Organic Chemistry 4 th Edition Chapter 28 Paula









- Slides: 46

Organic Chemistry 4 th Edition Chapter 28 Paula Yurkanis Bruice Synthetic Polymers Irene Lee Case Western Reserve University Cleveland, OH © 2004, Prentice Hall

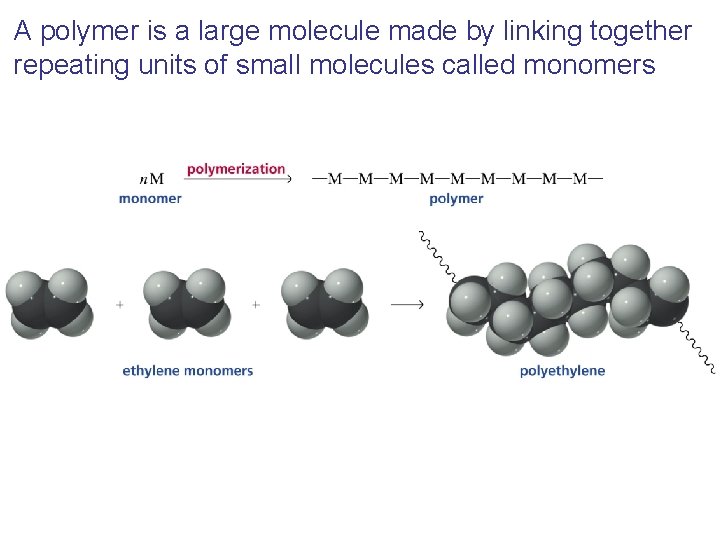
A polymer is a large molecule made by linking together repeating units of small molecules called monomers

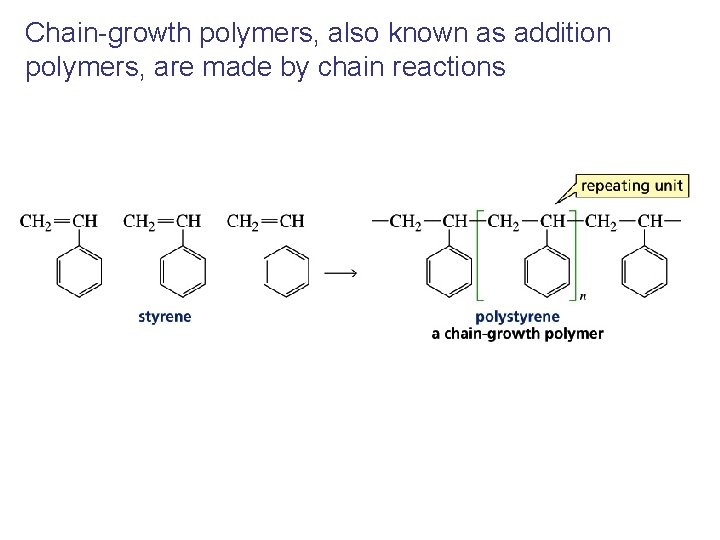
Chain-growth polymers, also known as addition polymers, are made by chain reactions

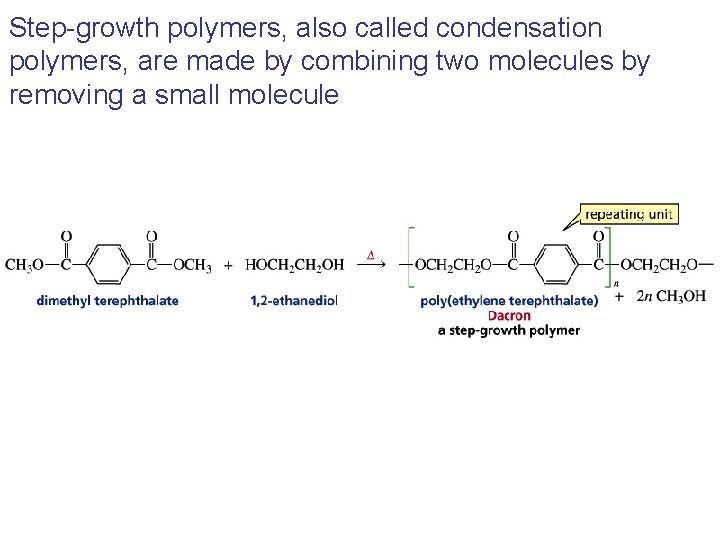
Step-growth polymers, also called condensation polymers, are made by combining two molecules by removing a small molecule

Chain-Growth Polymers Chain-growth polymers proceed by one of three mechanisms: radical polymerization cationic polymerization anionic polymerization


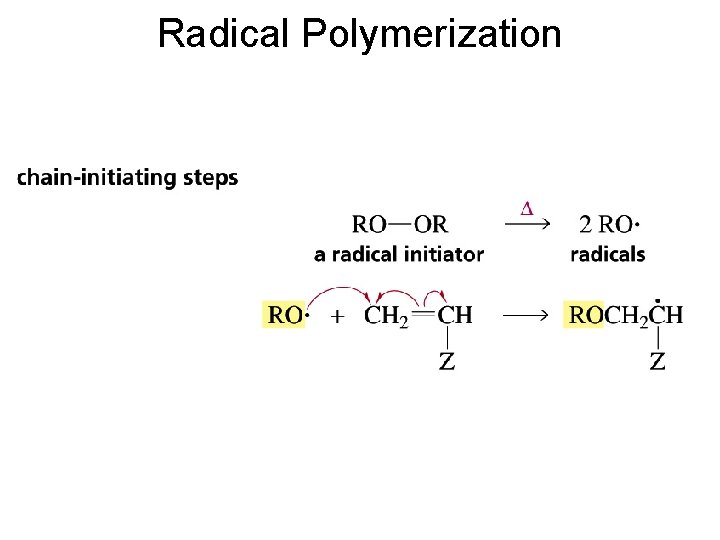
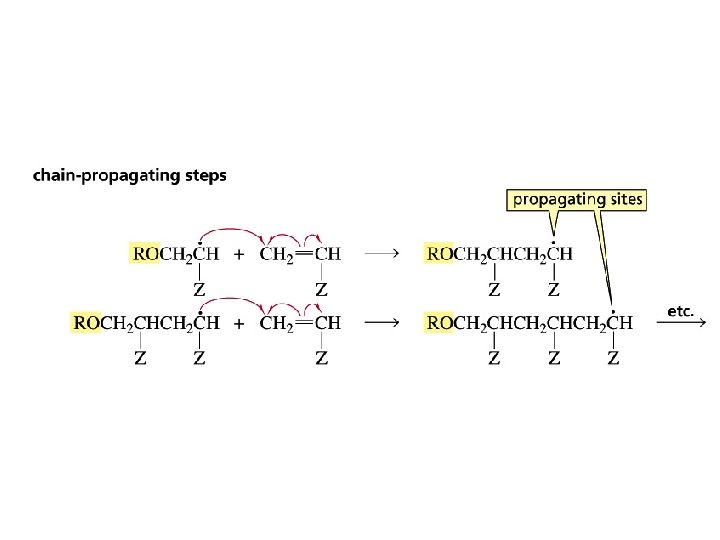
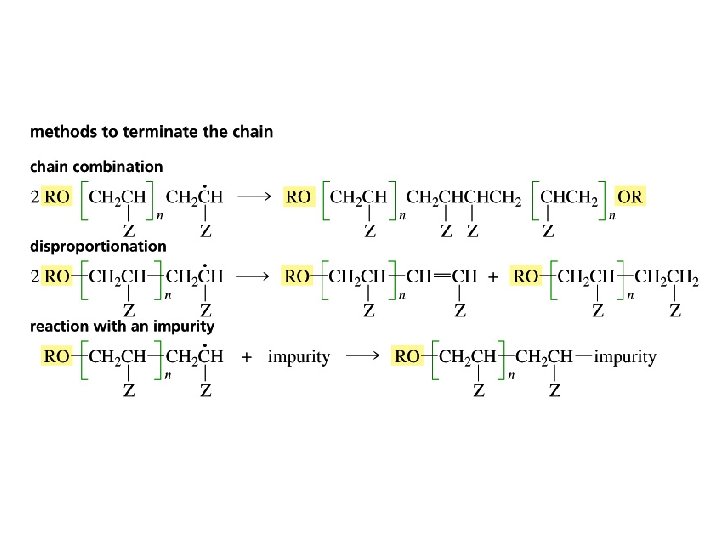
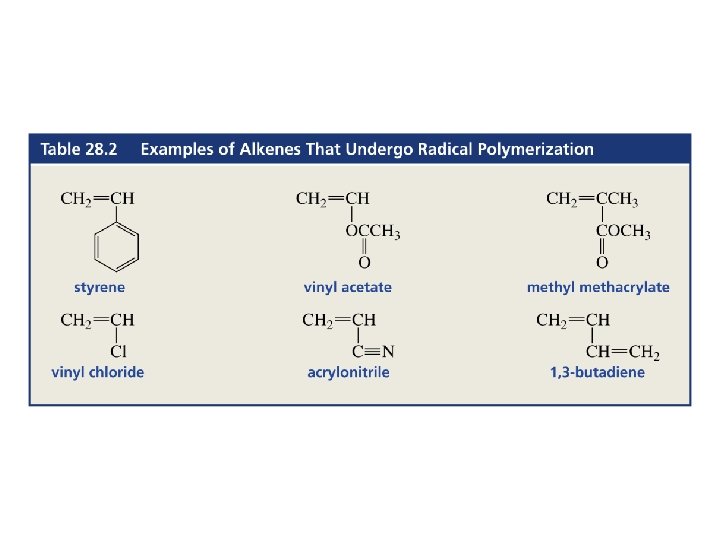
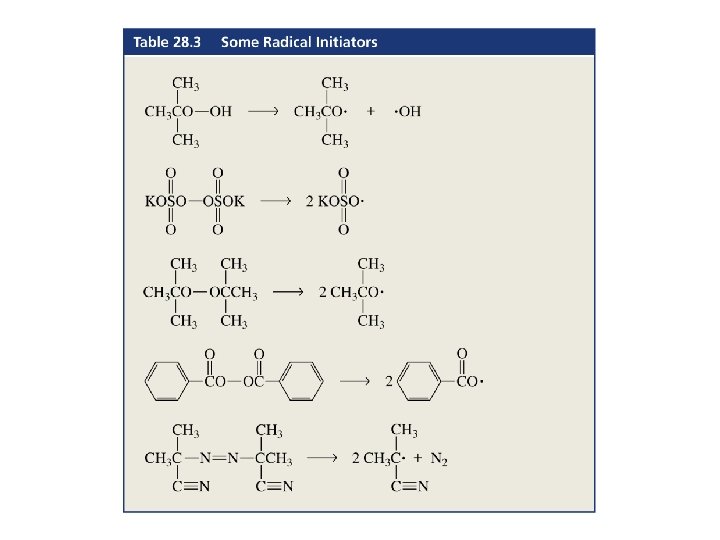
Radical Polymerization



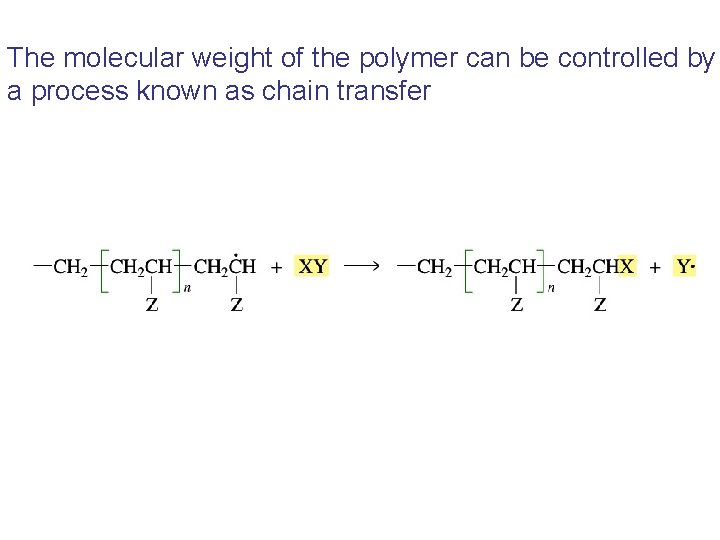
The molecular weight of the polymer can be controlled by a process known as chain transfer

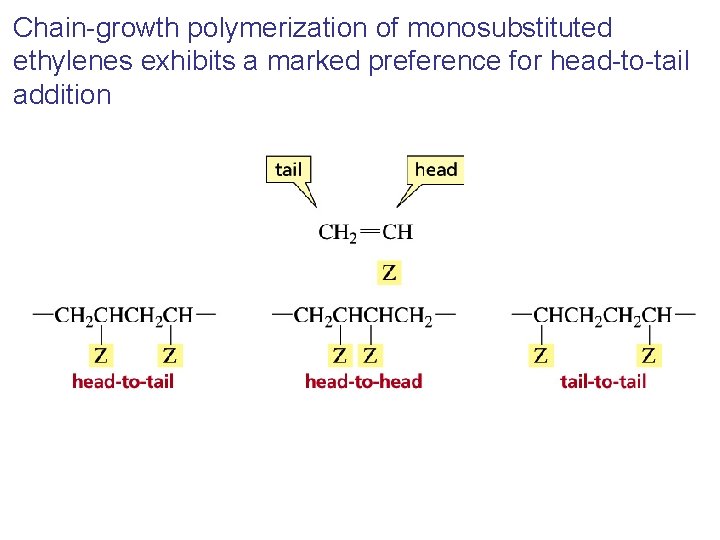
Chain-growth polymerization of monosubstituted ethylenes exhibits a marked preference for head-to-tail addition

Head-to-tail addition of a substituted ethylene results in a polymer in which every other carbon bears a substituent

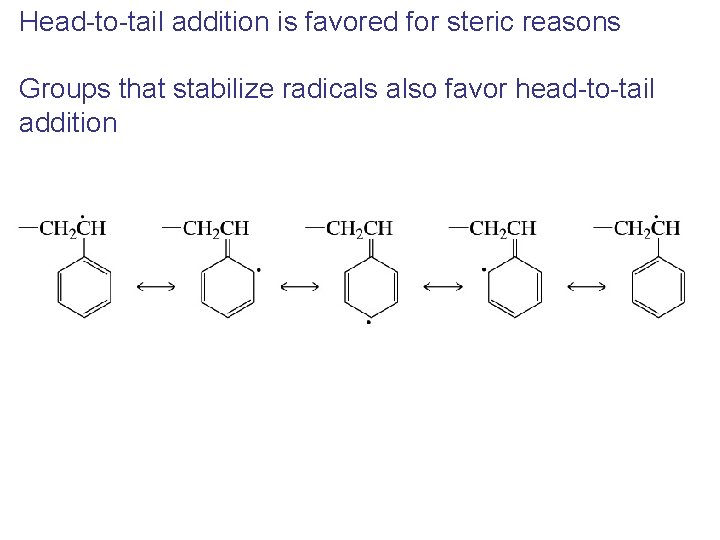
Head-to-tail addition is favored for steric reasons Groups that stabilize radicals also favor head-to-tail addition



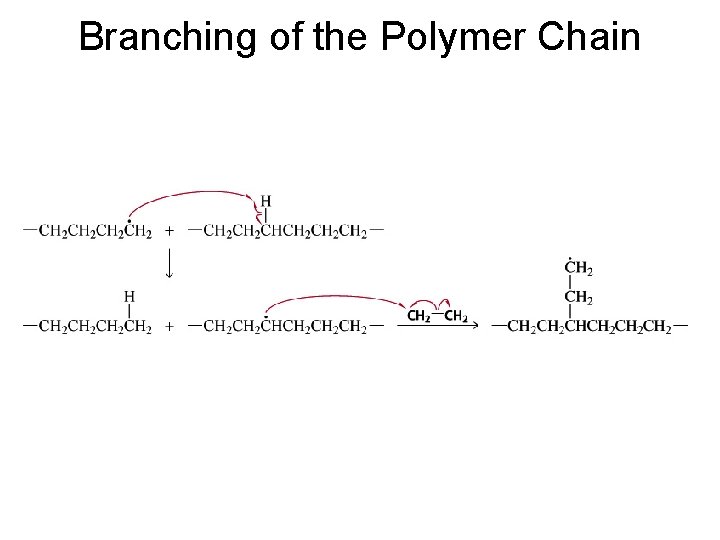
Branching of the Polymer Chain

Branched polymers are more flexible

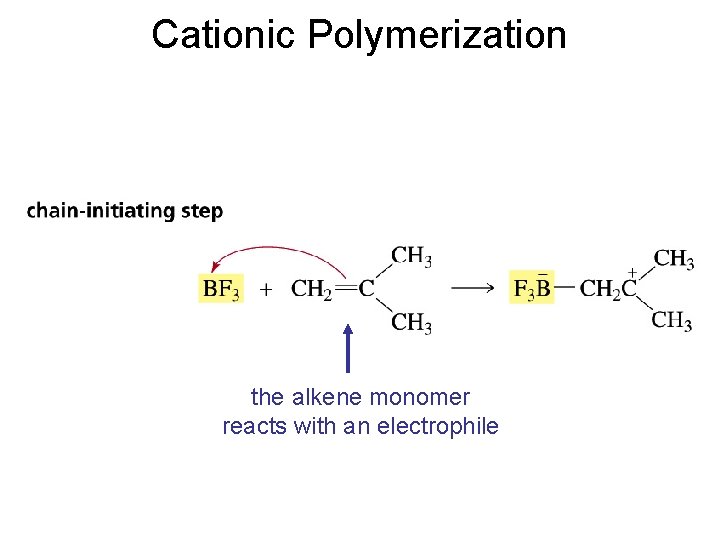
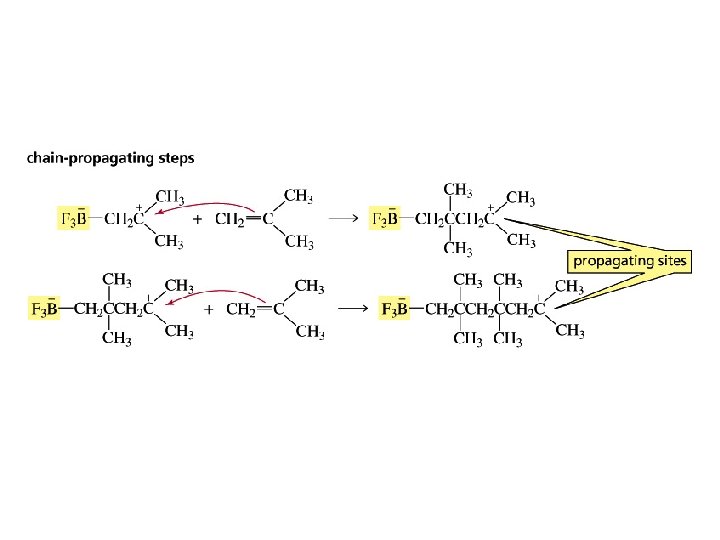
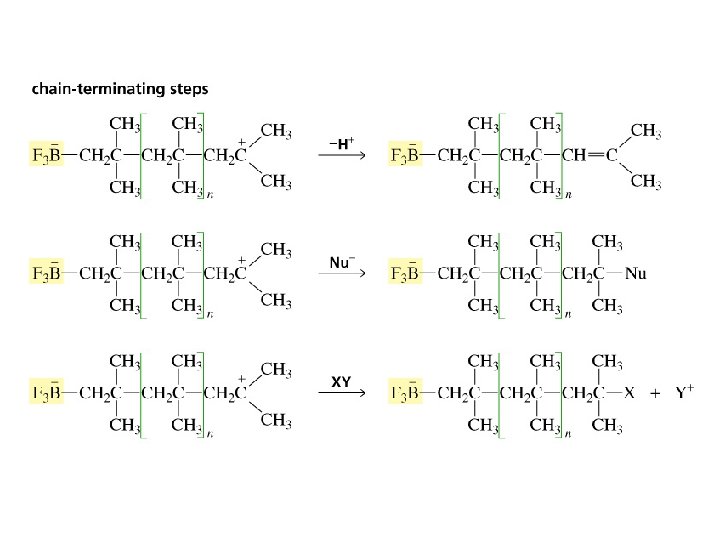
Cationic Polymerization the alkene monomer reacts with an electrophile



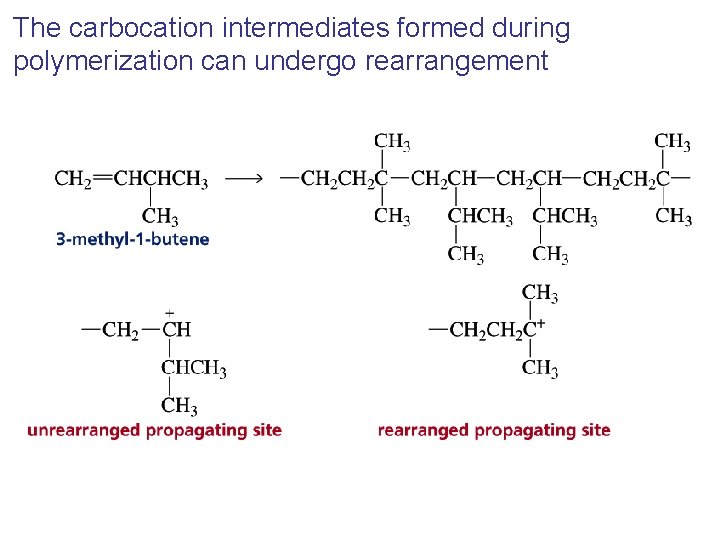
The carbocation intermediates formed during polymerization can undergo rearrangement

Monomers that are best able to undergo cationic polymerization are those with electron-donating substituents

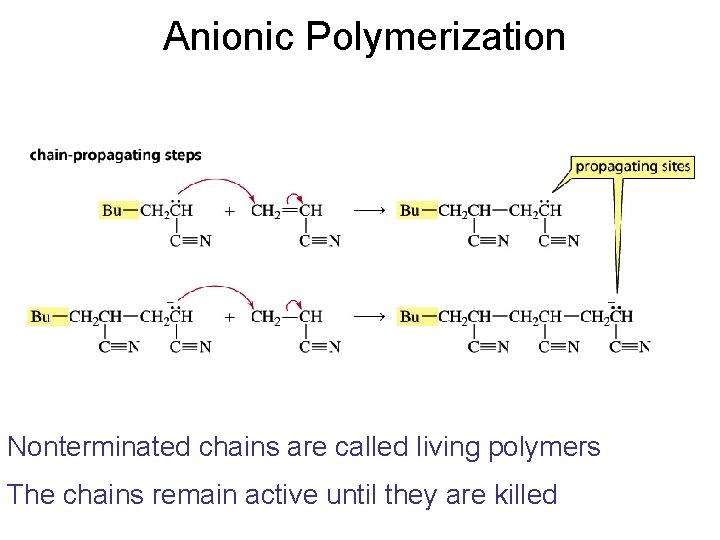
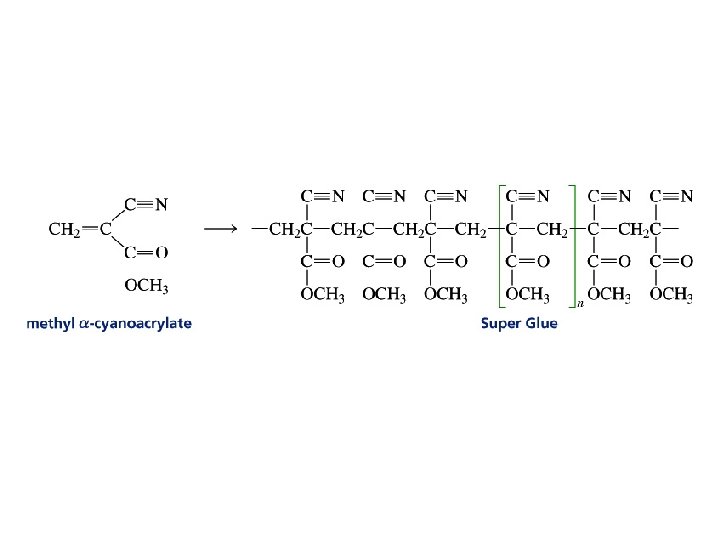
Anionic Polymerization Nonterminated chains are called living polymers The chains remain active until they are killed



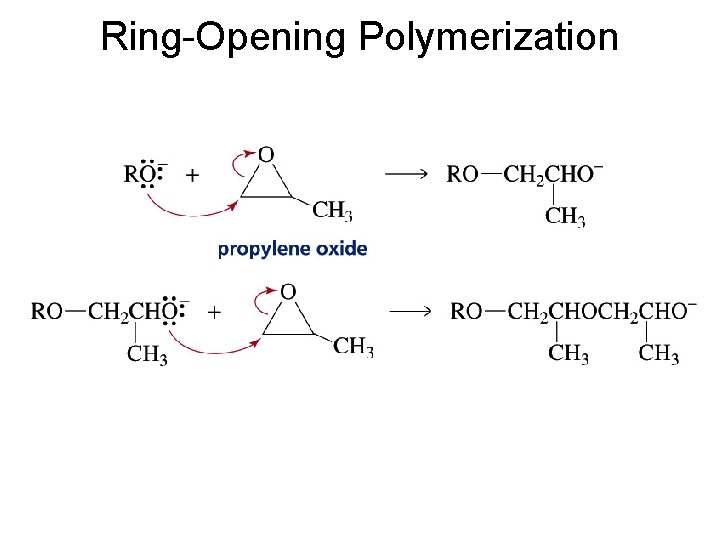
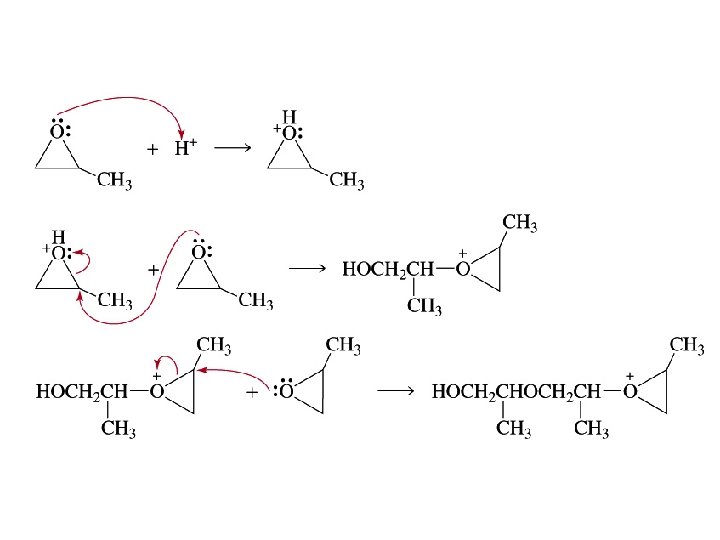
Ring-Opening Polymerization


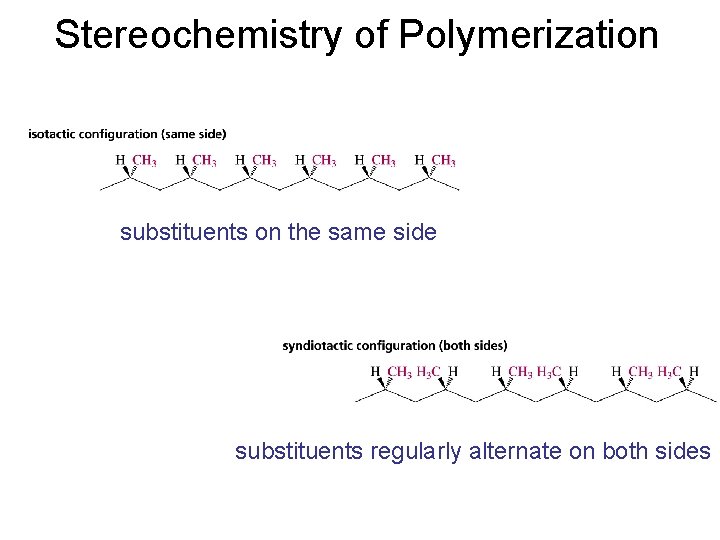
Stereochemistry of Polymerization substituents on the same side substituents regularly alternate on both sides

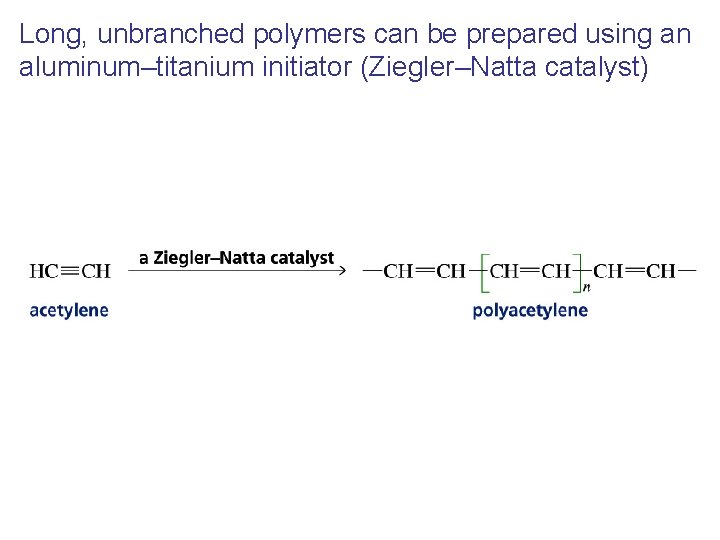
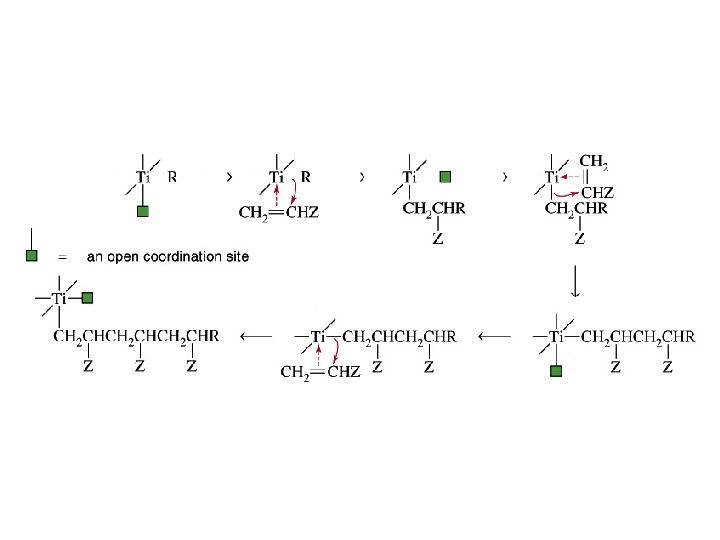
Long, unbranched polymers can be prepared using an aluminum–titanium initiator (Ziegler–Natta catalyst)


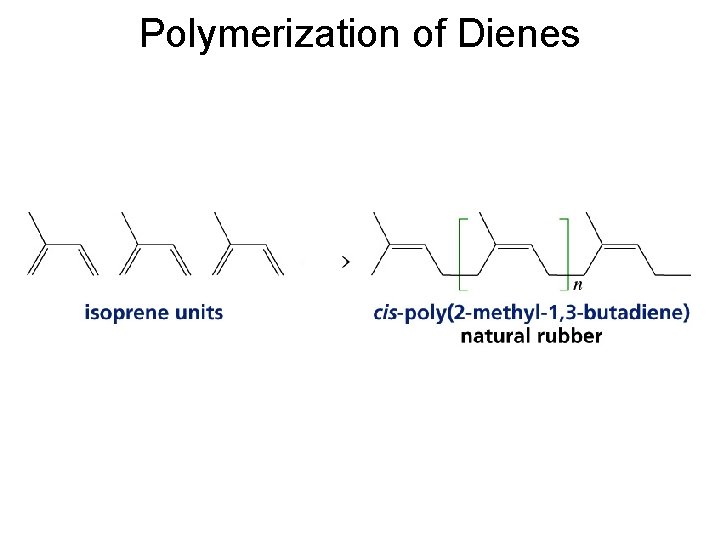
Polymerization of Dienes

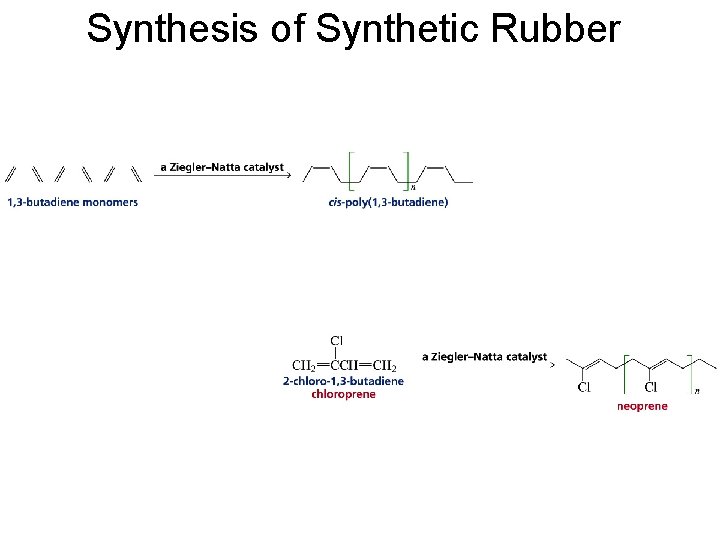
Synthesis of Synthetic Rubber

The product resulting from polymerization of different monomers is called a copolymer

Four Types of Copolymers

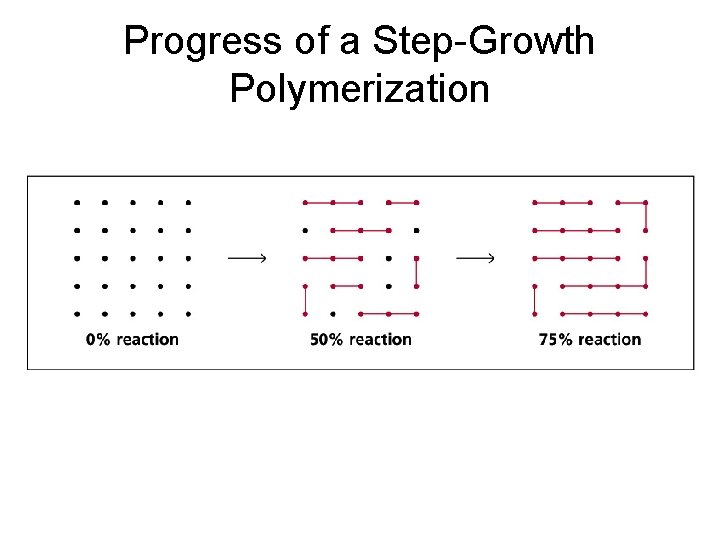
Progress of a Step-Growth Polymerization

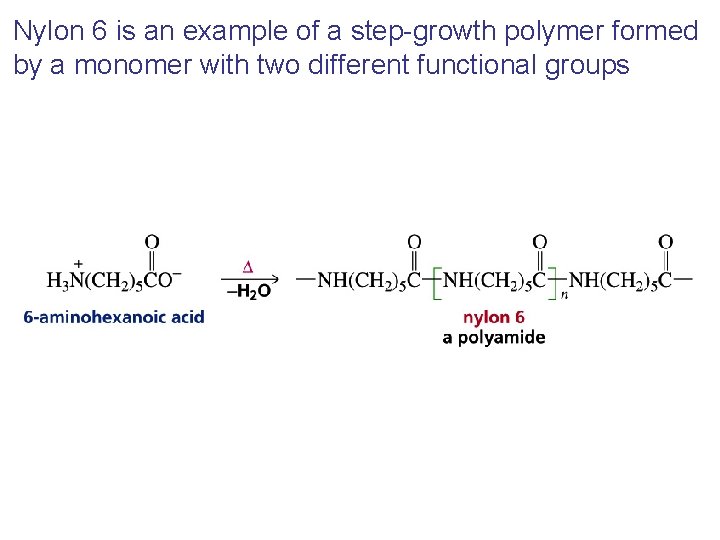
Nylon 6 is an example of a step-growth polymer formed by a monomer with two different functional groups

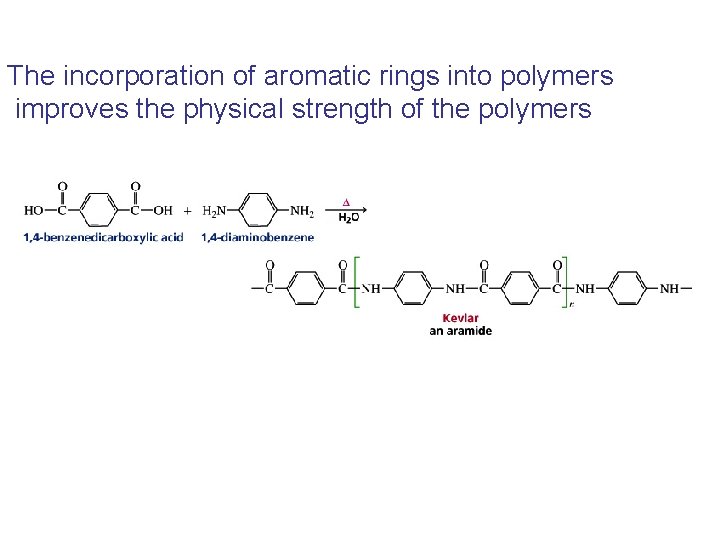
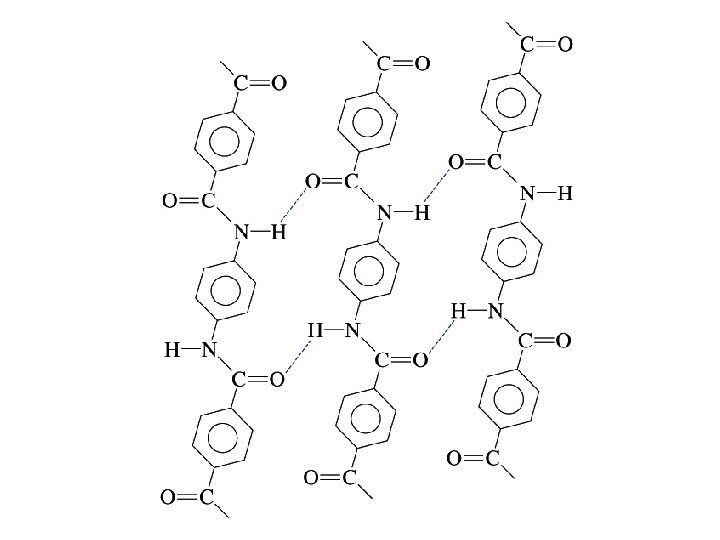
The incorporation of aromatic rings into polymers improves the physical strength of the polymers


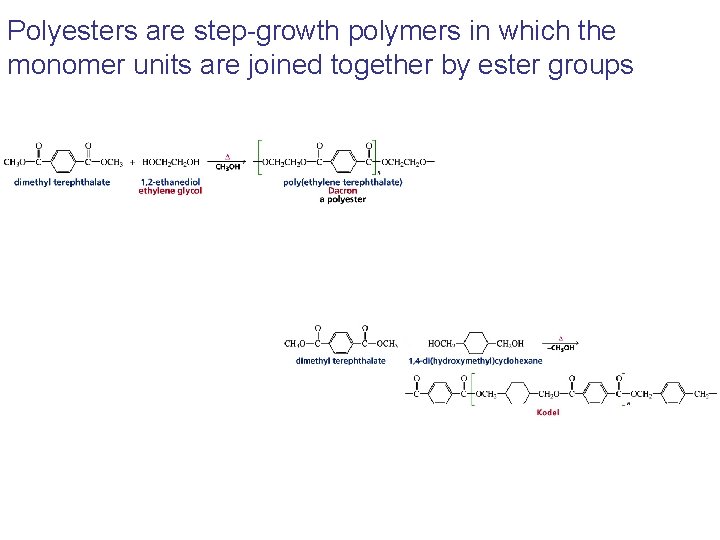
Polyesters are step-growth polymers in which the monomer units are joined together by ester groups

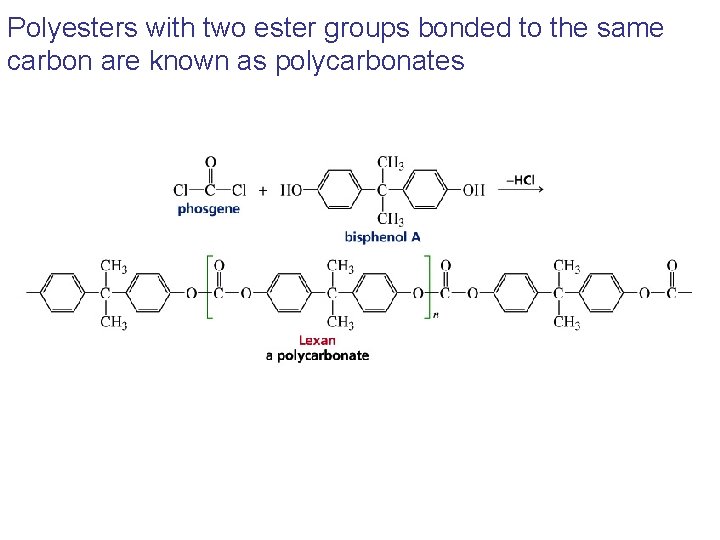
Polyesters with two ester groups bonded to the same carbon are known as polycarbonates

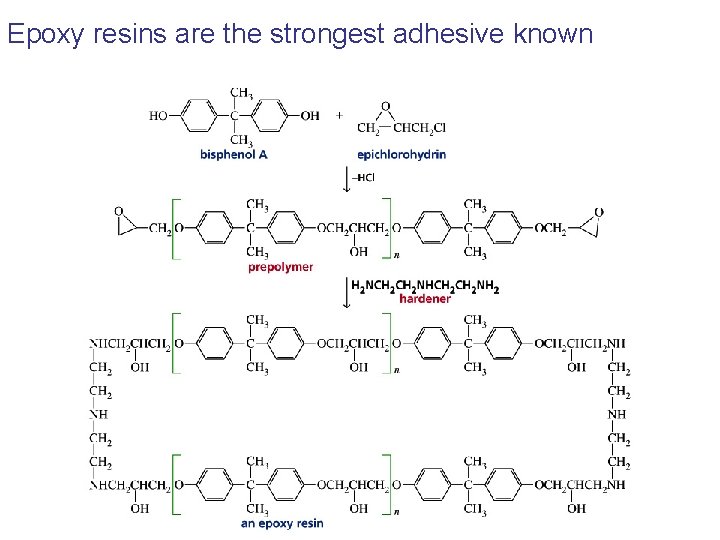
Epoxy resins are the strongest adhesive known

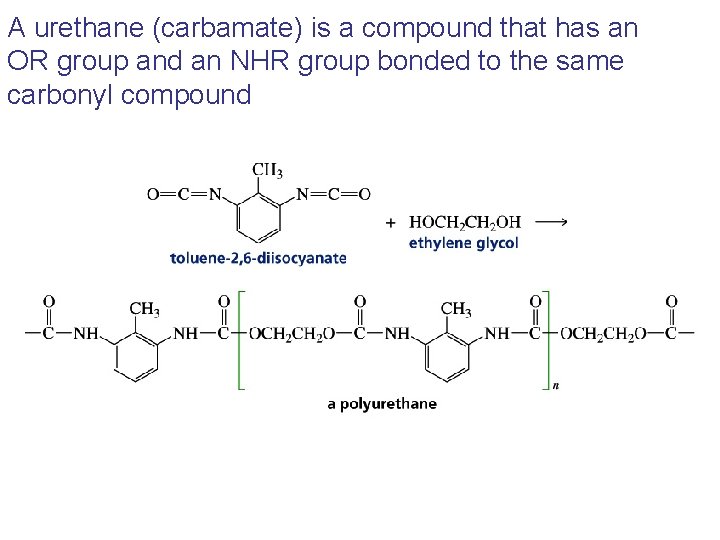
A urethane (carbamate) is a compound that has an OR group and an NHR group bonded to the same carbonyl compound

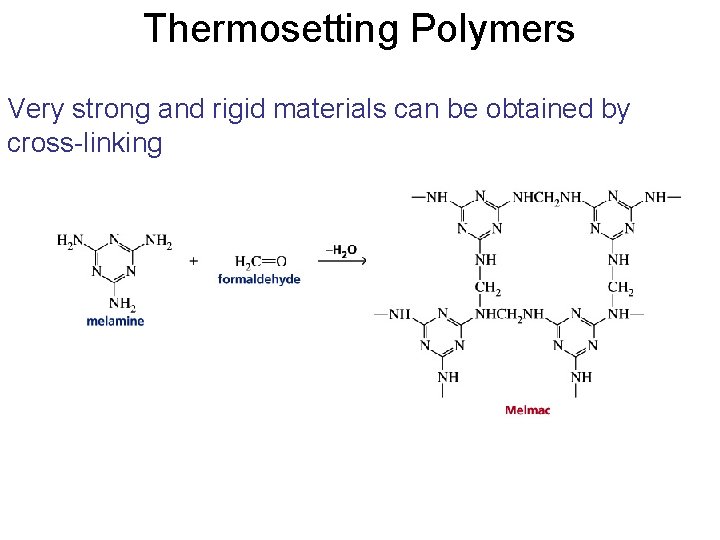
Thermosetting Polymers Very strong and rigid materials can be obtained by cross-linking

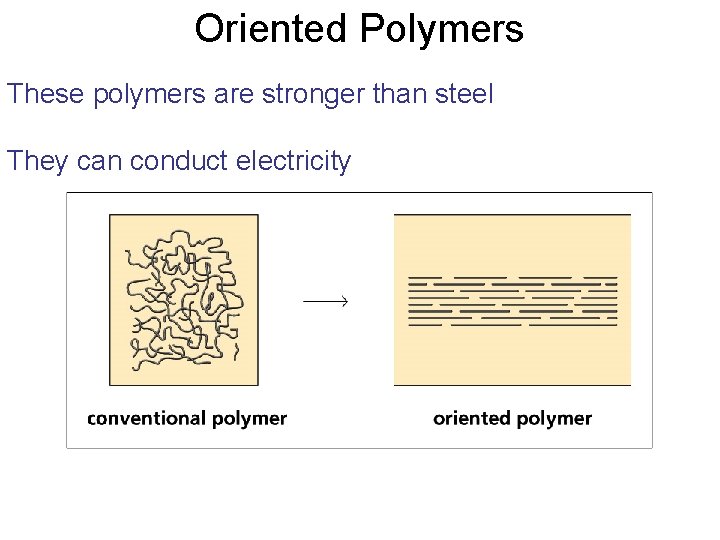
Oriented Polymers These polymers are stronger than steel They can conduct electricity

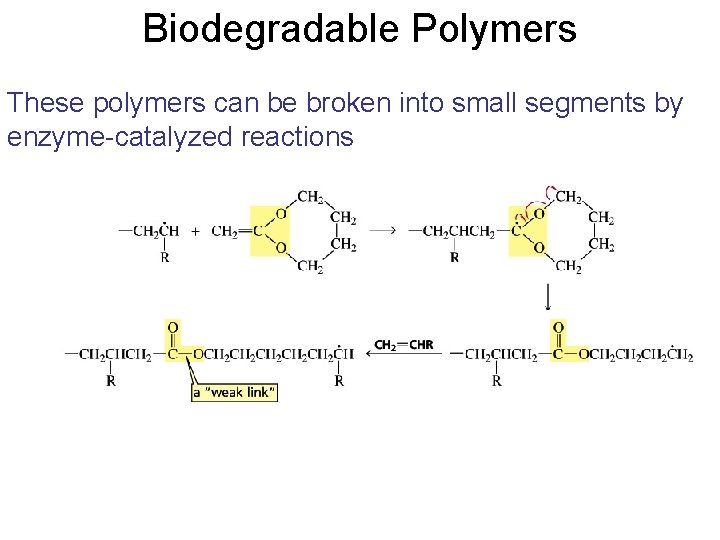
Biodegradable Polymers These polymers can be broken into small segments by enzyme-catalyzed reactions

Other Polymers Thermoplastic polymers have both ordered crystalline regions and amorphous, noncrystalline regions Elastomers are polymers that stretch and then revert to the original shape Plasticizer is an organic compound that is added to a polymer to make it more flexible