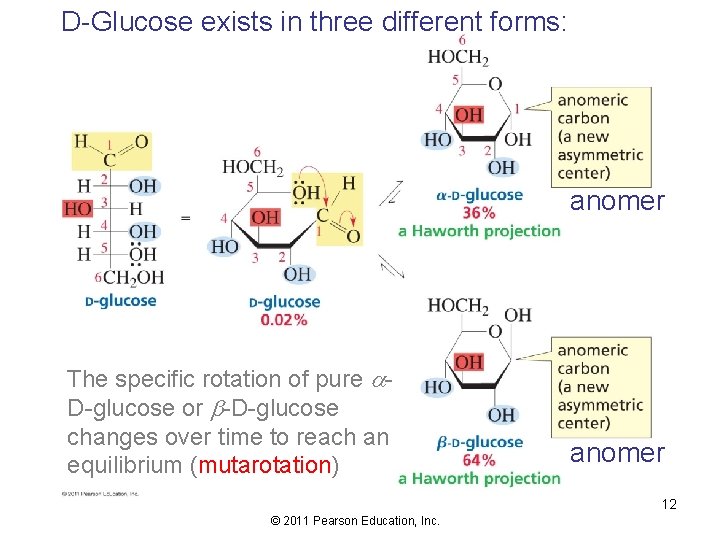
D-Glucose exists in three different forms: anomer The specific rotation of pure a. D-glucose or b-D-glucose changes over time to reach an equilibrium (mutarotation) anomer 12 © 2011 Pearson Education, Inc.
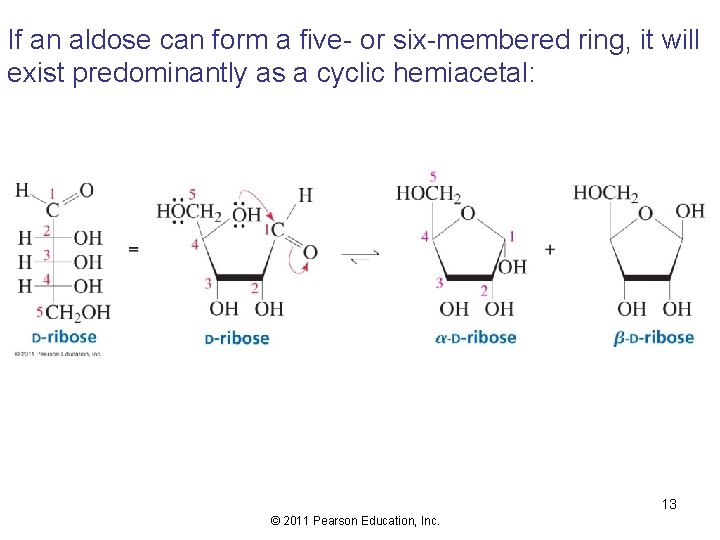
If an aldose can form a five- or six-membered ring, it will exist predominantly as a cyclic hemiacetal: 13 © 2011 Pearson Education, Inc.
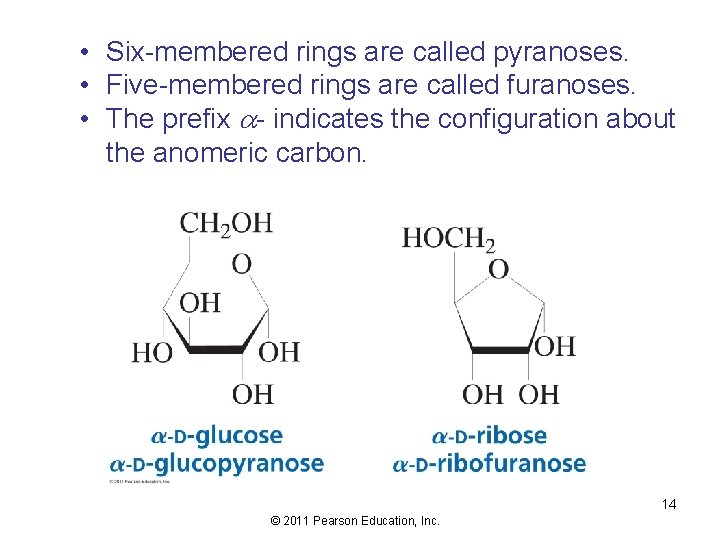
• Six-membered rings are called pyranoses. • Five-membered rings are called furanoses. • The prefix a- indicates the configuration about the anomeric carbon. 14 © 2011 Pearson Education, Inc.
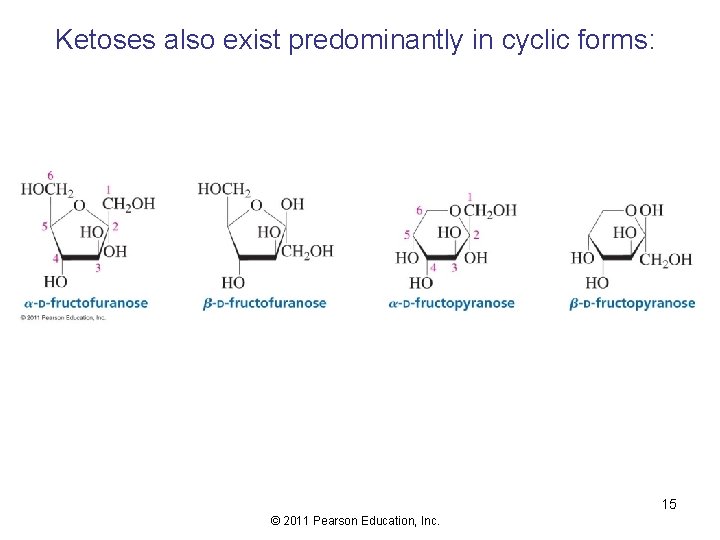
Ketoses also exist predominantly in cyclic forms: 15 © 2011 Pearson Education, Inc.
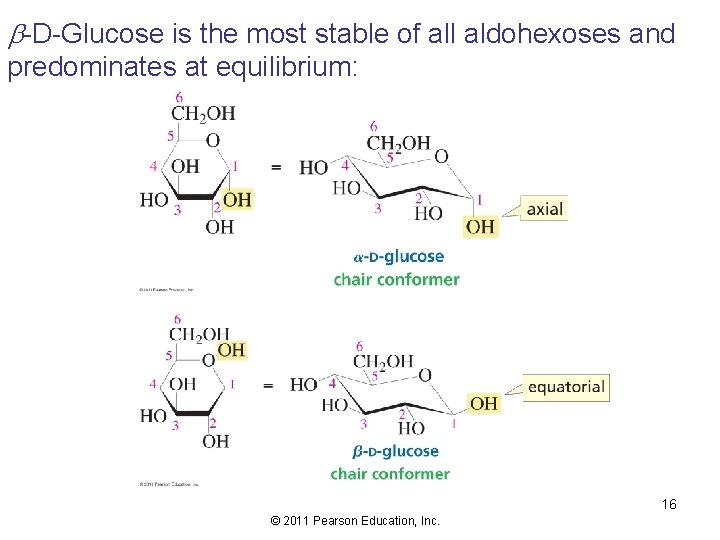
b-D-Glucose is the most stable of all aldohexoses and predominates at equilibrium: 16 © 2011 Pearson Education, Inc.
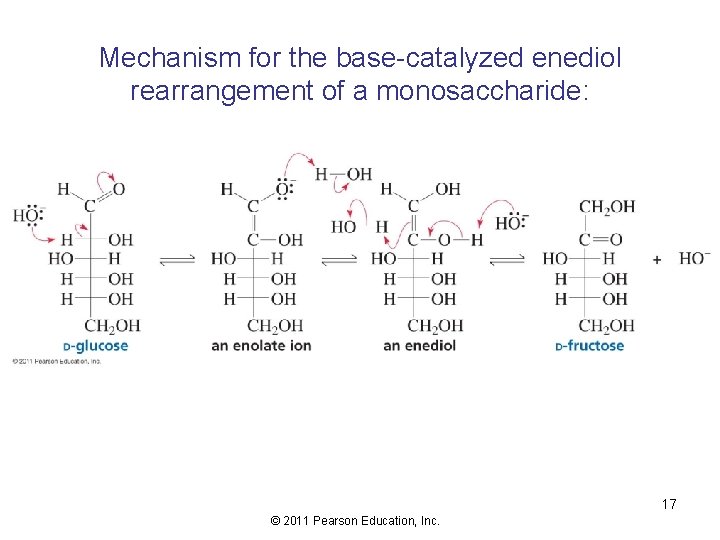
Mechanism for the base-catalyzed enediol rearrangement of a monosaccharide: 17 © 2011 Pearson Education, Inc.

Redox Reactions of Monosaccharides The carbonyl of aldoses and ketoses can be reduced by the carbonyl-group reducing agents: 18 © 2011 Pearson Education, Inc.
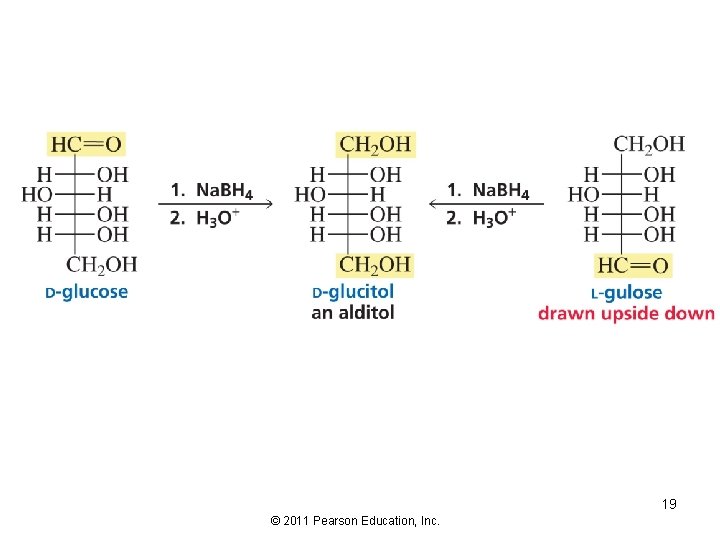
19 © 2011 Pearson Education, Inc.
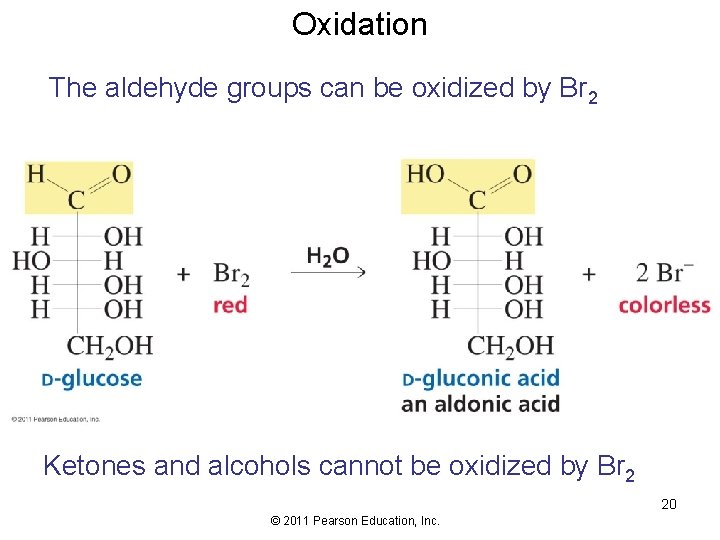
Oxidation The aldehyde groups can be oxidized by Br 2 Ketones and alcohols cannot be oxidized by Br 2 20 © 2011 Pearson Education, Inc.
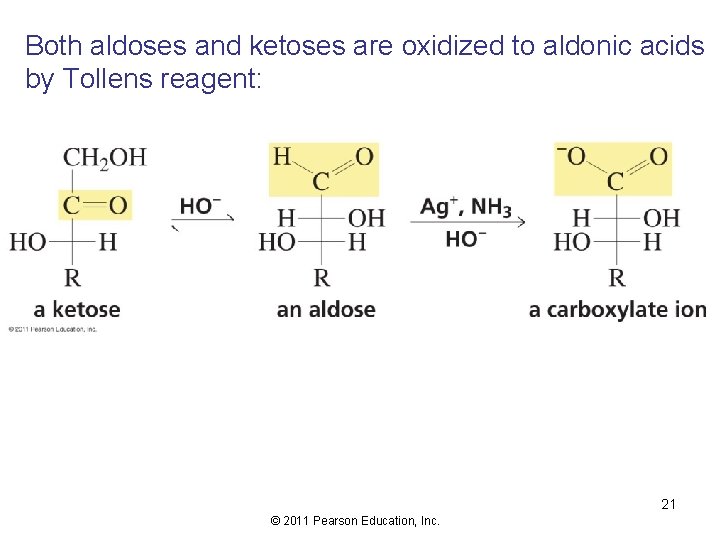
Both aldoses and ketoses are oxidized to aldonic acids by Tollens reagent: 21 © 2011 Pearson Education, Inc.
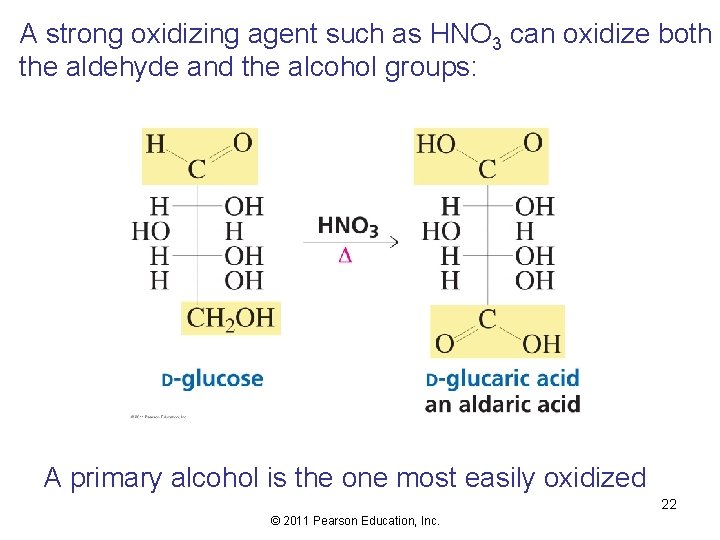
A strong oxidizing agent such as HNO 3 can oxidize both the aldehyde and the alcohol groups: A primary alcohol is the one most easily oxidized 22 © 2011 Pearson Education, Inc.
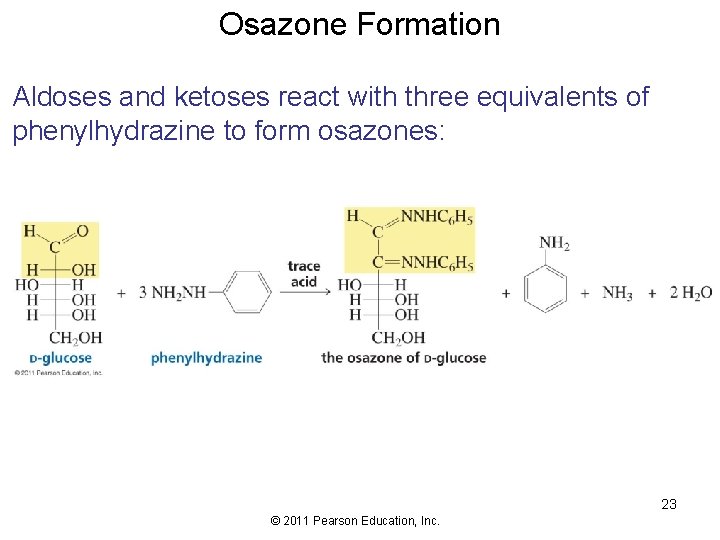
Osazone Formation Aldoses and ketoses react with three equivalents of phenylhydrazine to form osazones: 23 © 2011 Pearson Education, Inc.
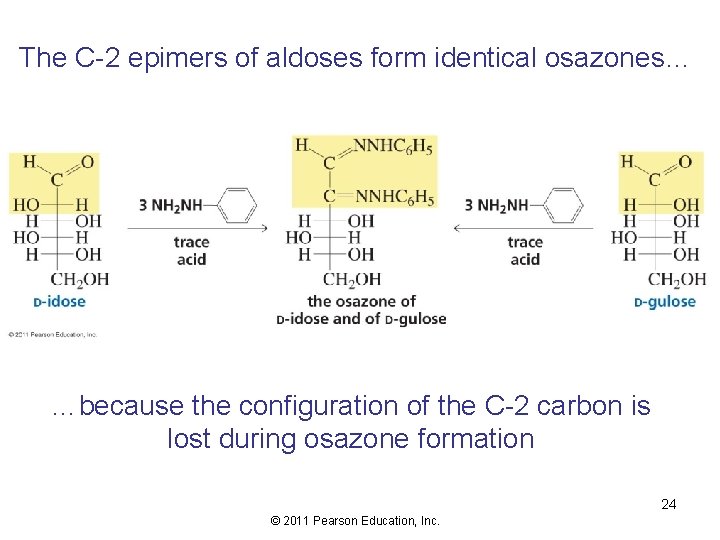
The C-2 epimers of aldoses form identical osazones… …because the configuration of the C-2 carbon is lost during osazone formation 24 © 2011 Pearson Education, Inc.
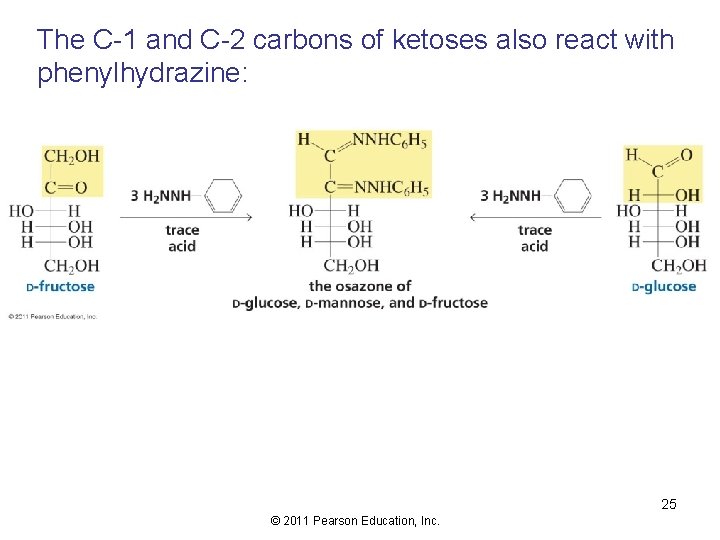
The C-1 and C-2 carbons of ketoses also react with phenylhydrazine: 25 © 2011 Pearson Education, Inc.
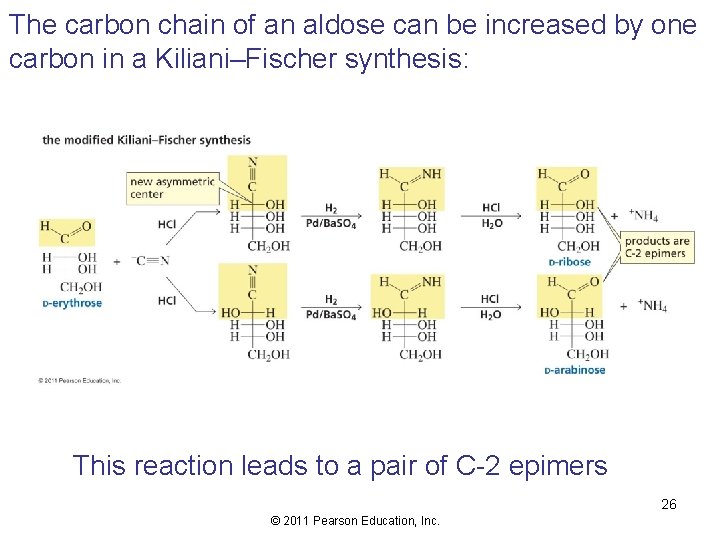
The carbon chain of an aldose can be increased by one carbon in a Kiliani–Fischer synthesis: This reaction leads to a pair of C-2 epimers 26 © 2011 Pearson Education, Inc.
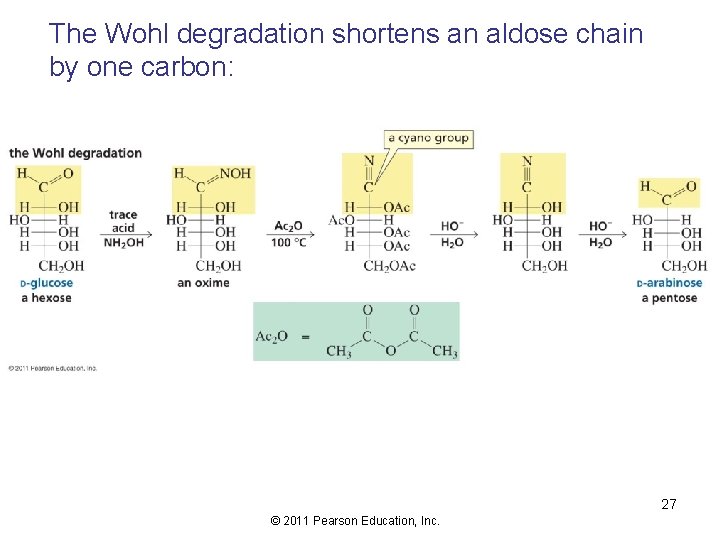
The Wohl degradation shortens an aldose chain by one carbon: 27 © 2011 Pearson Education, Inc.
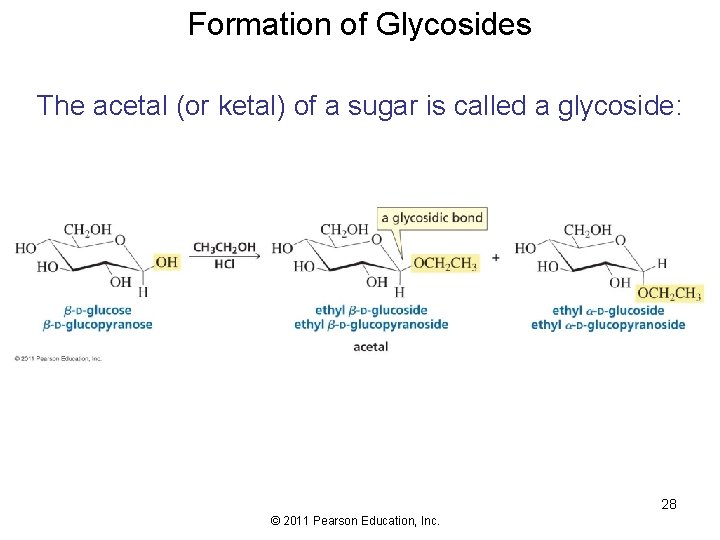
Formation of Glycosides The acetal (or ketal) of a sugar is called a glycoside: 28 © 2011 Pearson Education, Inc.
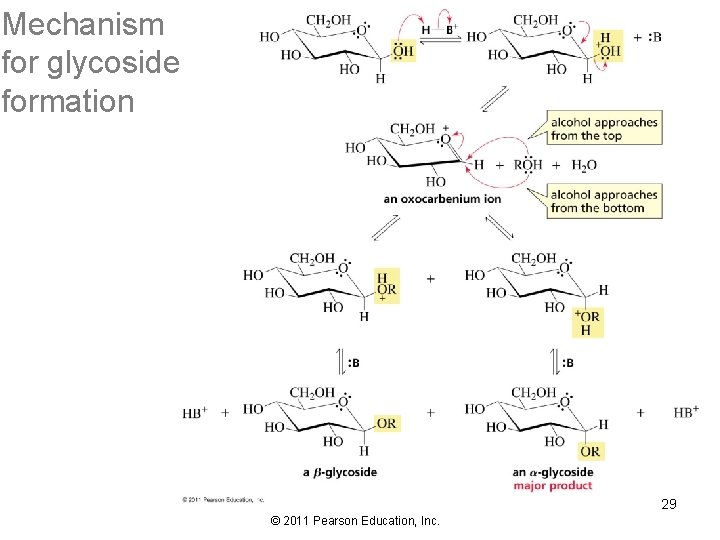
Mechanism for glycoside formation 29 © 2011 Pearson Education, Inc.
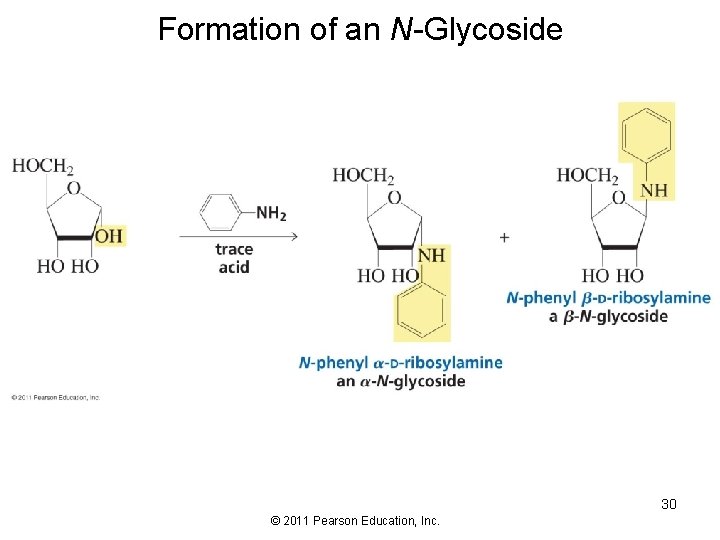
Formation of an N-Glycoside 30 © 2011 Pearson Education, Inc.
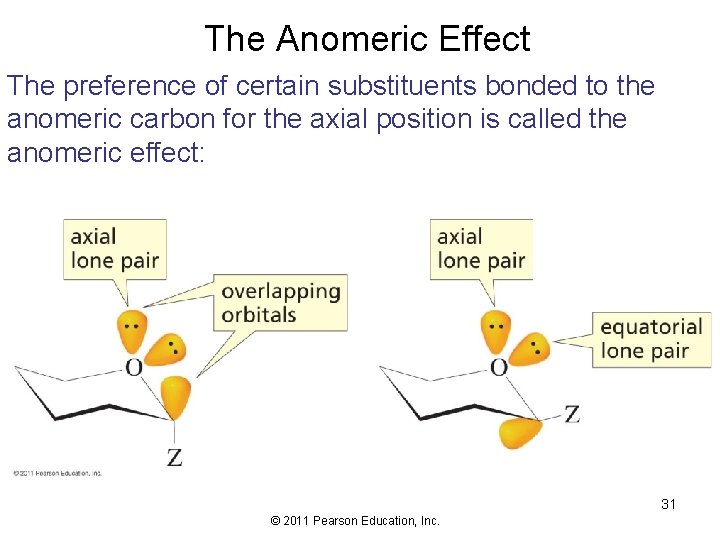
The Anomeric Effect The preference of certain substituents bonded to the anomeric carbon for the axial position is called the anomeric effect: 31 © 2011 Pearson Education, Inc.

Reducing and Nonreducing Sugars A sugar with an aldehyde, a ketone, a hemiacetal, or a hemiketal group is a reducing sugar A sugar without one of these groups is a nonreducing sugar Nonreducing sugar cannot reduce Ag+ or Br 2 32 © 2011 Pearson Education, Inc.
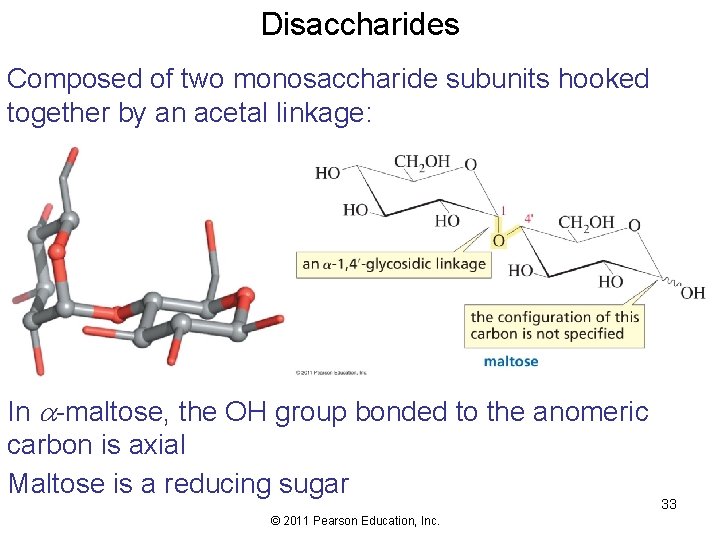
Disaccharides Composed of two monosaccharide subunits hooked together by an acetal linkage: In a-maltose, the OH group bonded to the anomeric carbon is axial Maltose is a reducing sugar © 2011 Pearson Education, Inc. 33
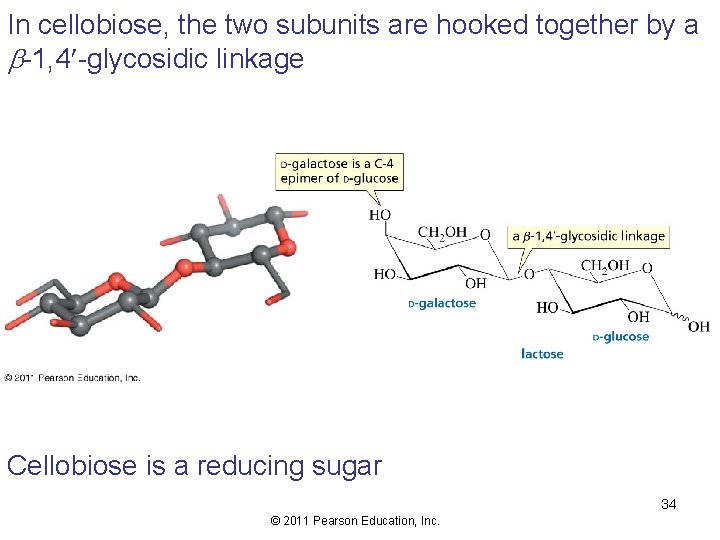
In cellobiose, the two subunits are hooked together by a b-1, 4 -glycosidic linkage Cellobiose is a reducing sugar 34 © 2011 Pearson Education, Inc.
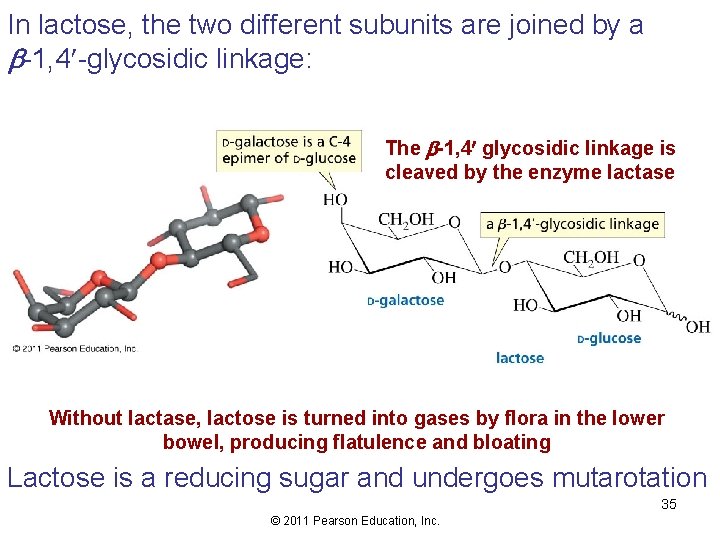
In lactose, the two different subunits are joined by a -1, 4 -glycosidic linkage: The -1, 4 glycosidic linkage is cleaved by the enzyme lactase Without lactase, lactose is turned into gases by flora in the lower bowel, producing flatulence and bloating Lactose is a reducing sugar and undergoes mutarotation 35 © 2011 Pearson Education, Inc.
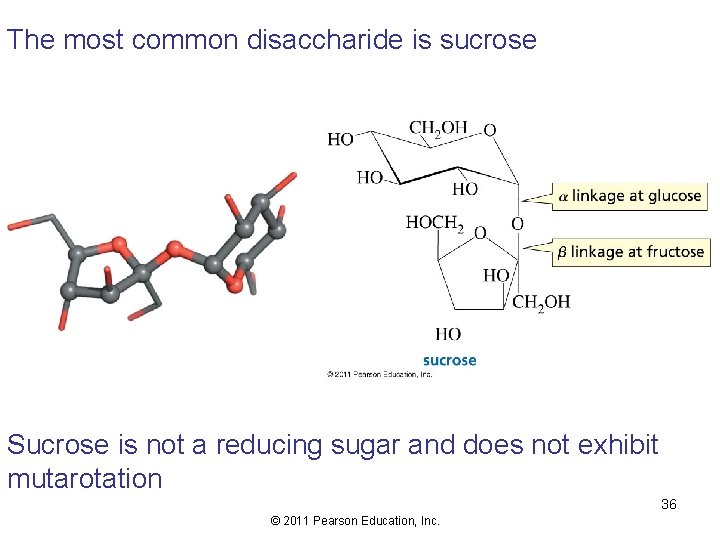
The most common disaccharide is sucrose Sucrose is not a reducing sugar and does not exhibit mutarotation 36 © 2011 Pearson Education, Inc.
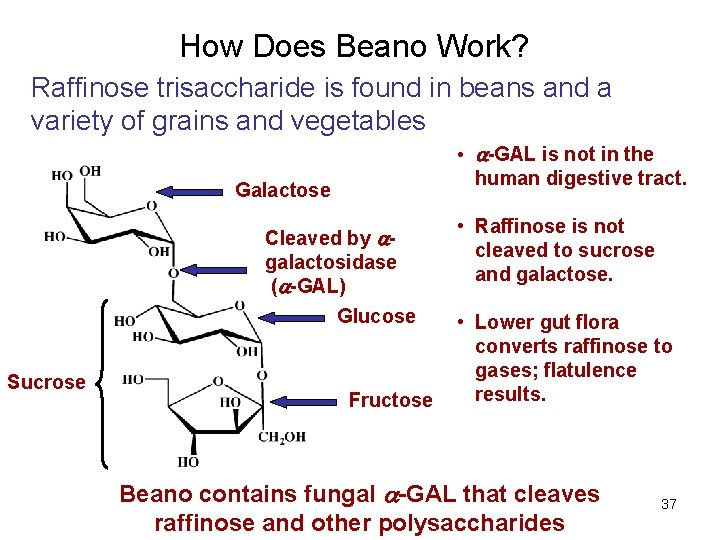
How Does Beano Work? Raffinose trisaccharide is found in beans and a variety of grains and vegetables • -GAL is not in the human digestive tract. Galactose Cleaved by galactosidase ( -GAL) Glucose Sucrose Fructose • Raffinose is not cleaved to sucrose and galactose. • Lower gut flora converts raffinose to gases; flatulence results. Beano contains fungal -GAL that cleaves © 2011 other Pearson Education, Inc. raffinose and polysaccharides 37
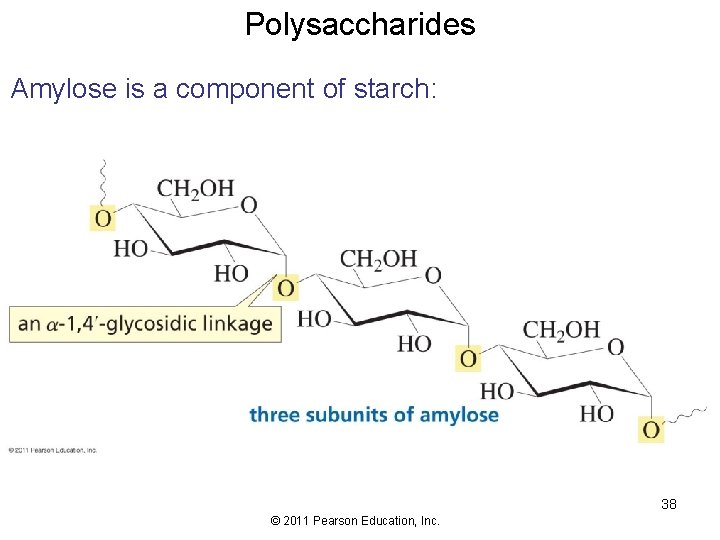
Polysaccharides Amylose is a component of starch: 38 © 2011 Pearson Education, Inc.
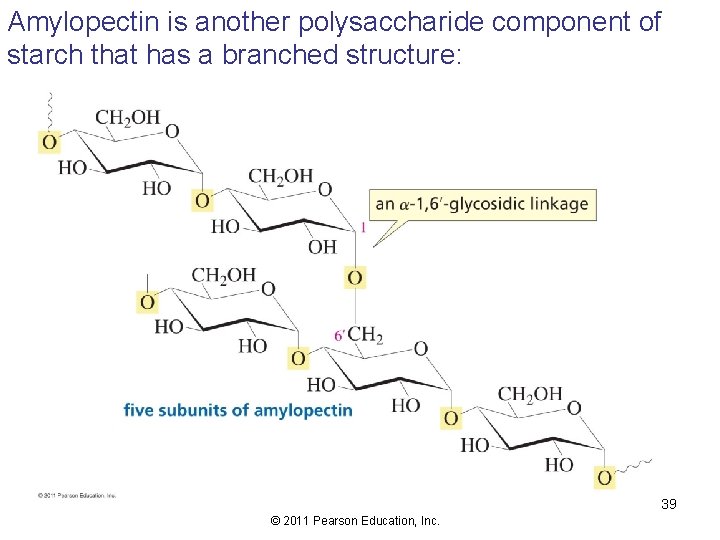
Amylopectin is another polysaccharide component of starch that has a branched structure: 39 © 2011 Pearson Education, Inc.
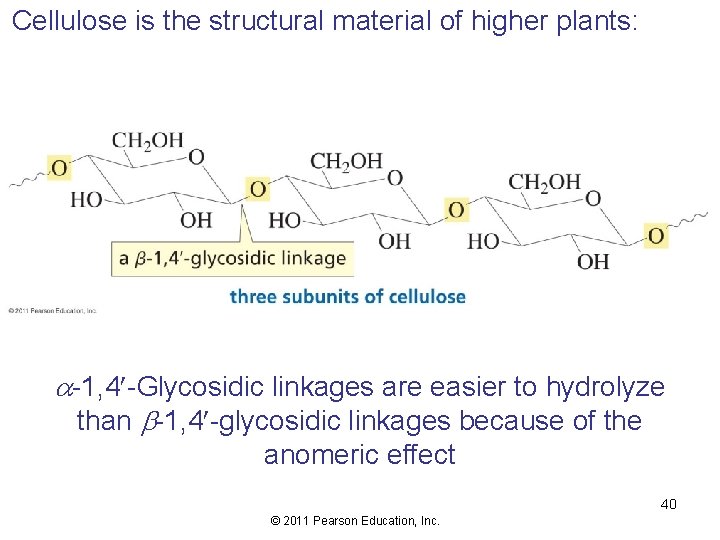
Cellulose is the structural material of higher plants: a-1, 4 -Glycosidic linkages are easier to hydrolyze than b-1, 4 -glycosidic linkages because of the anomeric effect 40 © 2011 Pearson Education, Inc.
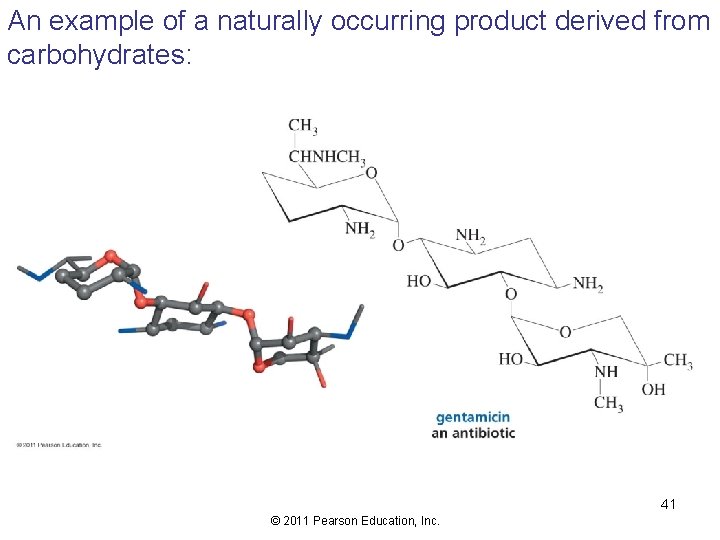
An example of a naturally occurring product derived from carbohydrates: 41 © 2011 Pearson Education, Inc.
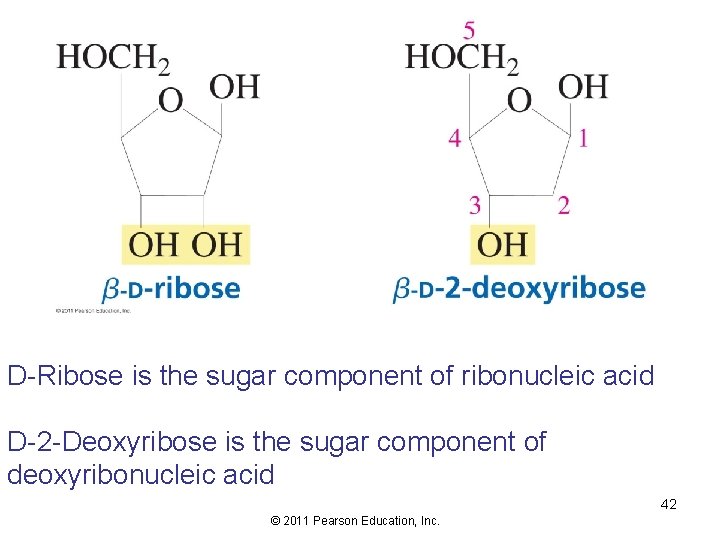
D-Ribose is the sugar component of ribonucleic acid D-2 -Deoxyribose is the sugar component of deoxyribonucleic acid 42 © 2011 Pearson Education, Inc.
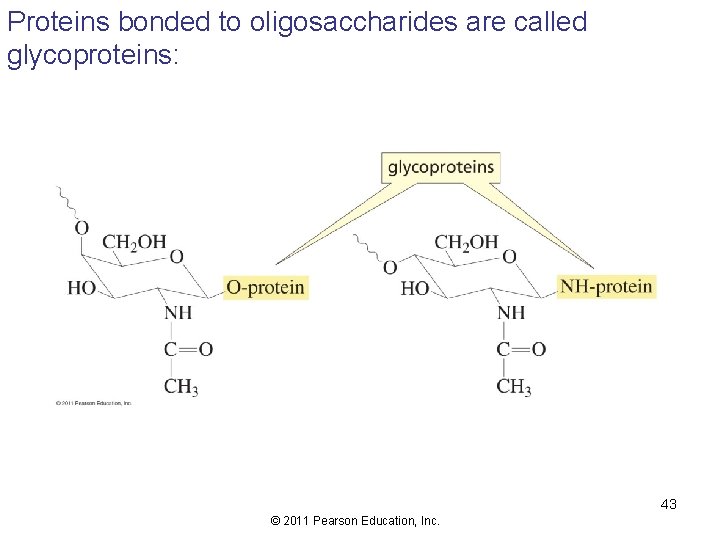
Proteins bonded to oligosaccharides are called glycoproteins: 43 © 2011 Pearson Education, Inc.
- Slides: 43