Organic Chemistry 14 Aromatic Hydrocarbons Leaving Certificate Chemistry

Organic Chemistry 14 – Aromatic Hydrocarbons Leaving Certificate Chemistry
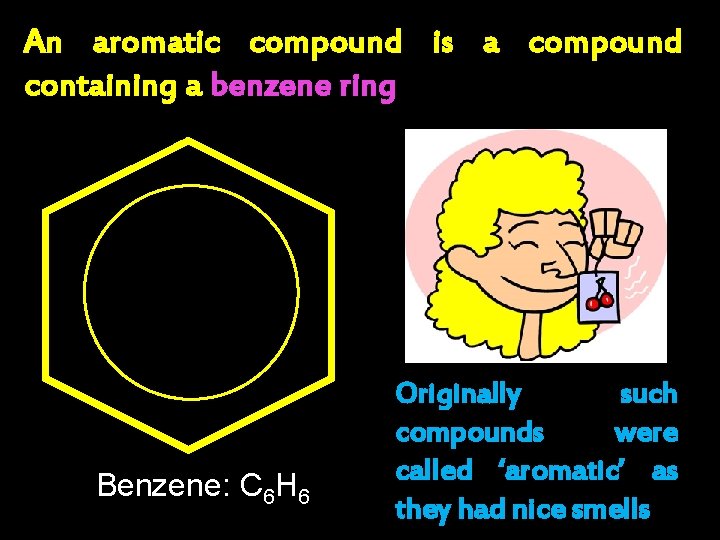
An aromatic compound is a compound containing a benzene ring Benzene: C 6 H 6 Originally such compounds were called ‘aromatic’ as they had nice smells

Benzene: C 6 H 6 Derived from whale oil – 1825 – Michael Faraday

Benzene: C 6 H 6 By 1834: Molecular formula known to be C 6 H 6 Chemical formula suggests a highly unsaturated compound But Benzene does not decolourise bromine water OR acidified potassium permanganate Confusion Benzene undergoes substitution reactions rather than addition.
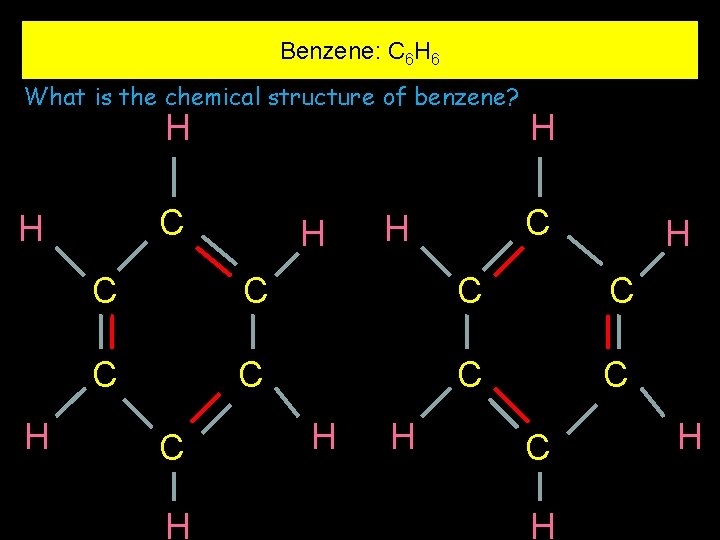
Benzene: C 6 H 6 What is the chemical structure of benzene? H C H H C C C C C H H H C H H

Benzene: C 6 H 6 Bond Lengths One very big problem C C 154 picometres C C 134 picometres But all of the bond lengths in Benzene are of the same length of 140 picometres 1 picometre is one trillionth of a metre

Benzene: C 6 H 6 H that He suggested six electrons revolve around the entire six 2007 Q. 4(i) (6) carbon atoms and this explains C why H all carbon-carbon bond H lengths are equal H Structure revealed by Kekule in 1865 C C C Benzene: C 6 H 6 H H
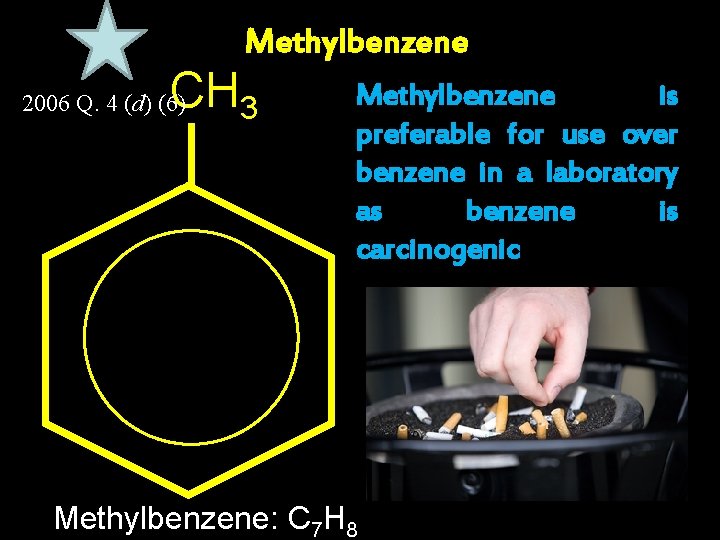
Methylbenzene CH 3 2006 Q. 4 (d) (6) Methylbenzene is preferable for use over benzene in a laboratory as benzene is carcinogenic Methylbenzene: C 7 H 8
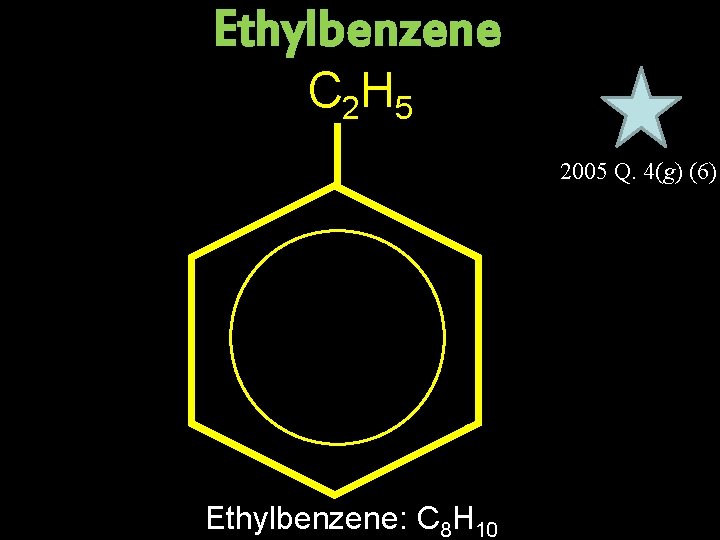
Ethylbenzene C 2 H 5 2005 Q. 4(g) (6) Ethylbenzene: C 8 H 10

Physical properties of benzene? Physical state: Benzene, Methylbenzene and ethylbenzene are liquid at room temperature Insoluble in water Soluble in non-polar solvents such as cyclohexane
- Slides: 10