Orbital Notation Valence Electrons Lewis Dot Structures Electron

Orbital Notation, Valence Electrons & Lewis Dot Structures

Electron Configuration Rules 1. Aufbau Principle Each electron MUST occupy the lowest energy orbital available first 2. Pauli Exclusion Principle A maximum of 2 electrons can occupy a single orbital, but only if the electrons have opposite spins 3. Hund’s Rule Single electrons with the same spin must occupy each equal energy orbital before additional electrons with opposite spins can occupy those same orbitals

Aufbau Principle Each electron MUST occupy the lowest energy level and sublevel available first Determine the order of energy levels, sub-levels, and the orbitals within the sub-levels Energy levels are in the order of the periods down the periodic table Order: 1, 2, 3, 4, 5, 6, and 7 Sub-levels: are in the order of s, p, d, and f

Aufbau Principle: Notice that d and f sub-levels are “behind” s and p sub-levels

Pauli Exclusion Principle A maximum of 2 electrons can occupy a single orbital, but only if the electrons have opposite spins Remember the amount of orbitals in each sub-level S-sublevel = 1 orbital = 2 electrons P-sublevel = 3 orbitals = 6 electrons D-sublevel = 5 orbitals = 10 electrons F=sublevel = 7 orbitals = 14 electrons Each energy level holds a set amount of electrons: 1 st energy level = 1 sublevel and 1 orbital = max 2 electrons 2 nd & 7 th energy level = 2 sublevels and 4 orbitals = max 8 electrons 3 rd & 6 th energy level = 3 sublevels and 9 orbitals = max 18 electrons 4 th – 5 th energy levels = 4 sublevels and 16 orbitals = max 32 electrons Remember that d and f sublevels are behind s and p

Hund’s Rule Single electrons with the same spin must occupy each equal energy orbital before additional electrons with opposite spins can occupy those same orbitals Orbital Notation: Shows the electrons in an orbital and their spin S- 1 orbital, p- 3 orbitals, d – 5 orbitals, and f – 7 orbitals Lines/boxes represent the orbitals The number of the energy level and its sublevel are written below the line Uses up and down arrows to represent the electrons and their spins Example: Lithium – 1 s 22 s 1 _____ 1 s _____ 2 s

Hund’s Rule Orbital Notation Let’s Try Nitrogen: 1 s 22 p 3 ______ 1 s _______ 2 s ___________ 2 p *This is called an orbital diagram/notation

A little more of a challenge: Sulfur What is Sulfur’s electron configuration? ____________________________ What is Sulfur’s orbital notation? *Remember: S- 1 orbital, p- 3 orbitals, d – 5 orbitals, and f – 7 orbitals

Your Turn: Complete these in your notes Write the orbital diagrams for the following elements: Manganese Potassium Gallium

Valence Electrons The electrons that determine the chemical properties of an element The electrons in the highest (outermost) energy level How do we know what is the highest/outermost energy level? The electron configuration for Chlorine is: 1 s 2 2 p 6 3 s 2 3 p 5 • The outermost energy level is the 3 rd energy level • Chlorine has 7 valence electrons (2 + 5 = 7)
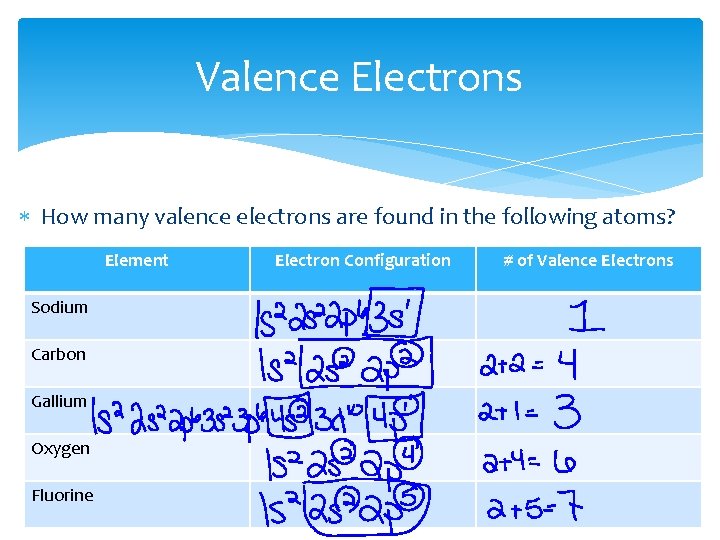
Valence Electrons How many valence electrons are found in the following atoms? Element Sodium Carbon Gallium Oxygen Fluorine Electron Configuration # of Valence Electrons
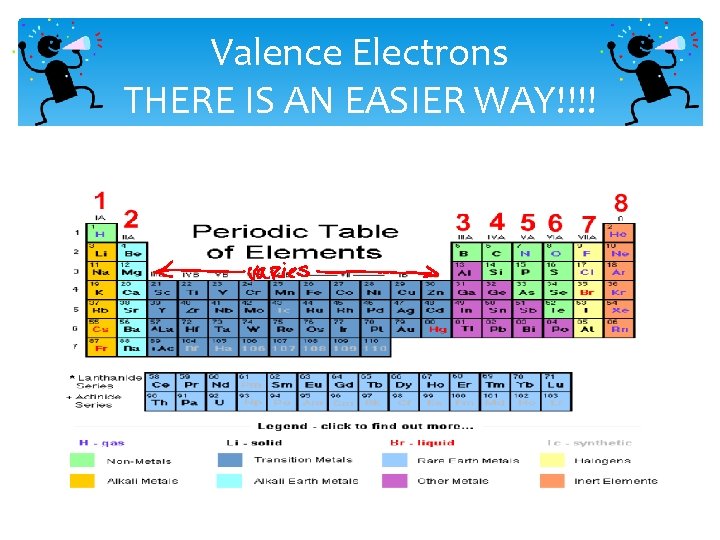
Valence Electrons THERE IS AN EASIER WAY!!!!
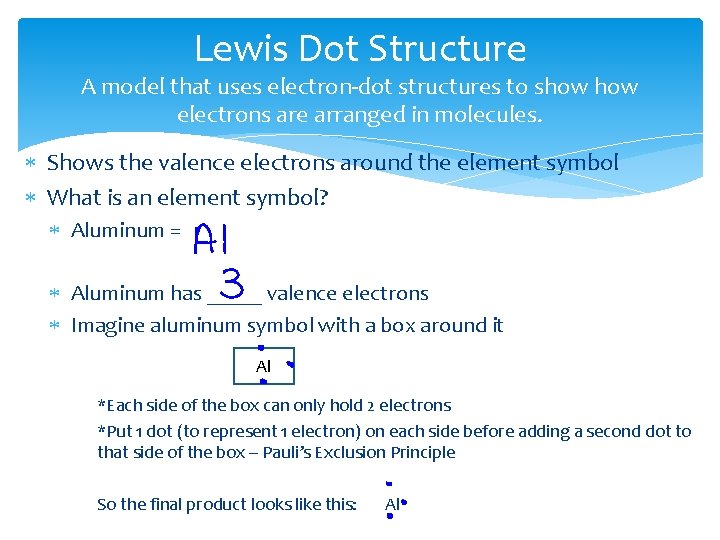
Lewis Dot Structure A model that uses electron-dot structures to show electrons are arranged in molecules. Shows the valence electrons around the element symbol What is an element symbol? Aluminum = Aluminum has _____ valence electrons Imagine aluminum symbol with a box around it Al *Each side of the box can only hold 2 electrons *Put 1 dot (to represent 1 electron) on each side before adding a second dot to that side of the box – Pauli’s Exclusion Principle So the final product looks like this: Al

Give Sulfur a try: How many valence electrons does Sulfur have? ______ S
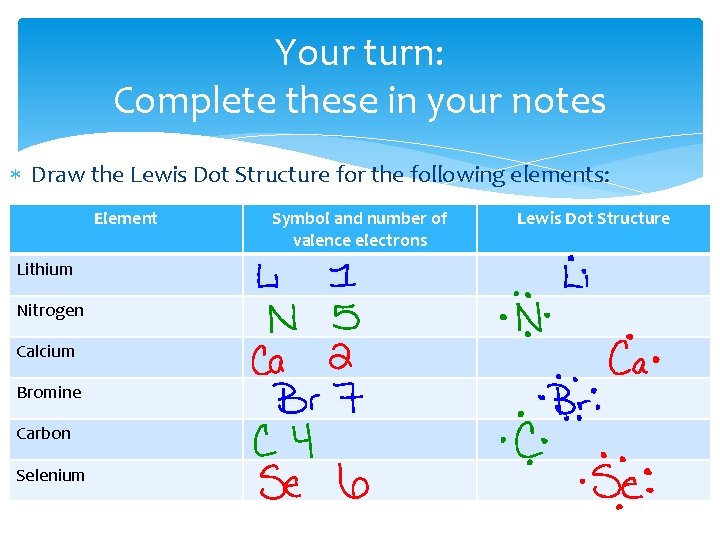
Your turn: Complete these in your notes Draw the Lewis Dot Structure for the following elements: Element Lithium Nitrogen Calcium Bromine Carbon Selenium Symbol and number of valence electrons Lewis Dot Structure
- Slides: 15