Ore deposit environments Magmatic Cumulate deposits fractional crystallization









- Slides: 14

Ore deposit environments • Magmatic – Cumulate deposits – fractional crystallization processes can concentrate metals (Cr, Fe, Pt) – Pegmatites – late staged crystallization forms pegmatites and many residual elements are concentrated (Li, Ce, Be, Sn, and U) • Hydrothermal – Magmatic fluid - directly associated with magma – Porphyries - Hot water heated by pluton – Skarn – hot water associated with contact metamorphisms – Exhalatives – hot water flowing to surface – Epigenetic – hot water not directly associated with pluton

Water-rock interactions • To concentrate a material, water must: – Transport the ions – A ‘trap’ must cause precipitation in a spatially constrained manner • Trace metals which do not go into igneous minerals easily get very concentrated in the last bit of melt • Leaching can preferentially remove materials, enriching what is left or having the leachate precipitate something further away

Metal Sulfide Mineral Solubility • Problem 1: Transport of Zn to ‘trap’: Zn. S + 2 H+ + 0. 5 O 2 = Zn 2+ + S 2 - + H 2 O Need to determine the redox state the Zn 2+ would have been at equilibrium with… What other minerals are in the deposit that might indicate that? define approximate f. O 2 and f. S 2 values and compute Zn 2+ conc. Pretty low Zn 2+

• Must be careful to consider what the conditions of water transporting the metals might have been how can we figure that out? ? • What other things might be important in increasing the amount of metal a fluid could carry? More metal a fluid can hold the quicker a larger deposit can be formed…

• How about the following: Zn. S + 2 H+ + 0. 5 O 2 + Cl- = Zn. Cl+ + S 2 - + H 2 O Compared to That is a BIG difference…

Geochemical Traps • Similar to chemical sedimentary rocks – must leach material into fluid, transport and deposit ions as minerals… • p. H, redox, T changes and mixing of different fluids results in ore mineralization • Cause metals to go from soluble to insoluble • Sulfide (reduced form of S) strongly binds metals many important metal ore minerals are sulfides!

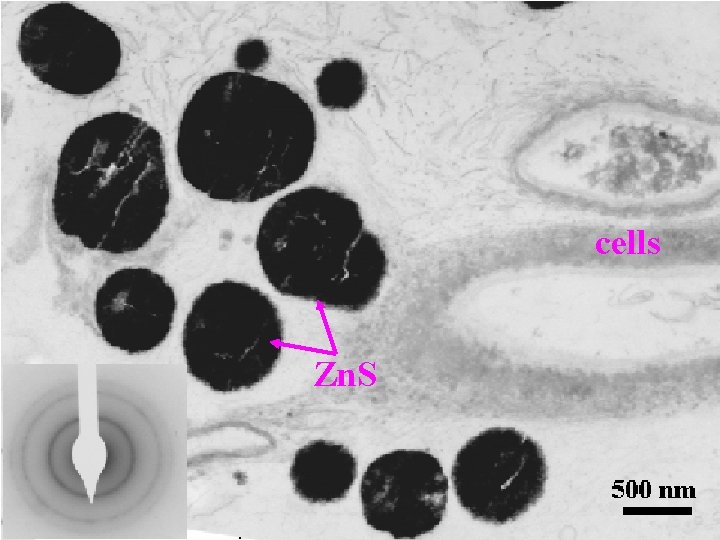
Piquette Mine • 1 -5 nm particles of Fe. OOH and Zn. S – biogenic precipitation • Tami collecting samples

cells Zn. S

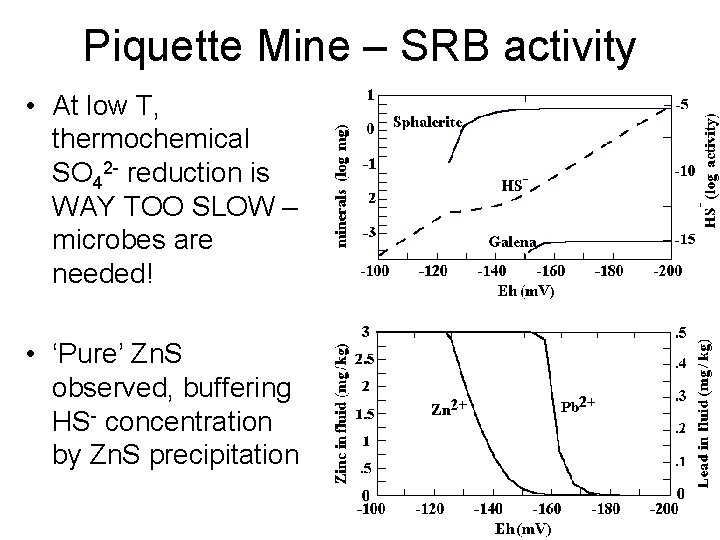
Piquette Mine – SRB activity • At low T, thermochemical SO 42 - reduction is WAY TOO SLOW – microbes are needed! • ‘Pure’ Zn. S observed, buffering HS- concentration by Zn. S precipitation

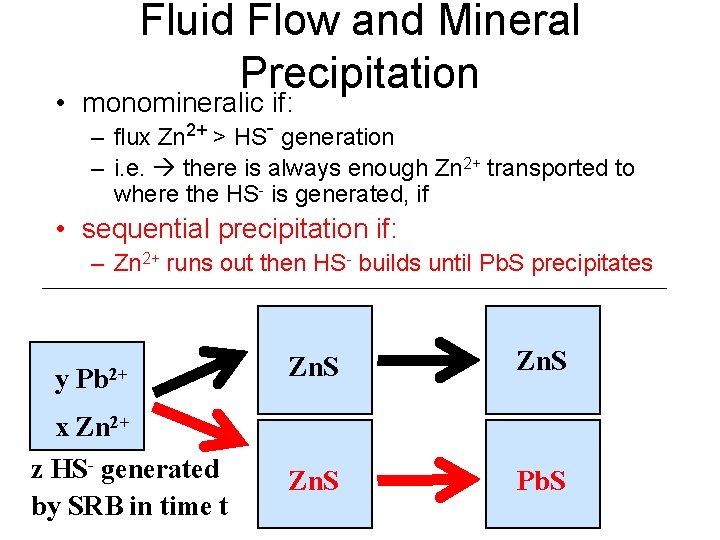
Fluid Flow and Mineral Precipitation • monomineralic if: – flux Zn 2+ > HS- generation – i. e. there is always enough Zn 2+ transported to where the HS- is generated, if • sequential precipitation if: – Zn 2+ runs out then HS- builds until Pb. S precipitates y Pb 2+ Zn. S Pb. S x Zn 2+ z HS- generated by SRB in time t

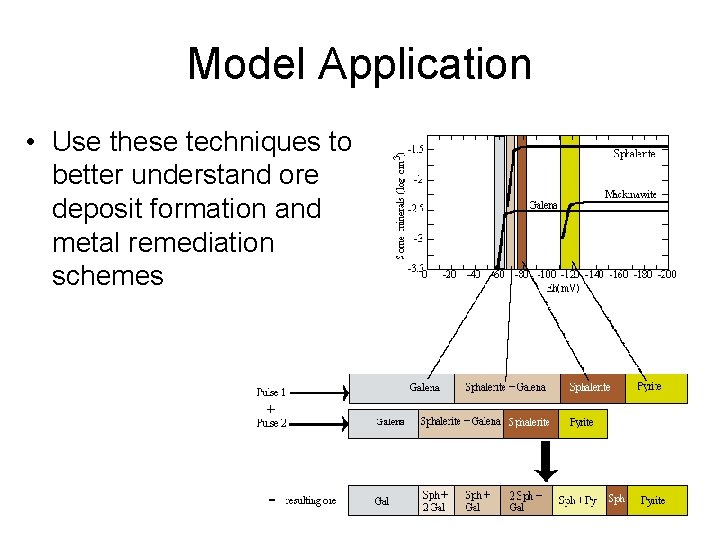
Model Application • Use these techniques to better understand ore deposit formation and metal remediation schemes

Sequential Precipitation Experiments • SRB cultured in a 125 ml septum flask containing equimolar Zn 2+ and Fe 2+ • Flask first develops a white precipitate (Zn. S) and only develops Fe. S precipitates after most of the Zn 2+ is consumed • Upcoming work in my lab will investigate this process using microelectrodes where observation of Zn. S and Fe. S molecular clusters will be possible!

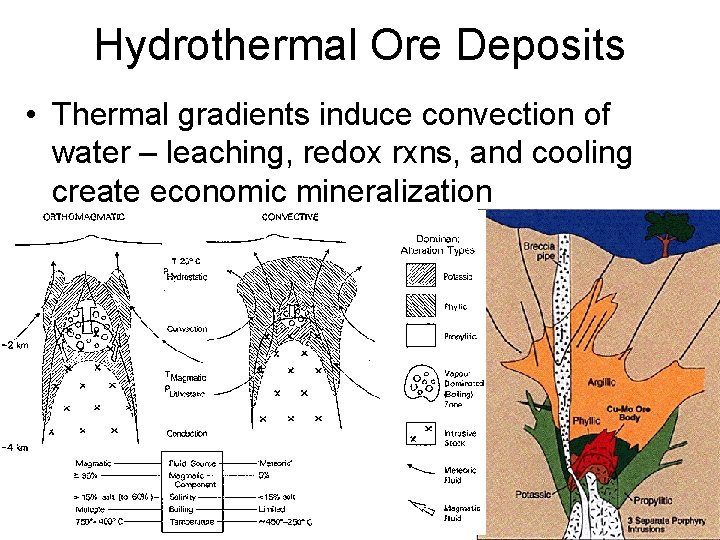
Hydrothermal Ore Deposits • Thermal gradients induce convection of water – leaching, redox rxns, and cooling create economic mineralization

Ore deposit environments • Sedimentary – Placer – weathering of primary mineralization and transport by streams (Gold, diamonds, other) – Banded Iron Formations – 90%+ of world’s iron tied up in these (more later…) – Evaporite deposits – minerals like gypsum, halite deposited this way – Laterites – leaching of rock leaves residual materials behind (Al, Ni, Fe) – Supergene – reworking of primary ore deposits remobilizes metals (often over short distances)