Orbital Diagrams Electron Configurations A 4 Quantum Numbers

Orbital Diagrams & Electron Configurations

A. 4 Quantum Numbers (used to describe electrons) 1. Principle Energy Levels (there are 7) 2. Sublevels (s, p, d, f) Principle energy levels are divided into these 3. Orbitals (1, 3, 5, 7) Region or space an electron occupies Different shapes depending on sublevel 4. Spin 2 electrons have opposite spin
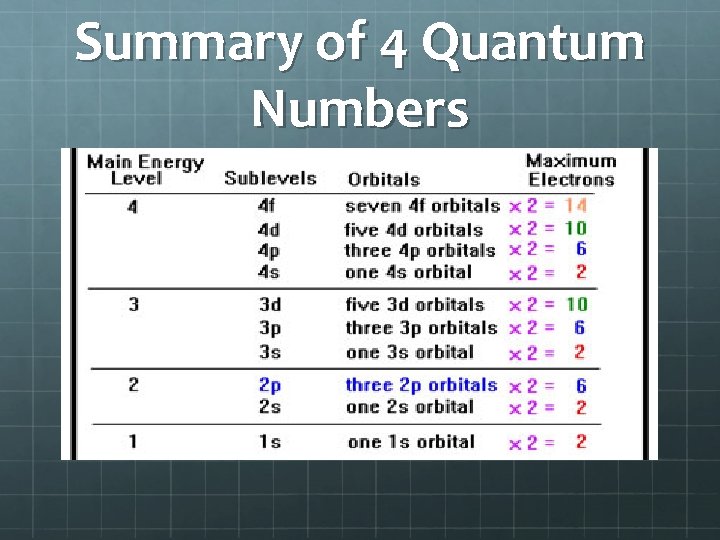
Summary of 4 Quantum Numbers
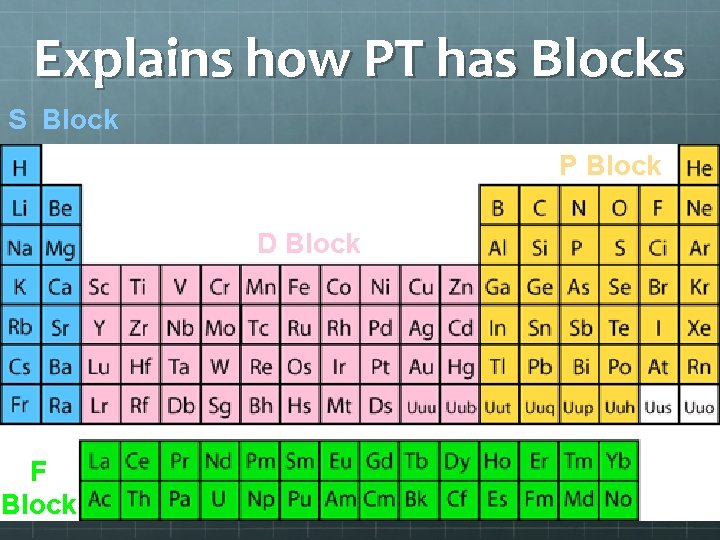
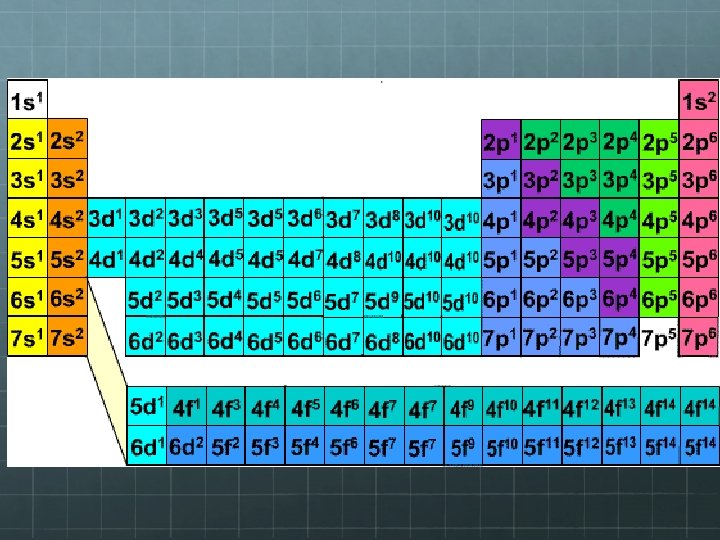
Explains how PT has Blocks S Block P Block D Block F Block
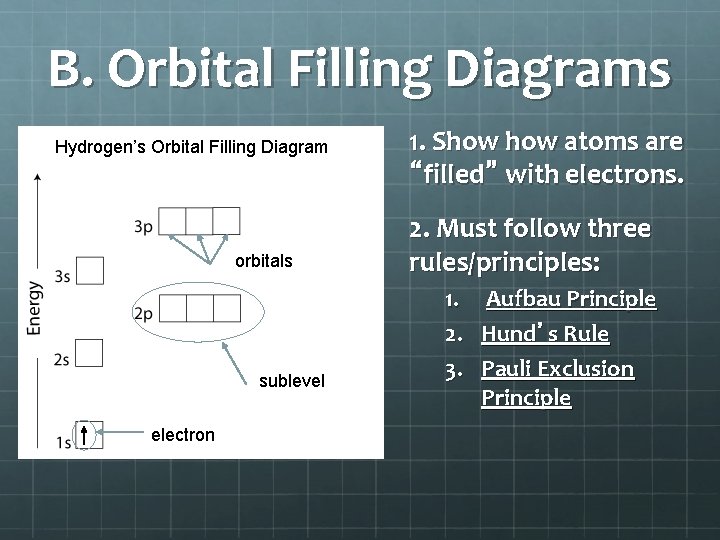
B. Orbital Filling Diagrams Hydrogen’s Orbital Filling Diagram orbitals sublevel electron 1. Show atoms are “filled” with electrons. 2. Must follow three rules/principles: 1. Aufbau Principle 2. Hund’s Rule 3. Pauli Exclusion Principle
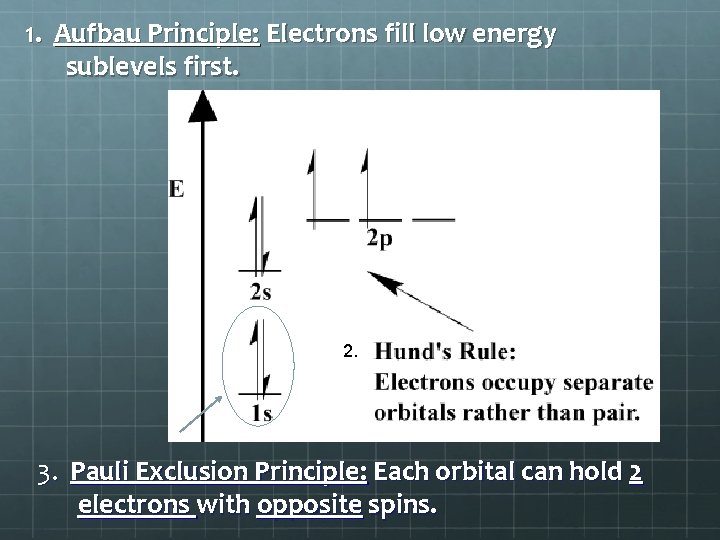
1. Aufbau Principle: Electrons fill low energy sublevels first. 2. 3. Pauli Exclusion Principle: Each orbital can hold 2 electrons with opposite spins.

3. Order of Filling Follow the diagonal arrows from top to bottom
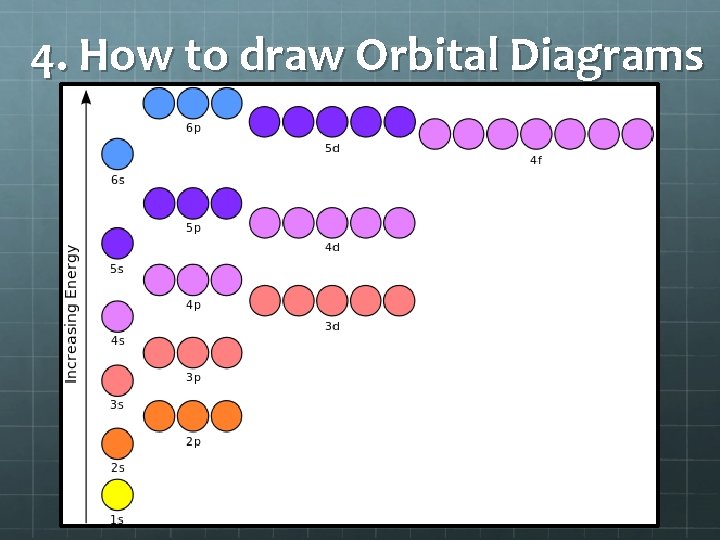
4. How to draw Orbital Diagrams

Example Problems Let’s do these problems together (write them down in your notebook): Helium (z = 2) (z = atomic #) Sodium (z = 11) Now try these on your own (write down in your notebook): Nitrogen (z = 7) Chlorine (z = 17)

C. Electron Configurations 1. What are they? Electron configurations are the arrangement of electrons in an atom Shorthand notation for information given in Orbital Diagrams

2. Rules for writing Electron Configurations A. Determine how many electrons are in the atom (DRAW ORBITAL DIAGRAM) B. Arrange the energy sublevels according to increasing energy C. Fill each sublevel with electrons until all the electrons in the atom are used up

3. Examples Let’s do these together: Li (z = 3) S (z = 16) V (z = 23) You do these on your own: Na (z = 11) Ar (z = 18) Mn (z = 25) • Orbital Diagram • Electron Configuration • Valence electron (if NOT a Transition Metal) • Honors only: Noble Gas Configuration

HONORS ONLY: Noble Gas Configuration SHORT HAND Instead of writing out all of the electrons in the configuration, you can write out just the ones since the last noble gas. Ex: Carbon Find C on PT Go backwards until you reach a noble gas. (He) Use shorthand to indicate all of the electrons that are identical to helium's configuration by putting He in square brackets [ ], and substituting it for those electrons. [He]2 s 22 p 2
- Slides: 14