ORAL NANOCARRIER FOR INSULIN COLON DELIVERY Miriam Colombo

ORAL NANOCARRIER FOR INSULIN COLON DELIVERY Miriam Colombo, Ph. D Nano. Bio. Lab Dipartimento di Biotecnologie e Bioscienze Università di Milano-Bicocca, Italy miriam. colombo@unimib. it www. nanobiolab. btbs. unimib. it

Nanobiolab Unimib ü Design, synthesis, bio-functionalization and characterization of nanoparticles. M. Colombo et al. Angew. Chem. Int. Ed. , 2012. M. Colombo et al. Small, 2012. M. Colombo et al. Angew. Chem. Int. Ed. , 2012. S. Mazzucchelli et al. Angew. Chem. , Int. Ed. 2013. ü in vitro and in vivo investigation of targeting, biodistribution and biomedical potential (diagnostics and therapeutics) of multifunctional nanoparticles M. Colombo et al. Biomat. Science, 2013 S. Mazzucchelli Biomat. Science, 2013. L. Fiandra, et al. ACS Nano, 2013. New insights: • I’m looking into new nanoparticles administration methods as an intravenous one alternative to INTRODUCTION - M. Colombo Cream Formulation Impact on Topical Administration of Engineered Colloidal Nanoparticles, PLo. S ONE, 2015. - Colombo Impact of semi-solid formulations on skin penetration of iron oxide nanoparticles, J. Nanobiotechnol. 2017. - M. Colombo Oral delivery of insulin via polyethylene imine-based nanoparticlesfor colonic release allows glycemic control in diabetic rats. Pharm Res 2016
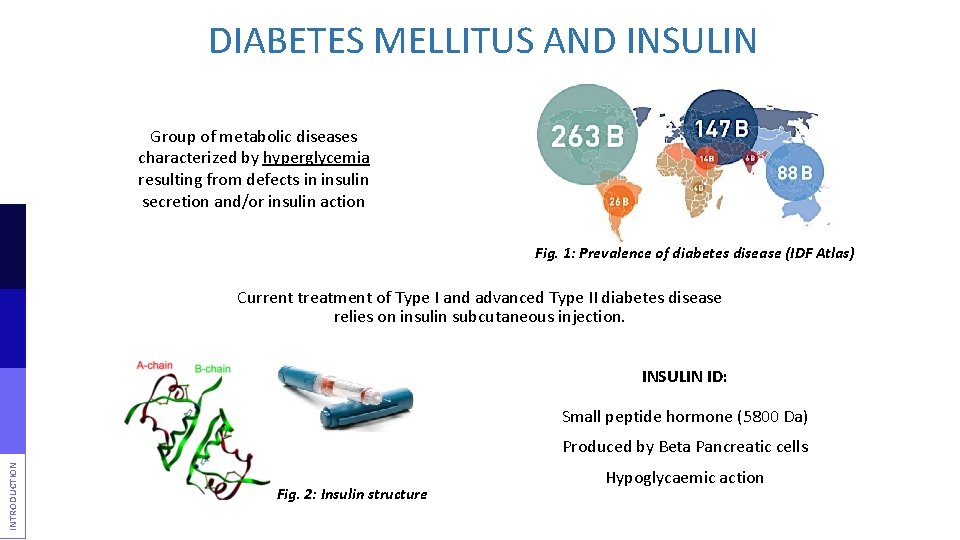
DIABETES MELLITUS AND INSULIN Group of metabolic diseases characterized by hyperglycemia resulting from defects in insulin secretion and/or insulin action Fig. 1: Prevalence of diabetes disease (IDF Atlas) Current treatment of Type I and advanced Type II diabetes disease relies on insulin subcutaneous injection. INSULIN ID: Small peptide hormone (5800 Da) INTRODUCTION Produced by Beta Pancreatic cells Fig. 2: Insulin structure Hypoglycaemic action
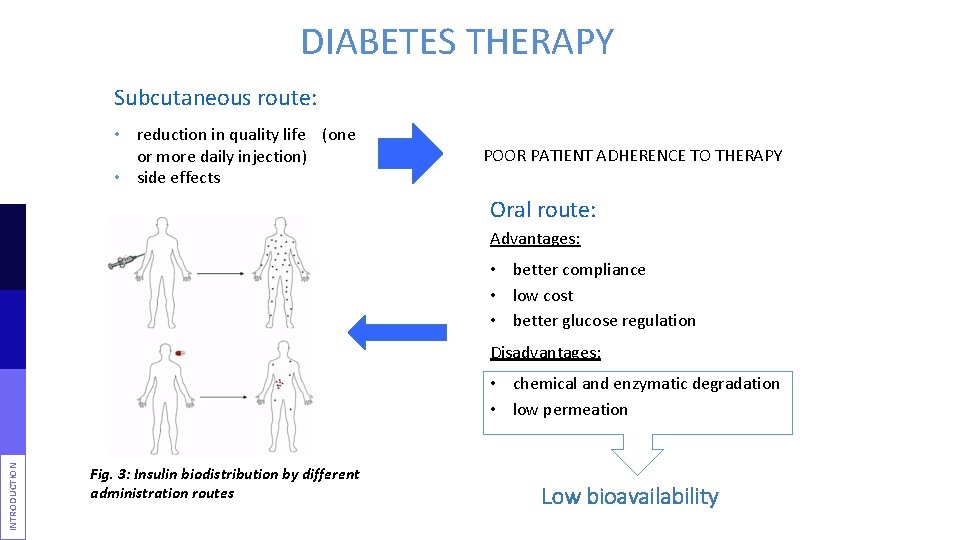
DIABETES THERAPY Subcutaneous route: • reduction in quality life (one or more daily injection) • side effects POOR PATIENT ADHERENCE TO THERAPY Oral route: Advantages: • better compliance • low cost • better glucose regulation Disadvantages: INTRODUCTION • chemical and enzymatic degradation • low permeation Fig. 3: Insulin biodistribution by different administration routes Low bioavailability
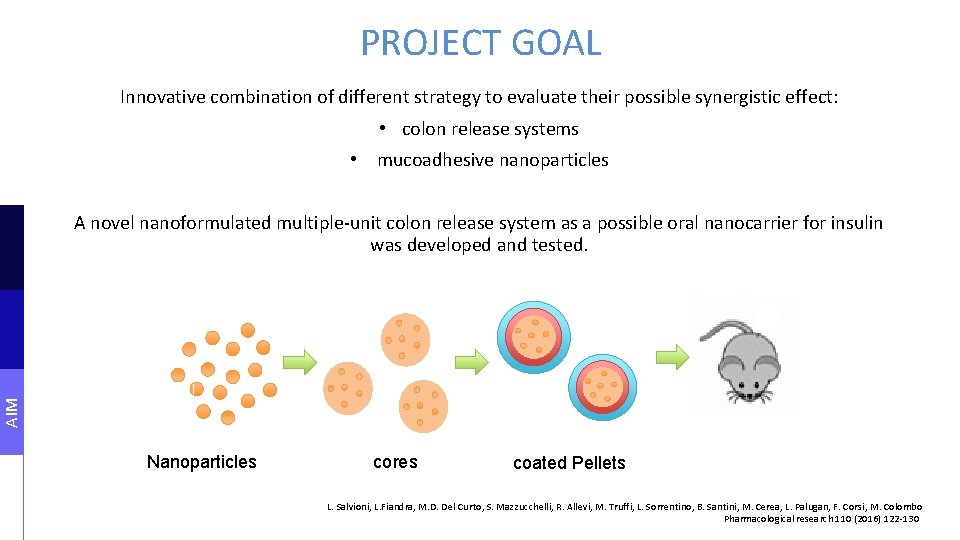
PROJECT GOAL Innovative combination of different strategy to evaluate their possible synergistic effect: • colon release systems • mucoadhesive nanoparticles A novel nanoformulated multiple-unit colon release system as a possible oral nanocarrier for insulin was developed and tested. AIM Nanoparticles cores coated Pellets L. Salvioni, L. Fiandra, M. D. Del Curto, S. Mazzucchelli, R. Allevi, M. Truffi, L. Sorrentino, B. Santini, M. Cerea, L. Palugan, F. Corsi, M. Colombo Pharmacological research 110 (2016) 122 -130
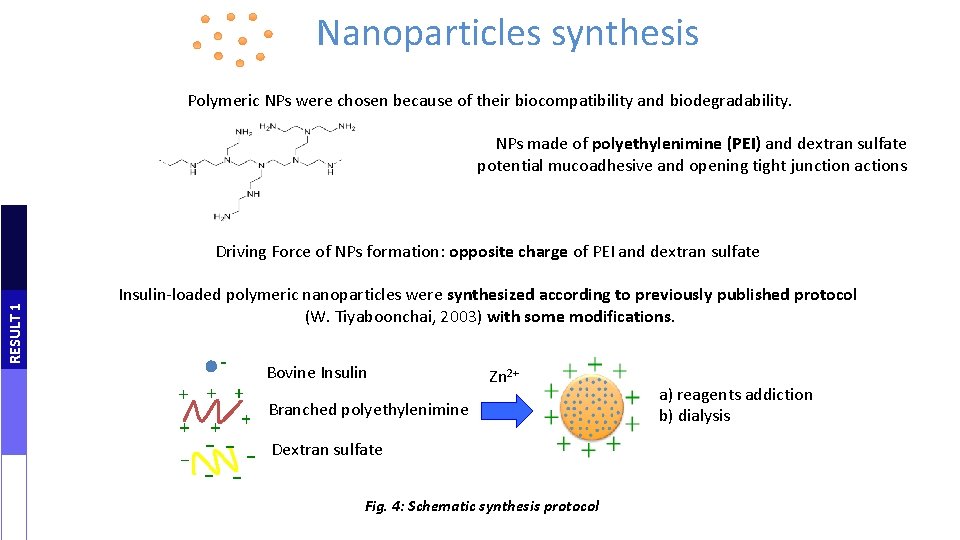
Nanoparticles synthesis Polymeric NPs were chosen because of their biocompatibility and biodegradability. NPs made of polyethylenimine (PEI) and dextran sulfate potential mucoadhesive and opening tight junction actions RESULT 1 Driving Force of NPs formation: opposite charge of PEI and dextran sulfate Insulin-loaded polymeric nanoparticles were synthesized according to previously published protocol (W. Tiyaboonchai, 2003) with some modifications. Bovine Insulin Zn 2+ Branched polyethylenimine Dextran sulfate Fig. 4: Schematic synthesis protocol a) reagents addiction b) dialysis
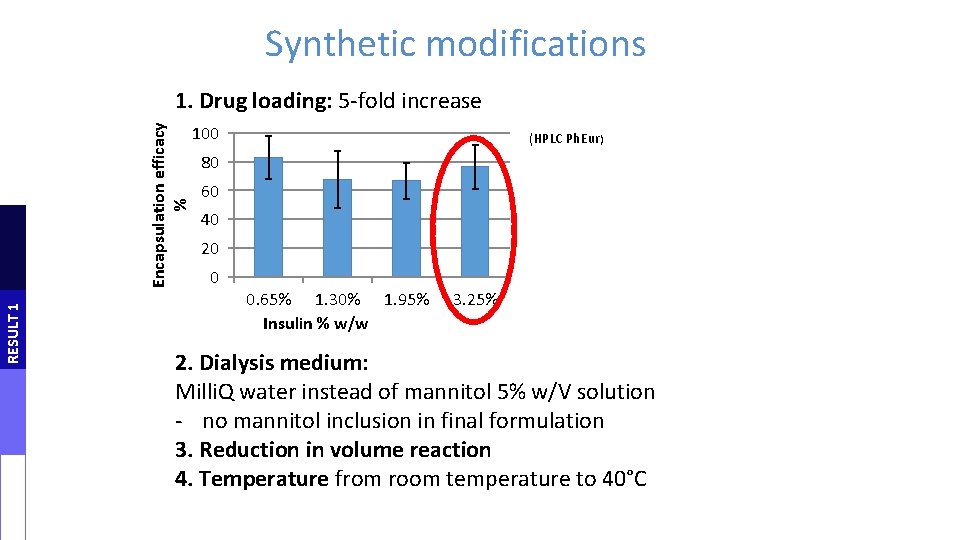
Synthetic modifications RESULT 1 Encapsulation efficacy % 1. Drug loading: 5 -fold increase 100 (HPLC Ph. Eur) 80 60 40 20 0 0. 65% 1. 30% 1. 95% Insulin % w/w 3. 25% 2. Dialysis medium: Milli. Q water instead of mannitol 5% w/V solution - no mannitol inclusion in final formulation 3. Reduction in volume reaction 4. Temperature from room temperature to 40°C
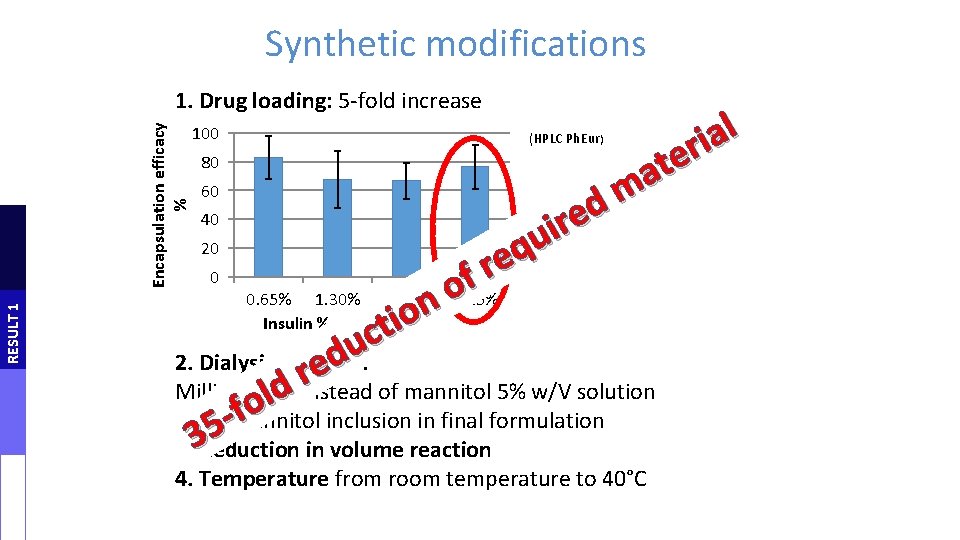
Synthetic modifications RESULT 1 Encapsulation efficacy % 1. Drug loading: 5 -fold increase 100 80 60 (HPLC Ph. Eur) l a i r e t a m d e r i u 20 q e r 0 f o 0. 65% 1. 30% 1. 95% 3. 25% n o Insulin % w/w i t c u d 2. Dialysis medium: e r Milli. Q water instead of mannitol 5% w/V solution d l o f - no mannitol inclusion in final formulation 5 3 3. Reduction in volume reaction 40 4. Temperature from room temperature to 40°C
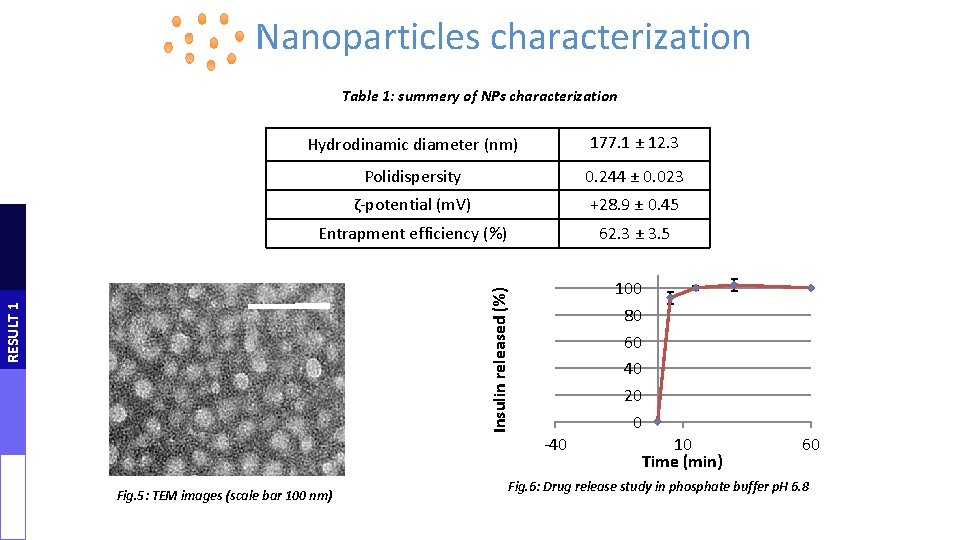
Nanoparticles characterization 177. 1 ± 12. 3 Polidispersity 0. 244 ± 0. 023 ζ-potential (m. V) +28. 9 ± 0. 45 Entrapment efficiency (%) 62. 3 ± 3. 5 Insulin released (%) Hydrodinamic diameter (nm) Time (min)
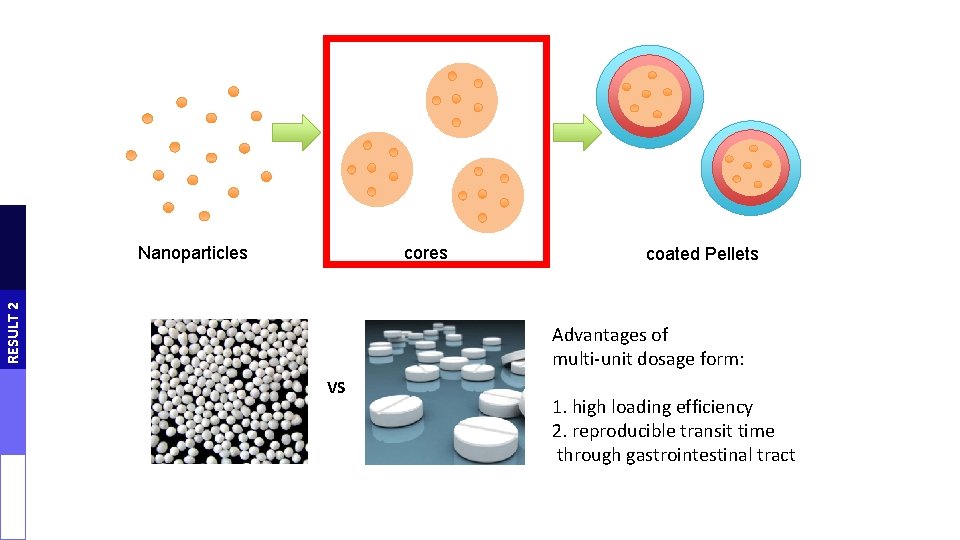
cores RESULT 2 Nanoparticles coated Pellets Advantages of multi-unit dosage form: VS 1. high loading efficiency 2. reproducible transit time through gastrointestinal tract

Preparation of cores Cores were prepared by extrusion and spheronization processes dough formation spheronization extrusion (850 mm sieve ) drying (40°C, 24 h) RESULT 2 Table 2: core composition Formulation 1 (%w/w) Formulation 2 (%w/w) Nanoparticles 43. 2 Avicel® CL 611 22. 7 43. 2 Lactose 13. 8 - Explotab® CLV 6. 7 - Sodium Glycocholate 13. 6 In formulation , Lactose acts as pore former and Explotab® is a superdisgregant To compare formulations, in vitro disintegration and dissolution test were performed.
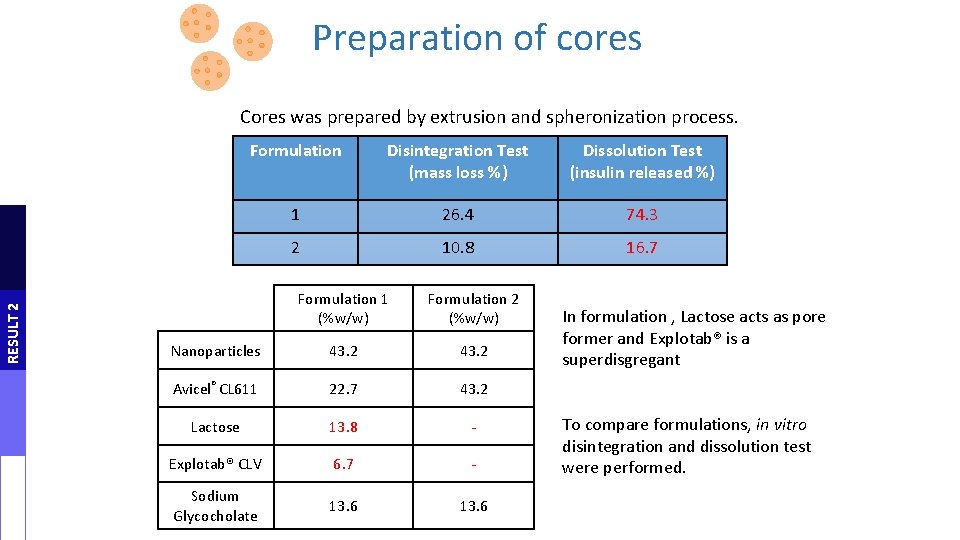
Preparation of cores RESULT 2 Cores was prepared by extrusion and spheronization process. spheronization dough extrusion Formulation Disintegration Test Dissolution Test (850 �� um sieve ) formation (mass loss %) (insulin released %) 1 26. 4 2 10. 8 Formulation 1 (%w/w) Formulation 2 (%w/w) Nanoparticles 43. 2 Avicel® CL 611 22. 7 43. 2 Lactose 13. 8 - Explotab® CLV 6. 7 - Sodium Glycocholate 13. 6 drying 74. 3 (40°C, 24 h) 16. 7 In formulation , Lactose acts as pore former and Explotab® is a superdisgregant To compare formulations, in vitro disintegration and dissolution test were performed.

Cores characterization RESULT 2 Table 2: core composition % w/w Nanoparticles 43. 2 Avicel® CL 611 22. 7 Lactose 13. 8 Explotab® CLV 6. 7 Sodium Glycocholate 13. 6 Fig. 7: Core digital photograph, scale bar 1 mm dgeo (μm) 1137 Aspect Ratio 1. 16 ± 0. 10 Yield (%) 61. 9 Insulin recovery (%) 88. 8 ± 3. 7 Re-dispersion studies were performed using DLS analysis: after disaggregation of cores, nanoparticles were detected in the aqueous medium
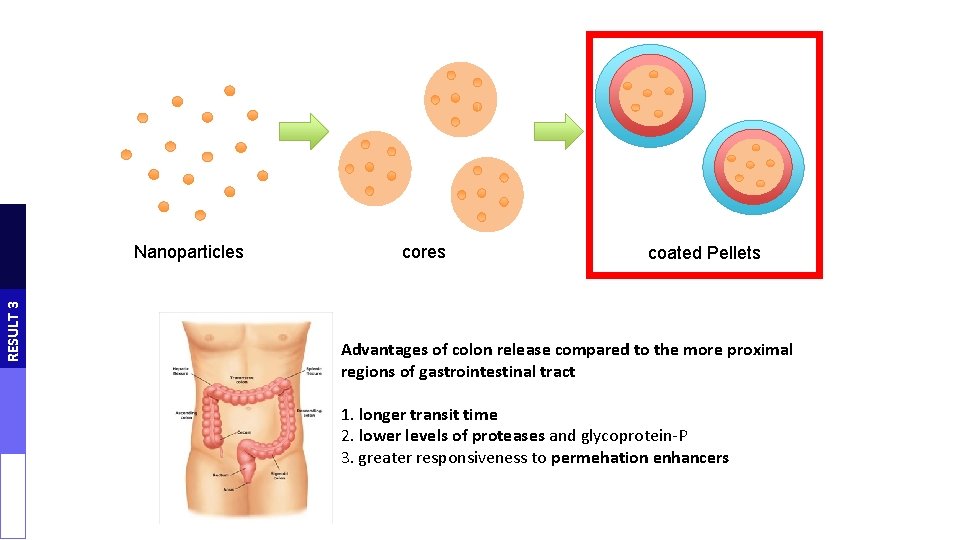
RESULT 3 Nanoparticles cores coated Pellets Advantages of colon release compared to the more proximal regions of gastrointestinal tract 1. longer transit time 2. lower levels of proteases and glycoprotein-P 3. greater responsiveness to permehation enhancers
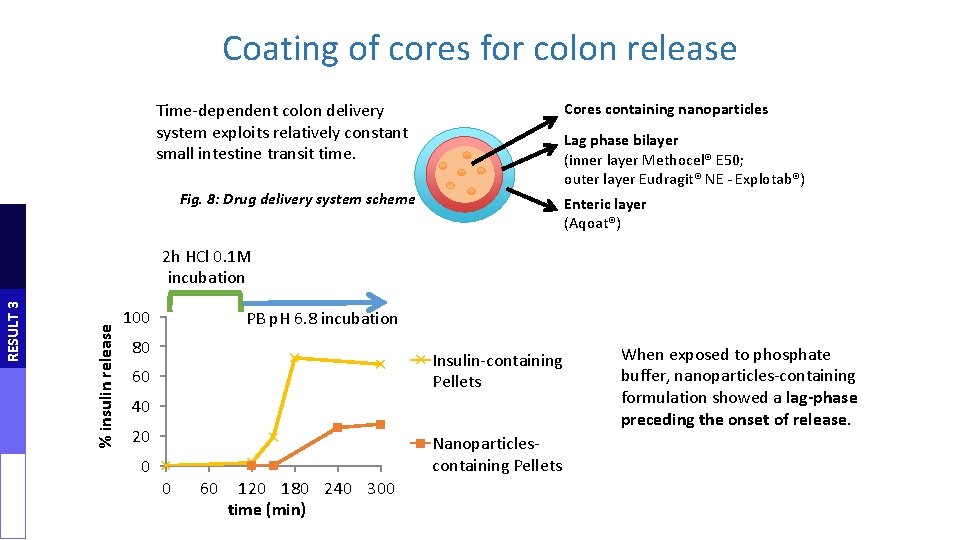
Coating of cores for colon release Time-dependent colon delivery system exploits relatively constant small intestine transit time. Cores containing nanoparticles Lag phase bilayer (inner layer Methocel® E 50; outer layer Eudragit® NE - Explotab®) Fig. 8: Drug delivery system scheme Enteric layer (Aqoat®) % insulin release RESULT 3 2 h HCl 0. 1 M incubation 100 PB p. H 6. 8 incubation 80 Insulin-containing Pellets 60 40 20 0 Nanoparticlescontaining Pellets 0 60 120 180 240 300 time (min) When exposed to phosphate buffer, nanoparticles-containing formulation showed a lag-phase preceding the onset of release.
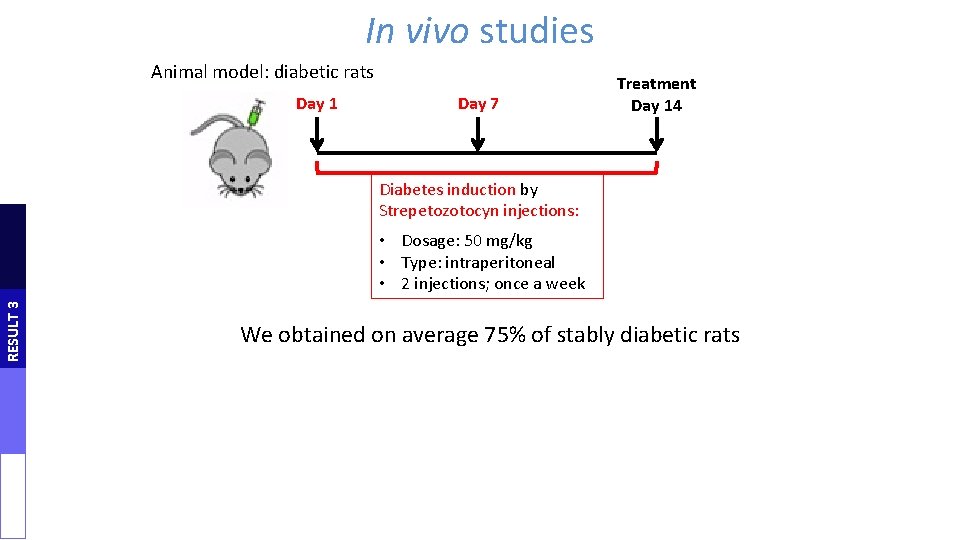
In vivo studies Animal model: diabetic rats Day 1 Day 7 Treatment Day 14 Diabetes induction by Strepetozotocyn injections: RESULT 3 • Dosage: 50 mg/kg • Type: intraperitoneal • 2 injections; once a week We obtained on average 75% of stably diabetic rats
Animal model: diabetic rats Day 1 In vivo studies Day 7 Treatment Day 14 RESULT 3 Evaluation of Colon-release formulation containing Insulin Nanoparticles in comparison with other formulations: • Subcutaneous injection (dose 0. 07 mg/Kg) • Others for oral administration (dose 1. 33 mg/Kg): 1. Free insulin 2. Insulin Nanoparticles 3. Gastroresistant formulation containing Insulin Nanoparticles 4. Gastroresistant formulation containing Insulin 5. Colon-release formulation containing Insulin
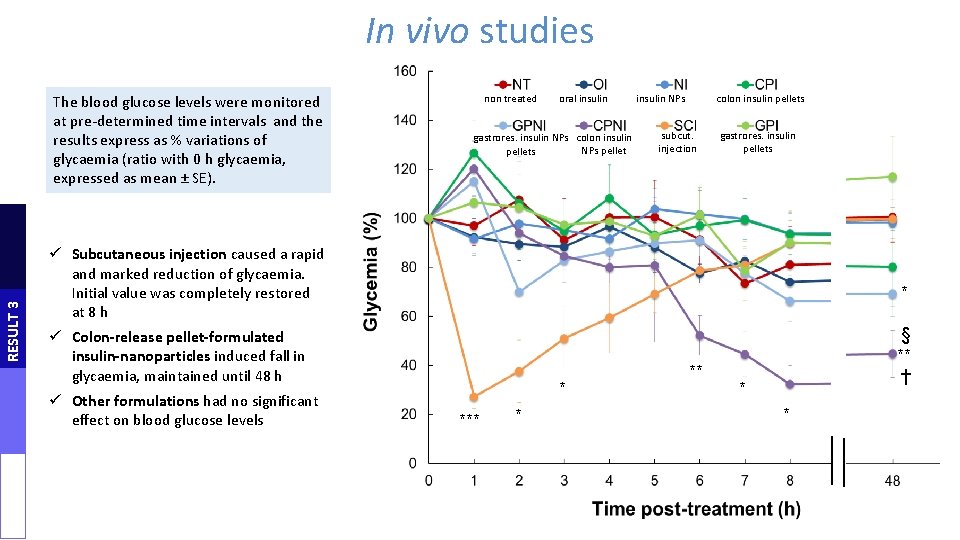
In vivo studies RESULT 3 The blood glucose levels were monitored at pre-determined time intervals and the results express as % variations of glycaemia (ratio with 0 h glycaemia, expressed as mean ± SE). non treated oral insulin gastrores. insulin NPs colon insulin NPs pellets insulin NPs colon insulin pellets subcut. injection gastrores. insulin pellets ü Subcutaneous injection caused a rapid and marked reduction of glycaemia. Initial value was completely restored at 8 h * ü Colon-release pellet-formulated insulin-nanoparticles induced fall in glycaemia, maintained until 48 h ü Other formulations had no significant effect on blood glucose levels * *** * ** § ** † * *

Conclusions CONCLUSIONS ü Novel nanoformulated multiple-unit colon release system of insulin was designed, developed and characterized ü Synergistic effect due of colon-release delivery and NPs use resulted in a significant and long-lasting hypoglycemic effect in vivo; ü Compared to standard treatment, our product showed prolonged hypoglycemic activity and a more physiological insulin metabolism. Perspective ü Delivery of other peptides with this system.

Acknowledgements Università di Milano-Bicocca Prof. Davide Prosperi Dr. Lucia Salvioni Dr. Benedetta Santini Università di Milano Dr. Luca Palugan Dr. Maria Dorly Del Curto L. Sacco Hospital M. D. Fabio Corsi Dr. Luisa Fiandra Nano. Bio. Lab Fondazione Regionale per la Ricerca Biomedica (FRRB)
- Slides: 20