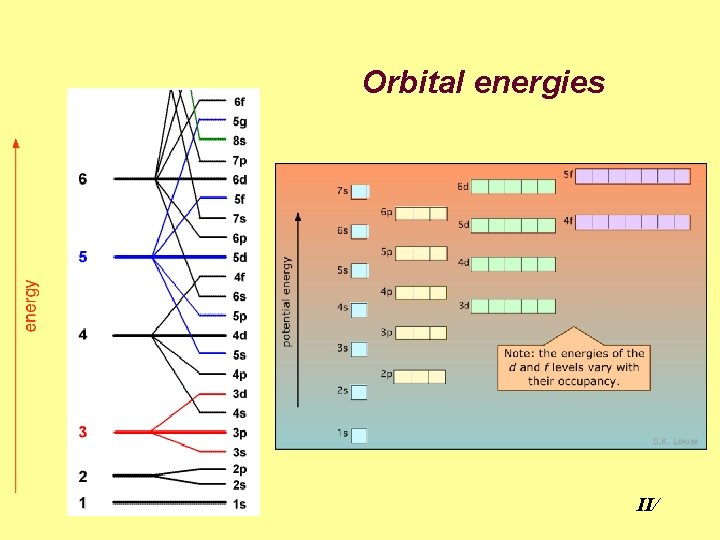
Orbital energies II Orbital Energy determined by n
Orbital energies II/

Orbital Energy determined by n and ℓ: üThe larger (n+ℓ), the higher the energy. üIf two orbitals have equal (n+ℓ), that with larger n has higher energy. 1 s 1 2 s 2 2 p 3 Orbital (n+ℓ) 3 s 3 3 p 4 3 d 5 4 s 4 4 p 5 4 d 6 4 f 7 5 s 5 5 p 6 5 d 7 5 f 8 5 g 9 6 s 6 6 p 7 6 d 8 6 f 9 6 g 10 6 h 11 c II/
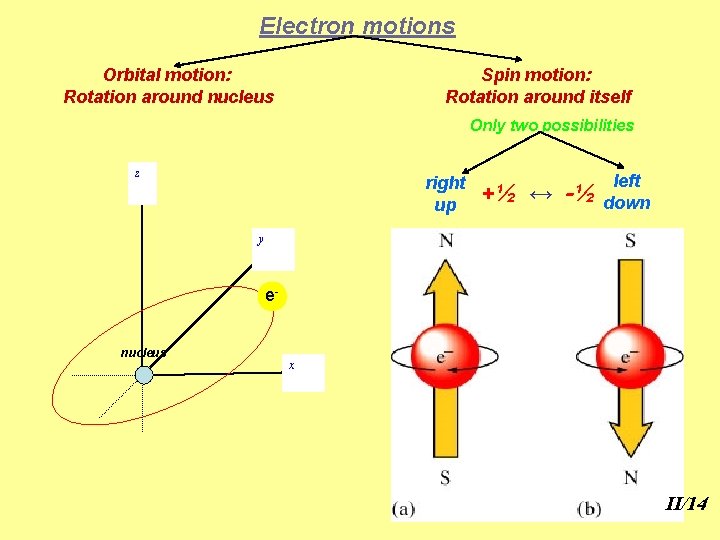
Electron motions Orbital motion: Rotation around nucleus Spin motion: Rotation around itself Only two possibilities z right up +½ ↔ -½ left down y enucleus x II/14

Pauli Exclusion Principle No two electrons in an atom can be equal in all four quantum numbers. → Only two electrons fit in one orbital. Energy 2 p 2 s 1 s 1 st e- 2 nd e- 3 rd e- n=1 ℓ=0 mℓ=0 ms=+½ n=1 ℓ=0 ms=-½ n=1 ℓ=0 ms=±½ II
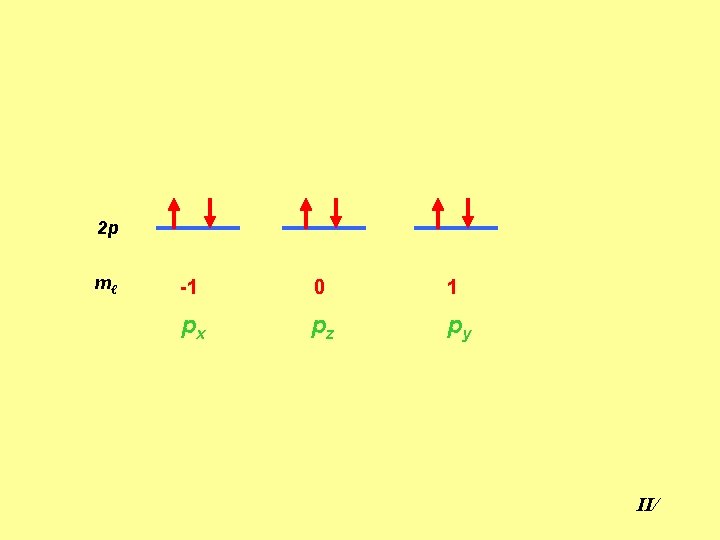
2 p mℓ -1 0 1 px pz py II/

Electron configuration and Aufbau Principle Ø Electrons are placed first in the lowest empty energy levels. Ø Two electrons with opposite spin in each orbital. Ø For electrons with the same ℓ-value, the most stable state is that with maximum spin (Hund’s rule). 2 p Spin=+½ -½=0 2 p Spin=+½ +½=1 II/
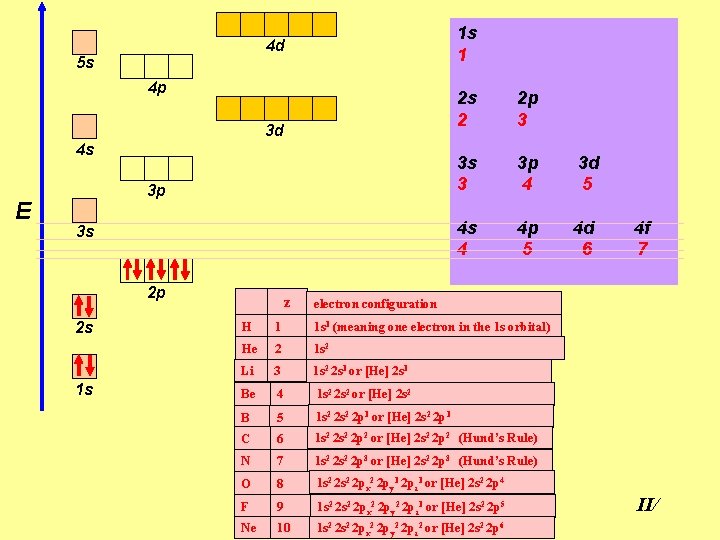
1 s 1 4 d 5 s 4 p 3 d 4 s E 3 p 3 s 2 p 2 s 1 s z 2 s 2 2 p 3 3 s 3 3 p 4 3 d 5 4 s 4 4 p 5 4 d 6 4 f 7 electron configuration H 1 1 s 1 (meaning one electron in the 1 s orbital) He 2 1 s 2 Li 3 1 s 2 2 s 1 or [He] 2 s 1 Be 4 1 s 2 2 s 2 or [He] 2 s 2 B 5 1 s 2 2 p 1 or [He] 2 s 2 2 p 1 C 6 1 s 2 2 p 2 or [He] 2 s 2 2 p 2 (Hund’s Rule) N 7 1 s 2 2 p 3 or [He] 2 s 2 2 p 3 (Hund’s Rule) O 8 1 s 2 2 px 2 2 py 1 2 pz 1 or [He] 2 s 2 2 p 4 F 9 1 s 2 2 px 2 2 py 2 2 pz 1 or [He] 2 s 2 2 p 5 Ne 10 1 s 2 2 px 2 2 py 2 2 pz 2 or [He] 2 s 2 2 p 6 II/
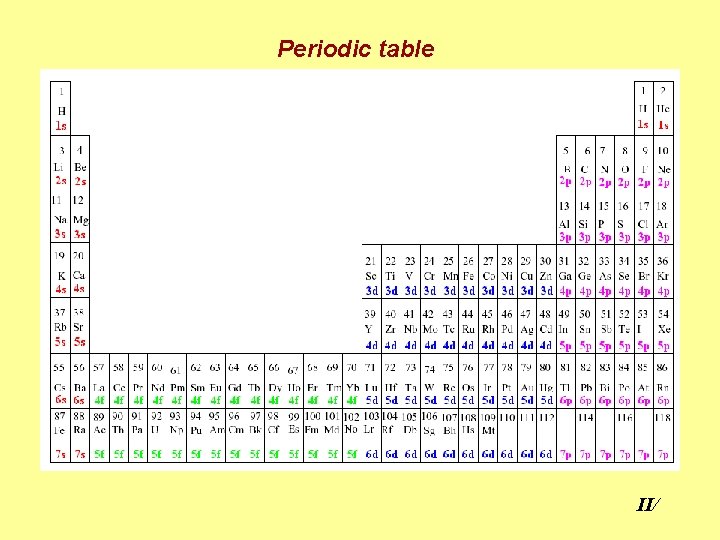
Periodic table II/
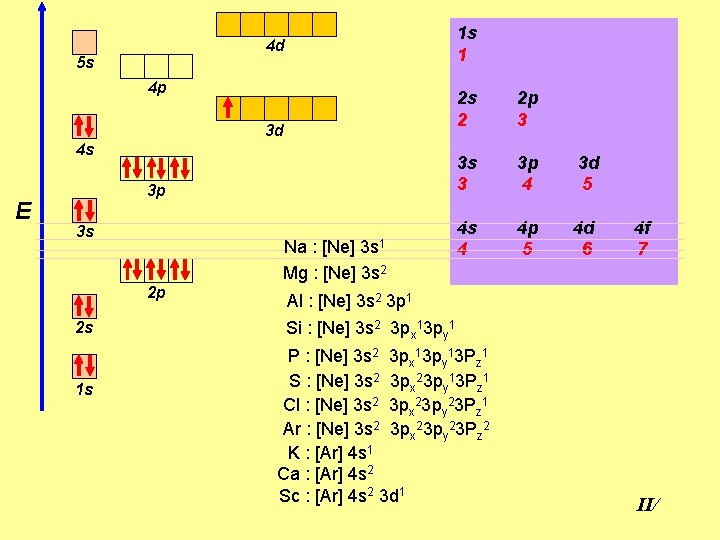
1 s 1 4 d 5 s 4 p 3 d 4 s E 3 p 3 s Na : [Ne] 3 s 1 Mg : [Ne] 3 s 2 2 p 2 s 1 s 2 s 2 2 p 3 3 s 3 3 p 4 3 d 5 4 s 4 4 p 5 4 d 6 4 f 7 Al : [Ne] 3 s 2 3 p 1 Si : [Ne] 3 s 2 3 px 13 py 1 P : [Ne] 3 s 2 3 px 13 py 13 Pz 1 S : [Ne] 3 s 2 3 px 23 py 13 Pz 1 Cl : [Ne] 3 s 2 3 px 23 py 23 Pz 1 Ar : [Ne] 3 s 2 3 px 23 py 23 Pz 2 K : [Ar] 4 s 1 Ca : [Ar] 4 s 2 Sc : [Ar] 4 s 2 3 d 1 II/
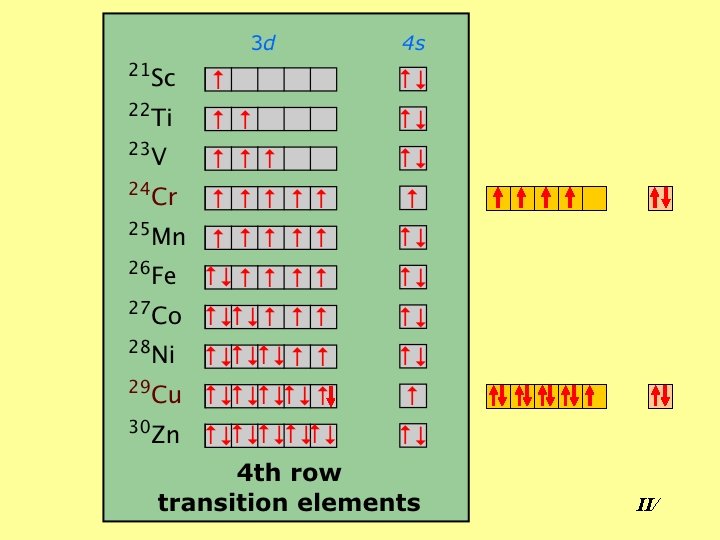
II/
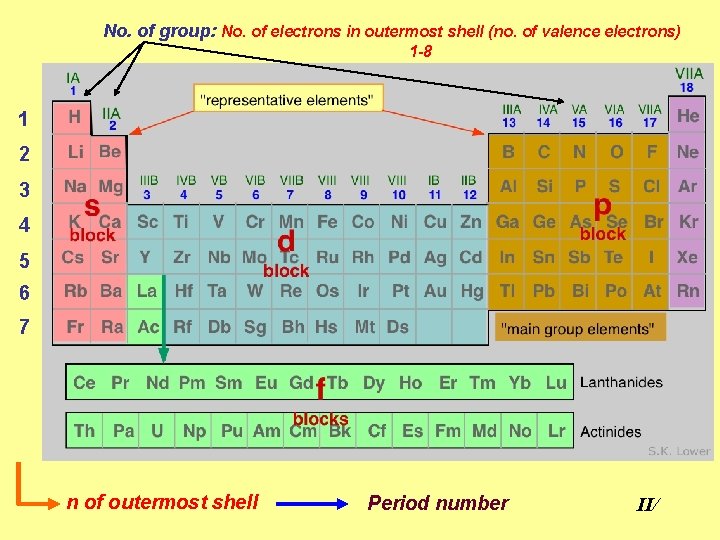
No. of group: No. of electrons in outermost shell (no. of valence electrons) 1 -8 1 2 3 4 5 6 7 n of outermost shell Period number II/

• Valence shell: outermost occupied main shell • Valence electrons: electrons that occupy the outermost main shell electrons with highest n determines the chemistry of element II/
![Electron configuration from periodic table • Se [Ar] 4 s 1 3 d 10 Electron configuration from periodic table • Se [Ar] 4 s 1 3 d 10](http://slidetodoc.com/presentation_image/423db93771e469cbb6ee8edde4faf183/image-13.jpg)
Electron configuration from periodic table • Se [Ar] 4 s 1 3 d 10 • Ru [Kr] 5 s 2 4 d 6 • In [Kr] 5 s 2 4 d 10 5 p 1 • Ba [Xe] 6 s 2 • Au [Xe] 6 s 2 [Xe] 6 s 1 4 f 14 5 d 9 5 d 10 4 p 4 II/
- Slides: 13