Oral Rivaroxaban for Symptomatic Venous Thromboembolism Background o

Oral Rivaroxaban for Symptomatic Venous Thromboembolism

Background o Tx ↓ risk of recurrence from 25% to 3% during first 6 -12 months of therapy o Risk after tx ends: 5 -10% during first year o Standard: Parenteral heparin initially + Vit K antagonist o Annual risk of major bleeding after first year 1 -2%

Background o Monitoring challenging for outpatients o Solution: oral anticoagulant without monitoring o Rivaroxaban: Direct factor Xa inhibitor

Methods: Acute DVT o Design: Randomized, open label, event driven, non-inferiority study o P: Pts with acute, symptomatic DVT o I: Rivaroxaban o C: Enoxaparin + Vit K antagonist o O: Symptomatic, recurrent VT

Acute DVT: Inclusion Criteria o Legal age for consent o Acute, symptomatic objectively confirmed proximal DVT, without symptomatic PE

Acute DVT: Exclusion Criteria o If therapeutic doses of LMWH, fondaparinux or UFH received for >48 hours or >1 dose of a VKA before randomization o Treated with thrombectomy, vena cava filter or a fibrinolytic agent for the current episode of thrombosis o Any CIs

Methods: Acute DVT o Duration of tx: determined by the treating physician o 15 mg BID X 3 wks o 20 mg OD for 3, 6 or 12 months o Standard: SC enoxaparin ( 1 mg/kg BID) o Warfarin or acenocoumarol started within 48 hrs of randomization

Methods: Continued Tx Study o Design: Randomized, double-blind (subject, caregiver, investigator, outcomes assessor), event-driven superiority study o P: Pts with DVT or PE treated x 6 -12 months with a Vit K antagonist or rivaroxaban o I: Rivaroxaban 20 mg OD o C: Placebo o O: Symptomatic, Recurrent VT

Continued Tx Study: Inclusion Criteria o Objectively confirmed symptomatic DVT or PE o Treated X 6 -12 months with acenocoumarol or warfarin or rivaroxaban o If there was equipoise with respect to the need for continued anticoagulation

Exclusion criteria for both studies o Another indication for Vit K antagonist o Cr. Cl< 30 ml/min o Clinically significant liver disease (acute hepatitis, chronic active hepatitis, or cirrhosis) or an ALT>3 ULN o Bacterial endocarditis o Active bleeding or a high risk of bleeding

Exclusion Criteria for both studies o CI anticoagulant treatment o SBP >180 mm Hg or DBP>110 mm Hg o Childbearing potential without proper contraception measures o Pregnancy or breast feeding o Concomitant use of strong P-450 3 A 4 inhibitors or inducers o Participation in another experimental pharmacotherapeutic program within 30 days before screening o Life expectancy <3 months

Methods o Continued Tx: Rivaroxaban 20 mg OD or matching placebo for 6 or 12 months o Both studies: NSAID & antiplatelet use discouraged o If indicated ASA (up to 100 mg), clopidogrel (75 mg) or both allowed

Outcome assessments o 1⁰ efficacy outcome: Symptomatic, recurrent VT o Acute DVT Study: n Principal safety outcome: Clinically relevant bleeding = composite of major or clinically relevant nonmajor bleeding o Continued Treatment Study: n Major bleeding

Outcome assessments o Predefined 2⁰ outcome: n All-cause mortality n Vascular events (ACS, ischemic stroke, TIA or SE) n Net clinical benefit (composite of primary efficacy outcome +major bleeding)

Statistical Analysis: Acute DVT o Event driven, Non-inferiority Study o Assumption: equal efficacy in 2 study groups n A total of 88 events would provide a power of 90% to demonstrate that rivaroxaban is non inferior to standard therapy o Margin = 2. 0, corresponds to maintenance of at least 50% of the proven efficacy of standard tx

Statistical Analysis: Continued Tx o Event driven, superiority study o Assumption: 70% RR with rivaroxaban n 30 events, power of 90%
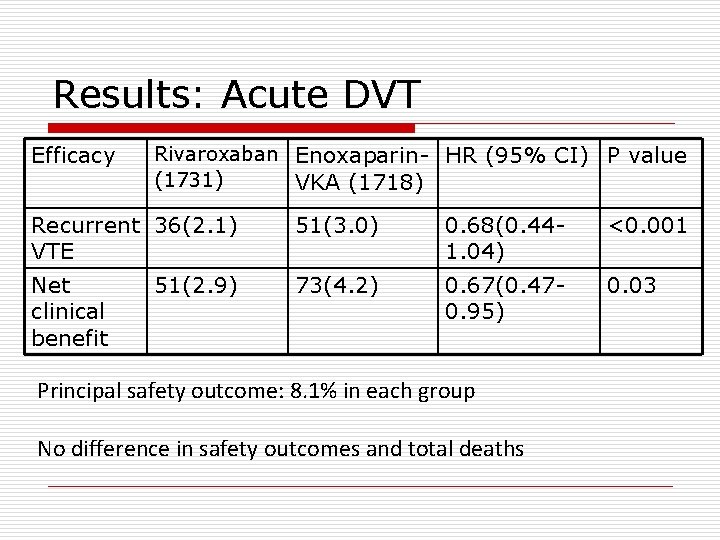
Results: Acute DVT Efficacy Rivaroxaban Enoxaparin- HR (95% CI) P value (1731) VKA (1718) Recurrent 36(2. 1) VTE 51(3. 0) 0. 68(0. 441. 04) <0. 001 Net clinical benefit 73(4. 2) 0. 67(0. 470. 95) 0. 03 51(2. 9) Principal safety outcome: 8. 1% in each group No difference in safety outcomes and total deaths
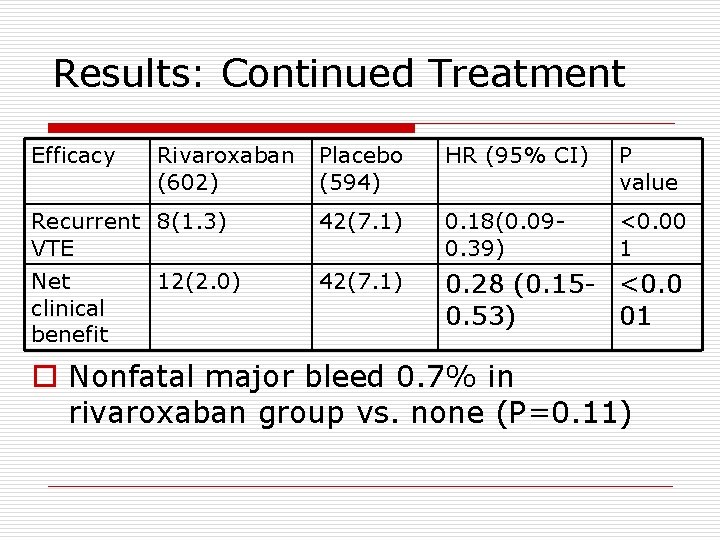
Results: Continued Treatment Efficacy Rivaroxaban (602) Placebo (594) HR (95% CI) P value Recurrent 8(1. 3) VTE 42(7. 1) 0. 18(0. 090. 39) <0. 00 1 Net clinical benefit 42(7. 1) 0. 28 (0. 15 - <0. 0 0. 53) 01 12(2. 0) o Nonfatal major bleed 0. 7% in rivaroxaban group vs. none (P=0. 11)

CASP SR Checklist o Did the study ask a clearly focused question? Yes o Was this a randomized controlled trial (RCT) and was it appropriately so? The first study is open label but the second one is RCT. Yes o Were participants appropriately allocated to intervention and control groups? Yes o Were participants, staff and study personnel ‘blind’ to participants’ study group? Outcomes Assessor blinded o Were all of the participants who entered the trial accounted for at its conclusion? Yes

CASP SR Checklist o Were the participants in all groups followed up and data collected in the same way? Yes o Did the study have enough participants to minimize the play of chance? Yes o How are the results presented and what is the main result? o How precise are these results? o Were all important outcomes considered so the results can be applied?

Limitations o Blinding: no protection from bias o. Suspected cases higher in rivaroxaban group o Margin of 2. 0 = at least 50% of proven efficacy of standard therapy. Acceptable? o On-treatment & per-protocol analyses similar to ITT but data not shown

Limitations o Safety: Bleeding events included in the analyses if occurred during tx or within 2 days after d/c o Compliance o Serious events not defined o Results of non-inferiority trial not as credible as a superiority trial

Implications to practice o Dose needs to be studied more o Single-drug approach to short-term & continued tx of VT n Option in patients not willing to do INR monitoring o Reversal of bleeding: no specific antidote, general hemostatic measures n Activated charcoal within 2 hours of dose o Highly protein bound not dialyzable
- Slides: 23