Option B Biochemistry Jeff Venables Northwestern High School

Option B Biochemistry Jeff Venables Northwestern High School

Introduction to Biochemistry • The chemistry of living organisms is called biochemistry. • Biochemical molecules tend to be very large and difficult to synthesize. • Living organisms are highly ordered. Therefore, living organisms have very low entropy. • Most biologically important molecules are polymers, called biopolymers. • Biopolymers fall into three classes: proteins, carbohydrates, and nucleic acids.

Calorimetry • Energy content of food can be determined by calorimetry. • The food is burned in a calorimeter, and the increase in temperature of surrounding water is measured: q = m. CΔT q = heat (in joules) m = mass of water (in grams) C = specific heat of water (4. 184 J g-1 ºC-1) T = temperature change of water (in ºC)

Calorimetry • Example: 1. 13 g of rice raises the temperature of 525 g of water by 3. 31ºC. Determine the energy content in k. J per 100 g of rice. Q = m. C T = (525 g) (4. 184 J g-1 ºC-1) (3. 31ºC) = 7260 k. J Energy content is 7260 k. J / 1. 13 g rice = 6. 42 k. J per gram of rice Multiply by 100 g to get 642 k. J per 100 g of rice.

Proteins Amino Acids • Proteins are large molecules present in all cells. • They are made up of 2 -amino acids. (this means that the amine group is on carbon number 2, while the carboxylic acid group is on carbon number 1) • There are two forms of an amino acid: one that is neutral (with -NH 2 and -COOH groups) and one that is zwitterionic (with -NH 3+ and -COO- groups). • A zwitterion has both positive and negative charge in one molecule.
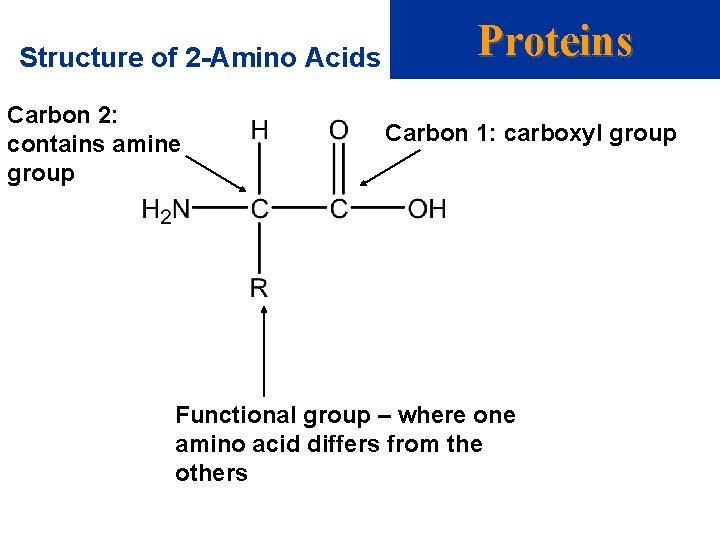
Structure of 2 -Amino Acids Carbon 2: contains amine group Proteins Carbon 1: carboxyl group Functional group – where one amino acid differs from the others
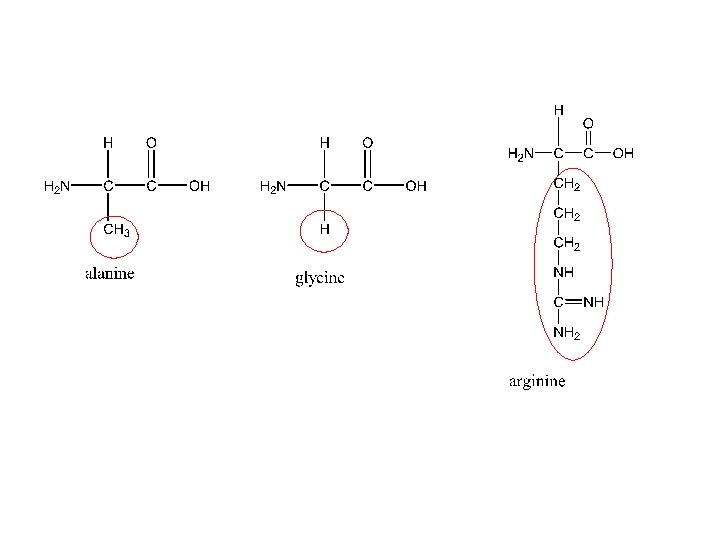

• There about 20 amino acids found in most proteins. • Each amino acid is assigned a three-letter abbreviation. • Amino acids are listed in the IB data booklet • Our bodies can synthesize about 10 amino acids. • Essential amino acids are the other 10 amino acids, which have to be ingested (part of our diet). • The -carbon (carbon 2) in all amino acids except glycine is chiral (has 4 different groups attached to it).
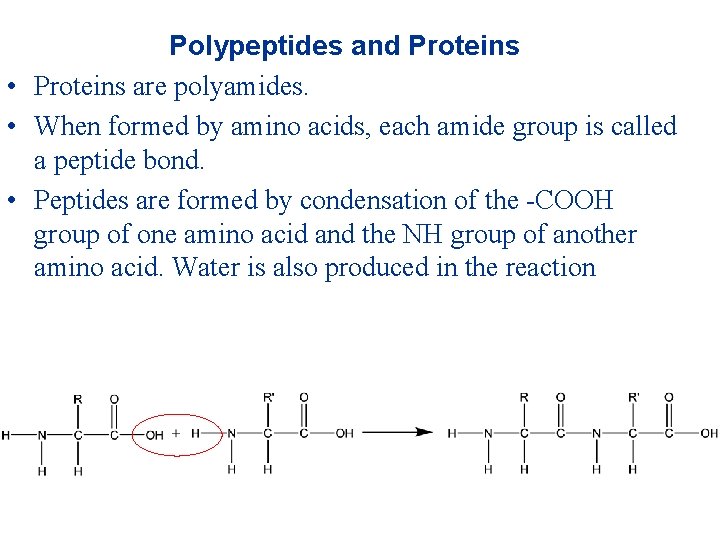
Polypeptides and Proteins • Proteins are polyamides. • When formed by amino acids, each amide group is called a peptide bond. • Peptides are formed by condensation of the -COOH group of one amino acid and the NH group of another amino acid. Water is also produced in the reaction

• The acid forming the peptide bond is named first. Example: if a dipeptide is formed from alanine and glycine so that the COOH group of glycine reacts with the NH group of alanine, then the dipeptide is called glycylalanine. • Glycylalanine is abbreviated gly-ala. • Polypeptides are formed with a large number of amino acids (usually result in proteins with molecular weights between 6000 and 50 million amu).
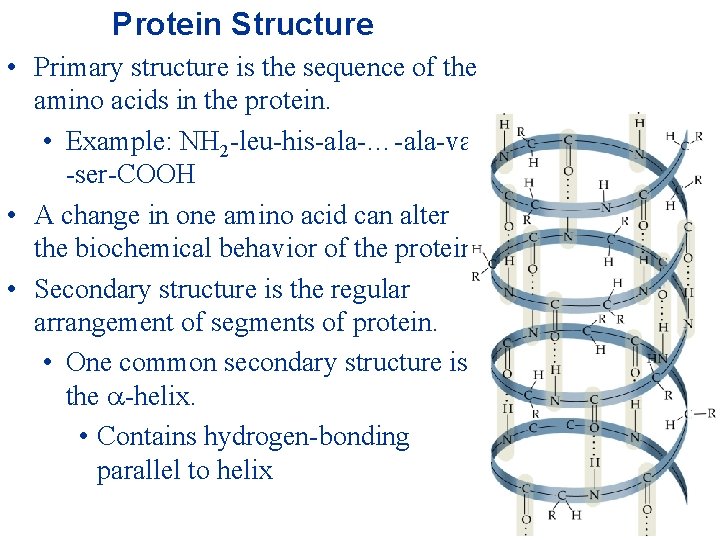
Protein Structure • Primary structure is the sequence of the amino acids in the protein. • Example: NH 2 -leu-his-ala-…-ala-val -ser-COOH • A change in one amino acid can alter the biochemical behavior of the protein. • Secondary structure is the regular arrangement of segments of protein. • One common secondary structure is the -helix. • Contains hydrogen-bonding parallel to helix
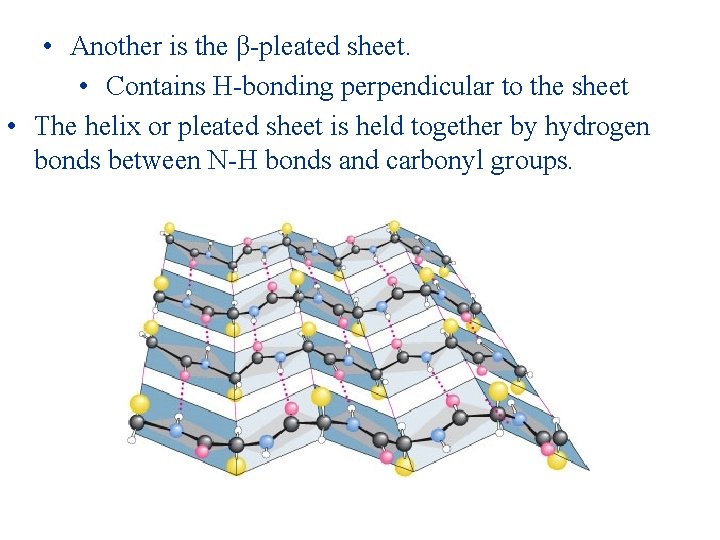
• Another is the β-pleated sheet. • Contains H-bonding perpendicular to the sheet • The helix or pleated sheet is held together by hydrogen bonds between N-H bonds and carbonyl groups.

Tertiary Structure is the overall shape of the protein. 1. Fibrous Proteins – provide strength for tissue (muscle, hair, cartilage) 2. Globular Proteins (sphere-shaped) • Transport and store nutrients • Catalyze reactions (enzymes) • Fight invasion • Participate in metabolism

Forces Affecting Tertiary Structure: • Ionic Bonding • Hydrogen Bonding • Covalent Bonds (disulfide linkage) • London Dispersion Forces • Dipole-dipole Forces Denaturation – A change in the function of a protein as a result of a change in tertiary structure.

Quaternary Structure • How multiple polypeptide chains are held together in large proteins containing more than one polypeptide molecule – Intermolecular forces (H-bonds, dipole, LDF)

Analysis of Proteins Paper Chromatography • Hydrolysis of protein (6 M HCl, 110°C) • Break peptide bonds, obtain amino acids • Place sample spot on paper, set paper in solvent • Spray ninhydrin to “develop” spots • Amino acids separate based on polarity

Electrophoresis – Separate amino acids based on isoelectric point (p. I) p. I = isoelectric point = the p. H at which positive and negative charges are balanced (no net charge on amino acid or polypeptide) – Depends on acid-base properties of “R” 1. Mixture of amino acids placed on gel (or paper) 2. Gel (or paper) is saturated with a buffer of known p. H. 3. Electric Current is applied

• If p. H = p. I, amino acid does not move • If p. H > p. I, amino acid moves toward “+” – Amino acid loses H+ in basic solution and becomes negative, moving toward anode. • If p. H < p. I, amino acid moves toward “-” – Amino acid gains H+ in acidic solution and becomes positive, moving toward cathode. • The further the p. H is from p. I, the faster the amino acid will move.

• Example – A mixture of 5 amino acids (shown below with p. I values) is to be separated by electrophoresis. A buffer with a p. H of 6. 0 is used. What will happen when the current is turned on? Cys Gln Gly His Lys 5. 1 5. 7 6. 0 7. 6 9. 7 + What if the buffer used has a p. H of 7. 0? -

Major Functions of Proteins 1. Structure – fibrous proteins • • Muscle, cartilage, skin, bones, hair, nails Collagen (skin), keratin (hair) 2. Enzymes – Catalyze specific chemical reactions in the body. 3. Movement and storage of energy and nutrients 4. Protection –antibodies 5. Control - hormones
- Slides: 20