Organic and Biochemical Compounds Organic compound a covalently

Organic and Biochemical Compounds

• Organic compound – a covalently bonded compound that contains carbon, excluding carbonates and oxides • Organic compounds contain Carbon and almost always Hydrogen • Other atoms such as Oxygen, Nitrogen Sulfur, and Phosphorus are also found in some organic compounds
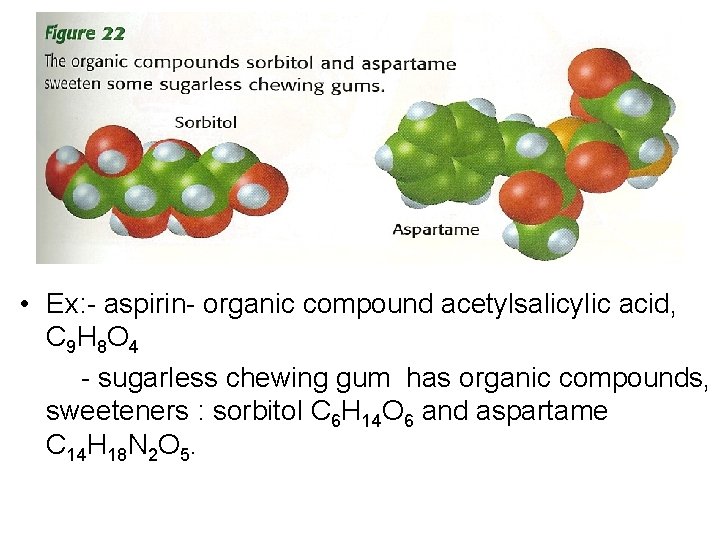
• Ex: - aspirin- organic compound acetylsalicylic acid, C 9 H 8 O 4 - sugarless chewing gum has organic compounds, sweeteners : sorbitol C 6 H 14 O 6 and aspartame C 14 H 18 N 2 O 5.

• When a compound is made of only carbon and hydrogen atoms, it is called Hydrocarbon • Ex: methane, carbon have 4 valence electrons, each of these electrons forms a different C-H single bond • Carbon atoms can never form more than a total of four covalent bonds in organic compounds
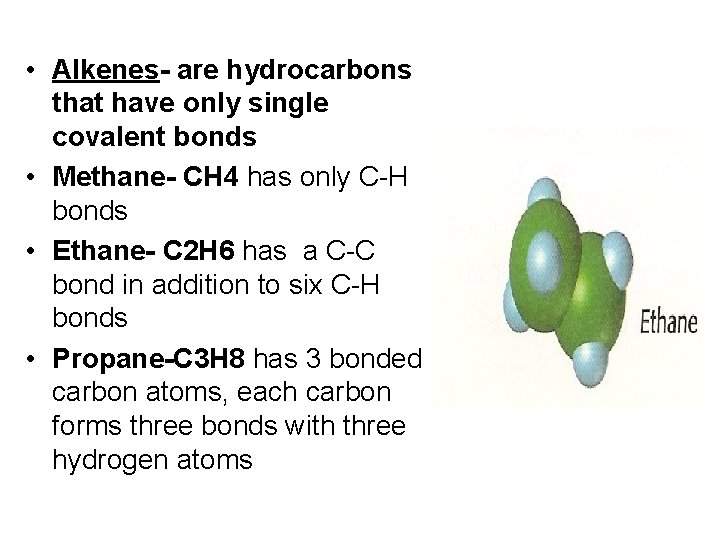
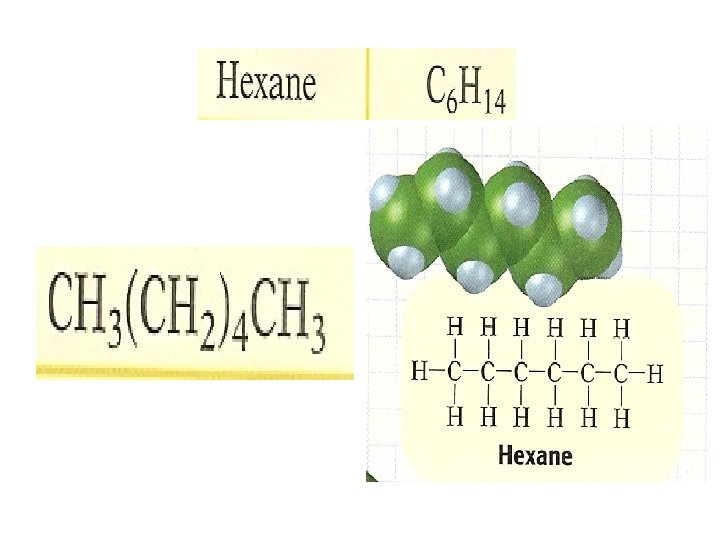
• Alkenes- are hydrocarbons that have only single covalent bonds • Methane- CH 4 has only C-H bonds • Ethane- C 2 H 6 has a C-C bond in addition to six C-H bonds • Propane-C 3 H 8 has 3 bonded carbon atoms, each carbon forms three bonds with three hydrogen atoms

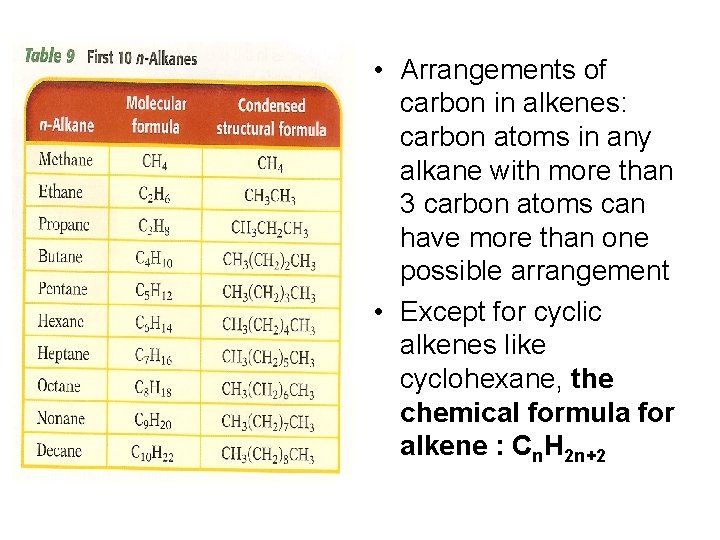
• Arrangements of carbon in alkenes: carbon atoms in any alkane with more than 3 carbon atoms can have more than one possible arrangement • Except for cyclic alkenes like cyclohexane, the chemical formula for alkene : Cn. H 2 n+2
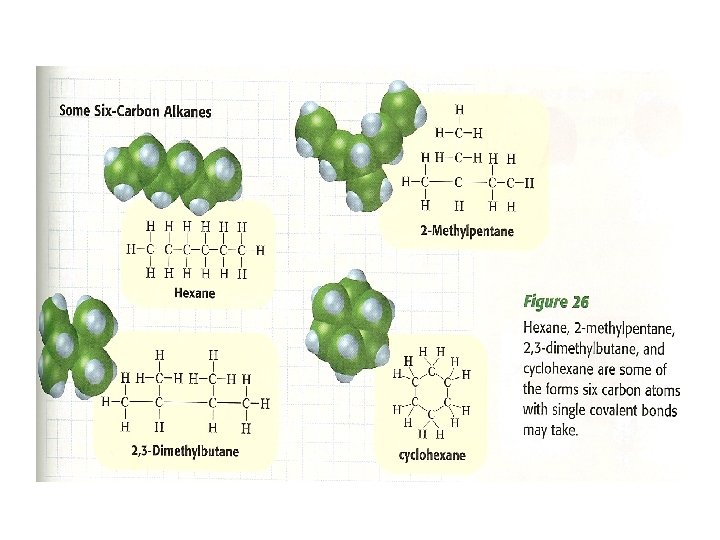

Alkenes have double C=C bonds

• Alkenes are also carbohydrates • They are called like alkanes but with the – ane ending replaced by –ene, they are different because they have at least one double covalent bond: C=C • Ex: - ethene (or ethylene) C 2 H 4 is formed when fruit ripens -propene (or propylene) C 3 H 6 is used to make rubbing alcohol and some plastics

• Alcohol have -OH groups (or hydroxyl) • Methanol CH 3 OH is added to ethanol CH 3 CH 2 OH, to make denatured alcohol • Isopropanol which is found in rubbing alcohol : C 3 H 8 O or (CH 3)2 CHOH
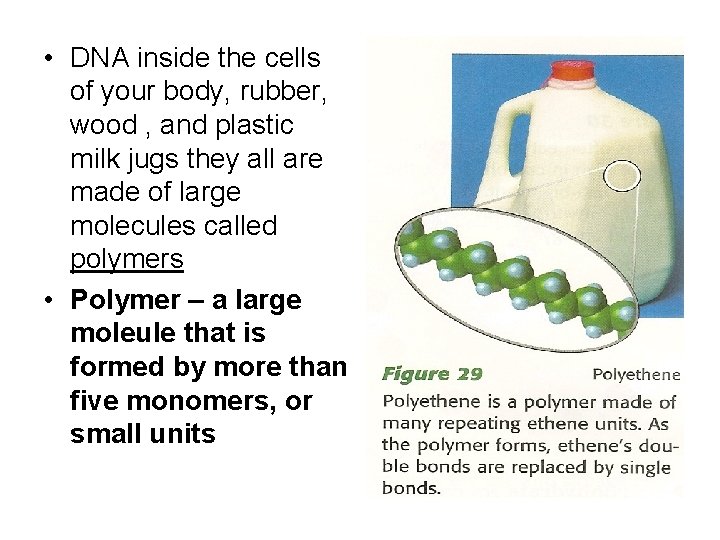
• DNA inside the cells of your body, rubber, wood , and plastic milk jugs they all are made of large molecules called polymers • Polymer – a large moleule that is formed by more than five monomers, or small units

• Many polymers have repeating subunits • Poly means “many” • Polyethene = “many ethenes”, in this case C 2 H 4 is called monomer • Some polymers are natural (wood, DNA, protein) others are man-made (plastic or fibers) • The elasticity of a polymer is determined by its structue (ex: if you crush a milk jug, it does not return to its original shape-is not elastic)

Biochemical Compounds • Carbohydrate- any organic compound that is made of carbon, hydrogen and oxygen and that provides nutrients to the cells of living things • Protein-an organic compound that is made of oneor more chains of amino cids and that is a principal component of all cell

• Amino acid any one of 20 different organic molecules that contain a carboxyl and an amino group and that to form proteins • Ex: insulin • Many proteins are made of thousands of bonded amino acid molecules
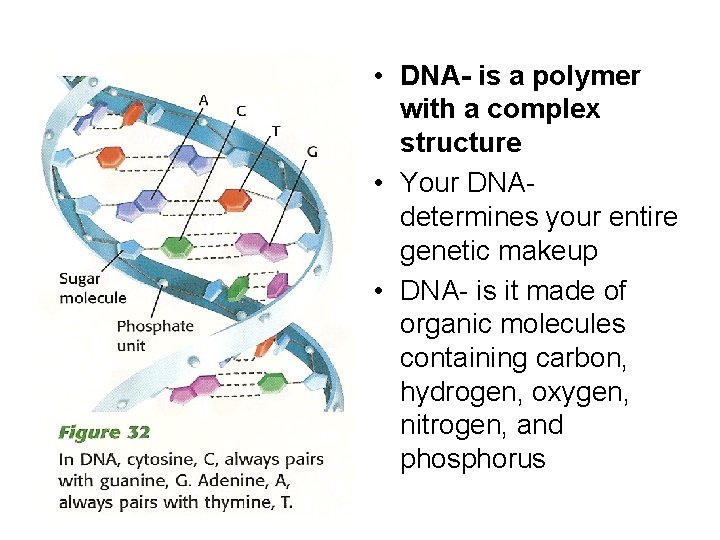
• DNA- is a polymer with a complex structure • Your DNAdetermines your entire genetic makeup • DNA- is it made of organic molecules containing carbon, hydrogen, oxygen, nitrogen, and phosphorus
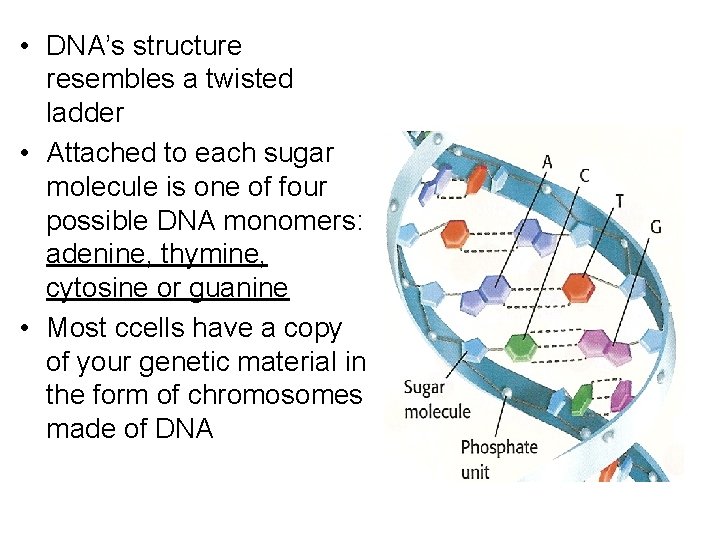
• DNA’s structure resembles a twisted ladder • Attached to each sugar molecule is one of four possible DNA monomers: adenine, thymine, cytosine or guanine • Most ccells have a copy of your genetic material in the form of chromosomes made of DNA
- Slides: 18