Organic and biochemistry q Basicity of aromatic amines

Organic and biochemistry q Basicity of aromatic amines. q Concept the carbanions. q Methods of generated of the carbanions. q Reactions involving carbanions. Assistancce Lecturer Amjad Ahmed Jumaa www. soran. edu. iq 1

Basicity of aromatic amines: q. The electron-releasing groups increases the basicity of aromatic amines, because the unshared-electron pair are more available to the proton and thus more stabilize the cation. üIn contract, the electron-withdrawing groups decrease the basicity of the aromatic amines because the unshared-electron pairs are unavailable to the proton and thus less stabilize the cation. www. soran. edu. iq

Problem: which of the following amines are more basic? Ethyl amine; diethyl amine; aniline; p-nitroaniline. Solution: Diethyl amine > Ethyl amine > aniline > p-nitroaniline. Problem: arrange the following aromatic amines according in increase in basicity: p-chloroaniline; m-methoxyaniline; p-nitroaniline; paminoaniline. Solution: p-aminoaniline > m-methoxyaniline > p-chloroaniline > pnitroaniline. www. soran. edu. iq

v. Some important organic chemical reactions: v. Concepts of the carbanions: üThe carbanion is the carbon atom that bears a negative charge. v. Methods of generated of the carbanions: §One of the methods to obtain carbanions is the ionization of a α-hydrogen which attached to the α-carbon (C-1) in the carbonyl compounds (aldehydes and ketones). Ionization achieved by a base. www. soran. edu. iq

o. The carbanion (I) is a resonance hybrid of two structures (II) and (III). ØAlso the carbonyl group affects the acidity of α-hydrogen in just the way it affects the acidity of carboxylic acid; by helping to accommodate the negative charge of the anion: www. soran. edu. iq

ØThe carbonyl group undergoes nucleophilic attack this is due to the ability of oxygen to accommodate the negative charge that develops as a result of the attack: www. soran. edu. iq
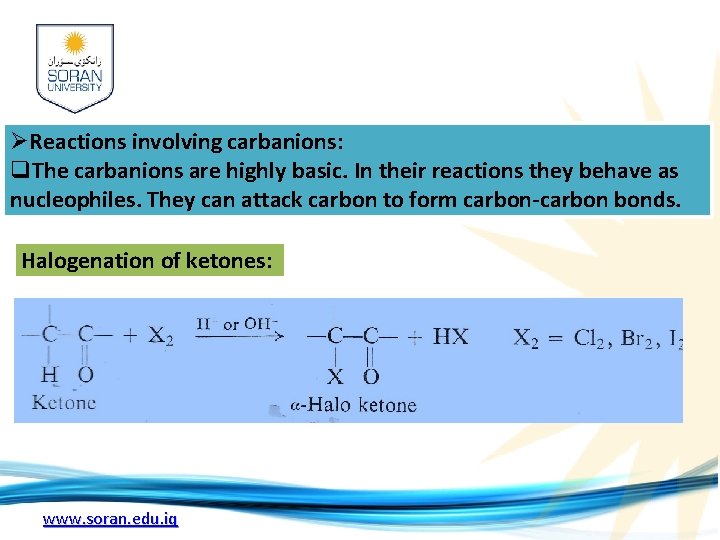
ØReactions involving carbanions: q. The carbanions are highly basic. In their reactions they behave as nucleophiles. They can attack carbon to form carbon-carbon bonds. Halogenation of ketones: www. soran. edu. iq
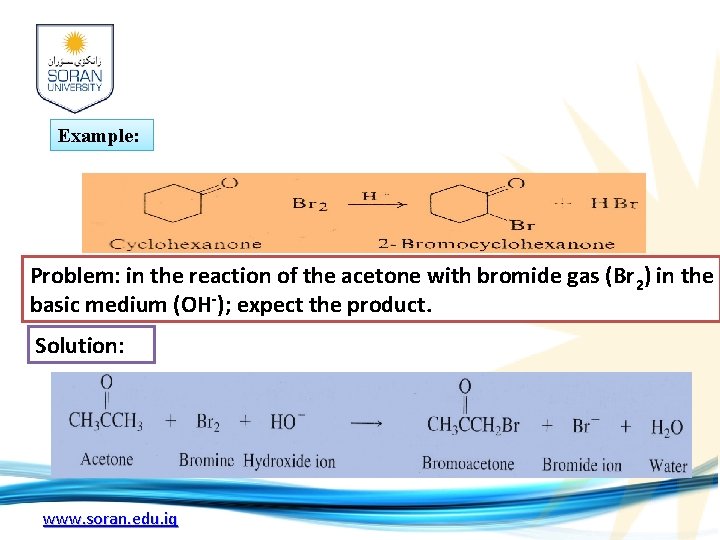
Example: Problem: in the reaction of the acetone with bromide gas (Br 2) in the basic medium (OH-); expect the product. Solution: www. soran. edu. iq

Follow problem: write balanced equations, naming all organic products for the chlorination in the basic medium for the following compounds. 1 - Phenyl acetaldehyde 2 - methyl ketone 3 - acetophenone. 4 - (3 -pentanone). www. soran. edu. iq
- Slides: 9