Organic and Biological Molecules 1 Organic Chemistry and

Organic and Biological Molecules 1

Organic Chemistry and Biochemistry • The study of carbon-containing compounds and their properties. • The vast majority of organic compounds contain chains of rings of carbon atoms. • The study of the chemistry of living matter 2

Organic Chemistry Today The study of compounds and reactions involving carbon, regardless of source (Kekulé, 1861) Is Vitalism “Dead”: “Natural products” – from living organisms “Biochemistry” – the chemistry that occurs in living organisms Organic Vegetables, Natural Vitamins from extracts 3
Why should we care? Biochemistry – Pharmaceuticals – Industry – Polymers • Explosives • 4
Organic Molecules 5

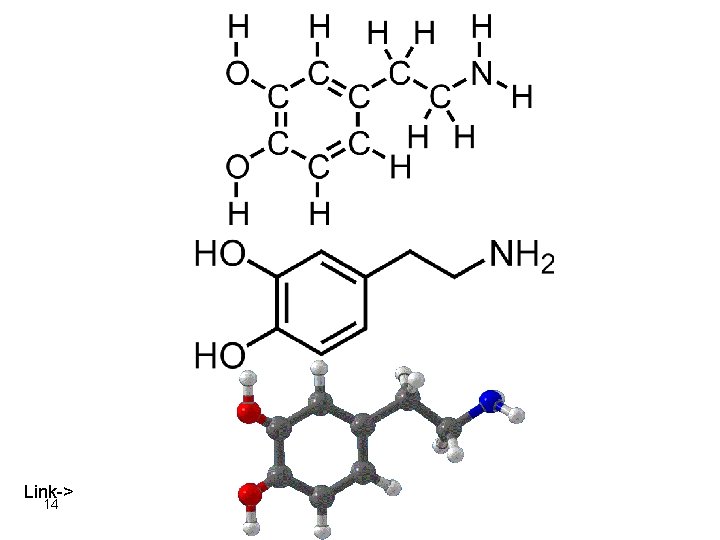
the brain 6 dopamine

Synaptic neurotransmission Dopamine neuron terminal Target Neurons dendrite axon cell body dopamine transporter vesicles tyrosine 7 l-dopamine presynaptic receptor synapse postsynaptic receptor

Dopamine a neurotransmitter present in the brain a space filling model 8
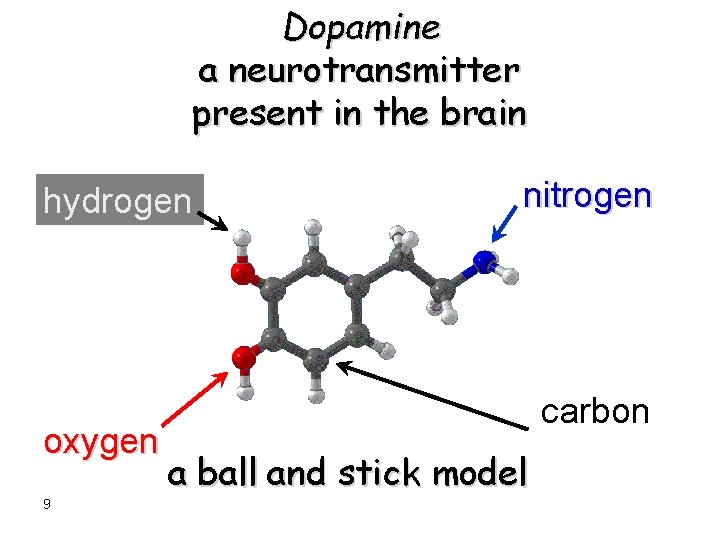
Dopamine a neurotransmitter present in the brain hydrogen oxygen 9 nitrogen carbon a ball and stick model

Dopamine 10

Dopamine. Dash Molecular formula two lines = 4 electrons one line = 2 electrons Condensed molecular formula = 11 C 8 H 11 NO 2

Dopamine. Line-Angle Molecular formula 12 all nodes are carbon add hydrogens until there are 4 bonds to each carbon atom
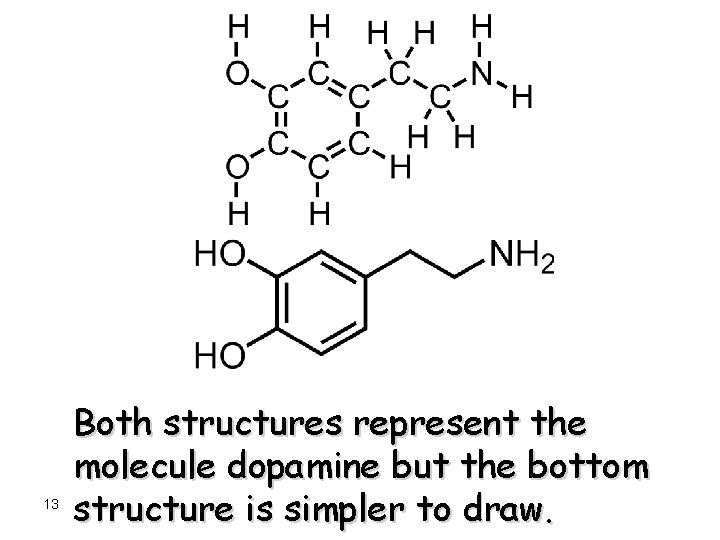
13 Both structures represent the molecule dopamine but the bottom structure is simpler to draw.
Link-> 14

Chemical Formulae Empirical formula • Smallest integer ratio between atoms of – different types in a molecule. Molecular formula • Actual number of atoms of each type in a – molecule. Example: • Ethene (C 2 H 4) and cyclohexane (C 6 H 12) both – have the empirical formula (CH 2) 15

Ibuprofen is the active ingredient in a number of analgesics. The structure of ibuprofen is given below. What is the molecular formula of ibuprofen? C H O 2 16
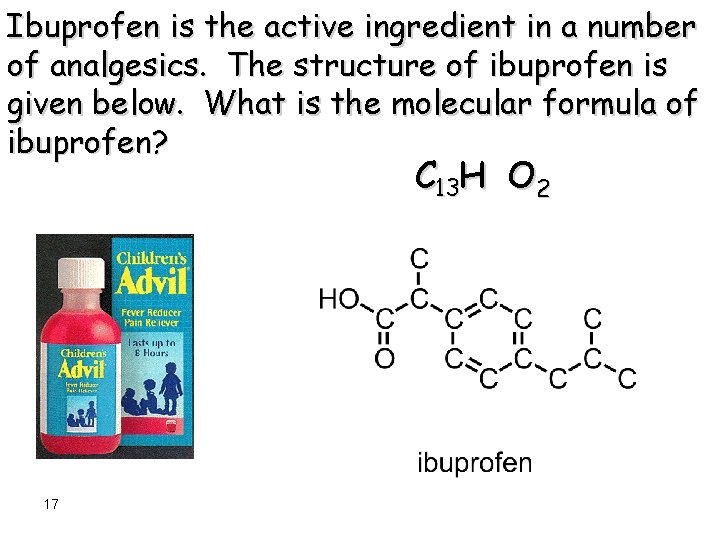
Ibuprofen is the active ingredient in a number of analgesics. The structure of ibuprofen is given below. What is the molecular formula of ibuprofen? C 13 H O 2 17
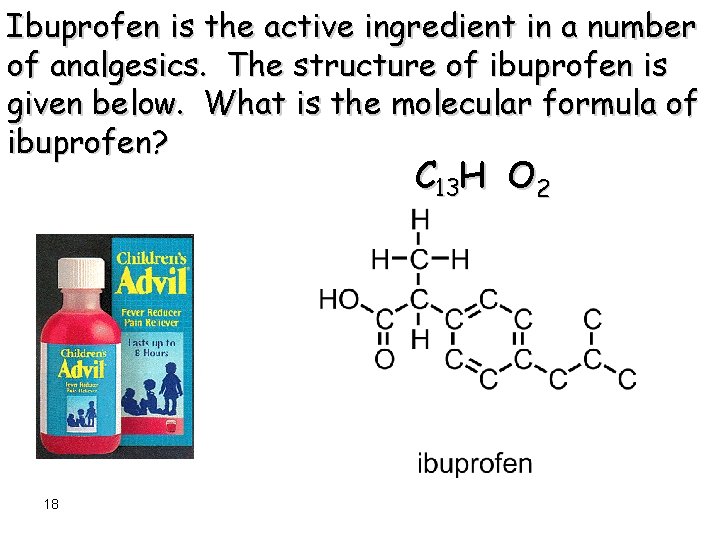
Ibuprofen is the active ingredient in a number of analgesics. The structure of ibuprofen is given below. What is the molecular formula of ibuprofen? C 13 H O 2 18
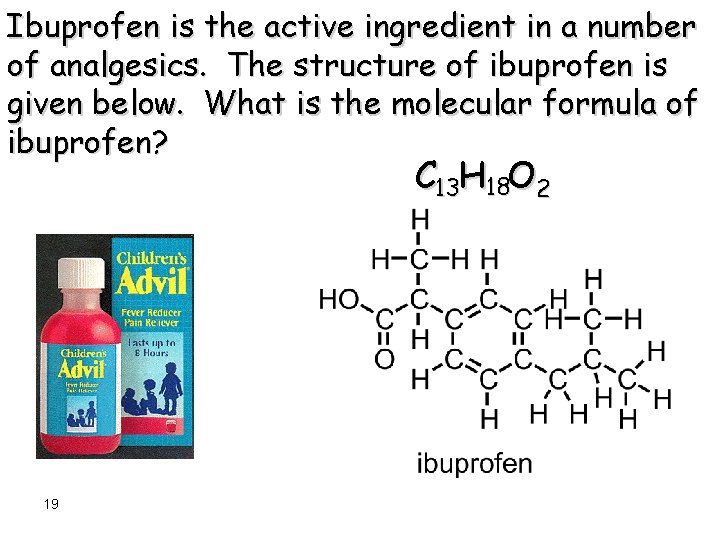
Ibuprofen is the active ingredient in a number of analgesics. The structure of ibuprofen is given below. What is the molecular formula of ibuprofen? C 13 H 18 O 2 19

Ibuprofen is the active ingredient in a number of analgesics. The structure of ibuprofen is given below. What is the molecular formula of ibuprofen? C 13 H 18 O 2 20
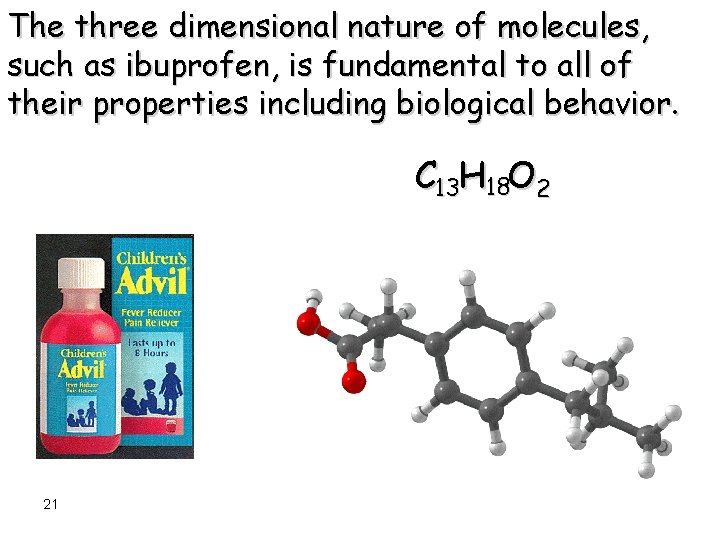
The three dimensional nature of molecules, such as ibuprofen, is fundamental to all of their properties including biological behavior. C 13 H 18 O 2 21

Bonding and Molecular Structure Structural formulas of organic compounds structural formula 22 condensed formula line-angle formula
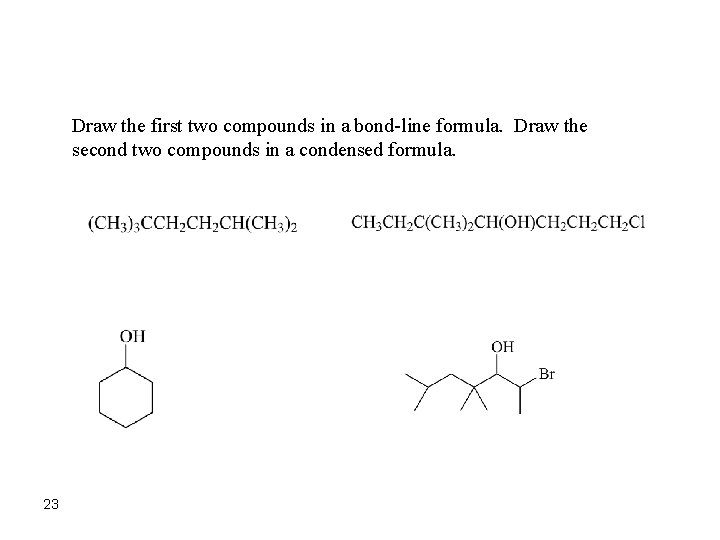
Draw the first two compounds in a bond-line formula. Draw the second two compounds in a condensed formula. 23
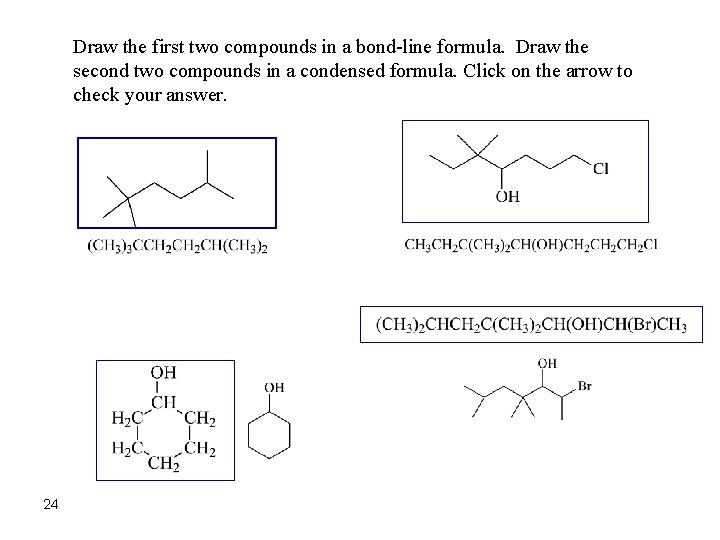
Draw the first two compounds in a bond-line formula. Draw the second two compounds in a condensed formula. Click on the arrow to check your answer. 24

Hydrocarbons • compounds composed of carbon and hydrogen. Saturated compounds (alkanes) have the maximum number of hydrogen atoms attached to each carbon atom Saturated: carbon-carbon bonds are all single - alkanes [Cn. H 2 n+2] • • 25

l Unsaturated compounds have fewer hydrogen atoms attached to the carbon chain than alkanes • Unsaturated: They contain carbon multiple bonds (double or triple) 26

Alkanes: Saturated hydrocarbons • Saturated hydrocarbons, Cn. H 2 n+2 – “Saturated” because they can’t take any more hydrogen atoms – Straight chains are H 3 C–(CH 2)n– 2–CH 3 – Waxes, oils, & fuel gases as n decreases. 27
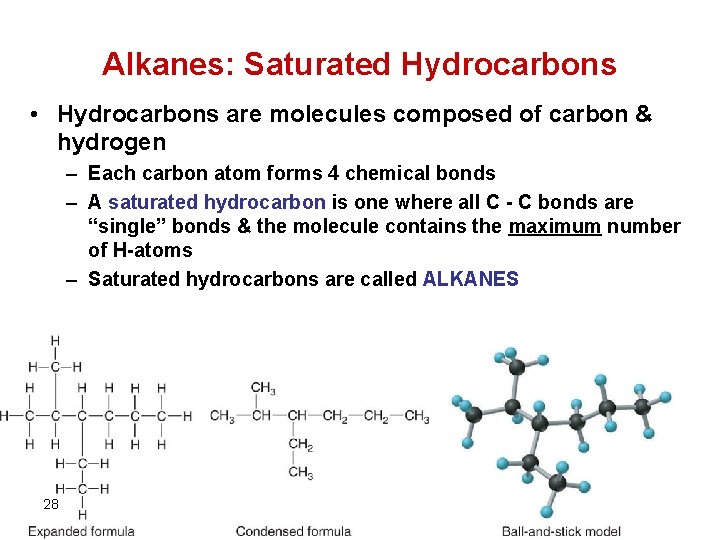
Alkanes: Saturated Hydrocarbons • Hydrocarbons are molecules composed of carbon & hydrogen – Each carbon atom forms 4 chemical bonds – A saturated hydrocarbon is one where all C - C bonds are “single” bonds & the molecule contains the maximum number of H-atoms – Saturated hydrocarbons are called ALKANES 28
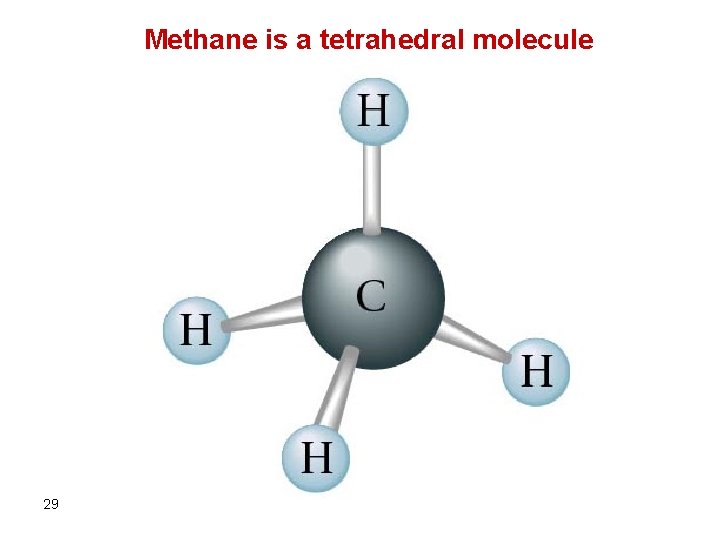
Methane is a tetrahedral molecule 29
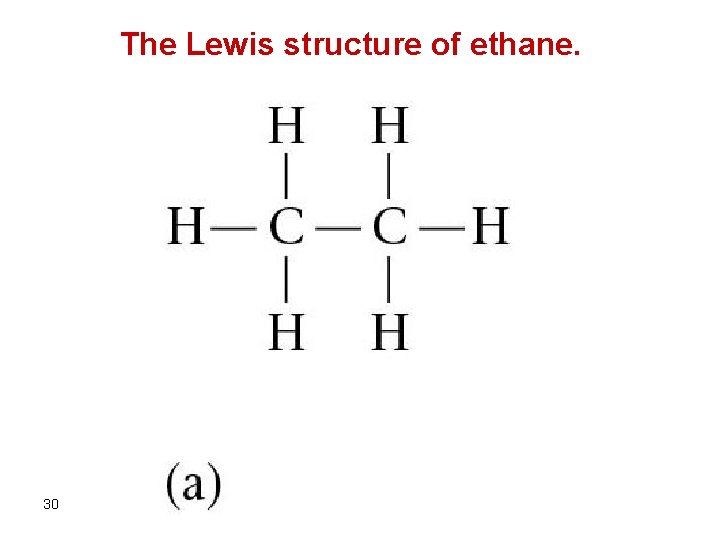
The Lewis structure of ethane. 30
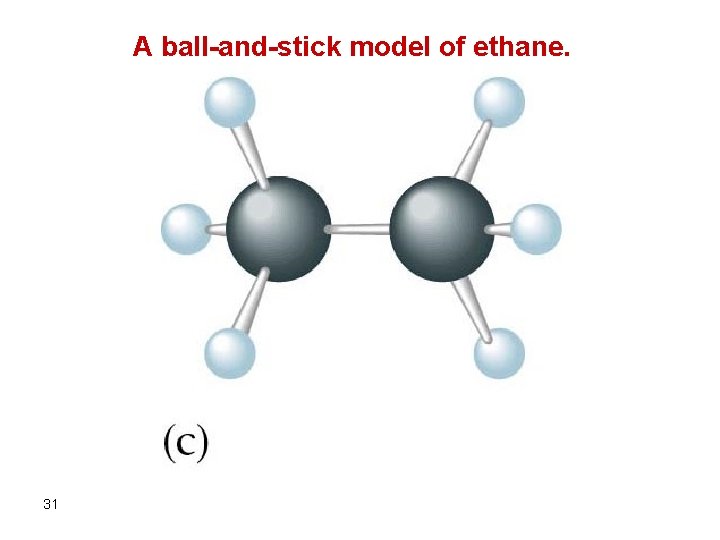
A ball-and-stick model of ethane. 31

Propane 32

Butane 33

The First 10 “Normal” Alkanes • • • 34 Name Formula M. P. B. P. Methane Ethane Propane Butane Pentane Hexane Heptane Octane Nonane Decane CH 4 C 2 H 6 C 3 H 8 C 4 H 10 C 5 H 12 C 6 H 14 C 7 H 16 C 8 H 18 C 9 H 20 C 10 H 22 -183 -172 -187 -138 -130 -95 -91 -57 -54 -30 -162 -89 -42 0 36 68 98 126 151 174 # Structural Isomers 1 1 1 2 3 5 9 18 35 75 C 1 - C 4 are Gases at Room Temperature C 5 - C 16 are Liquids at Room Temperature
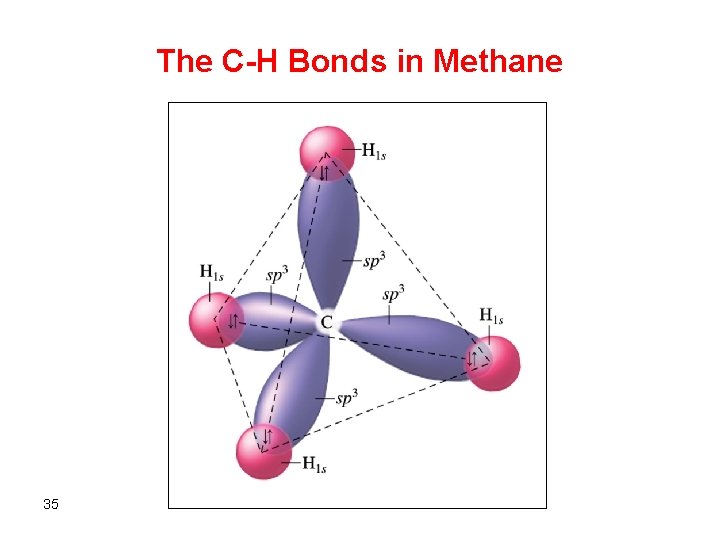
The C-H Bonds in Methane 35

IUPAC Rules for Naming Branched Alkanes 3/25 • Find and name the parent chain in the hydrocarbon this forms the root of the hydrocarbon name • Number the carbon atoms in the parent chain starting at the end closest to the branching • Name alkane branches by dropping the “ane” from the names and adding “yl”. A one-carbon branch is called “methyl”, a two-carbon branch is “ethyl”, etc… • When there are more than one type of branch (ethyl and methyl, for example), they are named alphabetically 36 • Finally, use prefixes to indicate multiple branches

Rules for Naming Alkanes 1. For alkanes beyond butane, add -ane to the Greek root for the number of carbons. C-C-C-C : hexane 2. Alkyl substituents: drop the -ane and add -yl -C 2 H 5 is ethyl 37
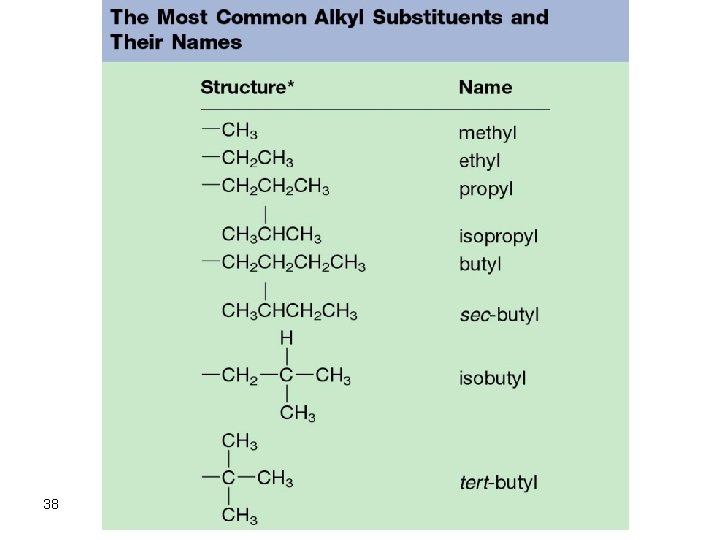
38

Rules for Naming Alkanes 3. Positions of substituent groups are specified by numbering the longest chain sequentially. C C-C-C-C 3 -methylhexane • Start numbering at the end closest to the branching 4. Location and name are followed by root alkane name. Substituents in alphabetical order and use di-, tri-, etc. 39
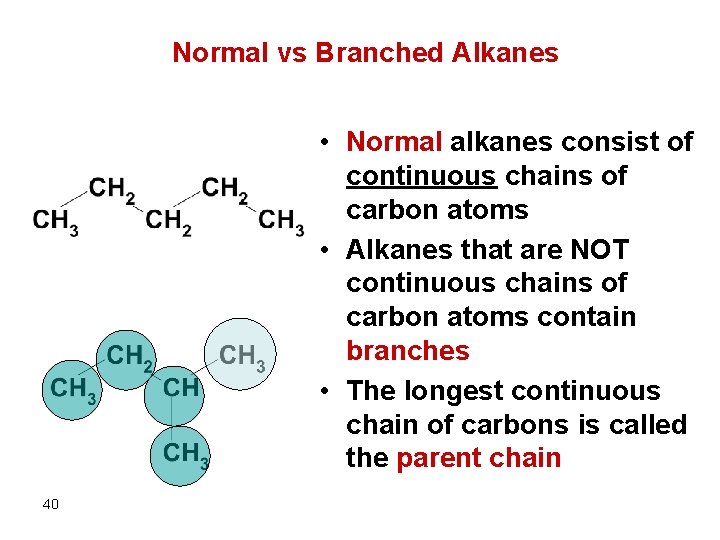
Normal vs Branched Alkanes • Normal alkanes consist of continuous chains of carbon atoms • Alkanes that are NOT continuous chains of carbon atoms contain branches • The longest continuous chain of carbons is called the parent chain 40
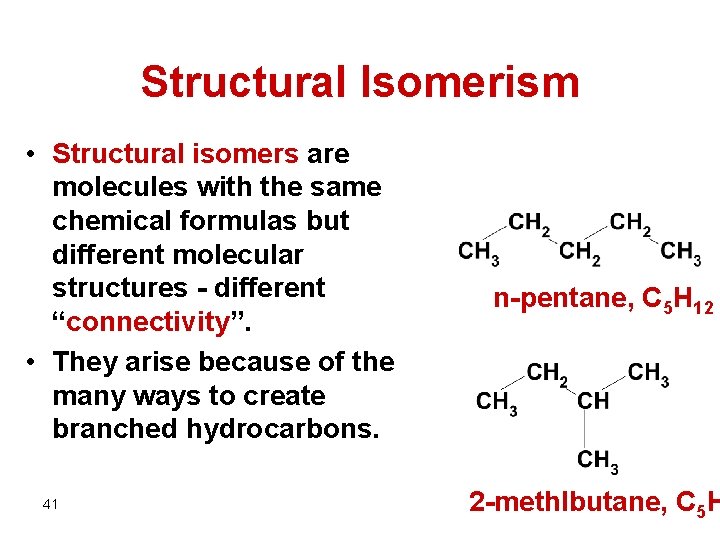
Structural Isomerism • Structural isomers are molecules with the same chemical formulas but different molecular structures - different “connectivity”. • They arise because of the many ways to create branched hydrocarbons. 41 n-pentane, C 5 H 12 2 -methlbutane, C 5 H

Isomer Naming • Older conventions would have that as “isooctane, ” but a good IUPAC name results from the following: – Name the longest C chain (pentane) – List the side groups in alphabetical order with Greek prefixes (trimethylpentane) – Supply (smallest possible) positional indices (2, 2, 4 trimethylpentane) 42
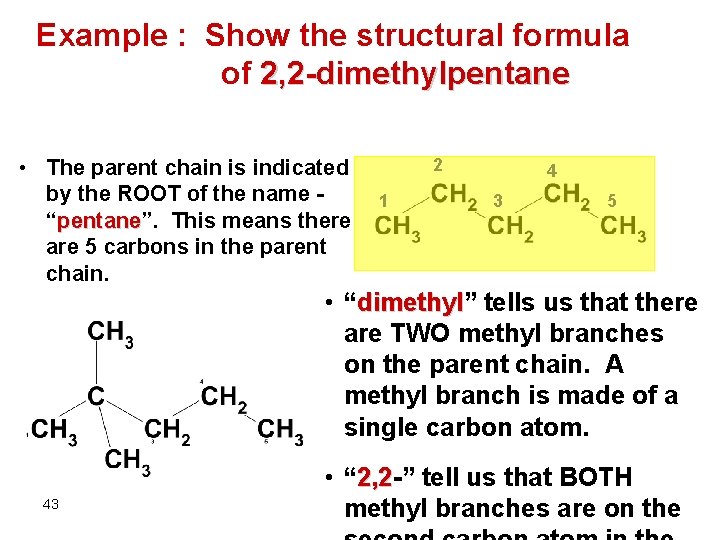
Example : Show the structural formula of 2, 2 -dimethylpentane • The parent chain is indicated by the ROOT of the name “pentane”. pentane This means there are 5 carbons in the parent chain. 2 1 4 3 5 • “dimethyl” dimethyl tells us that there are TWO methyl branches on the parent chain. A methyl branch is made of a single carbon atom. 43 • “ 2, 2 -” 2, 2 tell us that BOTH methyl branches are on the
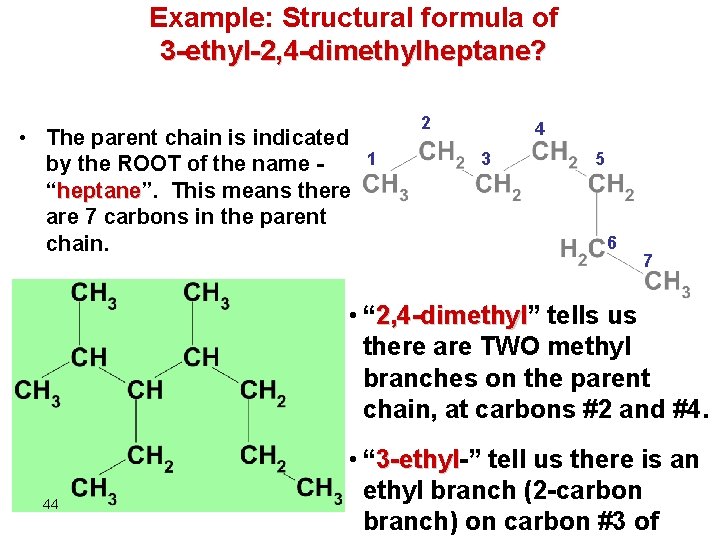
Example: Structural formula of 3 -ethyl-2, 4 -dimethylheptane? • The parent chain is indicated by the ROOT of the name “heptane”. heptane This means there are 7 carbons in the parent chain. 2 1 4 3 5 6 7 • “ 2, 4 -dimethyl” 2, 4 -dimethyl tells us there are TWO methyl branches on the parent chain, at carbons #2 and #4. 44 • “ 3 -ethyl-” 3 -ethyl tell us there is an ethyl branch (2 -carbon branch) on carbon #3 of
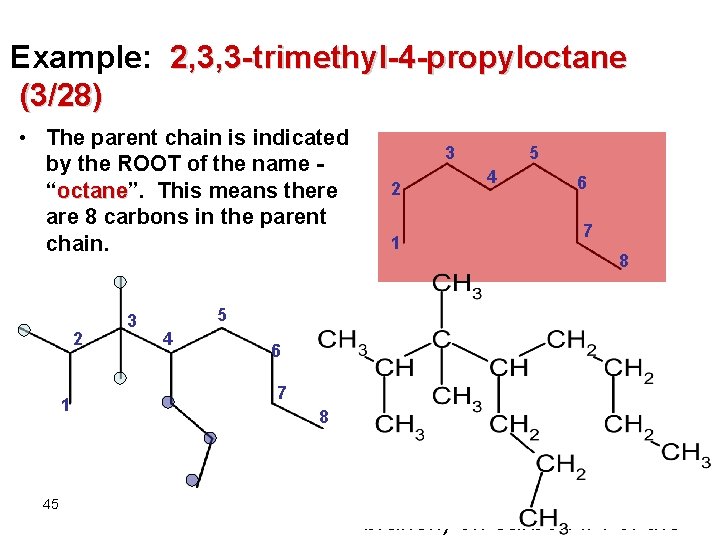
Example: 2, 3, 3 -trimethyl-4 -propyloctane (3/28) • The parent chain is indicated by the ROOT of the name “octane”. octane This means there are 8 carbons in the parent chain. 2 1 45 3 5 4 6 7 8 3 2 1 5 4 6 7 8 • “ 2, 3, 3 -trimethyl” 2, 3, 3 -trimethyl tells us there are THREE methyl branches - one on carbon #2 and two on carbon #3. • “ 4 -propyl-” 4 -propyl tell us there is a propyl branch (3 -carbon branch) on carbon #4 of the
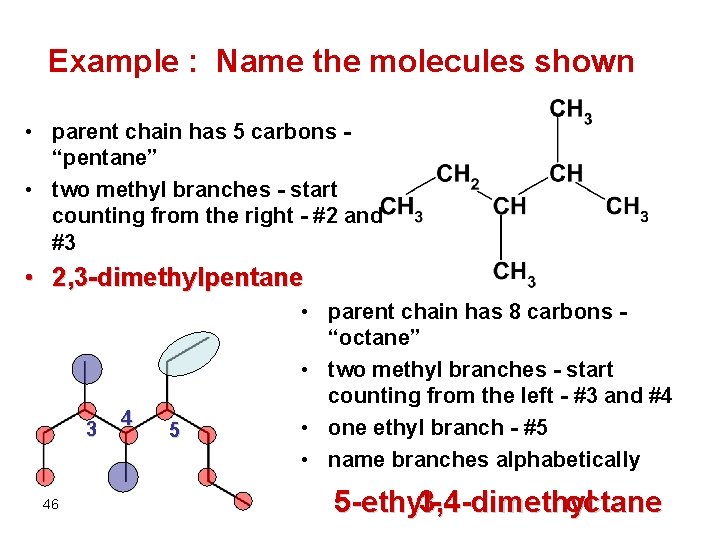
Example : Name the molecules shown • parent chain has 5 carbons “pentane” • two methyl branches - start counting from the right - #2 and #3 • 2, 3 -dimethylpentane 3 46 4 5 • parent chain has 8 carbons “octane” • two methyl branches - start counting from the left - #3 and #4 • one ethyl branch - #5 • name branches alphabetically 5 -ethyl 3, 4 -dimethyl octane
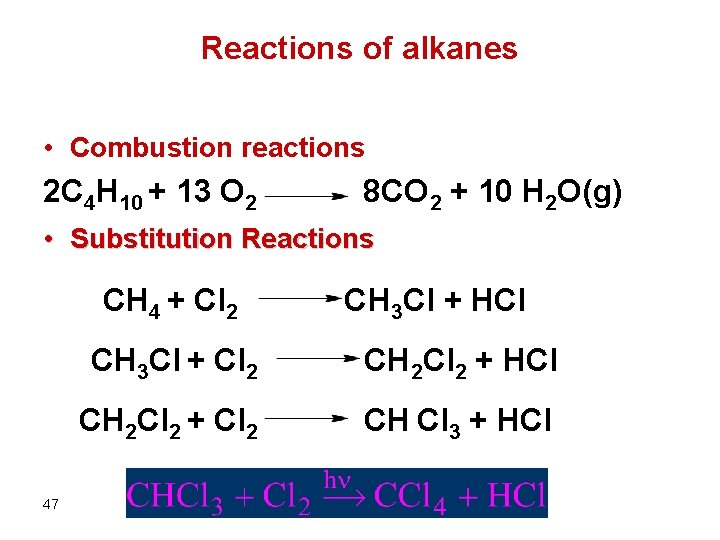
Reactions of alkanes • Combustion reactions 2 C 4 H 10 + 13 O 2 8 CO 2 + 10 H 2 O(g) • Substitution Reactions CH 4 + Cl 2 47 CH 3 Cl + HCl CH 3 Cl + Cl 2 CH 2 Cl 2 + HCl CH 2 Cl 2 + Cl 2 CH Cl 3 + HCl
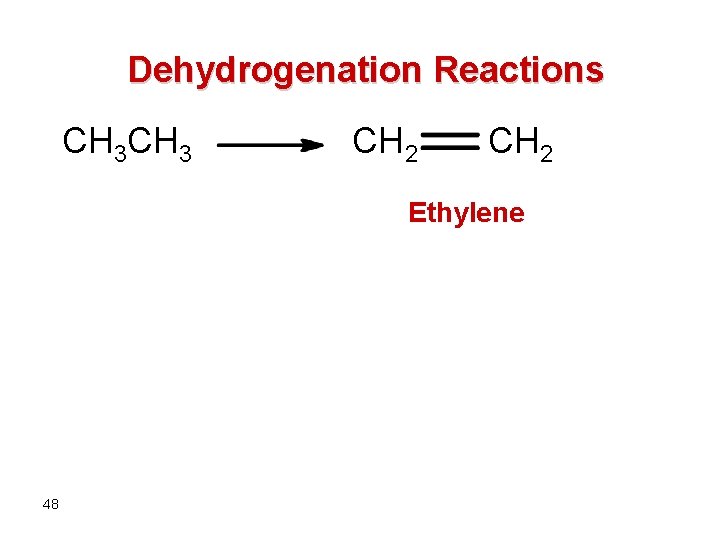
Dehydrogenation Reactions CH 3 CH 2 Ethylene 48
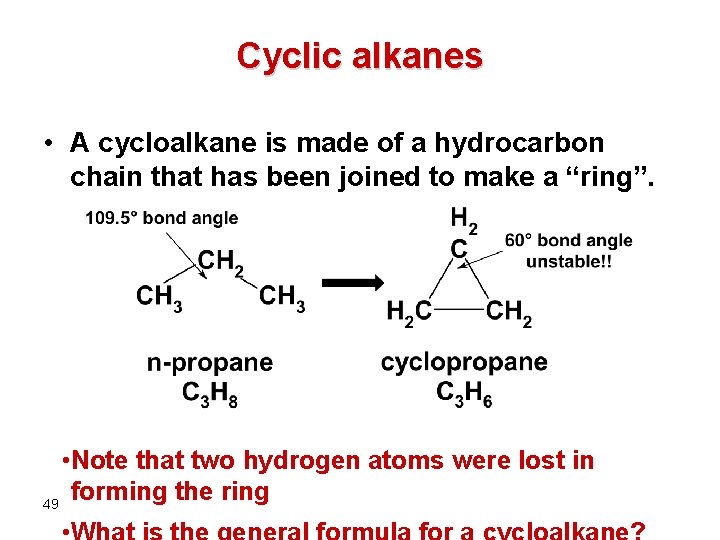
Cyclic alkanes • A cycloalkane is made of a hydrocarbon chain that has been joined to make a “ring”. • Note that two hydrogen atoms were lost in forming the ring 49

Cyclic alkanes, Cn. H 2 n • If the two end C’s lose 1 H each, they have free valence to close a ring • Again, properties similar to straight chains. – Can now have conformational isomers! – E. g. , BOAT cyclohexane versus CHAIR 50
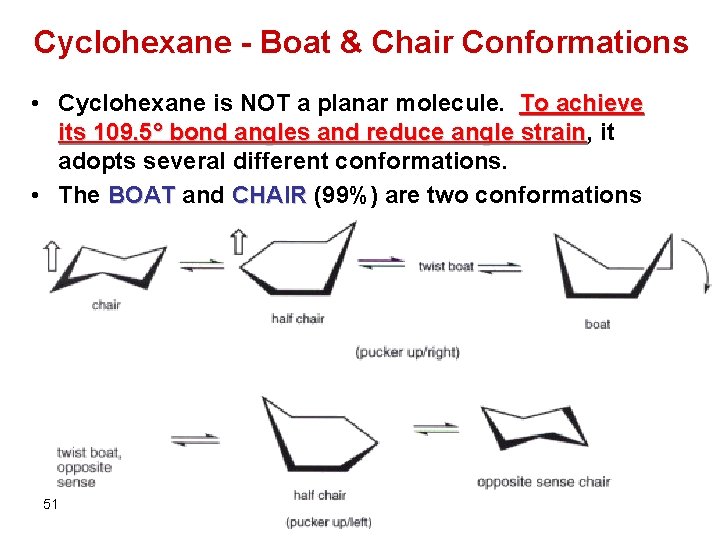
Cyclohexane - Boat & Chair Conformations • Cyclohexane is NOT a planar molecule. To achieve its 109. 5° bond angles and reduce angle strain, strain it adopts several different conformations. • The BOAT and CHAIR (99%) are two conformations 51

Unsaturated hydrocarbomns 22. 2 Alkenes and Alkynes Alkenes: hydrocarbons that contain a carbon-carbon double bond. [Cn. H 2 n] C=C Ethene C C=C propene Alkynes: hydrocarbons containing a carbon -carbon triple bond. [Cn. H 2 n-2] C C Ethyne C C C 2 -pentyne 52

Alkenes & Alkynes • The suffix for the • Alkenes are parent alkane chains hydrocarbons that are changed from contain at least one “ane” to “ene” and carbon-carbon “yne” double bond – e. g. ethene, ethyne • Alkynes are hydrocarbons that • Where it is ambiguous, the BONDS are contain at least one carbon-carbon triple numbered like branches so that the bond location of the multiple 53 bond may be indicated
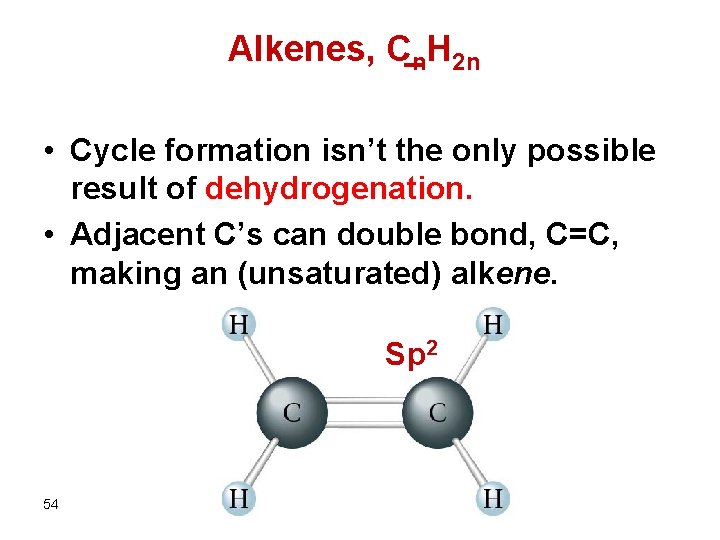
Alkenes, Cn. H 2 n • Cycle formation isn’t the only possible result of dehydrogenation. • Adjacent C’s can double bond, C=C, making an (unsaturated) alkene. Sp 2 54
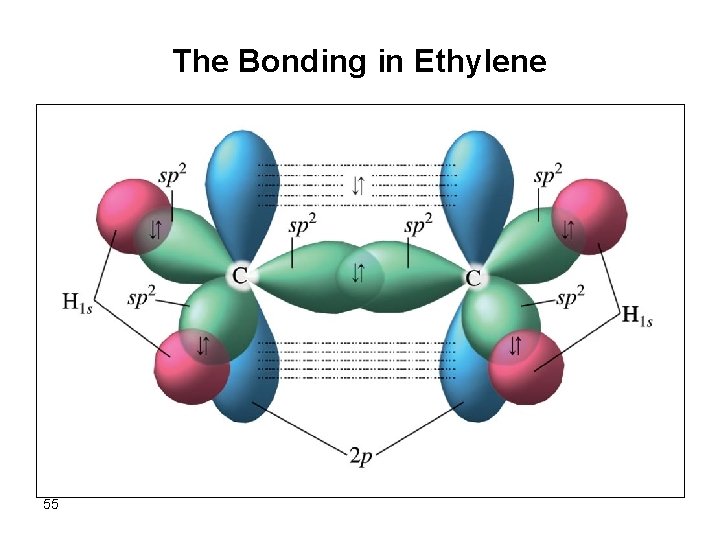
The Bonding in Ethylene 55

Nomenclature for Alkenes 1. Root hydrocarbon name ends in -ene C 2 H 4; CH 2=CH 2 is ethene 2. With more than 3 carbons, double bond is indicated by the lowest numbered carbon atom in the bond. C=C C C is 1 -butene 56

Alkene Isomers • While an sp 3 CX 2 Y 2 has only 1 isomer, • (every X and Y is adjacent to all the others) • the sp 2 alkene C 2 X 2 Y 2 has cis & trans isomers (where X is or isn’t on the same side of = as X). – For longer hydrocarbons, cis & trans refer to the side the chain extends: 57
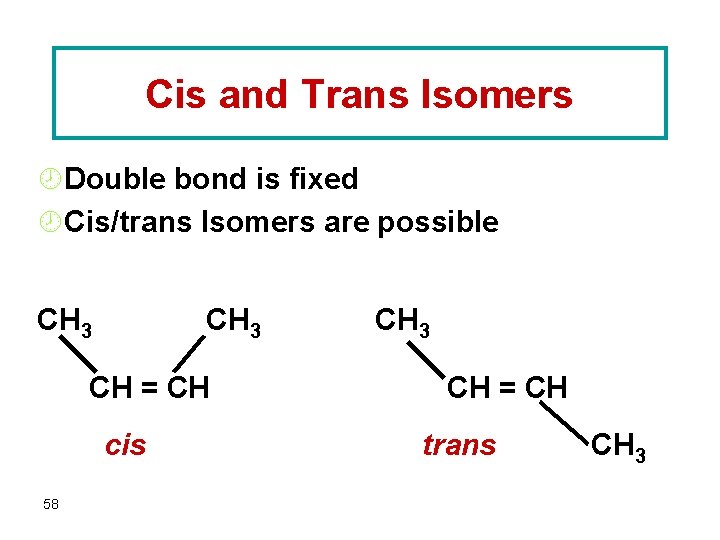
Cis and Trans Isomers ¾Double bond is fixed ¾Cis/trans Isomers are possible CH 3 CH = CH cis 58 CH 3 CH = CH trans CH 3

Addition Reactions • 59 Weaker bonds are broken and new (stronger) bonds are formed to atoms being added.
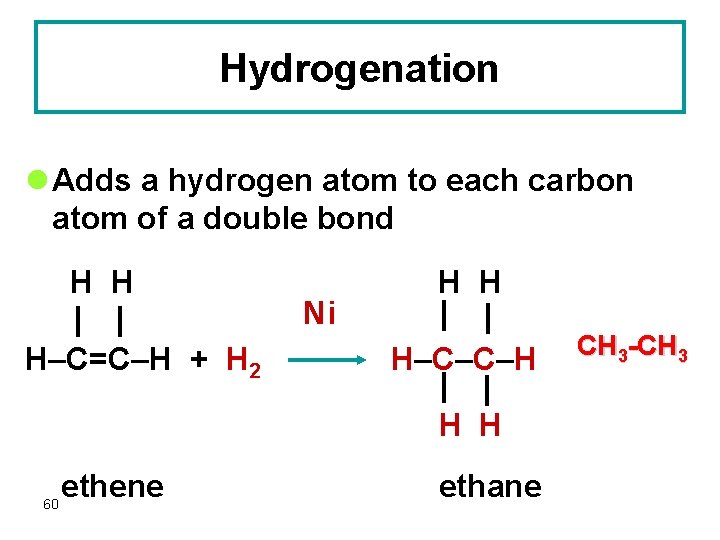
Hydrogenation l Adds a hydrogen atom to each carbon atom of a double bond H H H–C=C–H + H 2 Ni H H H–C–C–H H H 60 ethene ethane CH 3 -CH 3
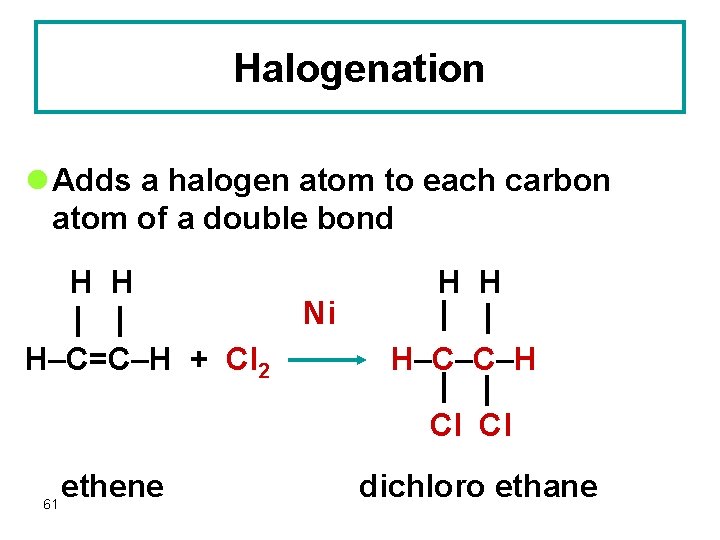
Halogenation l Adds a halogen atom to each carbon atom of a double bond H H H–C=C–H + Cl 2 Ni H H H–C–C–H Cl Cl 61 ethene dichloro ethane
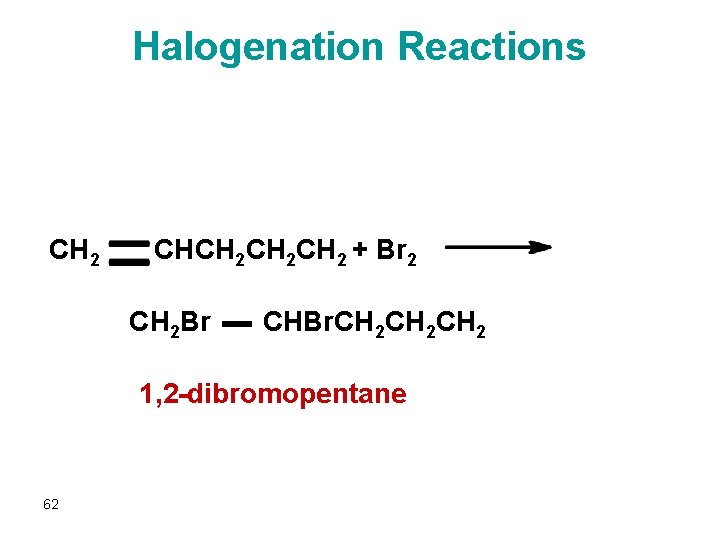
Halogenation Reactions CH 2 CHCH 2 CH 2 + Br 2 CH 2 Br CHBr. CH 2 CH 2 1, 2 -dibromopentane 62
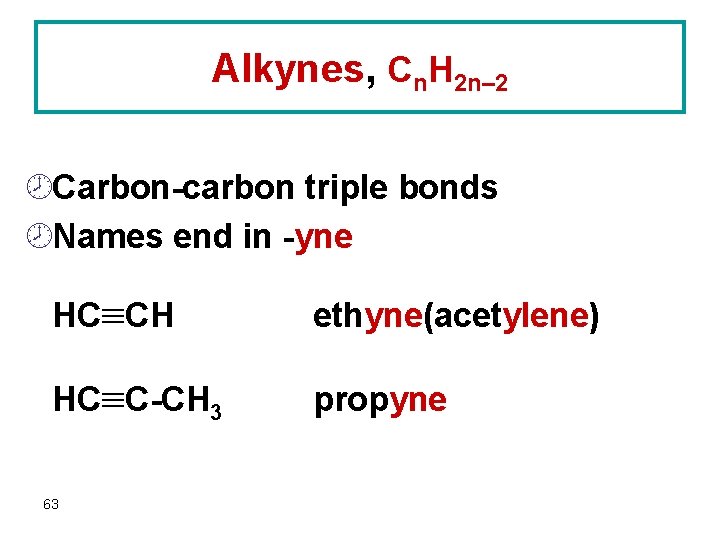
Alkynes, Cn. H 2 n– 2 ¾Carbon-carbon triple bonds ¾Names end in -yne HC CH ethyne(acetylene) HC C-CH 3 propyne 63
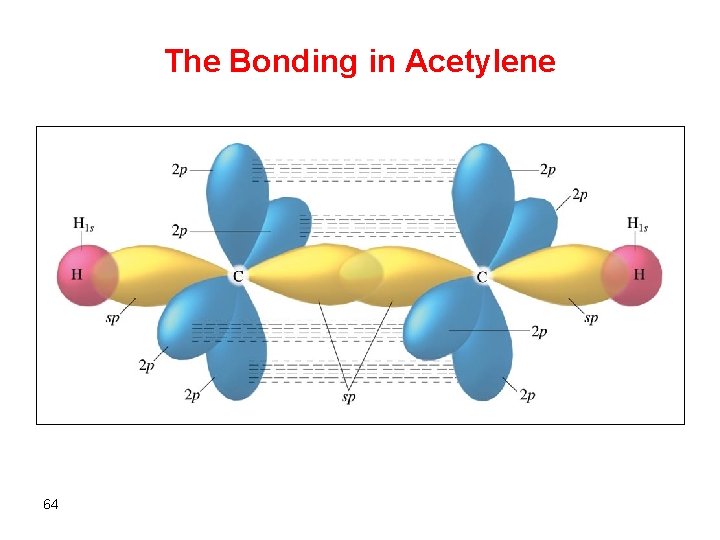
The Bonding in Acetylene 64

Naming Alkenes and Alkynes When the carbon chain has 4 or more C atoms, number the chain to give the lowest number to the double or triple bond. 1 2 3 4 CH 2=CHCH 2 CH 3 1 -butene CH 3 CH=CHCH 3 2 -butene CH 3 C CCH 3 65 2 -butyne
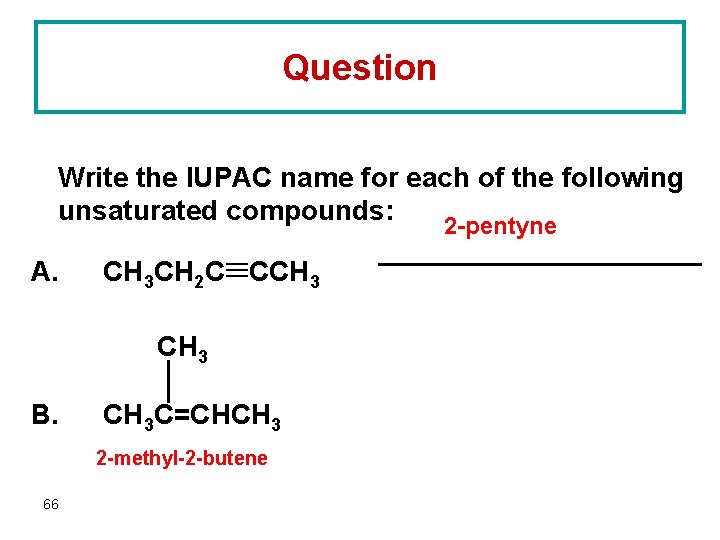
Question Write the IUPAC name for each of the following unsaturated compounds: 2 -pentyne A. CH 3 CH 2 C CCH 3 B. CH 3 C=CHCH 3 2 -methyl-2 -butene 66

Question • Name the following compound 67 5 -ethyl-3 -heptyne
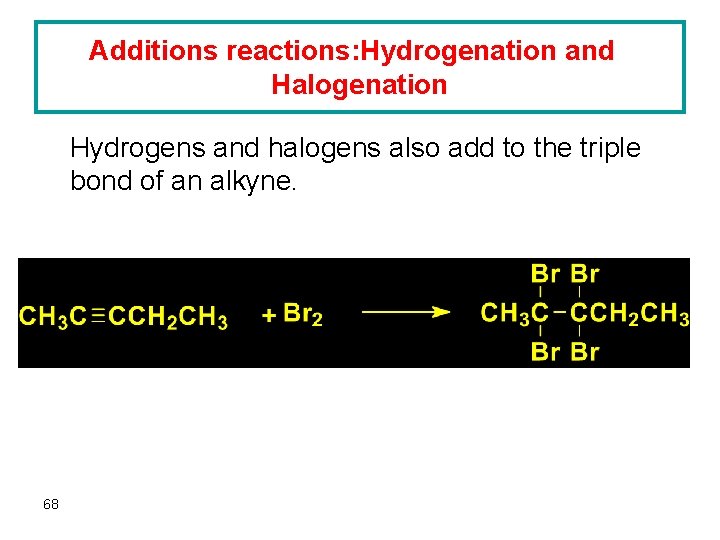
Additions reactions: Hydrogenation and Halogenation Hydrogens and halogens also add to the triple bond of an alkyne. 68

22. 3 Aromatic hydrocarbons Unsaturated Cyclic hydrocarbons • Alternating single/double bond cycles occur in many organic molecules – This class is called “aromatic” (by virtue of their aroma). – The structure is often preserved in their chemical reactions; they don’t add, they substitute instead. 69
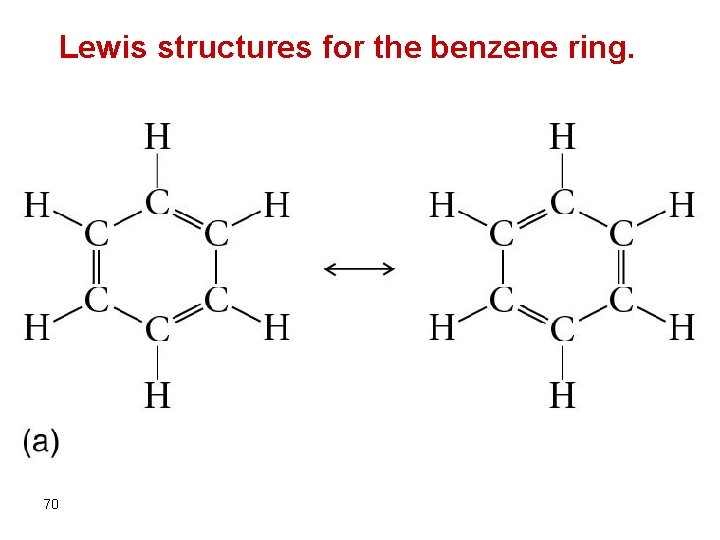
Lewis structures for the benzene ring. 70
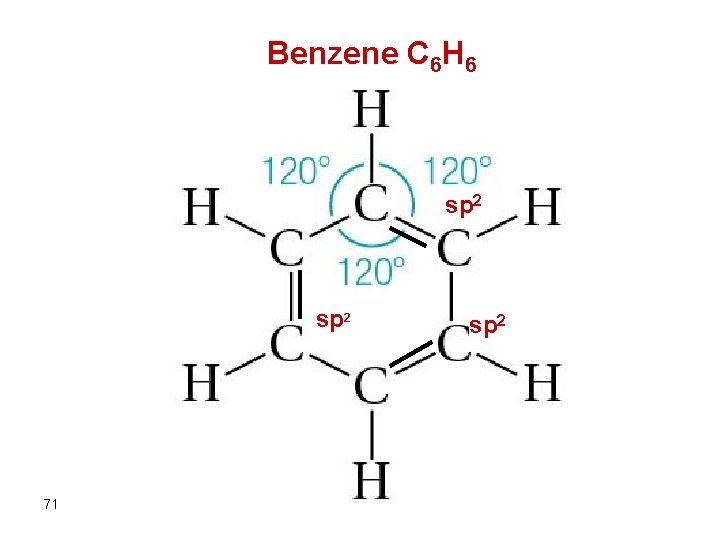
Benzene C 6 H 6 sp 2 71 sp 2
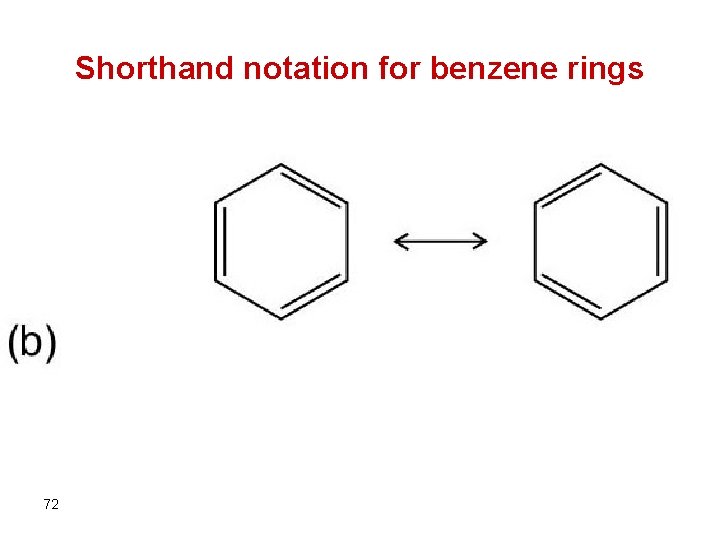
Shorthand notation for benzene rings 72
The bonding in the benzene ring is a combination of different Lewis structures. 73
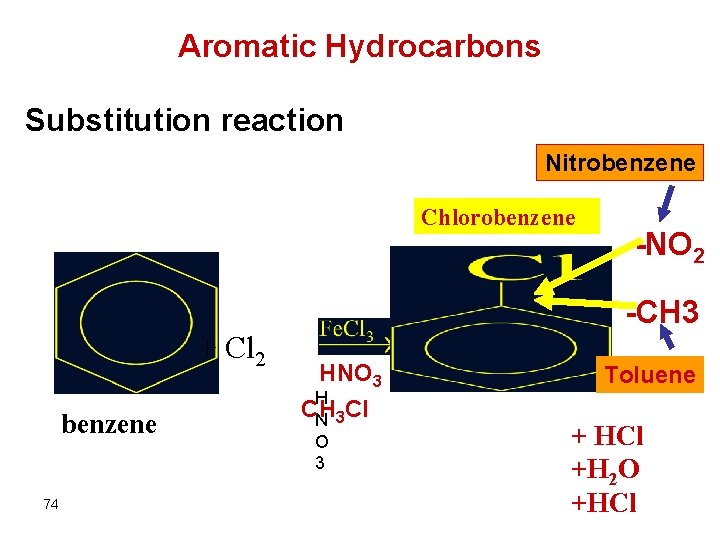
Aromatic Hydrocarbons Substitution reaction Nitrobenzene Chlorobenzene -NO 2 -CH 3 + Cl 2 benzene 74 HNO 3 H CH Cl N 3 O 3 Toluene + HCl +H 2 O +HCl
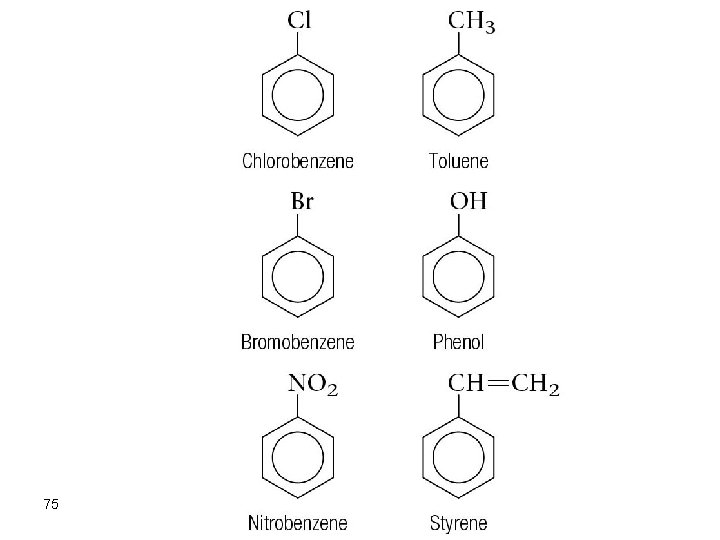
75
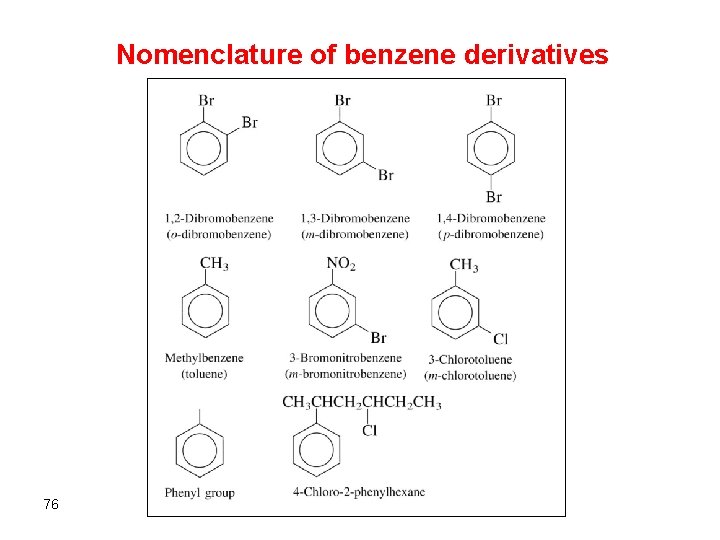
Nomenclature of benzene derivatives 76
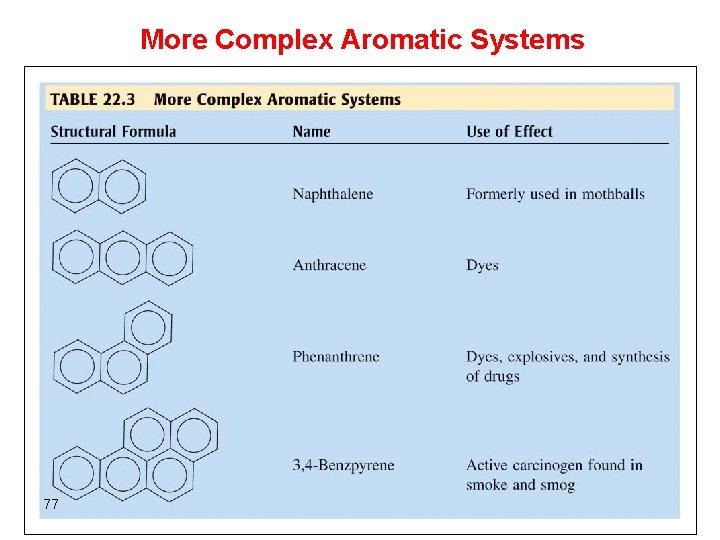
More Complex Aromatic Systems 77

22. 4 Hydrocarbon Derivatives (Functional Groups) § Molecules that are fundamentally § § § 78 hydrocarbons but have additional atoms or group of atoms called functional groups Part of an organic molecule where chemical reactions take place Replace an H in the corresponding alkane Provide a way to classify organic compounds

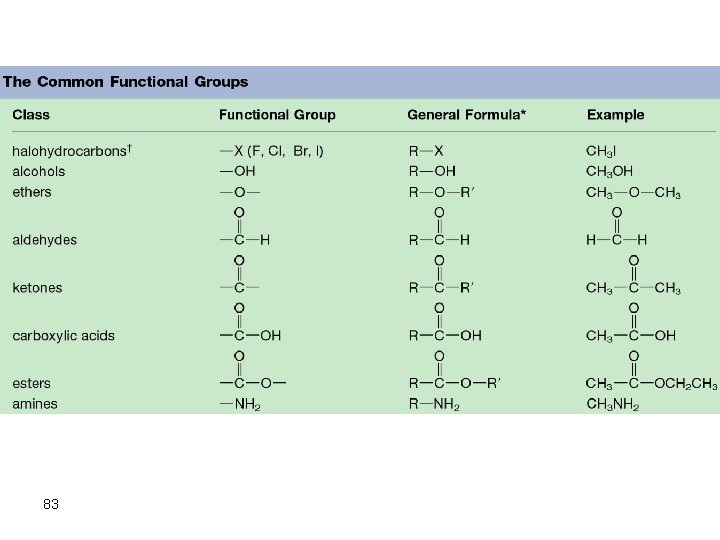
The Common Functional Groups Class Halohydrocarbons Alcohols Ethers Aldehydes 79 General Formula R X R OH R O R
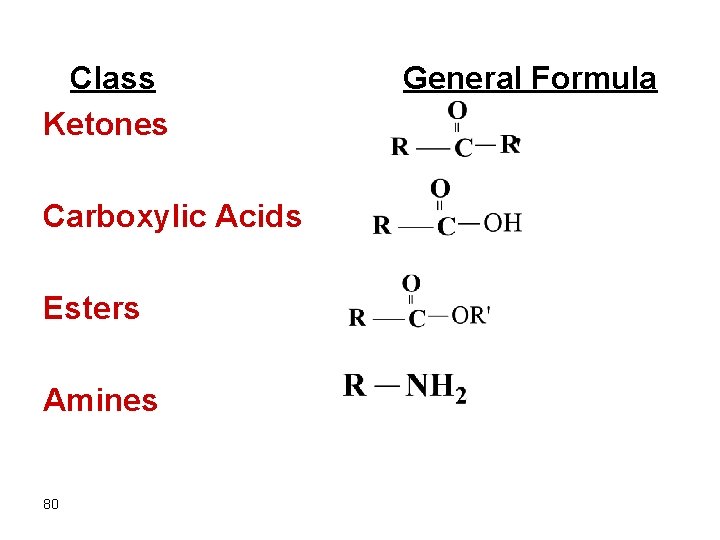
Class Ketones Carboxylic Acids Esters Amines 80 General Formula
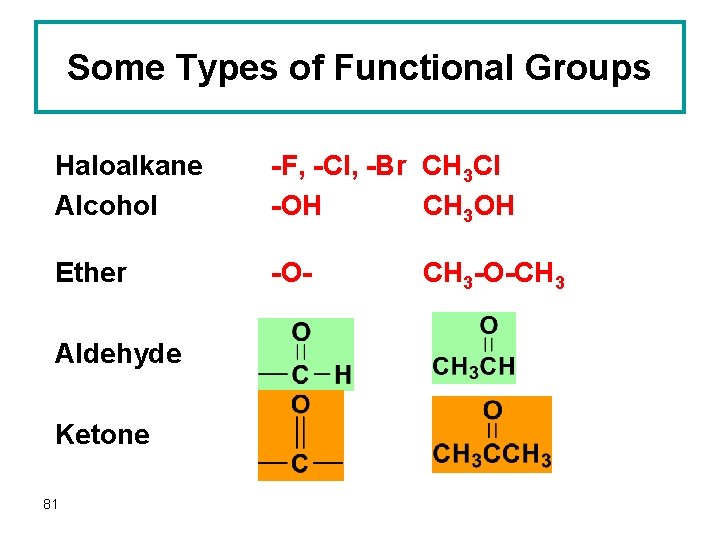
Some Types of Functional Groups Haloalkane Alcohol -F, -Cl, -Br CH 3 Cl -OH CH 3 OH Ether -O- Aldehyde Ketone 81 CH 3 -O-CH 3
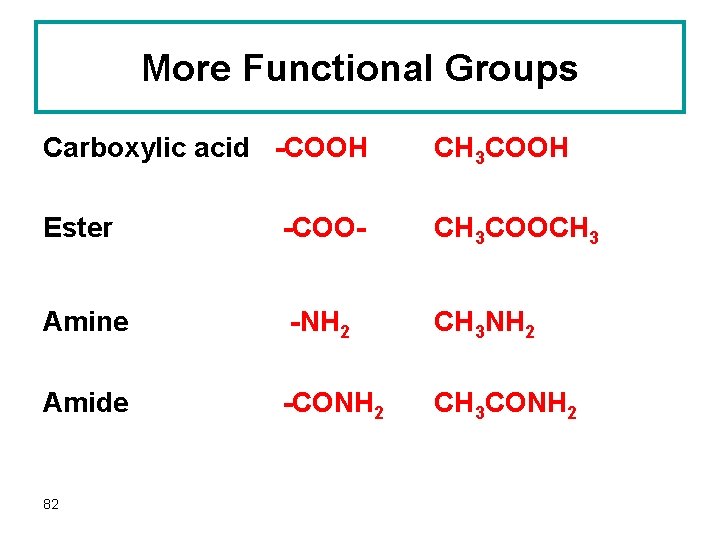
More Functional Groups Carboxylic acid -COOH CH 3 COOH Ester -COO- CH 3 COOCH 3 Amine -NH 2 CH 3 NH 2 Amide -CONH 2 CH 3 CONH 2 82
83
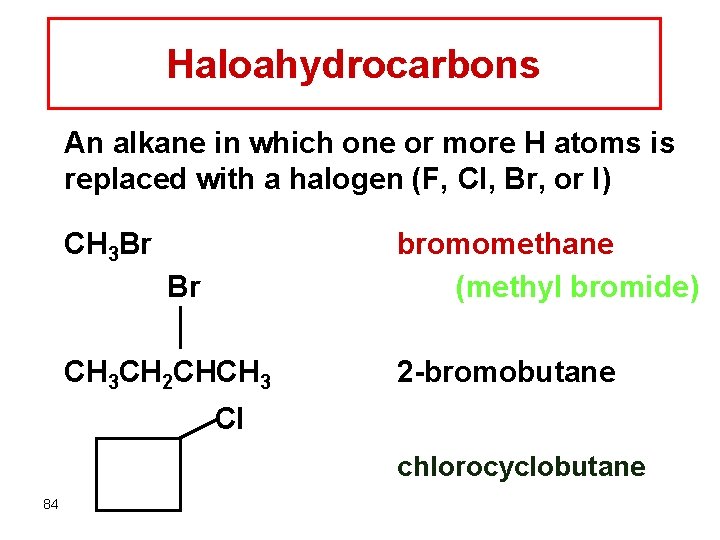
Haloahydrocarbons An alkane in which one or more H atoms is replaced with a halogen (F, Cl, Br, or I) CH 3 Br bromomethane (methyl bromide) Br CH 3 CH 2 CHCH 3 2 -bromobutane Cl chlorocyclobutane 84
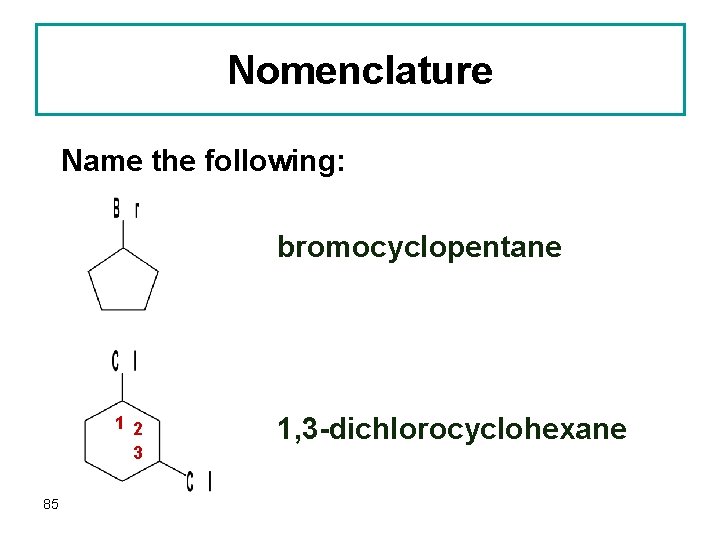
Nomenclature Name the following: bromocyclopentane 1 2 3 85 1, 3 -dichlorocyclohexane
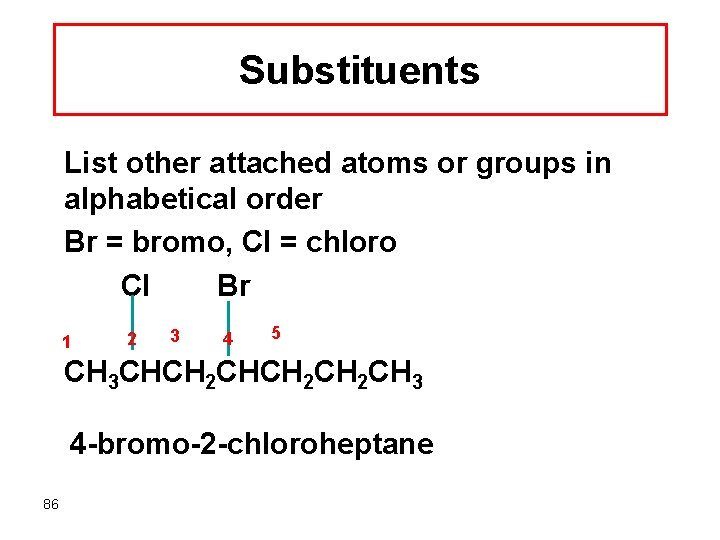
Substituents List other attached atoms or groups in alphabetical order Br = bromo, Cl = chloro Cl Br 1 2 3 4 5 CH 3 CHCH 2 CH 2 CH 3 4 -bromo-2 -chloroheptane 86
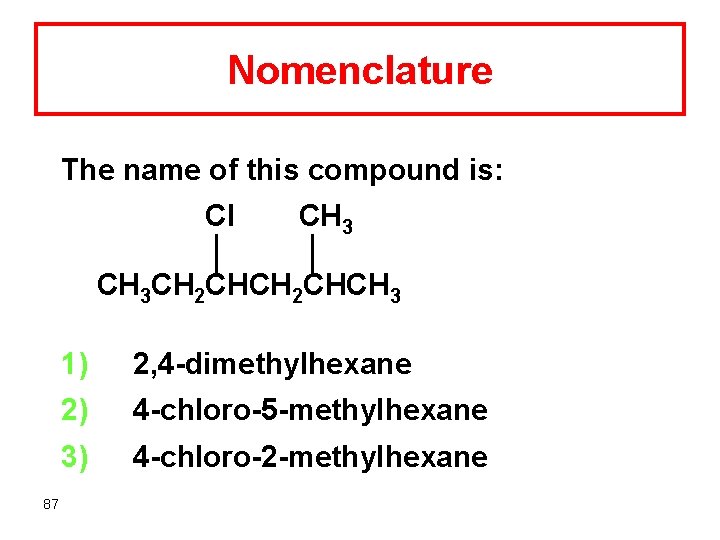
Nomenclature The name of this compound is: Cl CH 3 CH 2 CHCH 3 87 1) 2, 4 -dimethylhexane 2) 4 -chloro-5 -methylhexane 3) 4 -chloro-2 -methylhexane
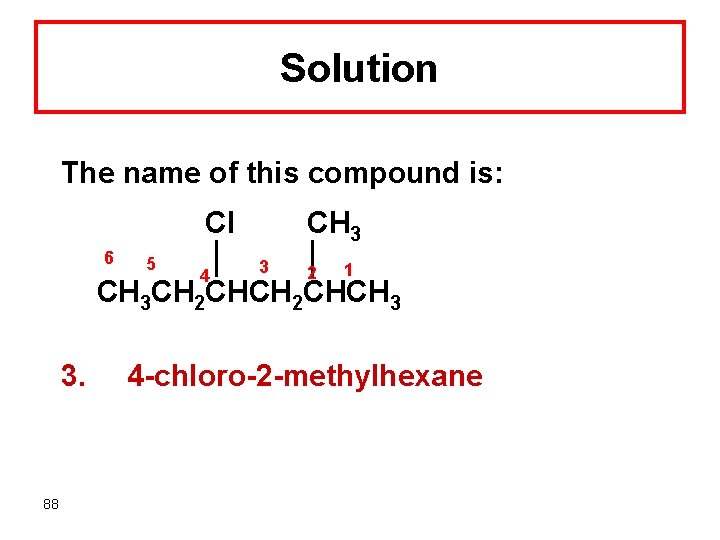
Solution The name of this compound is: Cl 6 5 4 CH 3 3 2 1 CH 3 CH 2 CHCH 3 3. 88 4 -chloro-2 -methylhexane

Alcohols: R–OH • The –OH makes alcohol polar enough to hydrogen bond • Thus, they are water soluble • Ethanol is a fermentation product acid. C 6 H 12 O 6 Glucose • 2 CH 3 CH 2 OH Ethanol + 2 CO 2 Methanol is produced industrially by hydrogenation of carbon monoxide CO + 2 H 2 O 89 yeast CH 3 OH Methanol

Uses of alcohols • Methanol is used to synthesize adhesives, fibers, plastics and recently as motor fuel • It is toxic to human and can lead to blindness and death • Ethanol can be added to gasoline to form gasohol and used in industry as solvent • Commercial production of ethanol: CH 2=CH 2 + H 2 O CH 3 CH 2 OH 90

Classes of alcohols Alcohols can be classified according to the number of hydrocarbon fragments bonded to the carbon where the –OH group is attached 91
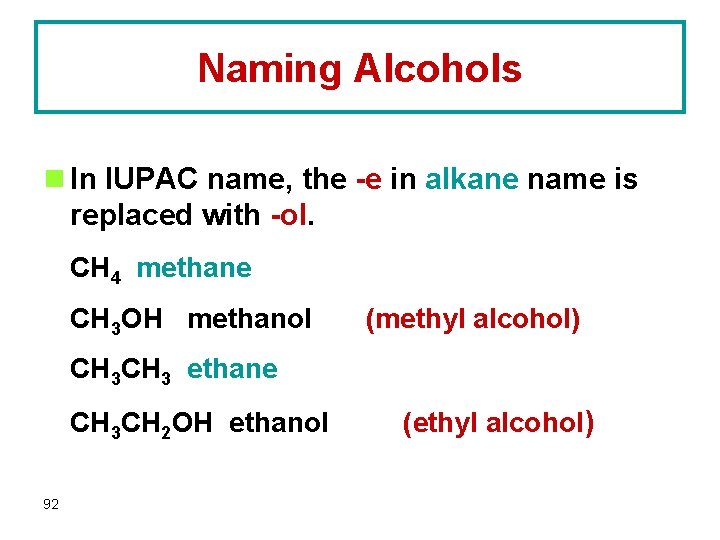
Naming Alcohols n In IUPAC name, the -e in alkane name is replaced with -ol. CH 4 methane CH 3 OH methanol (methyl alcohol) CH 3 ethane CH 3 CH 2 OH ethanol 92 (ethyl alcohol)
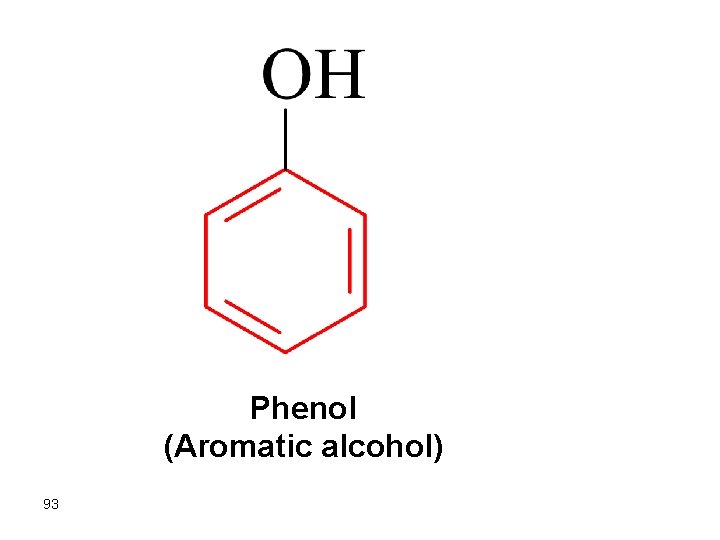
Phenol (Aromatic alcohol) 93
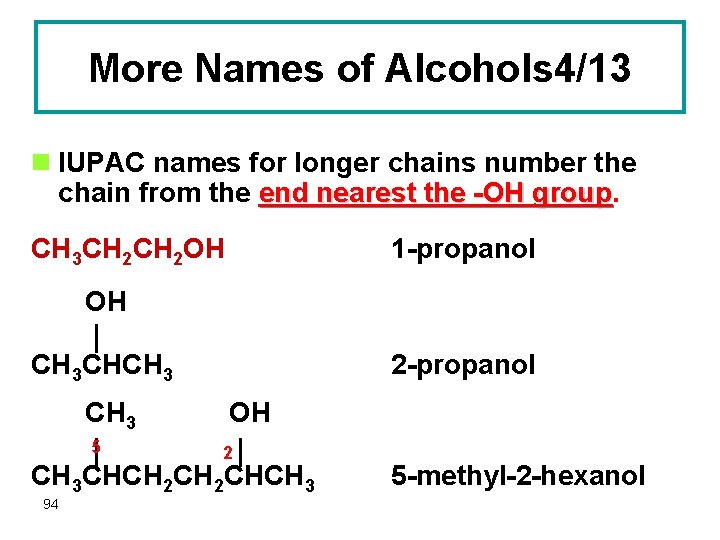
More Names of Alcohols 4/13 n IUPAC names for longer chains number the chain from the end nearest the -OH group CH 3 CH 2 OH 1 -propanol OH CH 3 CHCH 3 5 2 -propanol OH 2 CH 3 CHCH 2 CHCH 3 94 5 -methyl-2 -hexanol
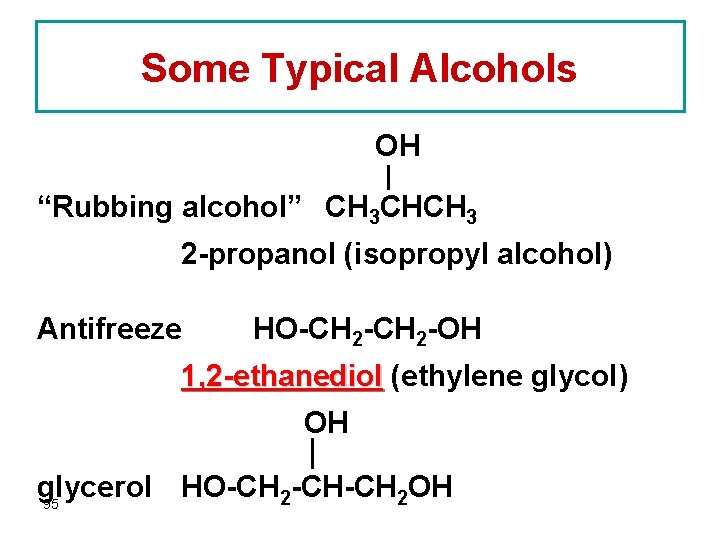
Some Typical Alcohols OH “Rubbing alcohol” CH 3 CHCH 3 2 -propanol (isopropyl alcohol) Antifreeze HO-CH 2 -OH 1, 2 -ethanediol (ethylene glycol) OH glycerol HO-CH 2 -CH-CH 2 OH 95
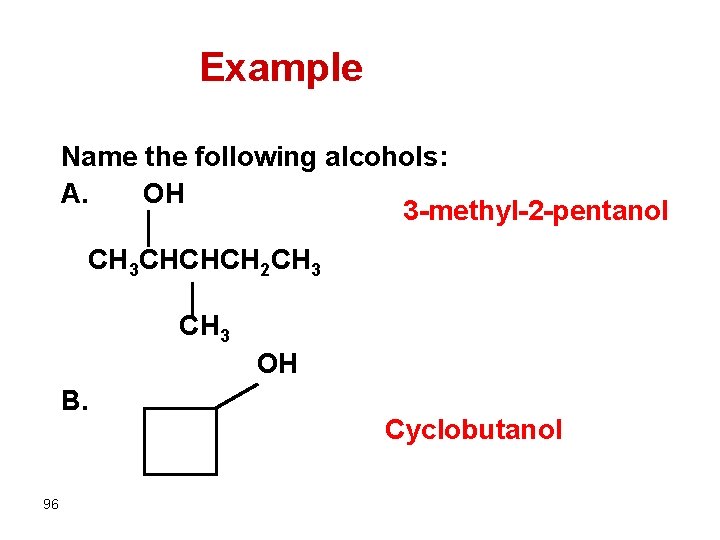
Example Name the following alcohols: A. OH 3 -methyl-2 -pentanol CH 3 CHCHCH 2 CH 3 OH B. 96 Cyclobutanol
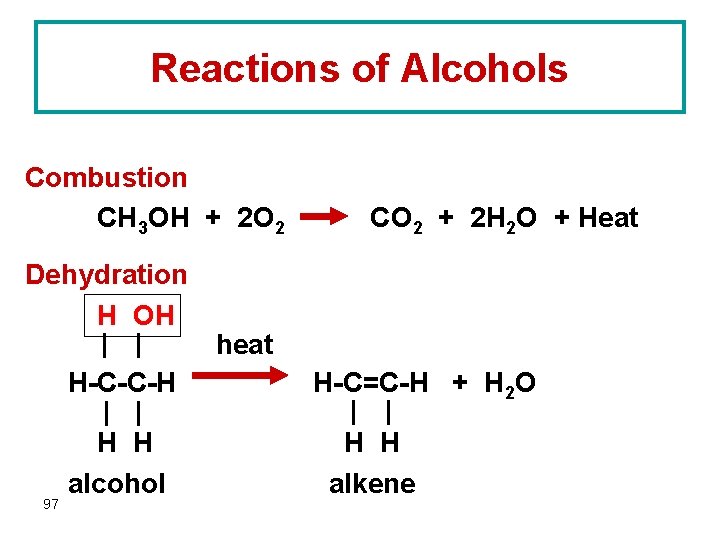
Reactions of Alcohols Combustion CH 3 OH + 2 O 2 Dehydration H OH H-C-C-H 97 H H alcohol CO 2 + 2 H 2 O + Heat heat H-C=C-H + H 2 O H H alkene

Ethers • Contain an -O- between two carbon groups • Simple ethers named from -yl names of the attached groups and adding ether CH 3 -O-CH 3 dimethyl ether CH 3 -O-CH 2 CH 3 ethyl methyl ether 98
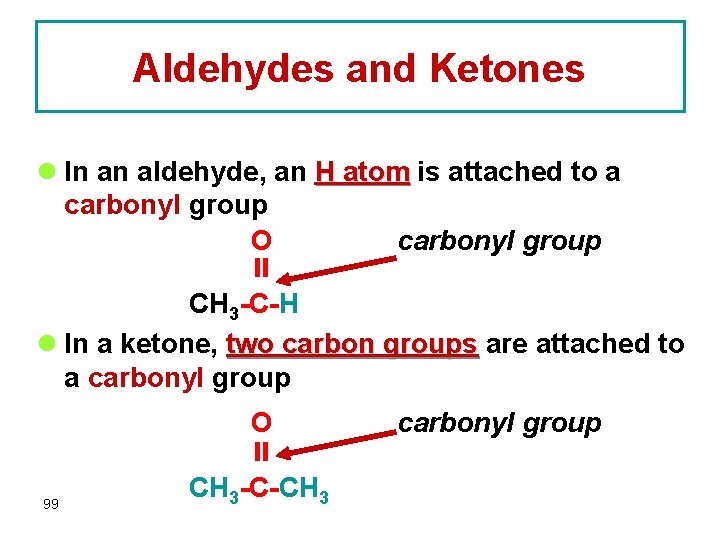
Aldehydes and Ketones l In an aldehyde, an H atom is attached to a carbonyl group O carbonyl group CH 3 -C-H l In a ketone, two carbon groups are attached to a carbonyl group 99 O CH 3 -C-CH 3 carbonyl group
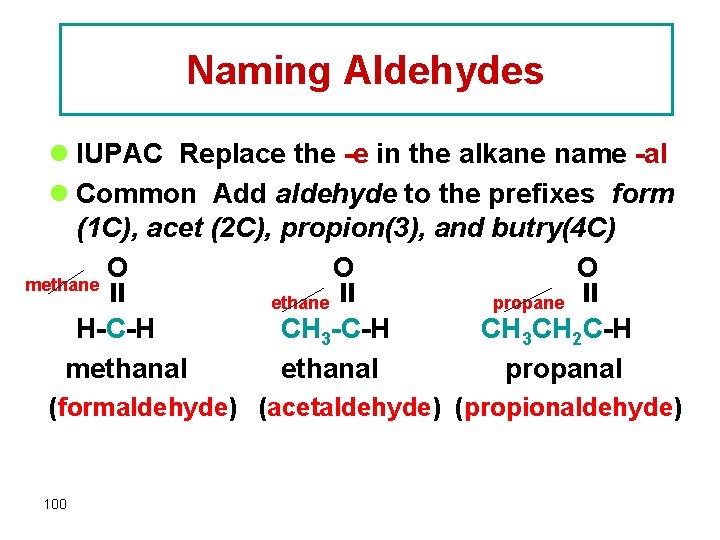
Naming Aldehydes l IUPAC Replace the -e in the alkane name -al l Common Add aldehyde to the prefixes form (1 C), acet (2 C), propion(3), and butry(4 C) O O O methane propane H-C-H CH 3 CH 2 C-H methanal propanal (formaldehyde) (acetaldehyde) (propionaldehyde) 100
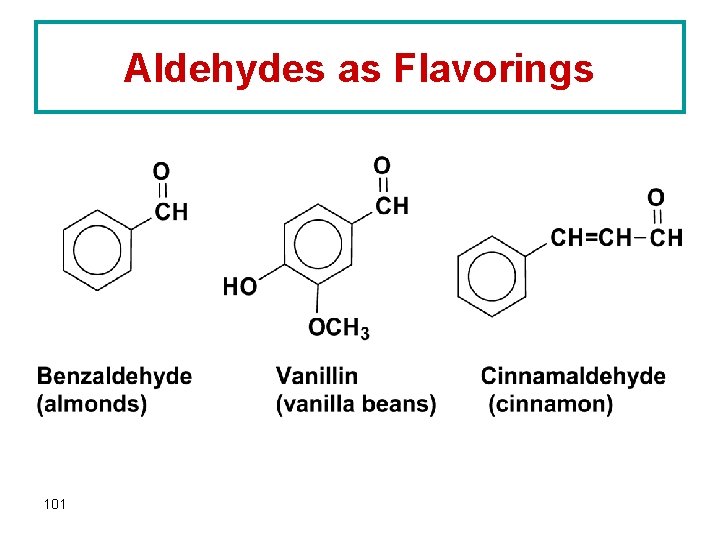
Aldehydes as Flavorings 101
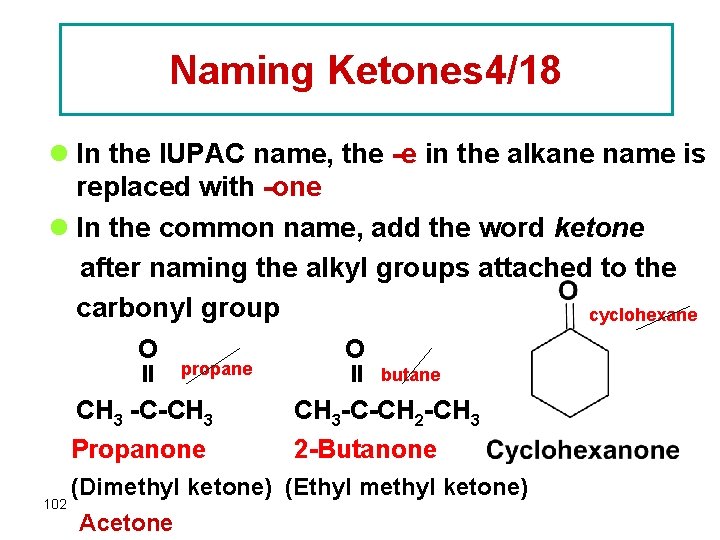
Naming Ketones 4/18 l In the IUPAC name, the -e in the alkane name is replaced with -one l In the common name, add the word ketone after naming the alkyl groups attached to the carbonyl group cyclohexane O propane CH 3 -C-CH 3 Propanone O butane CH 3 -C-CH 2 -CH 3 2 -Butanone (Dimethyl ketone) (Ethyl methyl ketone) 102 Acetone
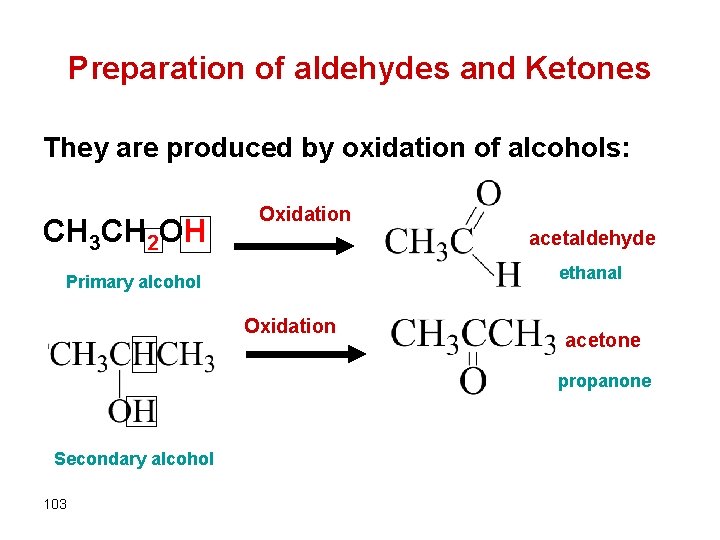
Preparation of aldehydes and Ketones They are produced by oxidation of alcohols: CH 3 CH 2 OH Oxidation acetaldehyde ethanal Primary alcohol Oxidation acetone propanone Secondary alcohol 103
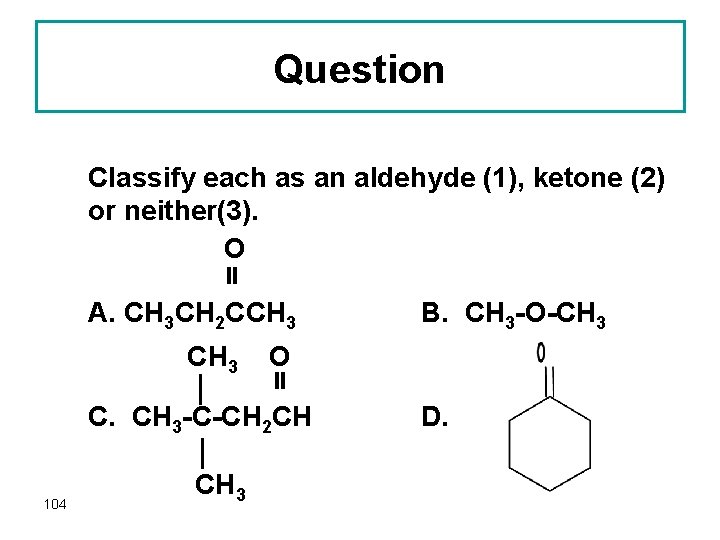
Question Classify each as an aldehyde (1), ketone (2) or neither(3). O A. CH 3 CH 2 CCH 3 O C. CH 3 -C-CH 2 CH 104 CH 3 B. CH 3 -O-CH 3 D.

Solution Classify each as an aldehyde (1), ketone (2) or neither(3). O A. CH 3 CH 2 CCH 3 2 O C. CH 3 -C-CH 2 CH 1 105 CH 3 B. CH 3 -O-CH 3 3 D. 2
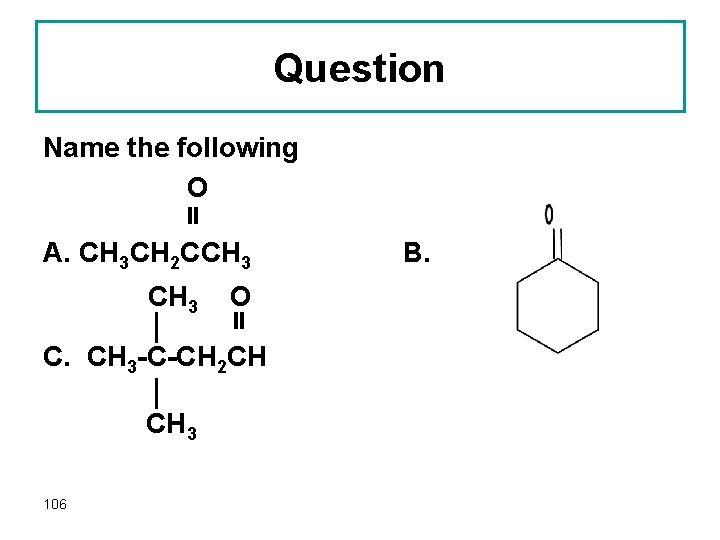
Question Name the following O A. CH 3 CH 2 CCH 3 O C. CH 3 -C-CH 2 CH CH 3 106 B.
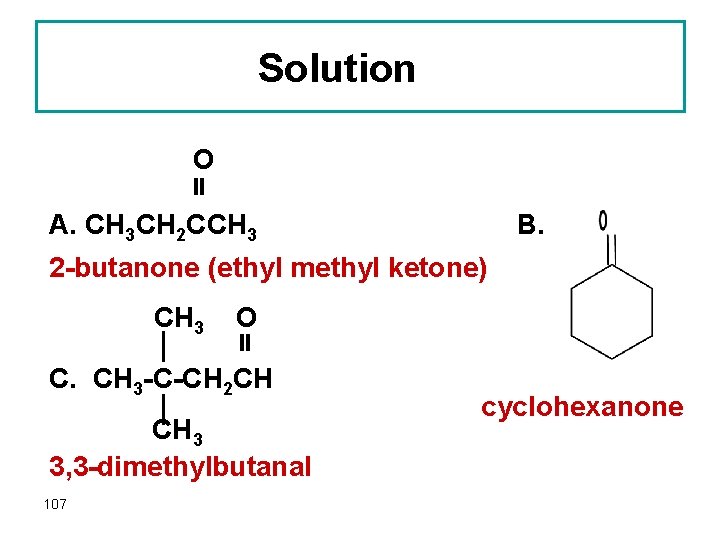
Solution O A. CH 3 CH 2 CCH 3 B. 2 -butanone (ethyl methyl ketone) CH 3 O C. CH 3 -C-CH 2 CH CH 3 3, 3 -dimethylbutanal 107 cyclohexanone

Question Draw the structural formulas for each: A. 3 -Methylpentanal B. 2, 3 -Dichloropropanal C. 3 -Methyl-2 -butanone 108
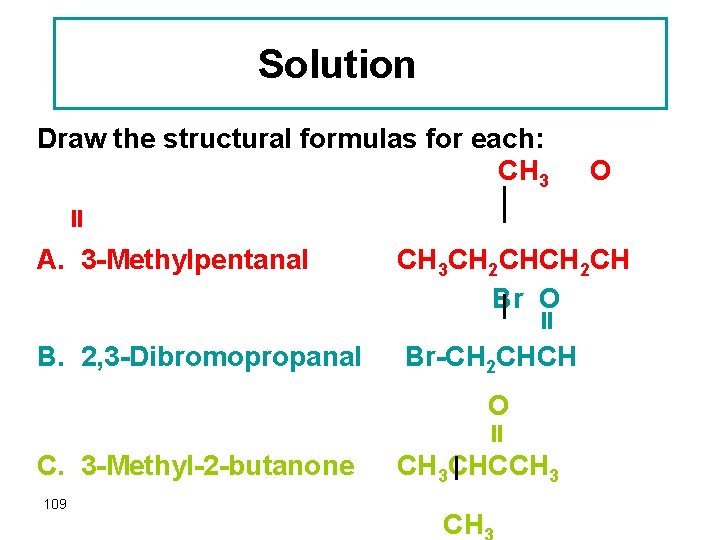
Solution Draw the structural formulas for each: CH 3 O A. 3 -Methylpentanal CH 3 CH 2 CH Br O B. 2, 3 -Dibromopropanal Br-CH 2 CHCH O C. 3 -Methyl-2 -butanone 109 CH 3 CHCCH 3 CH
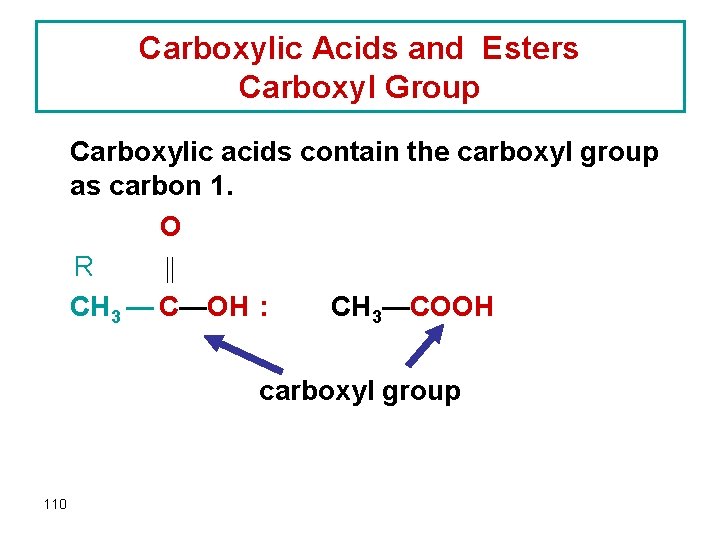
Carboxylic Acids and Esters Carboxyl Group Carboxylic acids contain the carboxyl group as carbon 1. O R CH 3 — C—OH : CH 3—COOH carboxyl group 110
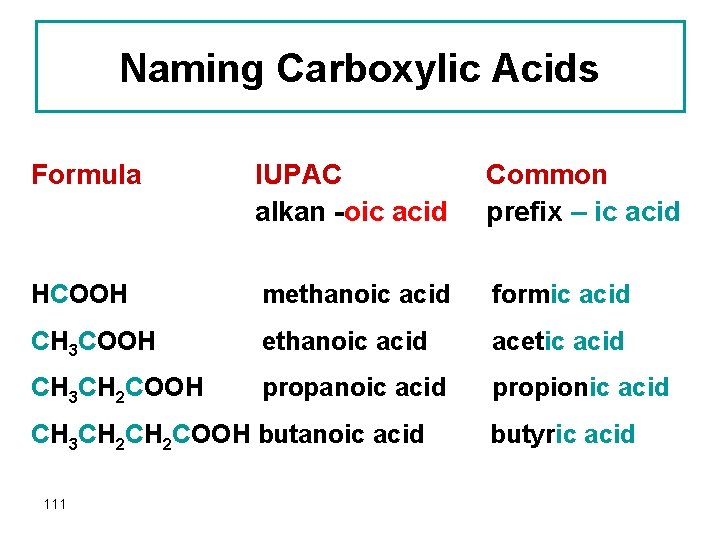
Naming Carboxylic Acids Formula IUPAC alkan -oic acid Common prefix – ic acid HCOOH methanoic acid formic acid CH 3 COOH ethanoic acid acetic acid CH 3 CH 2 COOH propanoic acid propionic acid CH 3 CH 2 COOH butanoic acid 111 butyric acid
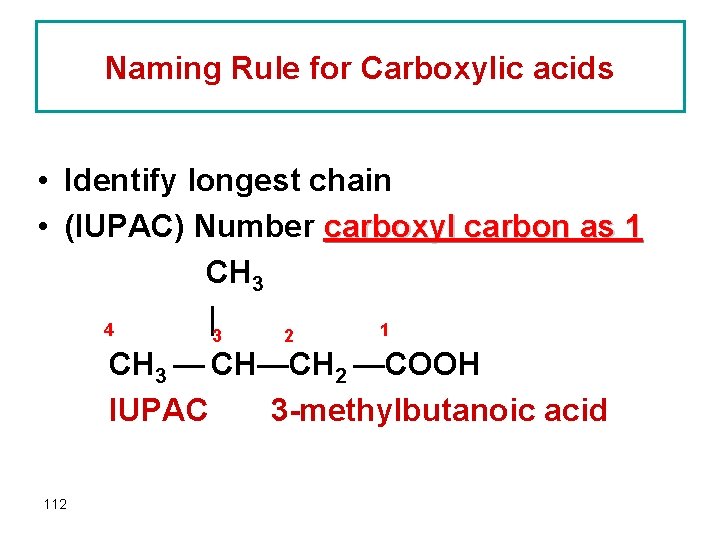
Naming Rule for Carboxylic acids • Identify longest chain • (IUPAC) Number carboxyl carbon as 1 CH 3 |3 4 1 2 CH 3 — CH—CH 2 —COOH IUPAC 3 -methylbutanoic acid 112
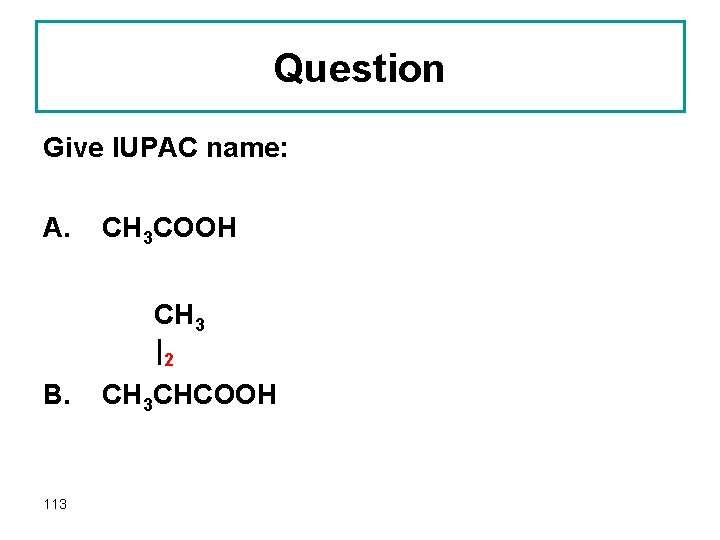
Question Give IUPAC name: A. CH 3 COOH CH 3 |2 B. 113 CH 3 CHCOOH

Solution A. CH 3 COOH ethanoic acid; acetic acid CH 3 | B. 114 CH 3 CHCOOH 2 -methylpropanoic acid;
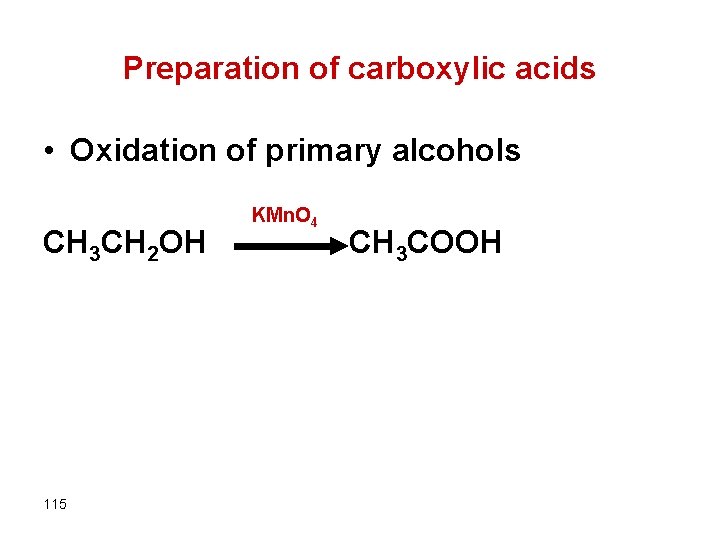
Preparation of carboxylic acids • Oxidation of primary alcohols CH 3 CH 2 OH 115 KMn. O 4 CH 3 COOH
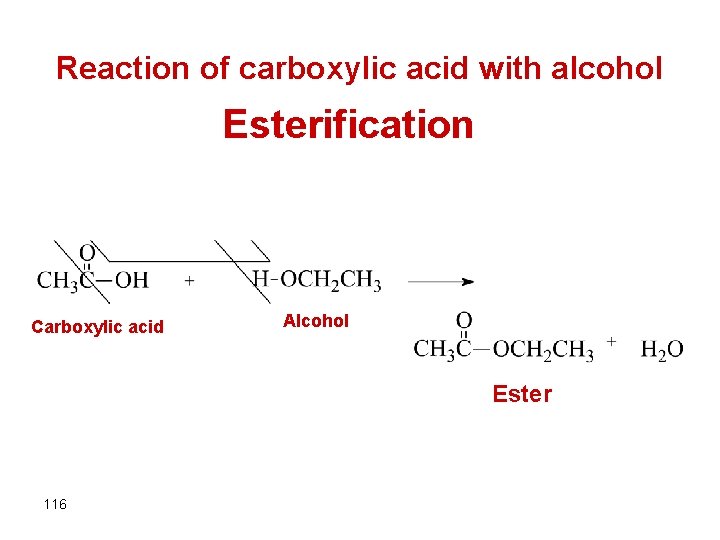
Reaction of carboxylic acid with alcohol Esterification Carboxylic acid Alcohol Ester 116
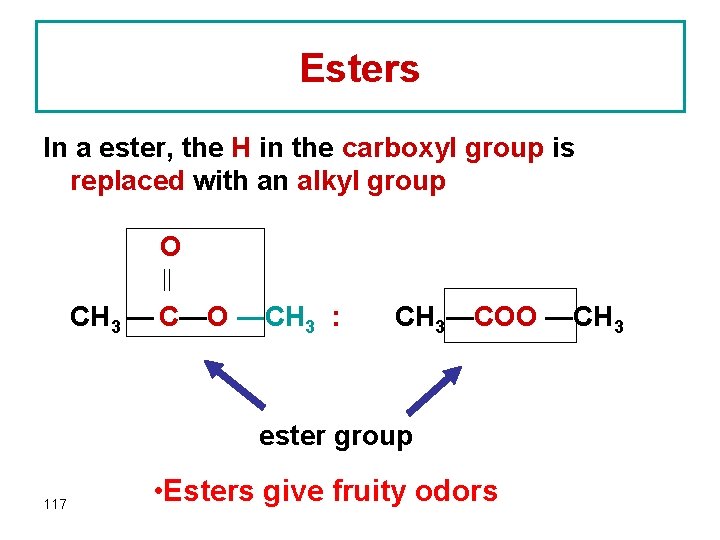
Esters In a ester, the H in the carboxyl group is replaced with an alkyl group O CH 3 — C—O —CH 3 : CH 3—COO —CH 3 ester group 117 • Esters give fruity odors
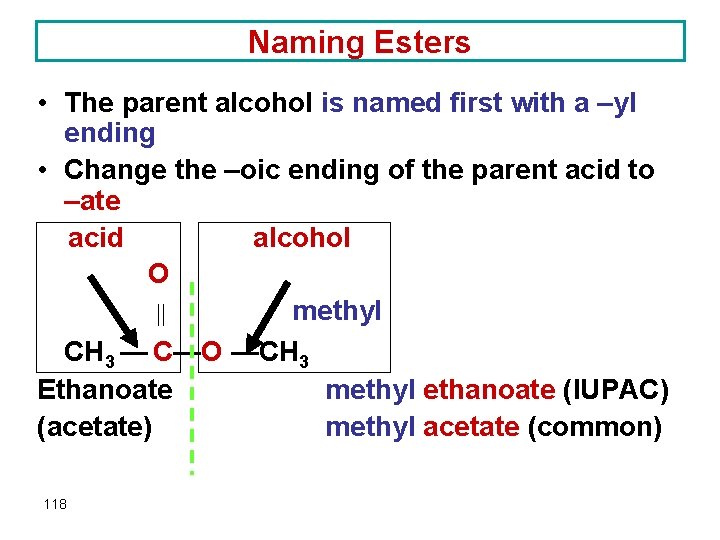
Naming Esters • The parent alcohol is named first with a –yl ending • Change the –oic ending of the parent acid to –ate acid alcohol O methyl CH 3 — C—O —CH 3 Ethanoate methyl ethanoate (IUPAC) (acetate) methyl acetate (common) 118

Some esters and their names Flavor/Odor Raspberries HCOOCH 2 CH 3 ethylmethanoate (IUPAC) ethylformate (common) Pineapples CH 3 CH 2 COOCH 2 CH 3 ethylbutanoate (IUPAC) ethylbutyrate (common) 119

Question Give the IUPAC and common names of the following compound, which is responsible for the flavor and odor of pears. O CH 3 — C— O —CH 2 CH 3 120
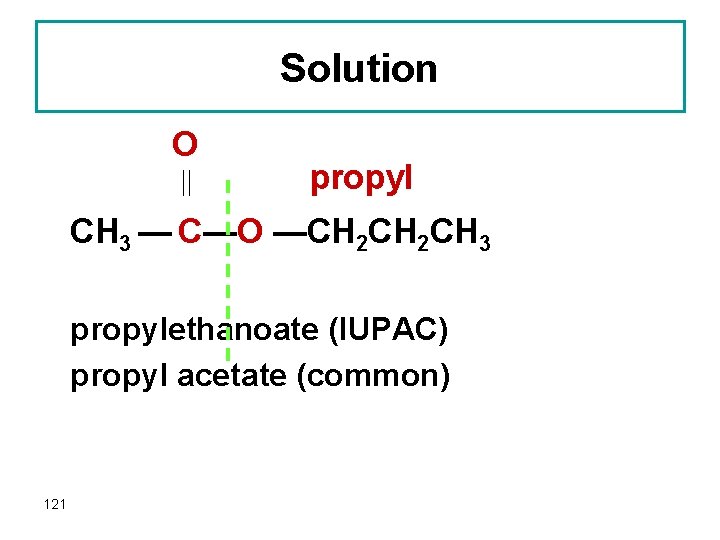
Solution O propyl CH 3 — C—O —CH 2 CH 3 propylethanoate (IUPAC) propyl acetate (common) 121

Question Draw the structure of the following compounds: A. 3 -bromobutanoic acid B. Ethyl propionoate 122
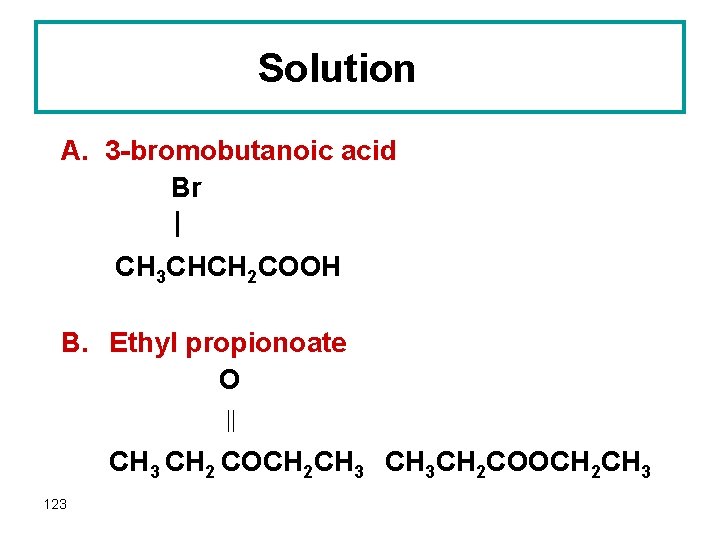
Solution A. 3 -bromobutanoic acid Br | CH 3 CHCH 2 COOH B. Ethyl propionoate O CH 3 CH 2 COCH 2 CH 3 CH 2 COOCH 2 CH 3 123
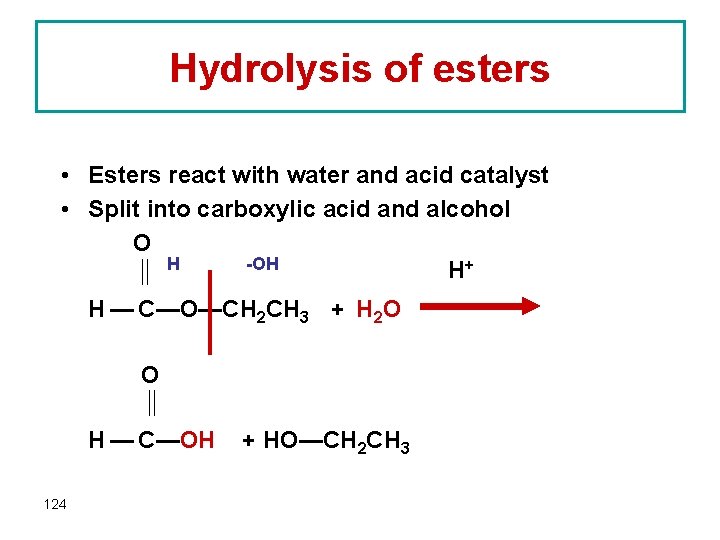
Hydrolysis of esters • Esters react with water and acid catalyst • Split into carboxylic acid and alcohol O -OH H H+ H — C—O—CH 2 CH 3 + H 2 O O H — C—OH 124 + HO—CH 2 CH 3
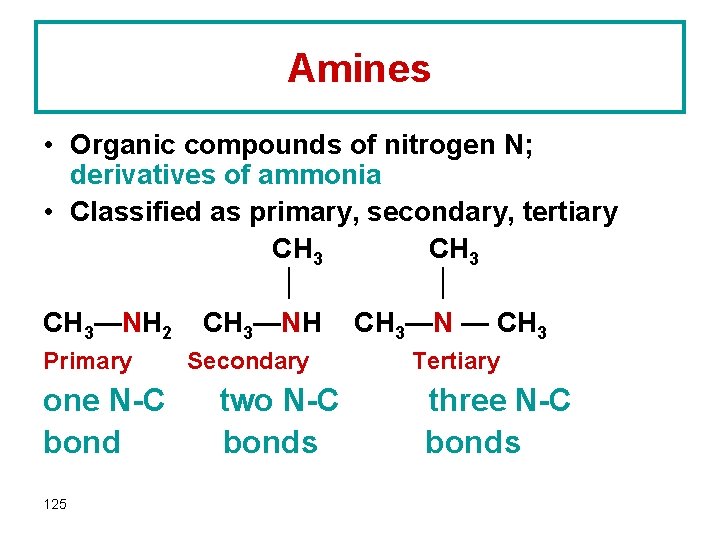
Amines • Organic compounds of nitrogen N; derivatives of ammonia • Classified as primary, secondary, tertiary CH 3—NH 2 CH 3—NH CH 3—N — CH 3 Primary one N-C bond 125 Secondary two N-C bonds Tertiary three N-C bonds
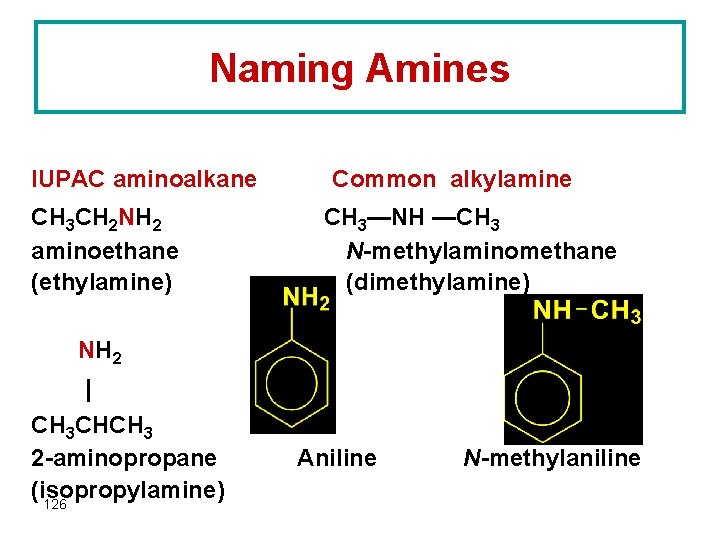
Naming Amines IUPAC aminoalkane CH 3 CH 2 NH 2 aminoethane (ethylamine) Common alkylamine CH 3—NH —CH 3 N-methylaminomethane (dimethylamine) NH 2 | CH 3 CHCH 3 2 -aminopropane (isopropylamine) 126 Aniline N-methylaniline
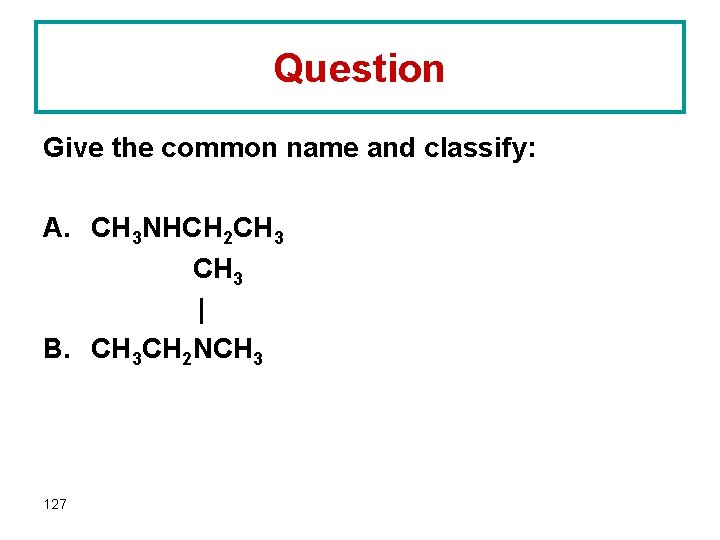
Question Give the common name and classify: A. CH 3 NHCH 2 CH 3 | B. CH 3 CH 2 NCH 3 127
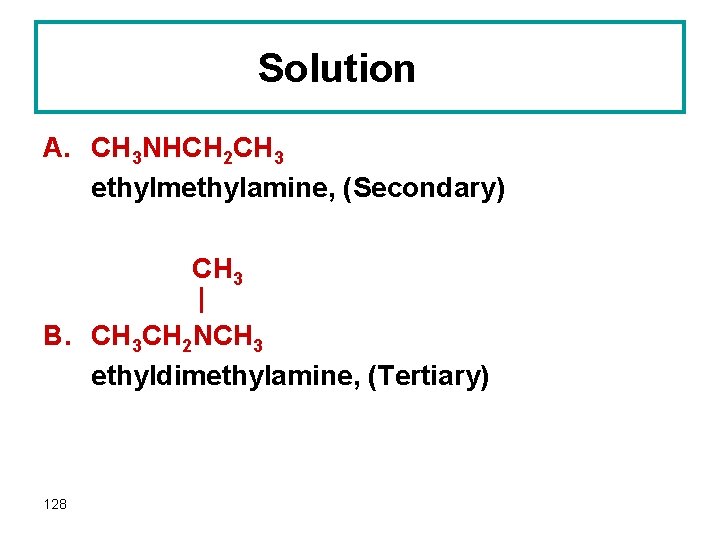
Solution A. CH 3 NHCH 2 CH 3 ethylmethylamine, (Secondary) CH 3 | B. CH 3 CH 2 NCH 3 ethyldimethylamine, (Tertiary) 128

Question Write a structural formula for A. 2 -aminopentane B. 1, 3 -diaminocyclohexane 129
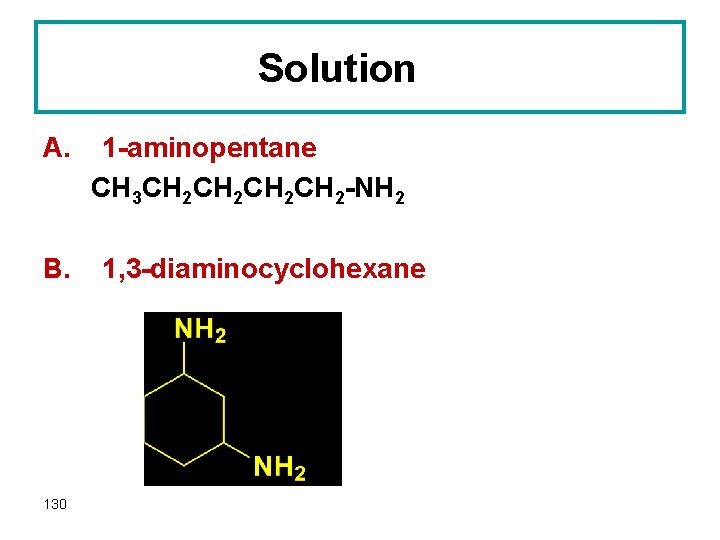
Solution A. B. 130 1 -aminopentane CH 3 CH 2 CH 2 -NH 2 1, 3 -diaminocyclohexane

Polymers Poly= many; mers=parts • Polymers are large, usually chainlike molecules that are built from small molecules called monomers joined by covalent bonds Monomer Polymer Ethylene Polyethylene Vinyl chloride Polyvinyl chloride Tetrafluoroethylene Teflon 131

132

Some common synthetic polymers, their monomers and applications 133

Types of Polymerization Addition Polymerization: monomers “add together” to form the polymer, with no other products. (Teflon) Condensation Polymerization: A small molecule, such as water, is formed for each extension of the polymer chain. (Nylon) 134
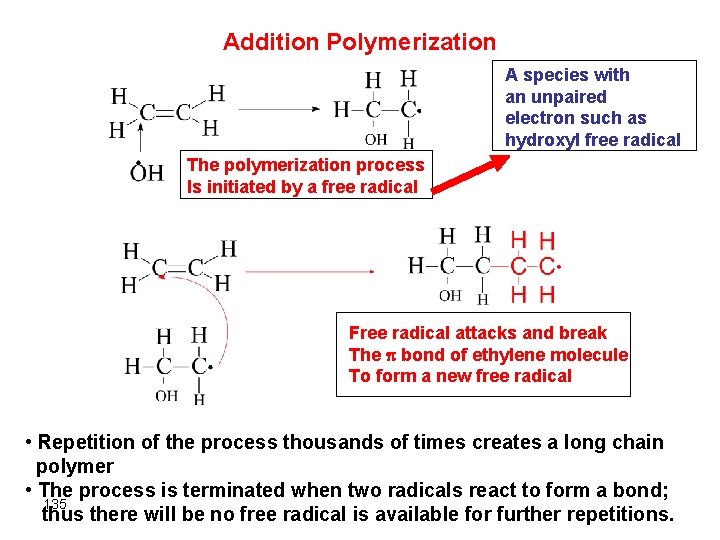
Addition Polymerization A species with an unpaired electron such as hydroxyl free radical The polymerization process Is initiated by a free radical Free radical attacks and break The bond of ethylene molecule To form a new free radical • Repetition of the process thousands of times creates a long chain polymer • The process is terminated when two radicals react to form a bond; 135 thus there will be no free radical is available for further repetitions.

another (Polythene) • Depending upon conditions of polymerization, the product may be branched or linear polyethylene 136
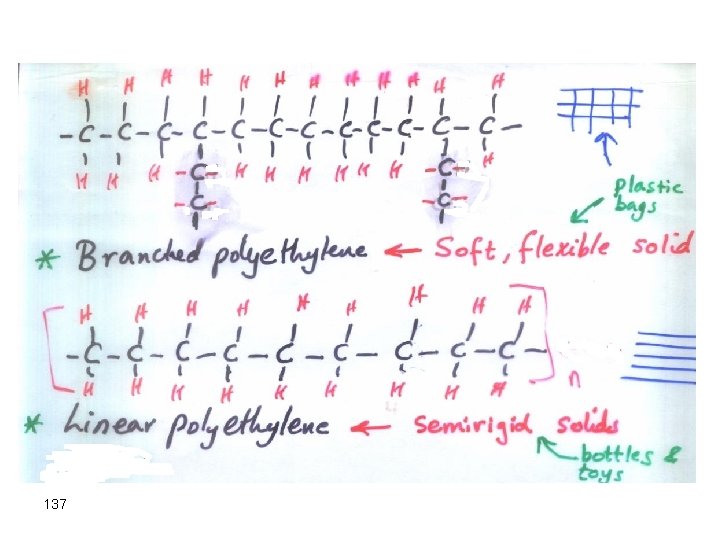
137
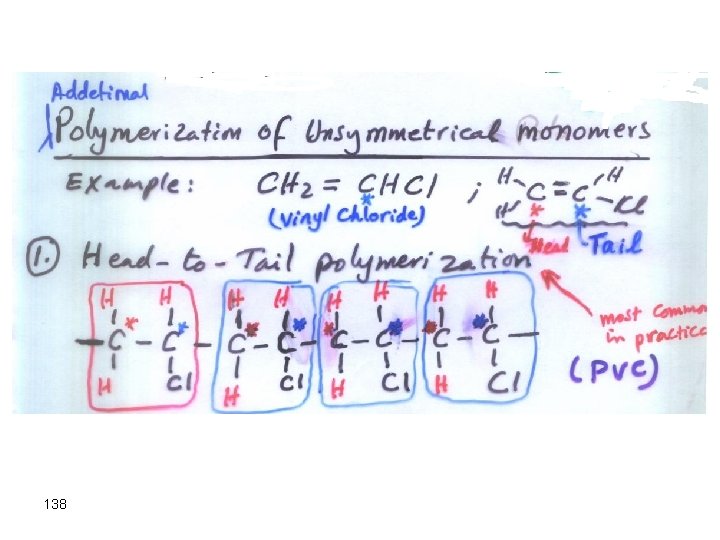
138
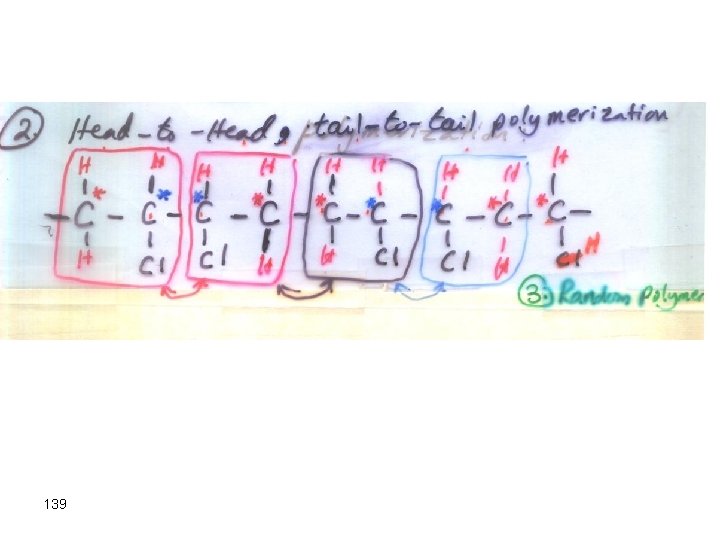
139
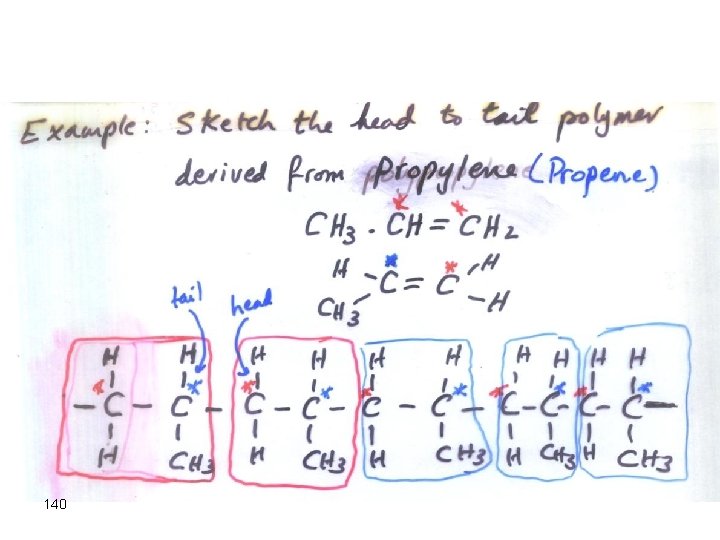
140
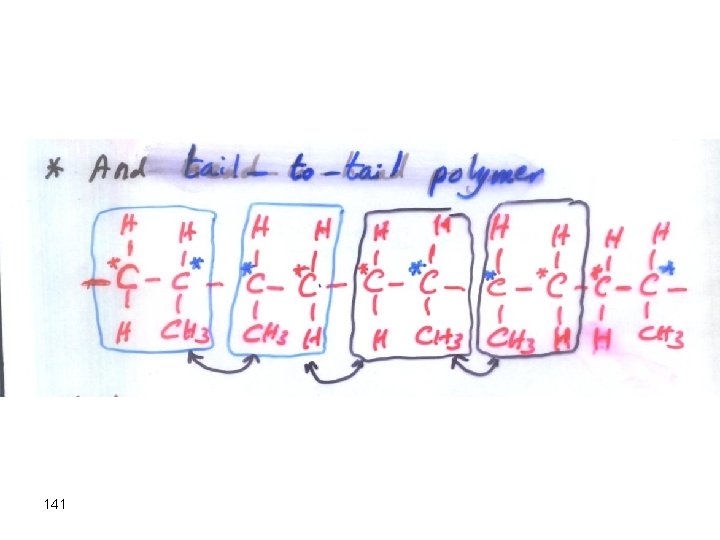
141

142
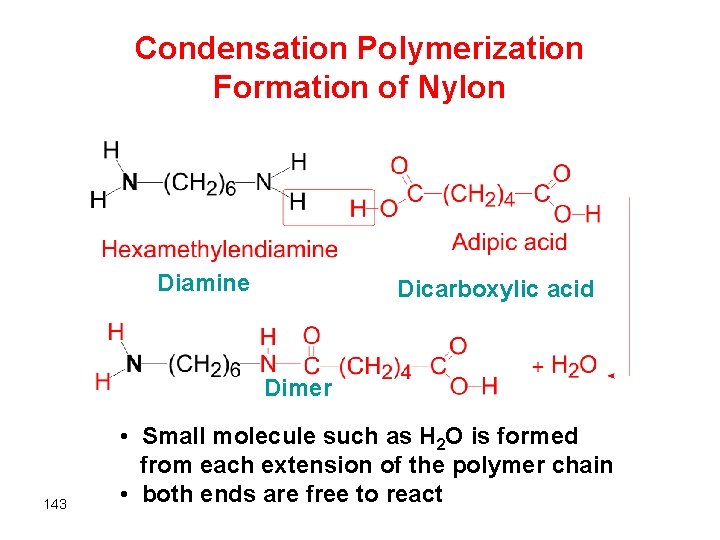
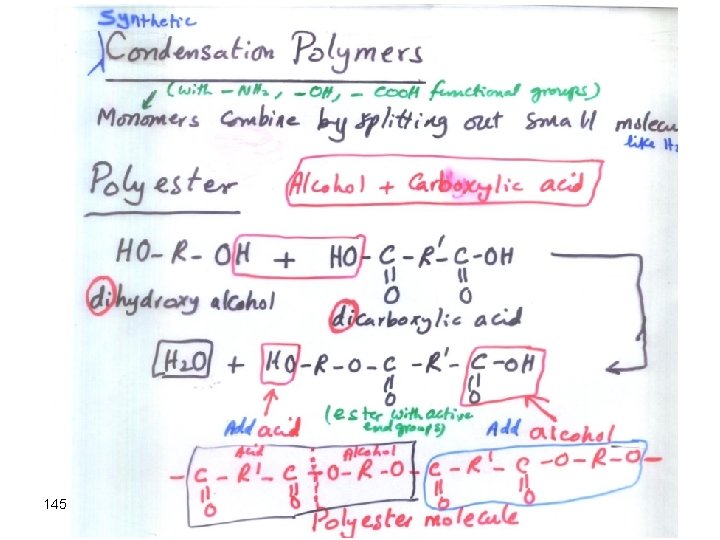
Condensation Polymerization Formation of Nylon Diamine Dicarboxylic acid Dimer 143 • Small molecule such as H 2 O is formed from each extension of the polymer chain • both ends are free to react

Nylon 144
145

Proteins • Natural polymers made up of -amino acids (molecular weight from 6000 to >1, 000 g/mol). 1. Fibrous Proteins: provide structural integrity and strength to muscle, hair and cartilage. 146

Proteins 2. Globular Proteins: § Roughly spherical shape § Transport and store oxygen and nutrients § Act as catalysts § Fight invasion by foreign objects § Participate in the body’s regulatory system § Transport electrons in metabolism 147

-Amino Acids • NH 2 always attached to the -carbon (the carbon attached to COOH) • C = -carbon 148
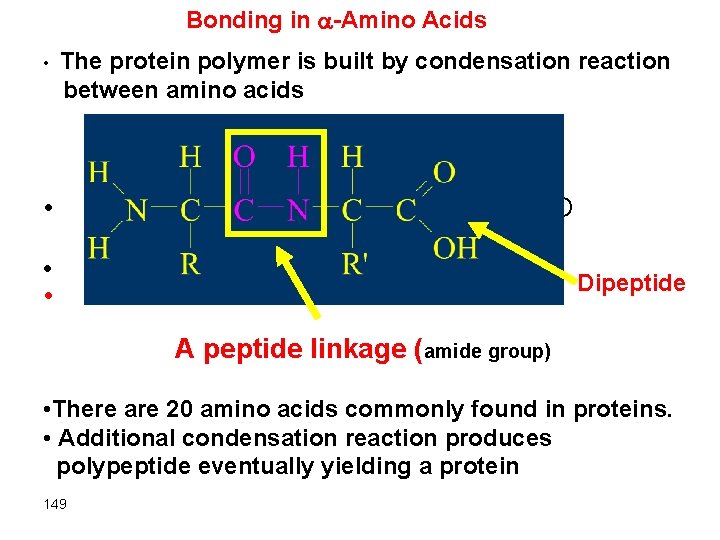
Bonding in -Amino Acids • The protein polymer is built by condensation reaction between amino acids • + H 2 O • • Dipeptide A peptide linkage (amide group) • There are 20 amino acids commonly found in proteins. • Additional condensation reaction produces polypeptide eventually yielding a protein 149
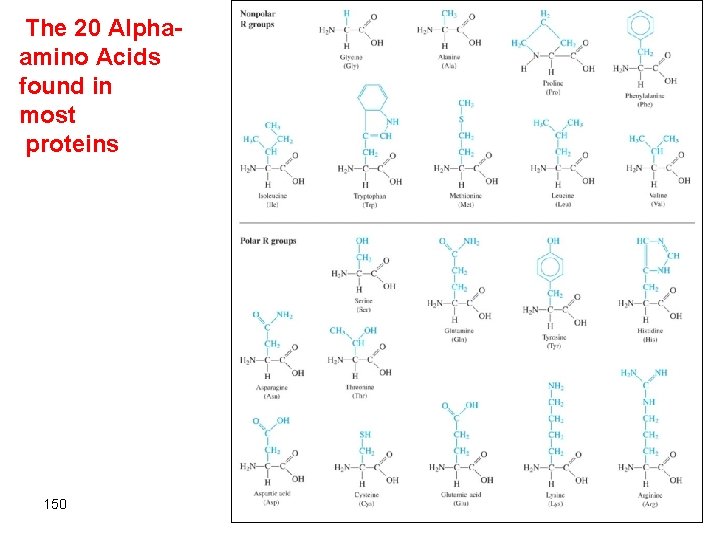
The 20 Alphaamino Acids found in most proteins 150
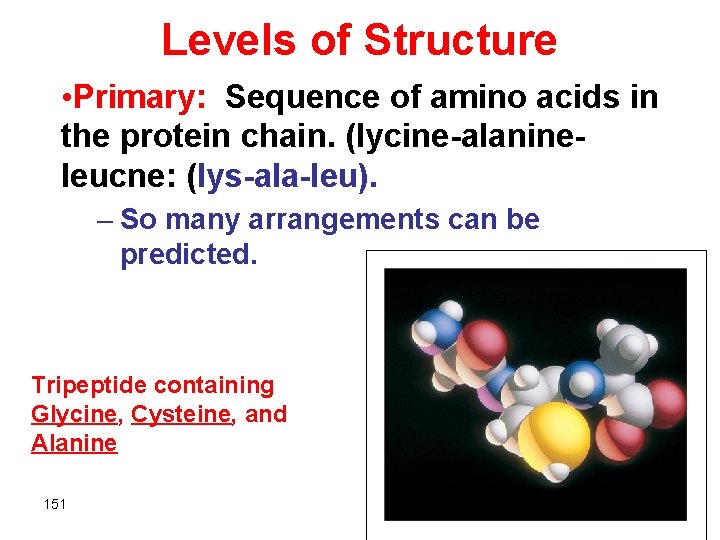
Levels of Structure • Primary: Sequence of amino acids in the protein chain. (lycine-alanineleucne: (lys-ala-leu). – So many arrangements can be predicted. Tripeptide containing Glycine, Cysteine, and Alanine 151
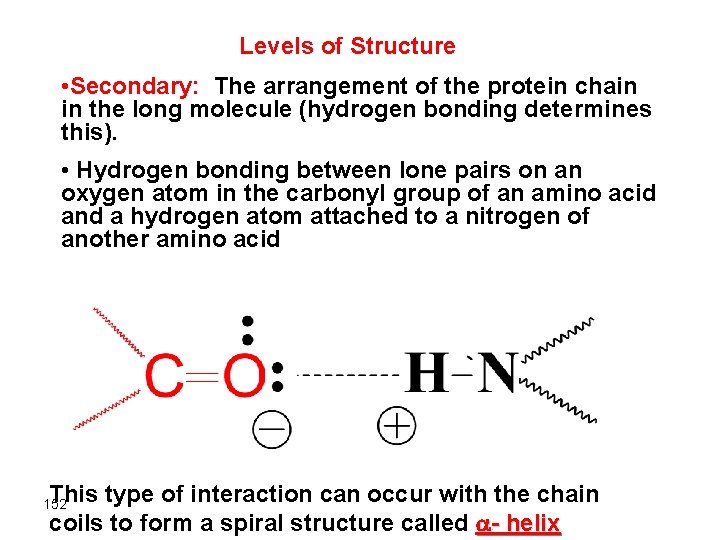
Levels of Structure • Secondary: The arrangement of the protein chain in the long molecule (hydrogen bonding determines this). • Hydrogen bonding between lone pairs on an oxygen atom in the carbonyl group of an amino acid and a hydrogen atom attached to a nitrogen of another amino acid This type of interaction can occur with the chain coils to form a spiral structure called - helix 152
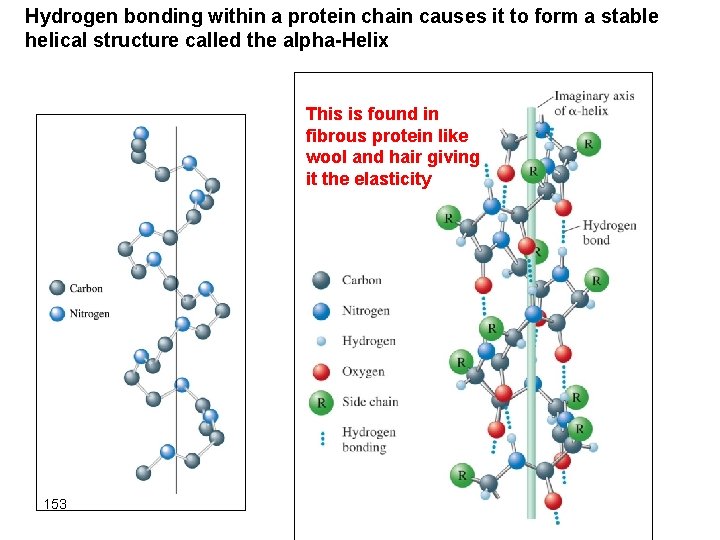
Hydrogen bonding within a protein chain causes it to form a stable helical structure called the alpha-Helix This is found in fibrous protein like wool and hair giving it the elasticity 153
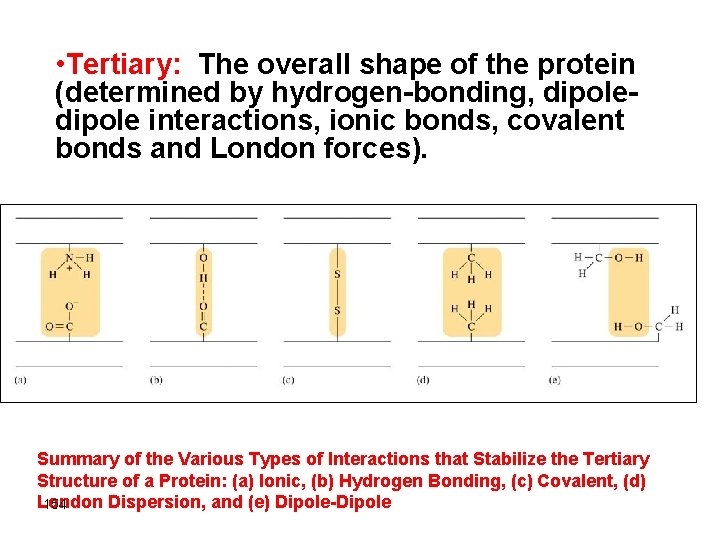
• Tertiary: The overall shape of the protein (determined by hydrogen-bonding, dipole interactions, ionic bonds, covalent bonds and London forces). Summary of the Various Types of Interactions that Stabilize the Tertiary Structure of a Protein: (a) Ionic, (b) Hydrogen Bonding, (c) Covalent, (d) London Dispersion, and (e) Dipole-Dipole 154
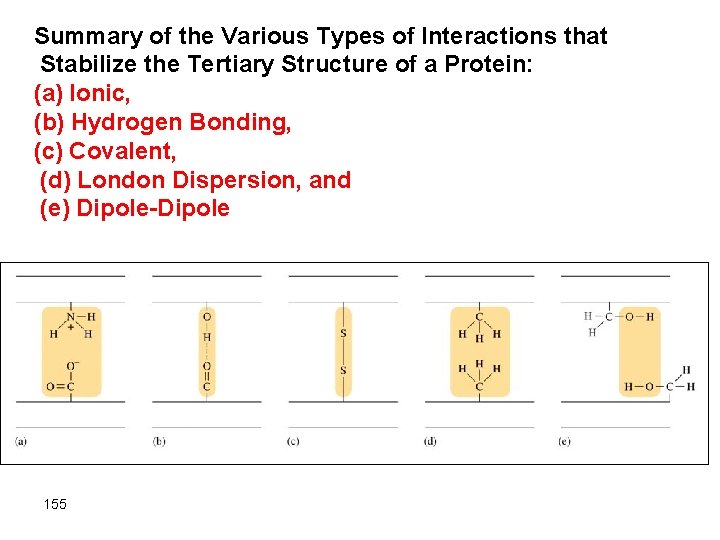
Summary of the Various Types of Interactions that Stabilize the Tertiary Structure of a Protein: (a) Ionic, (b) Hydrogen Bonding, (c) Covalent, (d) London Dispersion, and (e) Dipole-Dipole 155

Carbohydrates §Food source for most organisms and structural material for plants. §Empirical formula = (CH 2 O)n § Most carbohydrates such as starch and cellulose are polymers of monosacharides or simple sugar monomers §Monosaccharides (simple sugars) are polyhydroxy ketones and aldehydes Ø Pentoses (5 -carbon atoms) - ribose, arabinose Ø Hexoses (6 -carbon atoms) - fructose, glucose 156

D and L Nomenclature THIS IS NOT IN THE ORIGINAL NOTES!! • Used to represent the different • stereoisomers D=Dextrorotary • L=Levorotary • 157
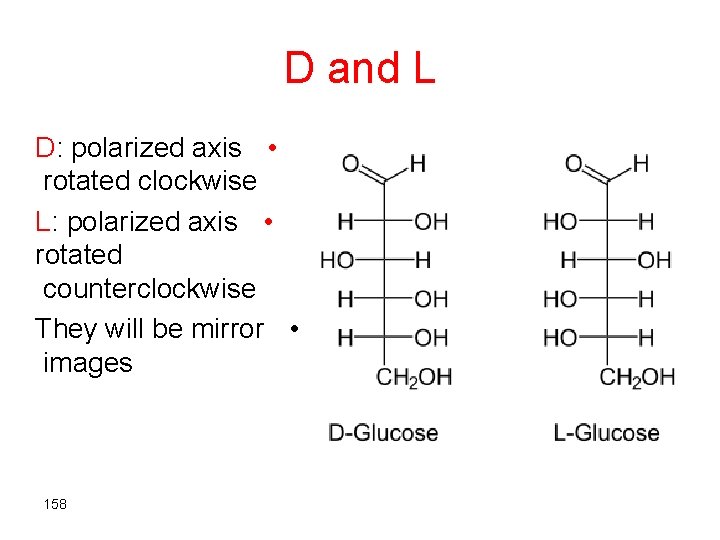
D and L D: polarized axis • rotated clockwise L: polarized axis • rotated counterclockwise They will be mirror • images 158
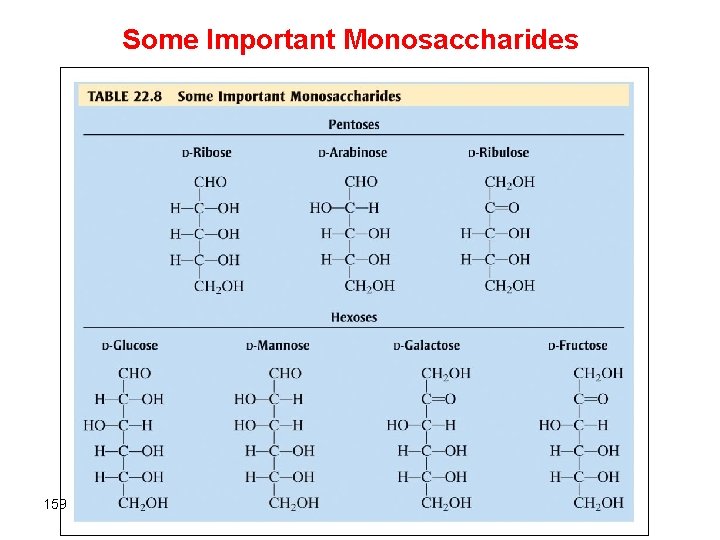
Some Important Monosaccharides 159

Chiral carbon atoms in fructose • Molecules with nonsuperimposable mirror images exhibit optical isomerism • A carbon atom with different groups bonded to it in a tetrahedral arrangement always has a nonsuperimposable mirror images which gives rise to a pair of optical isomers 160
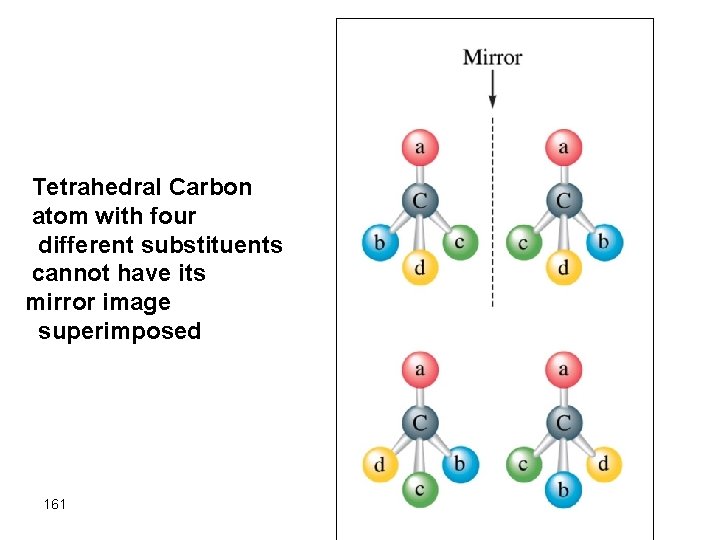
Tetrahedral Carbon atom with four different substituents cannot have its mirror image superimposed 161
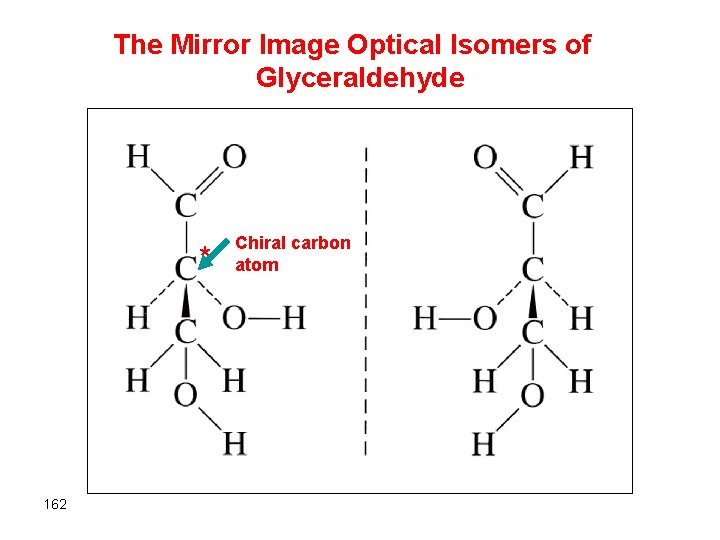
The Mirror Image Optical Isomers of Glyceraldehyde * 162 Chiral carbon atom

Fructose * * * 163 There are 3 chiral Carbon atoms There are 23 isomers That differ in the ability To rotate light

Complex carbohydrates §Disaccharides (formed from 2 monosaccharides joined by a glycoside linkage) Øsucrose (glucose + fructose) §Polysaccharides (many monosaccharide units) Østarch, cellulose 164
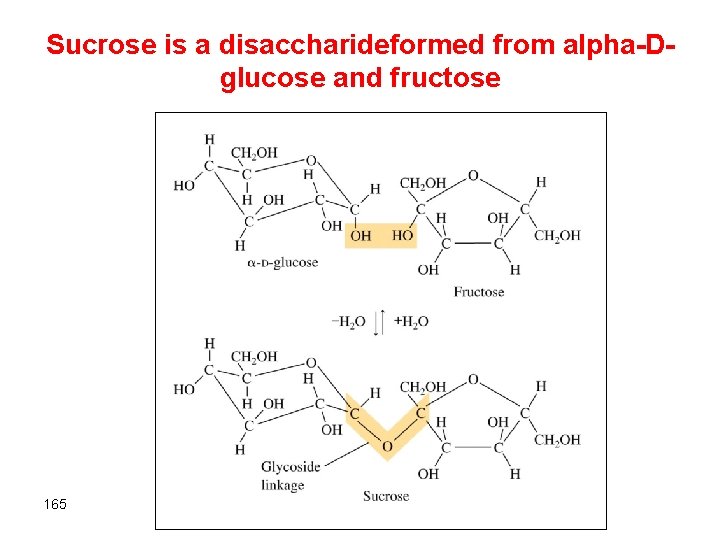
Sucrose is a disaccharideformed from alpha-Dglucose and fructose 165
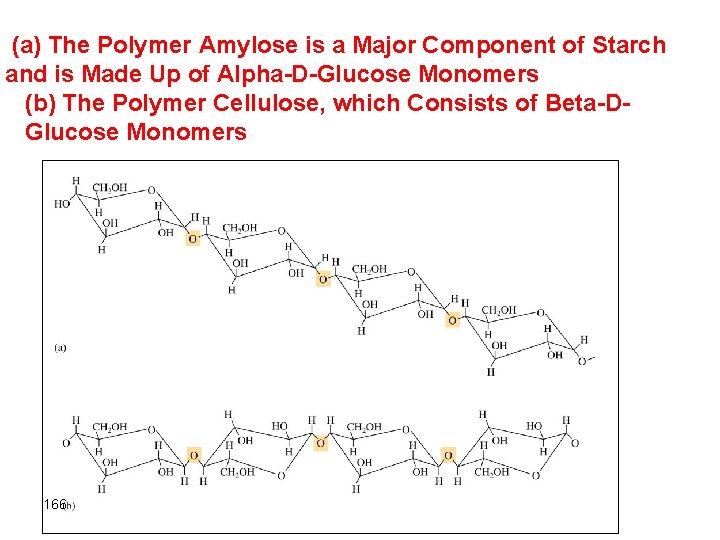
(a) The Polymer Amylose is a Major Component of Starch and is Made Up of Alpha-D-Glucose Monomers (b) The Polymer Cellulose, which Consists of Beta-DGlucose Monomers 166

Nucleic Acids • Life is possible because each cell when it divides can transmit the vital information about how it works to the next generation • The substance that stores and transmits information is a polymer called deoxyribonucleic acid (DNA) • DNA together with other similar nucleic acids called ribonucleic acids is responsible for the synthesis of various proteins needed by the cell to carry out its life functions 167

Nucleic Acids • DNA (deoxyribonucleic acids): stores and transmits genetic information, responsible (with RNA) for protein synthesis. (Molar mass = several billion) • RNA (ribonucleic acid): helps in protein synthesis. (Molecular weight = 20, 000 to 40, 000) 168

Monomers of nucleic acid Nucleotides 1. Five-carbon sugar, deoxyribose in DNA and ribose in RNA. 2. Nitrogen containing organic base 3. Phosphoric acid molecule, H 3 PO 4 • The base and the sugar combine to form a unit that in turn reacts with phosphoric acid to create a nucleotide • The nucleotides become connected through condensation reaction that eliminate water to give a polymer that contain a billion units. 169
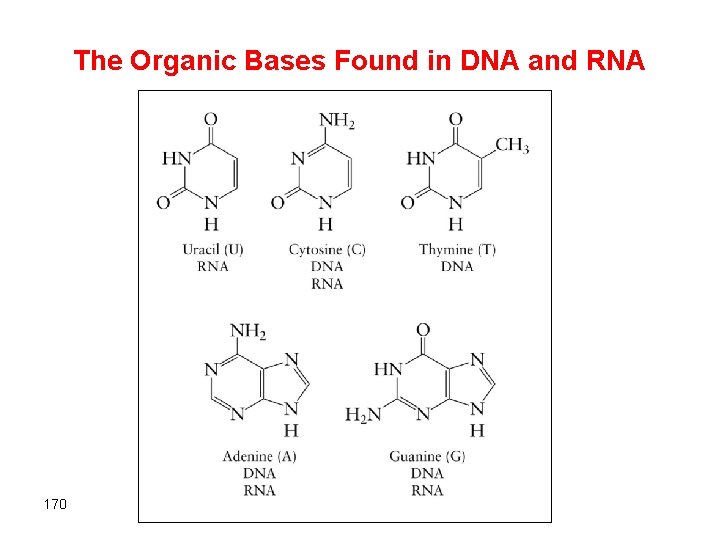
The Organic Bases Found in DNA and RNA 170
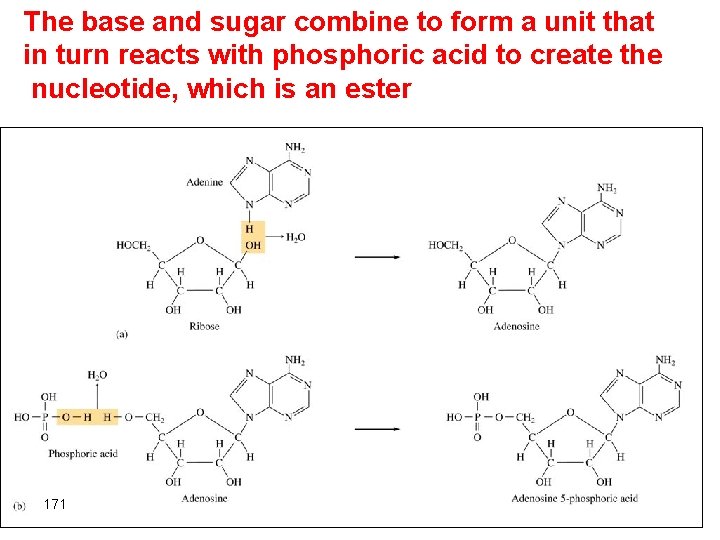
The base and sugar combine to form a unit that in turn reacts with phosphoric acid to create the nucleotide, which is an ester 171
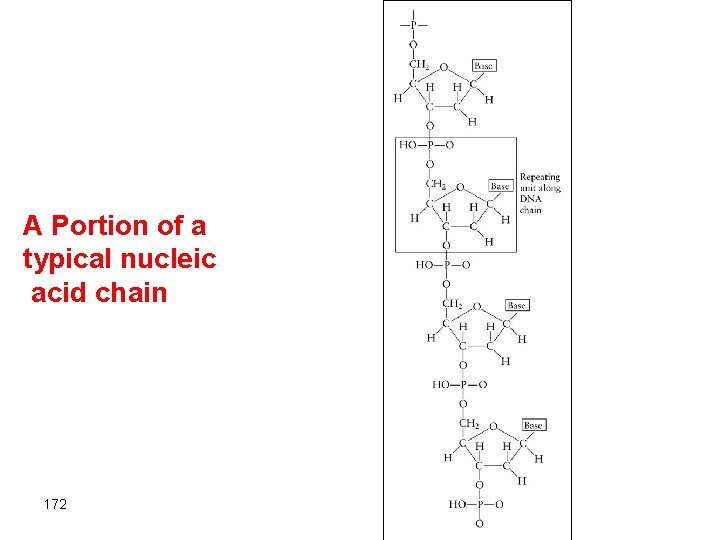
A Portion of a typical nucleic acid chain 172

Double helix formation • According to Watson and Crick (Nobel prize winners), CAN is composed of two strands (threads) running in opposite directions that are bridged by hydrogen bonds between specific pyrimidine groups on one strand purine group on the other • The two strands are twisted into a double -helix structure • The strongest hydrogen bonds form between adonine and thymine and between guanine and cystosine. Thus; A-T or G-C bonding interactions will take place • The sequence of nucleotides on one strand of the double helix determines the sequence of the other • The sequence of the bases determines what information is stored. 173
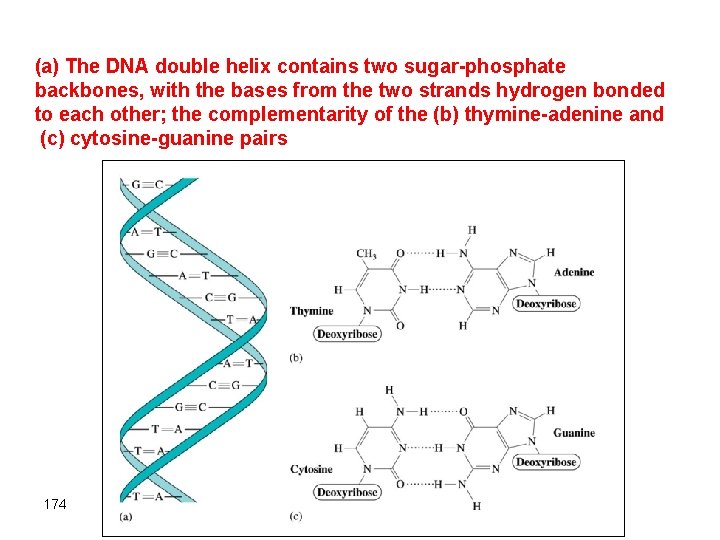
(a) The DNA double helix contains two sugar-phosphate backbones, with the bases from the two strands hydrogen bonded to each other; the complementarity of the (b) thymine-adenine and (c) cytosine-guanine pairs 174
- Slides: 174