ORGANIC CHEMISTRY 1 Alkyl Halides Preparation and Reactions















![SN 1 properties • Mechanism - Unimolecular Nucleophilic Substitution [SN 1] • Unimolecular because SN 1 properties • Mechanism - Unimolecular Nucleophilic Substitution [SN 1] • Unimolecular because](https://slidetodoc.com/presentation_image_h2/2eedcea8419eeff9cb5f649ea017b04e/image-41.jpg)


![Summary of SN reaction Unimolecular nucleophilic Substitution (SN 1) Rate = k [alkyl halide] Summary of SN reaction Unimolecular nucleophilic Substitution (SN 1) Rate = k [alkyl halide]](https://slidetodoc.com/presentation_image_h2/2eedcea8419eeff9cb5f649ea017b04e/image-51.jpg)




- Slides: 55

ORGANIC CHEMISTRY- 1 Alkyl Halides Preparation and Reactions BY Dr. Ghulam Abbas Assistant Professor UNIVERSITY OF NIZWA

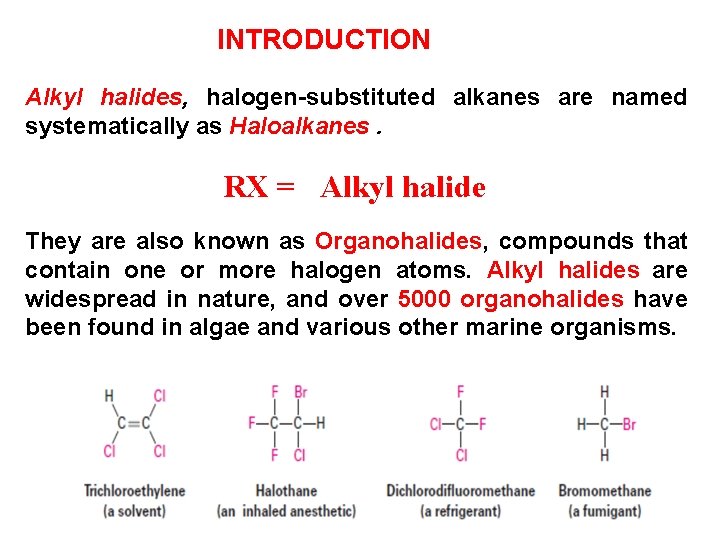
INTRODUCTION Alkyl halides, halogen-substituted alkanes are named systematically as Haloalkanes. RX = Alkyl halide They are also known as Organohalides, compounds that contain one or more halogen atoms. Alkyl halides are widespread in nature, and over 5000 organohalides have been found in algae and various other marine organisms.

Halogens increase in size going down the periodic table, so the lengths of carbon– halogen bonds increase accordingly. In addition, C -X bond strengths decrease going down the periodic table. Such as for CH 3 F bond length 139 pm and bond strength is 460 k. J/mol while CH 3 Br bond length is 178 pm and bond strength 294 k. J/mol.

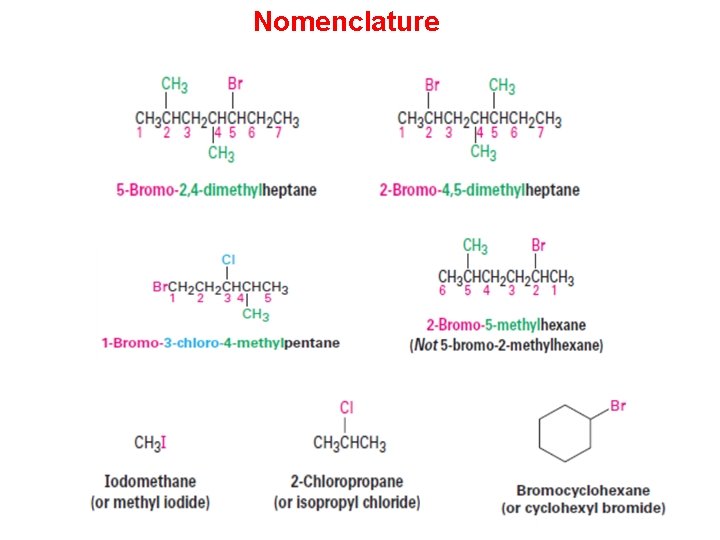
Nomenclature

Alkyl Halides • Alkyl halides are organic molecules containing a halogen atom bonded to an sp 3 hybridized carbon atom. • Alkyl halides are classified as primary (1°), secondary (2°), or tertiary (3°), depending on the number of carbons bonded to the carbon with the halogen atom. • The halogen atom in halides is often denoted by the symbol “X”.

• There are other types of organic halides. These include vinyl halides, aryl halides, allylic halides and benzylic halides. • Vinyl halides have a halogen atom (X) bonded to a C—C double bond (C=C-X). • Aryl halides have a halogen atom bonded to a benzene ring. (Ar-X). • Allylic halides have X bonded to the carbon atom adjacent to a C—C double bond. (C=C-C-X) • Benzylic halides have X bonded to the carbon atom adjacent to a benzene ring (Ar-C-X).

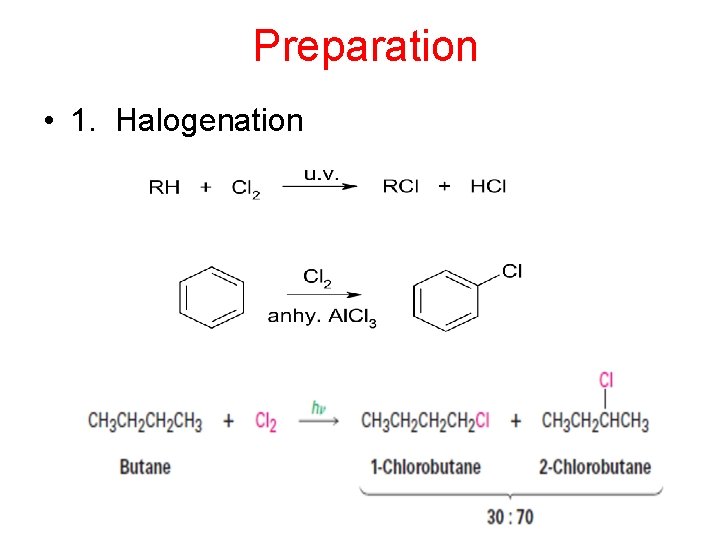
Preparation • 1. Halogenation


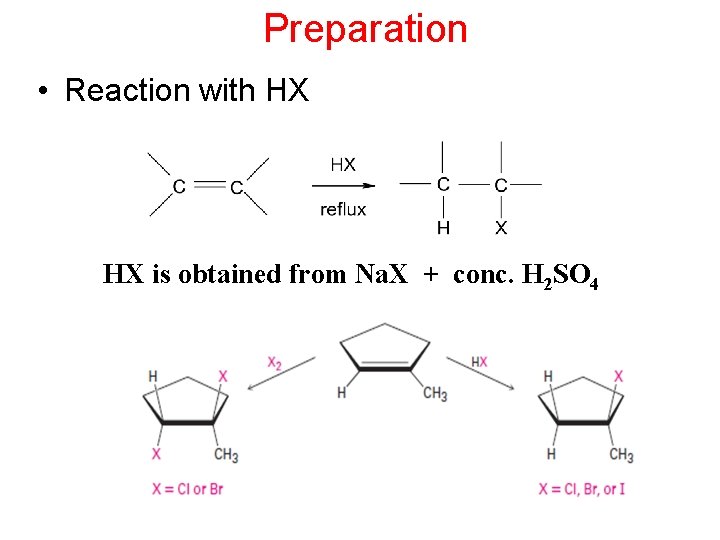
Preparation • Reaction with HX HX is obtained from Na. X + conc. H 2 SO 4

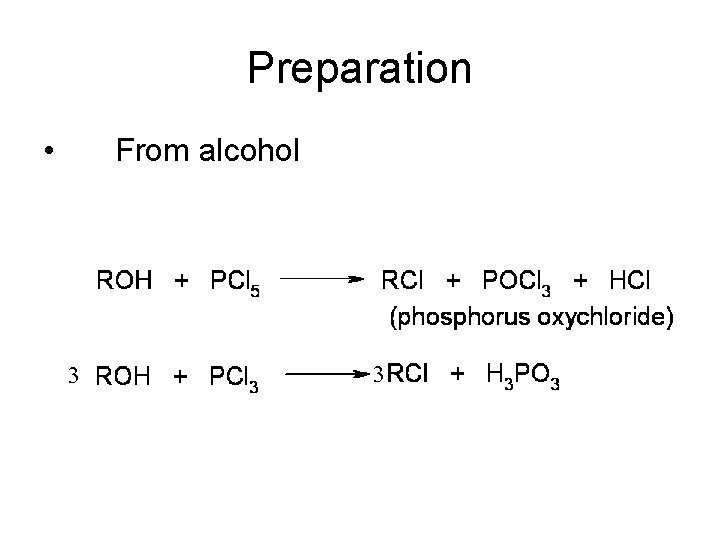
Preparation • From alcohol 3 3

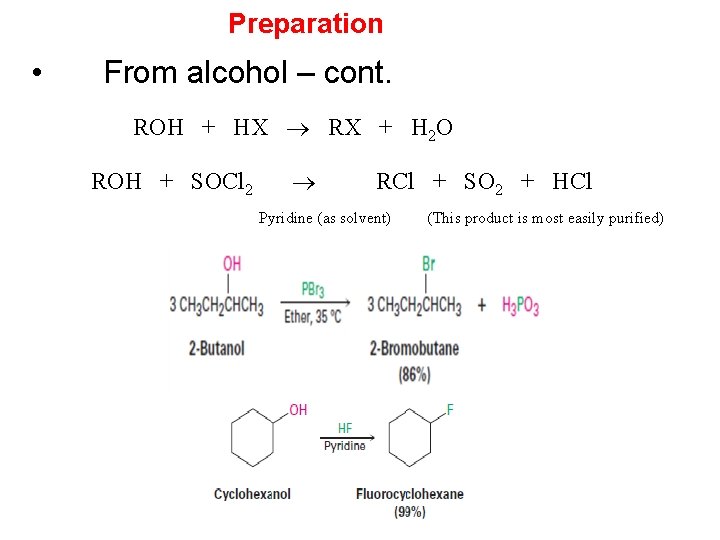
Preparation • From alcohol – cont. ROH + HX RX + H 2 O ROH + SOCl 2 RCl + SO 2 + HCl Pyridine (as solvent) (This product is most easily purified)

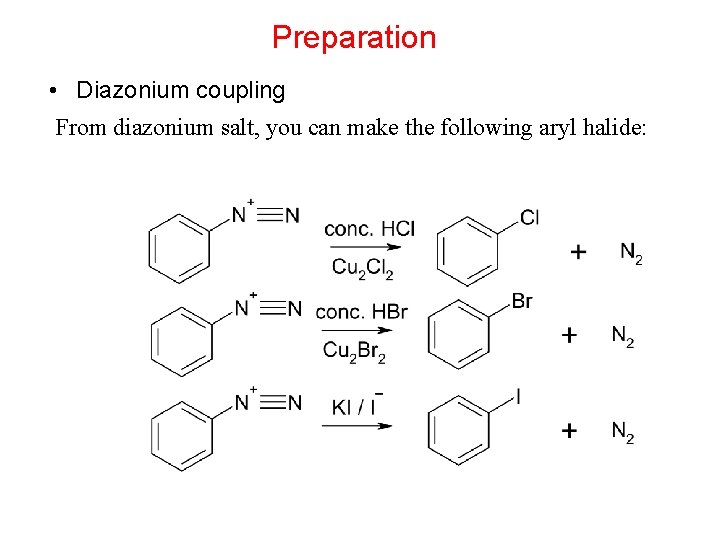
Preparation • Diazonium coupling From diazonium salt, you can make the following aryl halide:

Physical Properties It has a little higher boiling point than corresponding alkane of comparable molecular mass. This is due to the dipole-dipole attraction between the molecules as they are polar. CH 3 Cl, CH 3 Br and C 2 H 5 Cl are gases in room temperature while other members are liquids. Chlorobenzene is colourless liquid. All alkyl and aryl halides are insoluble in water due to the inability to form extensive H-bond with water molecules.

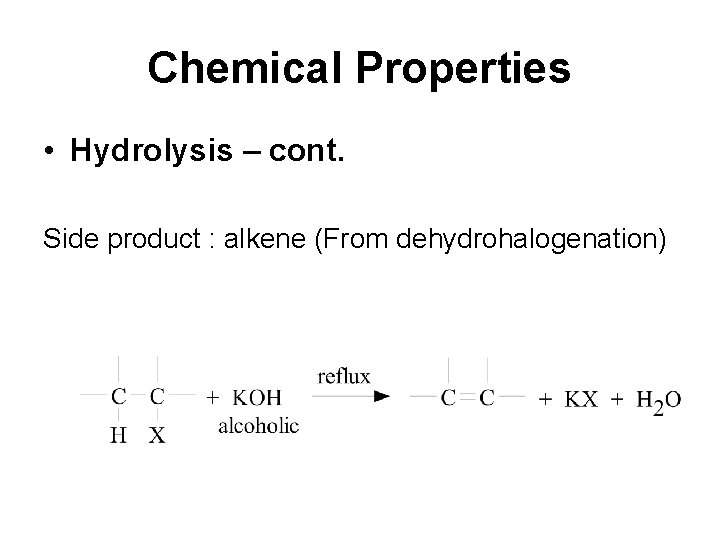
Chemical Properties • Hydrolysis – cont. Side product : alkene (From dehydrohalogenation)

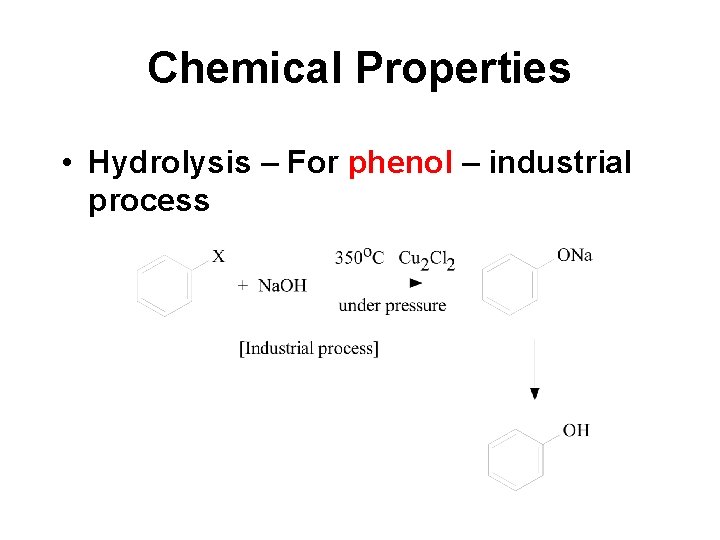
Chemical Properties • Hydrolysis – For phenol – industrial process

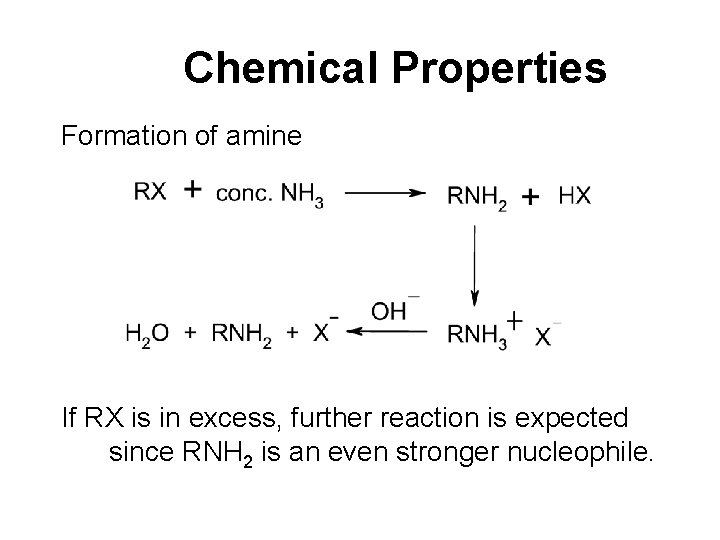
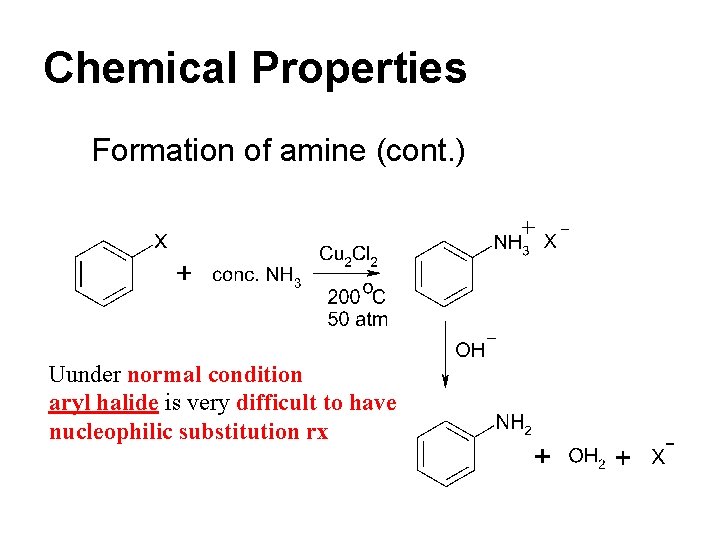
Chemical Properties Formation of amine If RX is in excess, further reaction is expected since RNH 2 is an even stronger nucleophile.

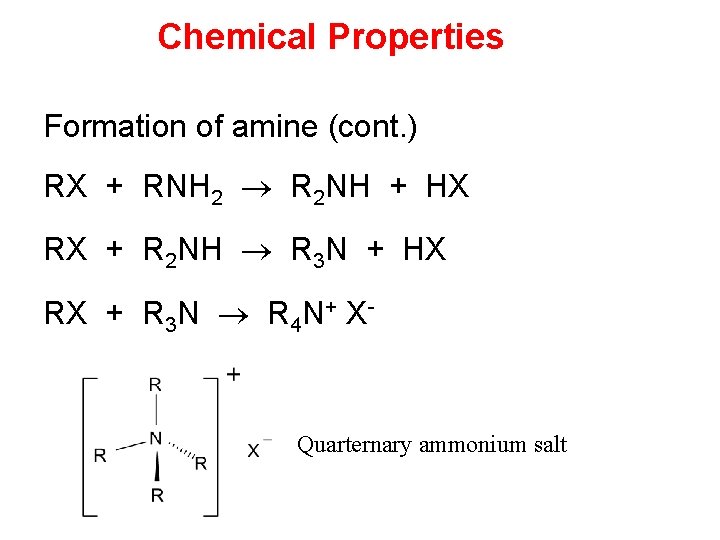
Chemical Properties Formation of amine (cont. ) RX + RNH 2 R 2 NH + HX RX + R 2 NH R 3 N + HX RX + R 3 N R 4 N+ X- Quarternary ammonium salt

Chemical Properties Formation of amine (cont. ) Uunder normal condition aryl halide is very difficult to have nucleophilic substitution rx

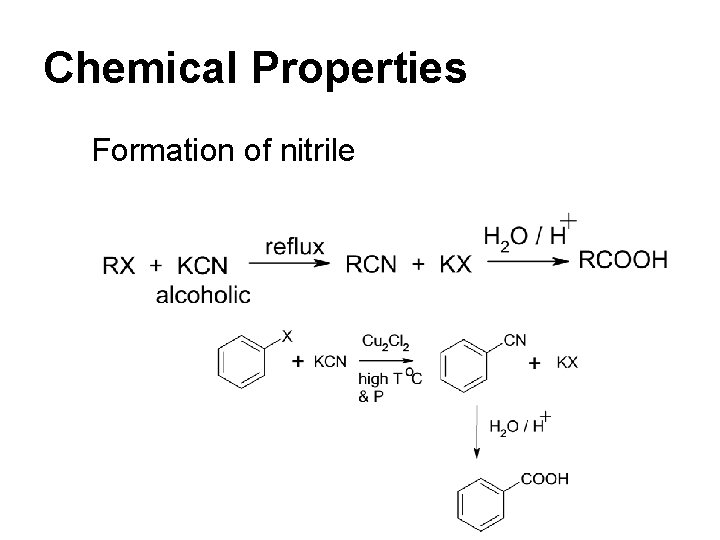
Chemical Properties Formation of nitrile

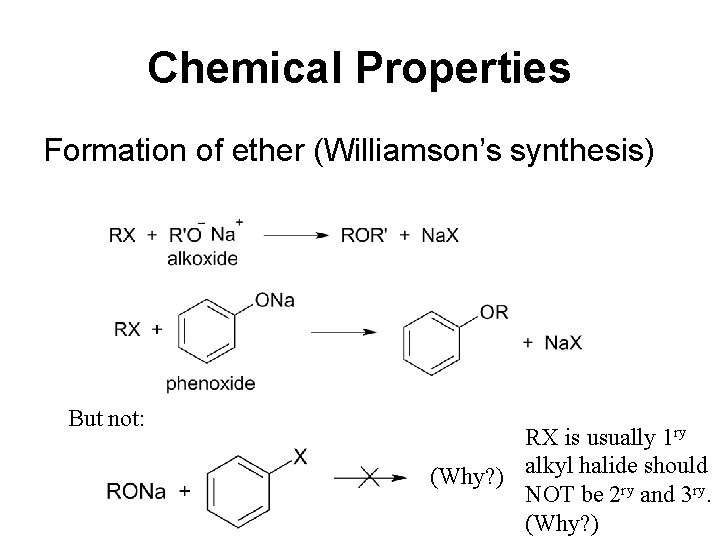
Chemical Properties Formation of ether (Williamson’s synthesis) But not: RX is usually 1 ry should (Why? ) alkyl halide NOT be 2 ry and 3 ry. (Why? )

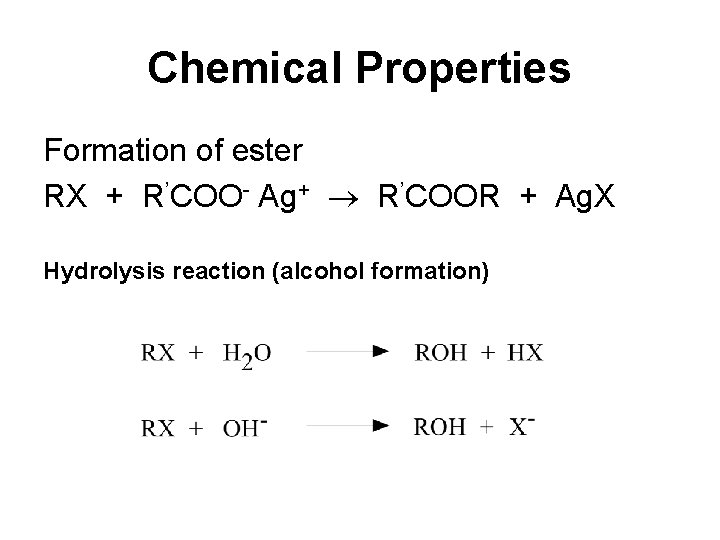
Chemical Properties Formation of ester RX + R’COO- Ag+ R’COOR + Ag. X Hydrolysis reaction (alcohol formation)

Chemical properties Formation of Grignard Reagent excess

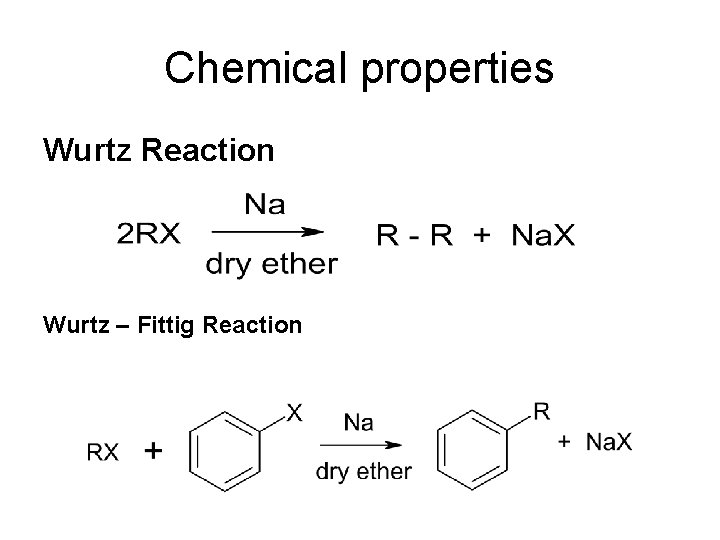
Chemical properties Wurtz Reaction Wurtz – Fittig Reaction

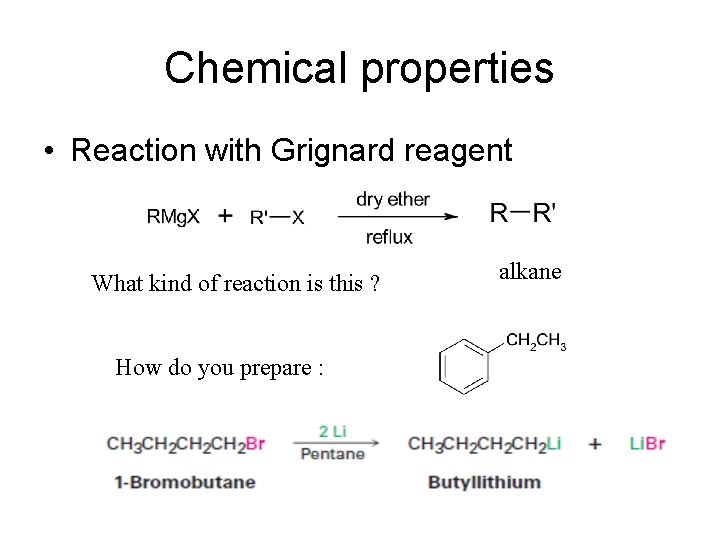
Chemical properties • Reaction with Grignard reagent What kind of reaction is this ? How do you prepare : alkane

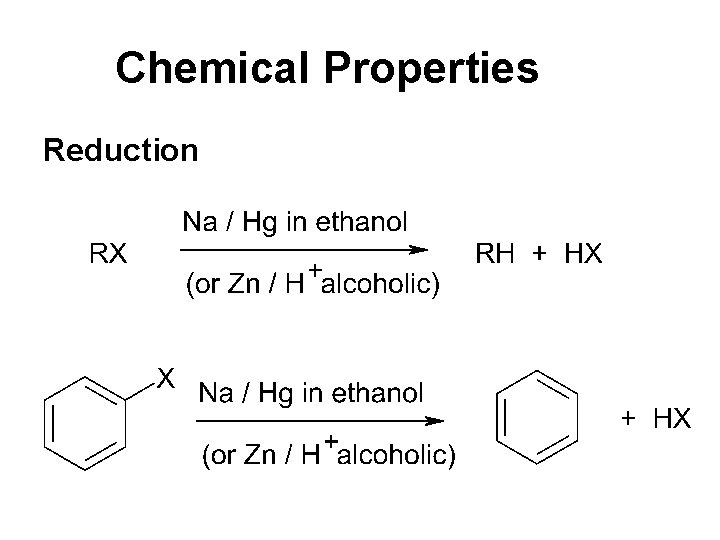
Chemical Properties Reduction

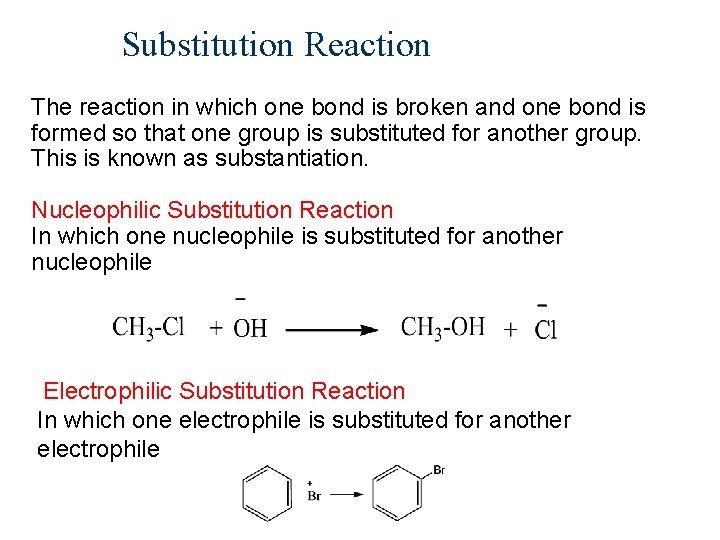
Substitution Reaction The reaction in which one bond is broken and one bond is formed so that one group is substituted for another group. This is known as substantiation. Nucleophilic Substitution Reaction In which one nucleophile is substituted for another nucleophile Electrophilic Substitution Reaction In which one electrophile is substituted for another electrophile

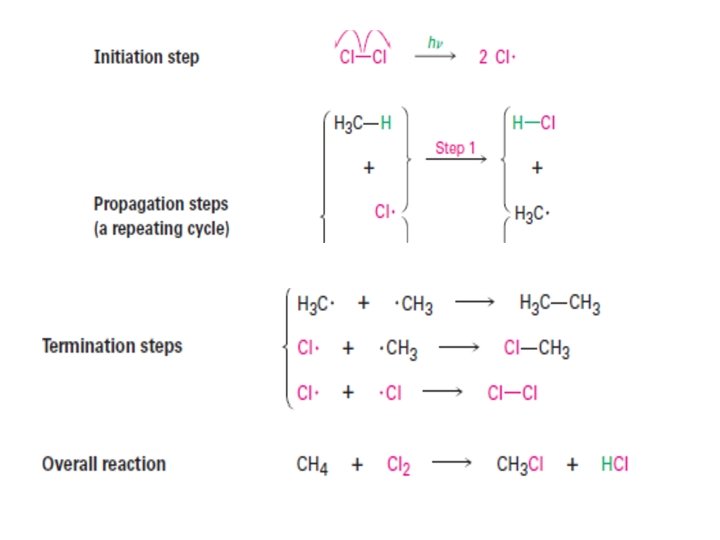
Nucleophilic Substitution Reactions Nucleophilic Substitution (1896 by the German chemist Paul Walden) Halogen compounds are polar compounds. The electron deficient carbon attach to the halogen is susceptible to the attack of an electron rich species (nucleophile) and undergo nucleophilic substitution.

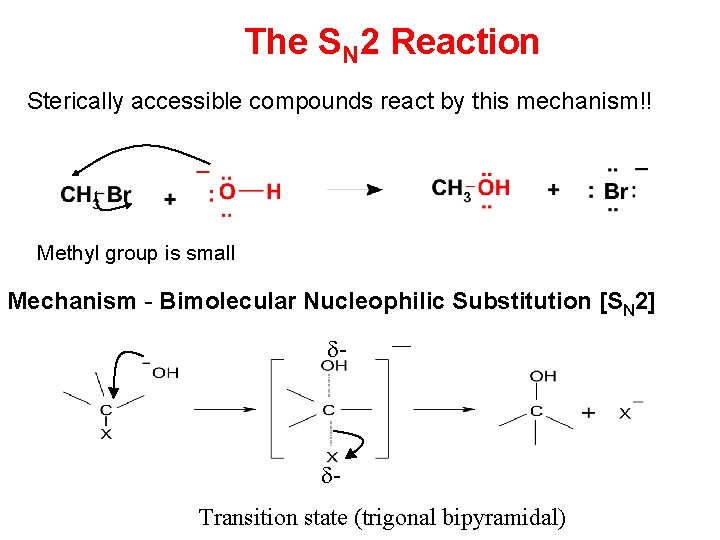
SN 2 Reaction SN 2 reaction: It represents nucleophilic, bimolecular reaction, (Bimolecular means that two molecules i. e. nucleophile and alkyl halide, take part in the step whose kinetics are measured. ) • Two species in the rate determine step • When Nu─ attacks on a substrate the breaking of old bond and formation of a new bond takes place simultaneously and the reaction proceed through the formation of transition state. • Transition state (T. S. ) is a slow step and is called rate determing step.

The SN 2 Reaction Sterically accessible compounds react by this mechanism!! Methyl group is small Mechanism - Bimolecular Nucleophilic Substitution [SN 2] - Transition state (trigonal bipyramidal)

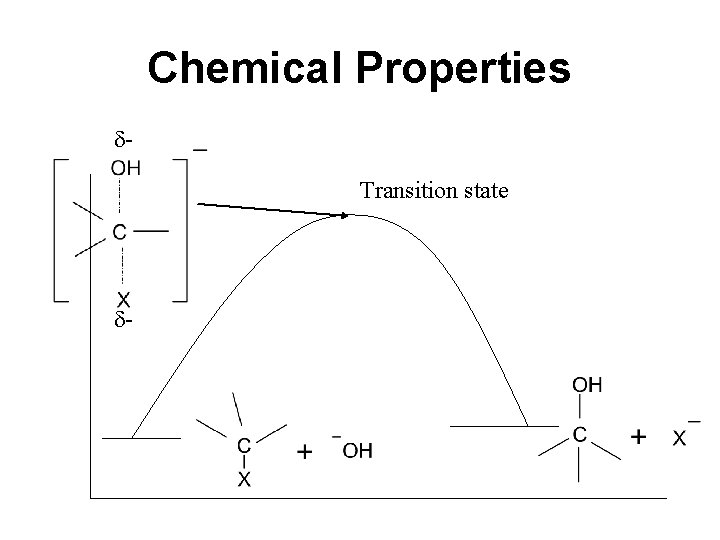
Chemical Properties Transition state -

Chemical Properties • Bimolecular: Molecularity refers to the number of species that are undergoing bond-making and / or bond-breaking process in the rate determining step. Rate = k [alkyl halide]1 [OH-]1 Second order reaction If concentration of any of the two species is doubled the rate of reaction will be doubled and if conc. of both the substrate and nucleophile is doubled the rate of reaction will increases four times.

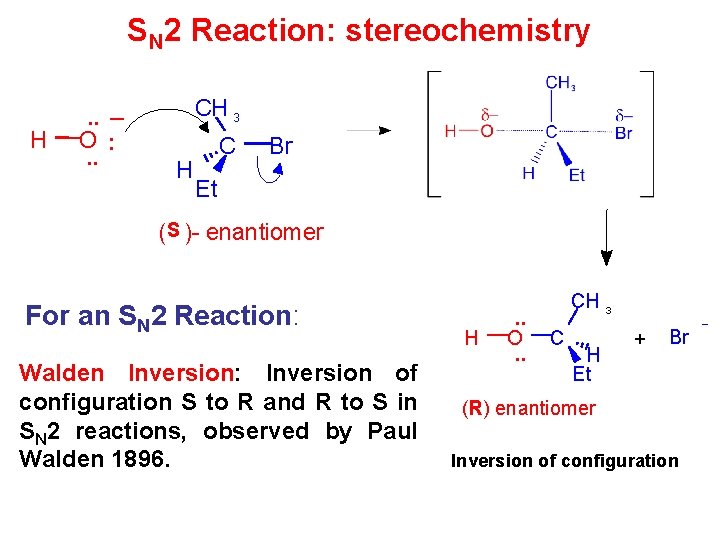
SN 2 Reaction: stereochemistry H . . _ O : . . CH 3 H C Br Et (S )- enantiomer For an SN 2 Reaction: Walden Inversion: Inversion of configuration S to R and R to S in SN 2 reactions, observed by Paul Walden 1896. H . . O. . CH 3 C H Et _ + Br (R) enantiomer Inversion of configuration

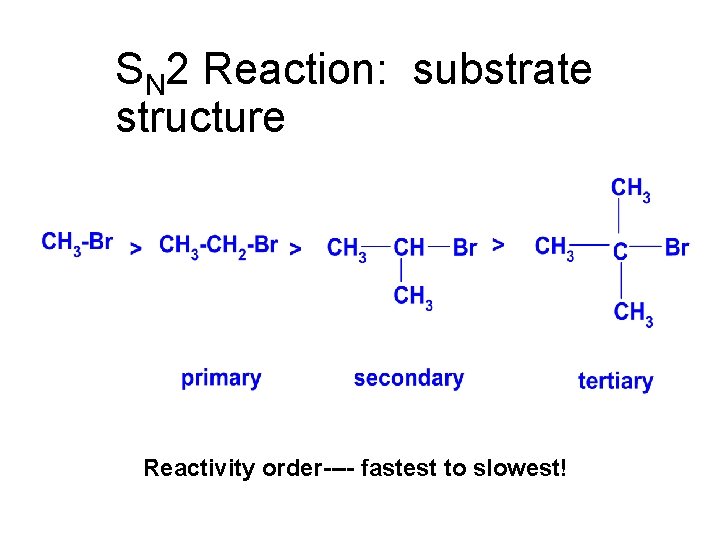
SN 2 Reaction: substrate structure Reactivity order---- fastest to slowest!

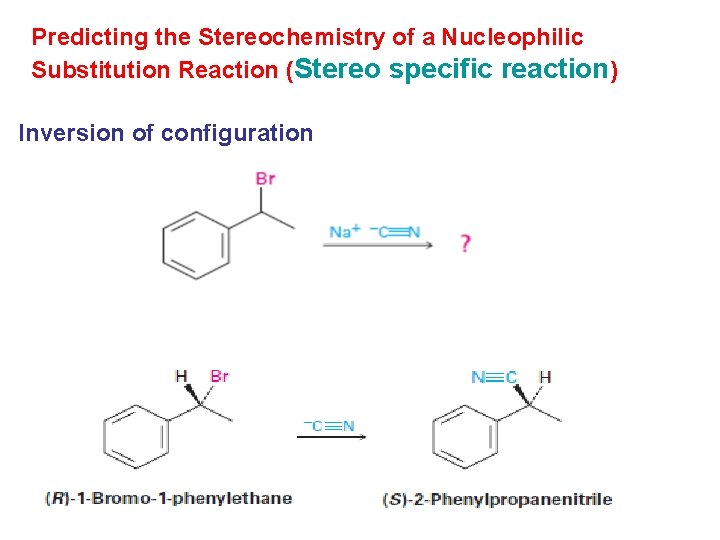
Predicting the Stereochemistry of a Nucleophilic Substitution Reaction (Stereo specific reaction) Inversion of configuration

The Substrate: Steric Effects in the SN 2 Reaction v Hindered and bulky substrate prevent easy approach of the nucleophile, making bond formation difficult. v The transition state of a sterically hindered substrate, is higher in energy and forms more slowly than the corresponding transition state for a less hindered substrate.

The Nucleophile Any species, either neutral or negatively charged, can act as a nucleophile as long as it has an unshared pair of electrons; that is, as long as it is a Lewis base. Nucleophilicity” is usually taken as the affinity of a Lewis base for a carbon atom in the SN 2 reaction and “basicity” is the affinity of a base for a proton. Thus a nucleophile attacks on carbon (C) while base attacks on proton (H+) in SN 2 reactions.

leaving group Since the leaving group is expelled with a negative charge in most SN 2 reactions, the best leaving groups are those that best stabilize the negative charge in the transition state. The greater the extent of charge stabilization by the leaving group, the lower the energy of the transition state and the more rapid the reaction.

In a reaction, the exact nucleophilicity of a species depends on the substrate, the solvent, and the reactant concentrations. • Nucleophilicity usually increases going down a column of the periodic table. Thus, H 2 S is more nucleophilic than H 2 O, and the halide reactivity order is I 2> Br 2> Cl 2.

Down the periodic table, elements have their valence electrons less tightly held, and consequently more reactive. The nucleophilicity order can change depending on the solvent. • Negatively (─ve) charged nucleophiles are usually more reactive than neutral ones. As a result, SN 2 reactions are often carried out under basic conditions rather than neutral or acidic conditions.

carbocation
![SN 1 properties Mechanism Unimolecular Nucleophilic Substitution SN 1 Unimolecular because SN 1 properties • Mechanism - Unimolecular Nucleophilic Substitution [SN 1] • Unimolecular because](https://slidetodoc.com/presentation_image_h2/2eedcea8419eeff9cb5f649ea017b04e/image-41.jpg)
SN 1 properties • Mechanism - Unimolecular Nucleophilic Substitution [SN 1] • Unimolecular because in rate determining step, only one molecule is involved. Rate = k [alkyl halide]1 [OH-]0 Rate = k [R-Br]1 Thus it follows first order (unimolecular) kinetics.

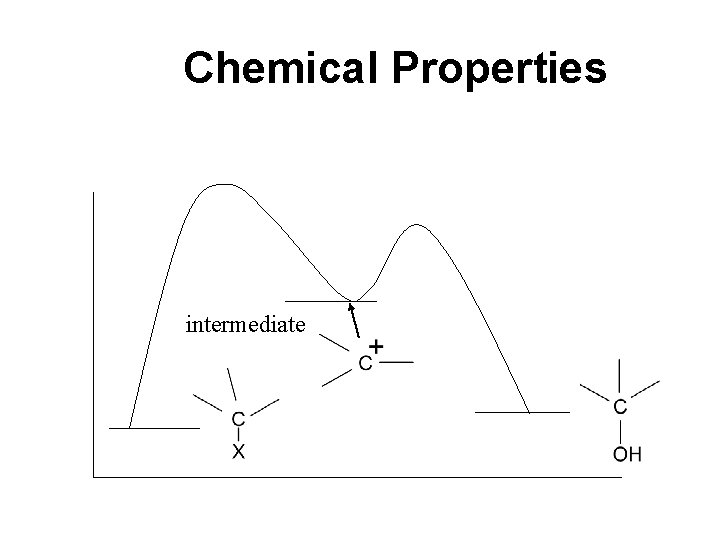
Chemical Properties intermediate

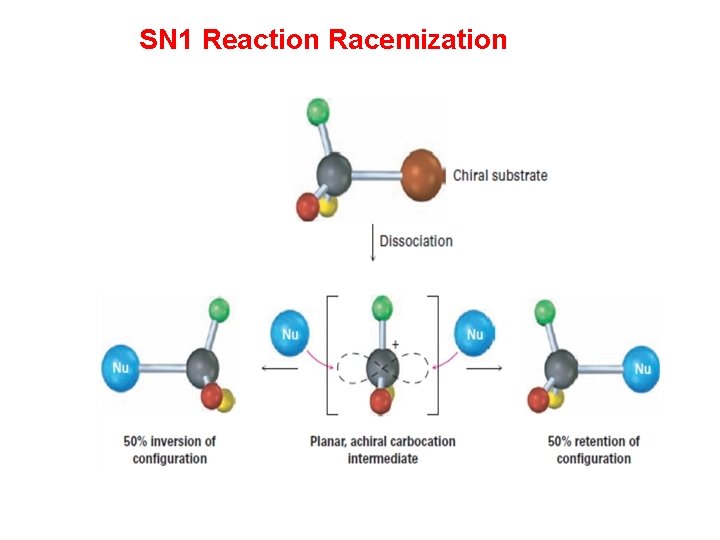
SN 1 Reaction: stereochemistry With chiral R-X compounds, the product will be racemic (50% of each enantiomer). Racemization is the conversion of one enantiomer in a 50: 50 mixture of the two enantiomers (+ and −, or R and S) of a substance. Racemization is normally associated with the loss of optical activity over a period of time since 50: 50 mixtures of enantiomers are optically inactive.

SN 1 Reaction Racemization

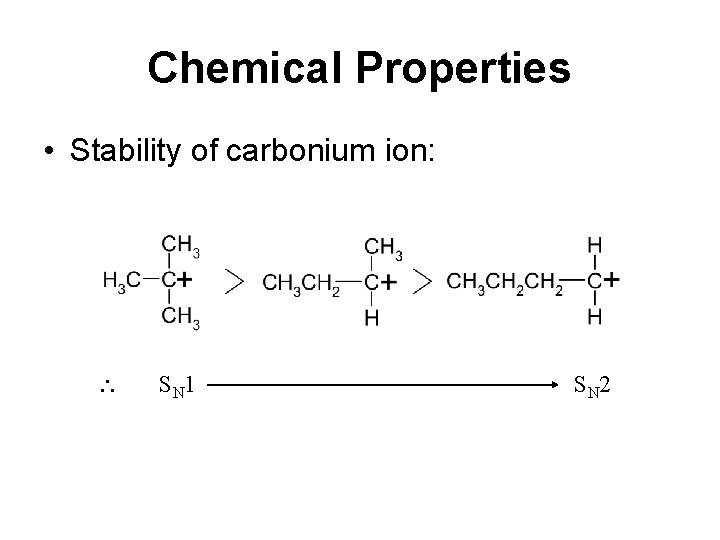
Chemical Properties • Stability of carbonium ion: S N 1 S N 2

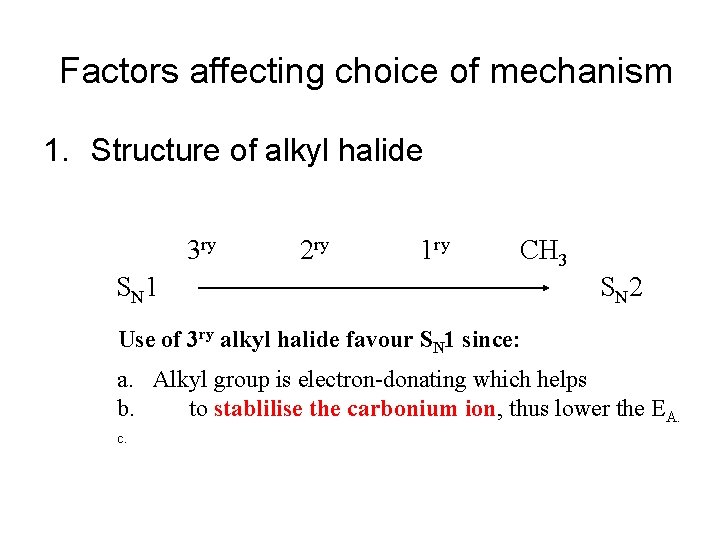
Factors affecting choice of mechanism 1. Structure of alkyl halide 3 ry 2 ry 1 ry CH 3 S N 1 S N 2 Use of 3 ry alkyl halide favour SN 1 since: a. Alkyl group is electron-donating which helps b. to stablilise the carbonium ion, thus lower the EA. c.

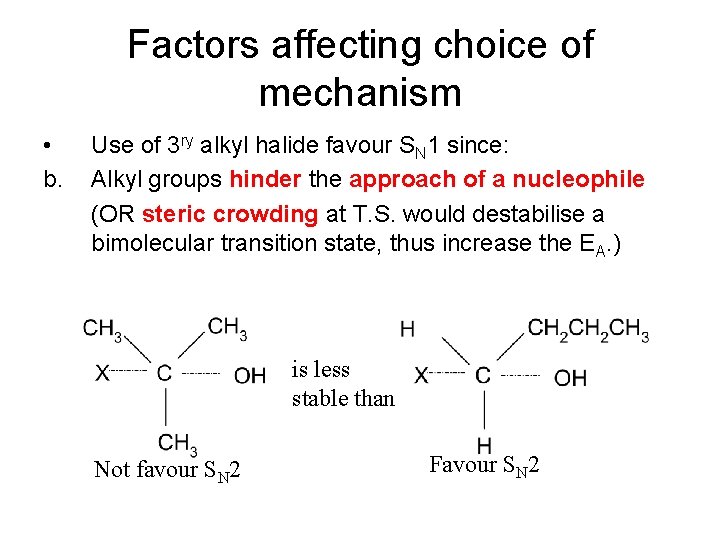
Factors affecting choice of mechanism • b. Use of 3 ry alkyl halide favour SN 1 since: Alkyl groups hinder the approach of a nucleophile (OR steric crowding at T. S. would destabilise a bimolecular transition state, thus increase the EA. ) is less stable than Not favour SN 2 Favour SN 2

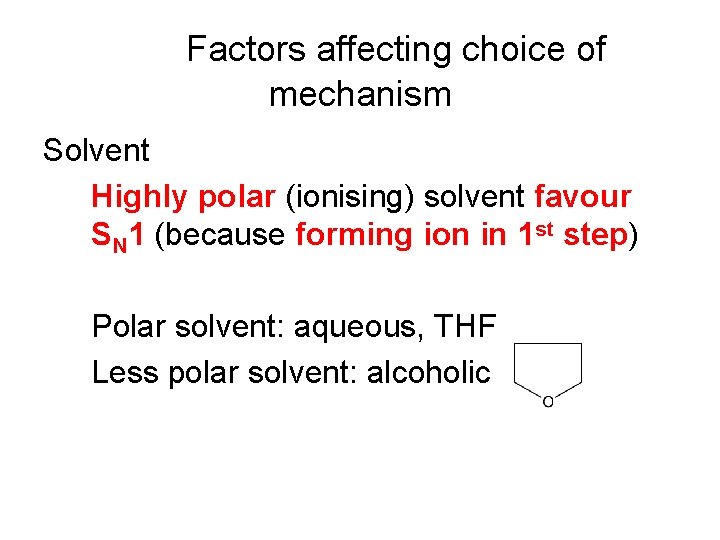
Factors affecting choice of mechanism Solvent Highly polar (ionising) solvent favour SN 1 (because forming ion in 1 st step) Polar solvent: aqueous, THF Less polar solvent: alcoholic

Chemical Properties Factors affecting choice of mechanism Choice of nucleophile Strong nucleophile in high conc. favour SN 2 while weak nucleophile in dilute solution favour SN 1. Strong nucleophile Weak nucleophile OHH 2 O NH 2 NH 3 CNHCN ROROH Presence of Ag+ ion favour SN 1

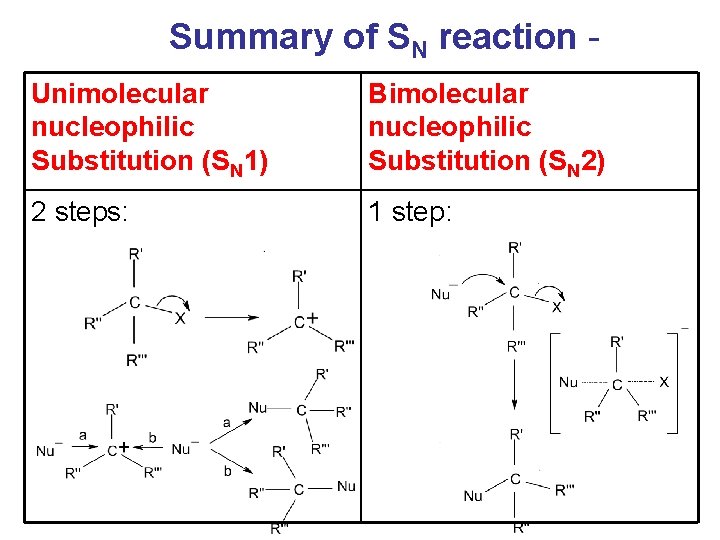
Summary of SN reaction Unimolecular nucleophilic Substitution (SN 1) Bimolecular nucleophilic Substitution (SN 2) 2 steps: 1 step:
![Summary of SN reaction Unimolecular nucleophilic Substitution SN 1 Rate k alkyl halide Summary of SN reaction Unimolecular nucleophilic Substitution (SN 1) Rate = k [alkyl halide]](https://slidetodoc.com/presentation_image_h2/2eedcea8419eeff9cb5f649ea017b04e/image-51.jpg)
Summary of SN reaction Unimolecular nucleophilic Substitution (SN 1) Rate = k [alkyl halide] Bimolecular nucleophilic Substitution (SN 2) Rate = k [alkyl halide] [Nu] Carbonium ion formed as No intermediate (stabilized by carbonium ions but only inductive effect) transition states are involved. Usually occur with tertiary Usually occur with primary alkyl halide Energy profile: 2 peaks Energy profile: 1 peak

Summary of SN reaction Unimolecular Bimolecular nucleophilic Substitution nucleophilic (SN 1) Substitution (SN 2) Because of the equal chance of attack from both sides of carbonium ions, a racemic mixture of enantiomers obtained, i. e. optically inactive. Rate of Rx: Ph. CH 2 X > RCH=CHCH 2 X > 3 o > 2 o > 1 o Configuration of the carbon centre attacked inverted (inversion of configuration). If the original alkyl halide is optically active, optically active product will be obtained. Rate of Rx: 1 o > 2 o > 3 o

Vinyl and Phenyl Compounds


Chemical Properties Effect of halogen: Since the electronagativity of halogen decreased down the group, C-Cl bond is more polar than the others. Hence, the carbon join to Cl is the most electron deficient, so the carbon in R-Cl in most susceptible to the attack of nucleophile. The bond strength is also important in determining the rate since bond strength decreased rapidly from C-Cl to C-I bond, the reaction rate decreases in the order : R – I > R – Br > R – Cl