Optimizing the Management of Chronic Obstructive Pulmonary Disease

Optimizing the Management of Chronic Obstructive Pulmonary Disease (COPD) ©Astra. Zeneca LP. All rights reserved. 275086 2/09
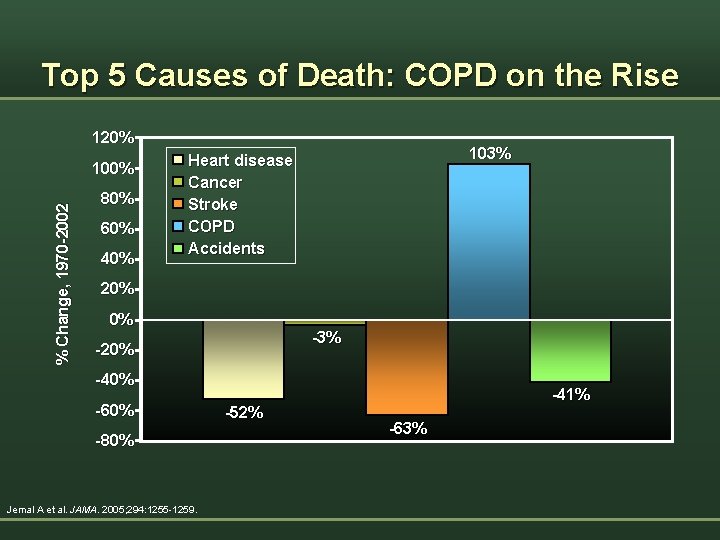
Top 5 Causes of Death: COPD on the Rise 120% % Change, 1970 -2002 100% 80% 60% 40% 103% Heart disease Cancer Stroke COPD Accidents 20% 0% -3% -20% -40% -60% -80% Jemal A et al. JAMA. 2005; 294: 1255 -1259. -52% -41% -63%
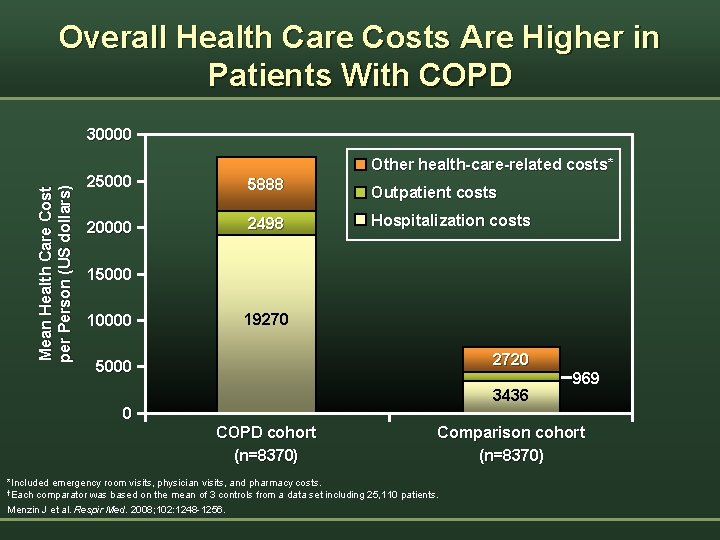
Overall Health Care Costs Are Higher in Patients With COPD Mean Health Care Cost per Person (US dollars) 30000 Other health-care-related costs* 25000 5888 20000 2498 Outpatient costs Hospitalization costs 15000 10000 19270 2720 5000 3436 0 COPD cohort (n=8370) 969 Comparison cohort (n=8370) *Included emergency room visits, physician visits, and pharmacy costs. †Each comparator was based on the mean of 3 controls from a data set including 25, 110 patients. Menzin J et al. Respir Med. 2008; 102: 1248 -1256.

Where Can We Improve COPD Management? COPD remains underdiagnosed 1 • Awareness of COPD guidelines suboptimal 2 • Spirometry used inconsistently 2 • Mortality increasing among women 3 • Present in the fifth decade of life, 4 though early symptoms may be missed 2 Early diagnosis and treatment may lead to better outcomes 5 Diagnosis and treatment may prevent or delay progression of lung function decline and symptoms 5 • Spirometry is key to the diagnosis of COPD 5 • Education represents an opportunity to improve outcomes for COPD patients 2, 5 1. Mannino DM et al. MMWR Surveillance Summary. 2002; 51: 1 -16. 2. Yawn BP, Wollan PC. Int J COPD. 2008; 3: 311 -317. 3. Deaths from chronic obstructive pulmonary disease—United States, 2000 -2005. MMWR Morb Mortal Wkly Rep. 2008; 57: 1229 -1232. 4. American Association for Respiratory Care. http: //www. aarc. org/resources/confronting_copd/exesum. pdf. Accessed February 11, 2009. 5. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. Updated 2008. http: //www. goldcopd. com/Guidelineitem. asp? l 1=2&l 2=1&int. Id=989. Accessed November 21, 2008.
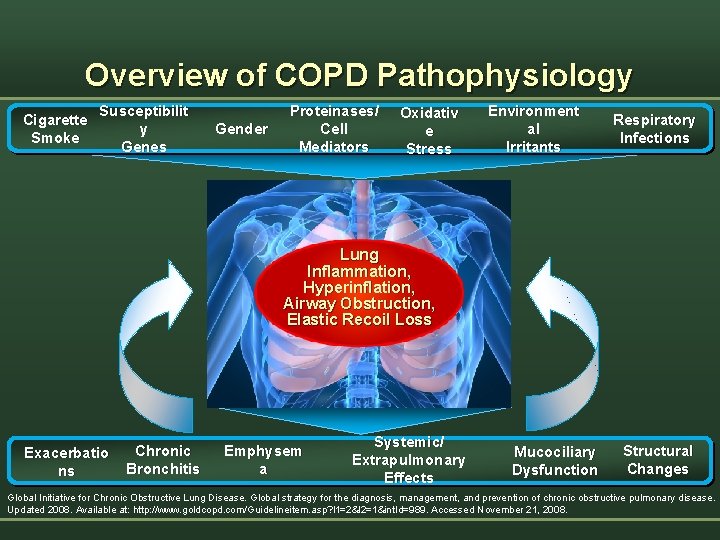
Overview of COPD Pathophysiology Cigarette Smoke Susceptibilit y Genes Gender Proteinases/ Cell Mediators Oxidativ e Stress Environment al Irritants Respiratory Infections Lung Inflammation, Hyperinflation, Airway Obstruction, Elastic Recoil Loss Exacerbatio ns Chronic Bronchitis Emphysem a Systemic/ Extrapulmonary Effects Mucociliary Dysfunction Structural Changes Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. Updated 2008. Available at: http: //www. goldcopd. com/Guidelineitem. asp? l 1=2&l 2=1&int. Id=989. Accessed November 21, 2008.
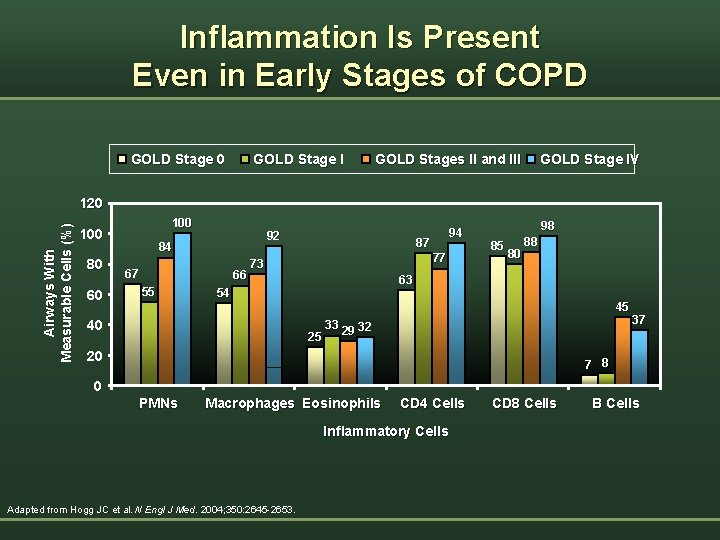
Inflammation Is Present Even in Early Stages of COPD GOLD Stage 0 GOLD Stage I GOLD Stages II and III GOLD Stage IV Airways With Measurable Cells (%) 120 100 80 60 92 84 67 66 55 94 87 77 73 98 85 80 88 63 54 45 40 25 33 29 32 20 37 7 8 0 PMNs Macrophages Eosinophils CD 4 Cells Inflammatory Cells Adapted from Hogg JC et al. N Engl J Med. 2004; 350: 2645 -2653. CD 8 Cells B Cells
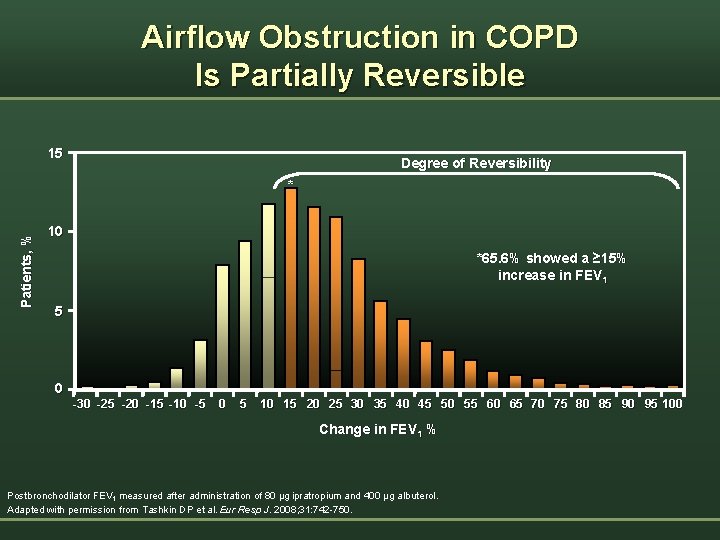
Airflow Obstruction in COPD Is Partially Reversible 15 Degree of Reversibility Patients, % * 10 *65. 6% showed a ≥ 15% increase in FEV 1 5 0 -30 -25 -20 -15 -10 -5 0 5 10 15 20 25 30 35 40 45 50 55 60 65 70 75 80 85 90 95 100 Change in FEV 1 % Postbronchodilator FEV 1 measured after administration of 80 µg ipratropium and 400 µg albuterol. Adapted with permission from Tashkin DP et al. Eur Resp J. 2008; 31: 742 -750.

Spirometry Is Essential for Diagnosing COPD If. . . Chronic symptoms = cough, sputum, and/or shortness of breath And. . . Exposure to risk factors = tobacco, occupational irritants, and/or indoor/outdoor pollution Then. . . Spirometry* to confirm COPD diagnosis • FEV 1/FVC <0. 70 • FEV 1 determines staging *Additional testing: chest x-ray, echocardiogram, arterial blood gas, sputum analysis, computed tomography (CT) scan. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. Updated 2008. http: //www. goldcopd. com/Guidelineitem. asp? l 1=2&l 2=1&int. Id=989. Accessed November 21, 2008.
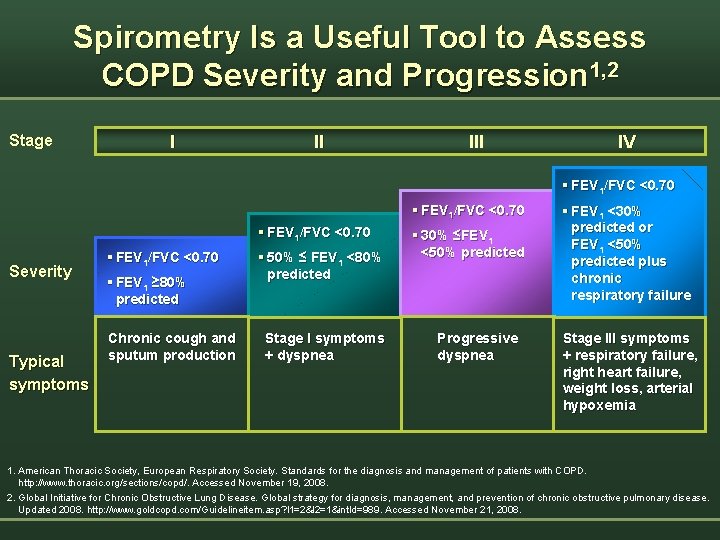
Spirometry Is a Useful Tool to Assess COPD Severity and Progression 1, 2 Stage I II IV § FEV 1/FVC <0. 70 Severity Typical symptoms § FEV 1/FVC <0. 70 § FEV 1 ≥ 80% predicted Chronic cough and sputum production § 50% ≤ FEV 1 <80% predicted Stage I symptoms + dyspnea § 30% ≤FEV 1 <50% predicted Progressive dyspnea § FEV 1 <30% predicted or FEV 1 <50% predicted plus chronic respiratory failure Stage III symptoms + respiratory failure, right heart failure, weight loss, arterial hypoxemia 1. American Thoracic Society, European Respiratory Society. Standards for the diagnosis and management of patients with COPD. http: //www. thoracic. org/sections/copd/. Accessed November 19, 2008. 2. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for diagnosis, management, and prevention of chronic obstructive pulmonary disease. Updated 2008. http: //www. goldcopd. com/Guidelineitem. asp? l 1=2&l 2=1&int. Id=989. Accessed November 21, 2008.

Goals of COPD Management • Relieve symptoms Short Term • Minimize side effects • Improve exercise tolerance and • Prevent and treat exacerbations and complications • Improve health status Long Term • Prevent disease progression • Reduce mortality COPD management includes both pharmacologic and nonpharmacologic treatment. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. Updated 2008. http: //www. goldcopd. com/Guidelineitem. asp? l 1=2&l 2=1&int. Id=989. Accessed November 21, 2008.
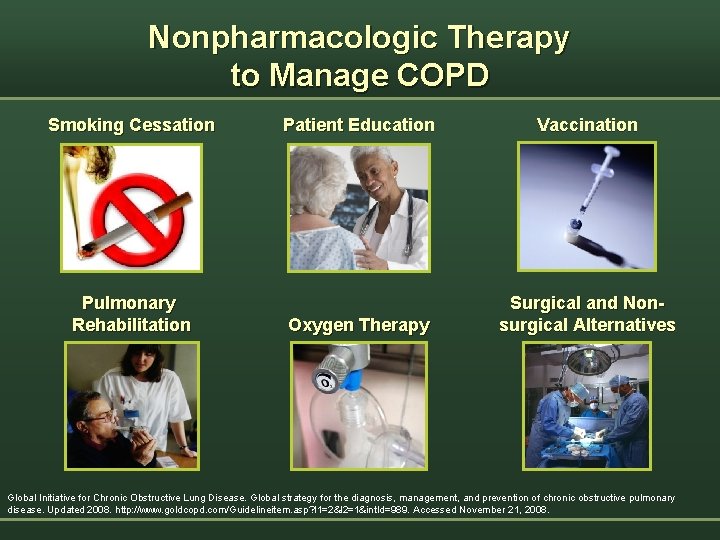
Nonpharmacologic Therapy to Manage COPD Smoking Cessation Pulmonary Rehabilitation Patient Education Vaccination Oxygen Therapy Surgical and Nonsurgical Alternatives Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. Updated 2008. http: //www. goldcopd. com/Guidelineitem. asp? l 1=2&l 2=1&int. Id=989. Accessed November 21, 2008.

Pharmacologic Therapy to Manage COPD Stage I II IV § FEV 1/FVC <0. 70 § 30% ≤FEV 1 <50% predicted § FEV 1/FVC <0. 70 Severity § FEV 1/FVC <0. 70 § FEV 1 ≥ 80% predicted § 50% ≤FEV 1 <80% predicted § FEV 1 <30% predicted or FEV 1 <50% predicted plus chronic respiratory failure Active reduction of risk factor(s); influenza vaccination Add short-acting bronchodilator (when needed) Add regular treatment with one or more long-acting bronchodilators (when needed); Add rehabilitation Add inhaled glucocorticosteroids if repeated exacerbations Add long-term oxygen if chronic respiratory failure; Consider surgery Adapted from Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. Updated 2008. http: //www. goldcopd. com/Guidelineitem. asp? l 1=2&l 2=1&int. Id=989. Accessed November 21, 2008.

Symbicort® (budesonide/formoterol fumarate dihydrate) Inhalation Aerosol for COPD SYMBICORT 160/4. 5 μg is indicated for the twice-daily maintenance treatment of airflow obstruction in patients with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and emphysema Does not replace fast-acting inhalers and should not be used to treat acute symptoms of COPD ©Astra. Zeneca LP. All rights reserved. 277761 5/09

SYMBICORT Has Been Extensively Studied in Patients With Moderate to Very Severe COPD Overview of Key Phase III Studies Study Duration (weeks) N SUN 1 52 1964 SYMBICORT p. MDI 160/4. 5 μg × 2 inhalations BID and SYMBICORT p. MDI 80/4. 5 μg × 2 inhalations BID versus formoterol* and placebo* SHINE 2 26 1704 SYMBICORT p. MDI 160/4. 5 μg × 2 inhalations BID and SYMBICORT p. MDI 80/4. 5 μg × 2 inhalations BID versus its monocomponents† and placebo* Purpose Was to Compare the Efficacy and Safety of. . . *Administered as 2 inhalations BID. †Monocomponents refers to administration of budesonide monotherapy, formoterol monotherapy, and budesonide + formoterol administered in separate devices, all administered as 2 inhalations BID. 1. Rennard SI et al. Drugs. 2009; 69: 549 -565. 2. Tashkin DP et al. Drugs. 2008; 68: 1975 -2000.

SUN and SHINE Study Design Features • Patients receiving concomitant medications prior to study initiation 1, 2 – Allowed to remain on ICS during run-in – Long-acting anticholinergics were changed to stable doses of ipratropium during run-in and throughout the study – Long-acting β 2 -agonists (LABAs) were changed to short-acting β 2 -agonists (SABAs) as needed • Subjects with underlying comorbidities were included 1, 2 – – Hypertension (42%) Dyslipidemia (22%-24%) Cardiac disease (18%) Diabetes (10%-11%) 1. Rennard SI et al. Drugs. 2009; 69: 549 -565. 2. Tashkin DP et al. Drugs. 2008; 68: 1975 -2000.
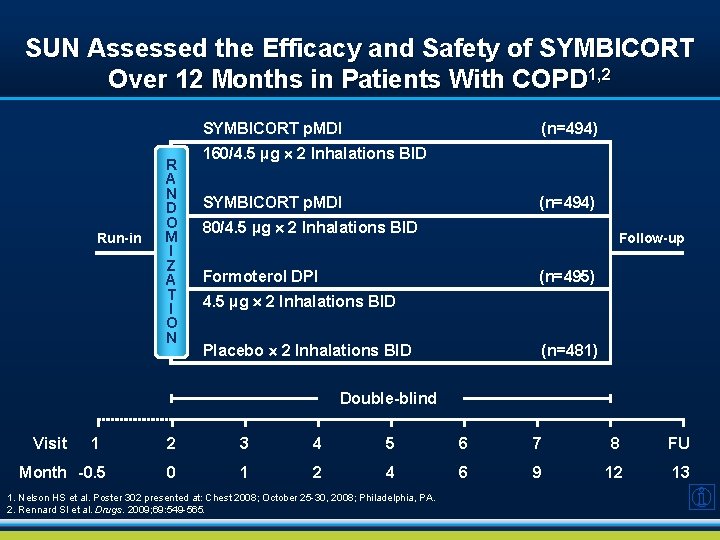
SUN Assessed the Efficacy and Safety of SYMBICORT Over 12 Months in Patients With COPD 1, 2 SYMBICORT p. MDI Run-in R A N D O M I Z A T I O N (n=494) 160/4. 5 µg 2 Inhalations BID SYMBICORT p. MDI (n=494) 80/4. 5 µg 2 Inhalations BID Follow-up Formoterol DPI (n=495) 4. 5 µg 2 Inhalations BID Placebo 2 Inhalations BID (n=481) Double-blind Visit 1 Month -0. 5 2 3 4 5 6 7 8 FU 0 1 2 4 6 9 12 13 1. Nelson HS et al. Poster 302 presented at: Chest 2008; October 25 -30, 2008; Philadelphia, PA. 2. Rennard SI et al. Drugs. 2009; 69: 549 -565.
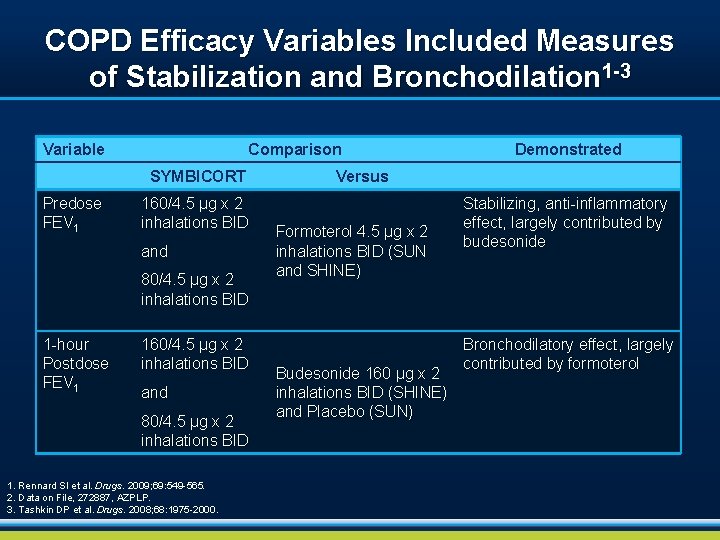
COPD Efficacy Variables Included Measures of Stabilization and Bronchodilation 1 -3 Variable Comparison SYMBICORT Predose FEV 1 160/4. 5 µg x 2 inhalations BID and 80/4. 5 µg x 2 inhalations BID 1 -hour Postdose FEV 1 160/4. 5 µg x 2 inhalations BID and 80/4. 5 µg x 2 inhalations BID 1. Rennard SI et al. Drugs. 2009; 69: 549 -565. 2. Data on File, 272887, AZPLP. 3. Tashkin DP et al. Drugs. 2008; 68: 1975 -2000. Demonstrated Versus Formoterol 4. 5 µg x 2 inhalations BID (SUN and SHINE) Budesonide 160 µg x 2 inhalations BID (SHINE) and Placebo (SUN) Stabilizing, anti-inflammatory effect, largely contributed by budesonide Bronchodilatory effect, largely contributed by formoterol
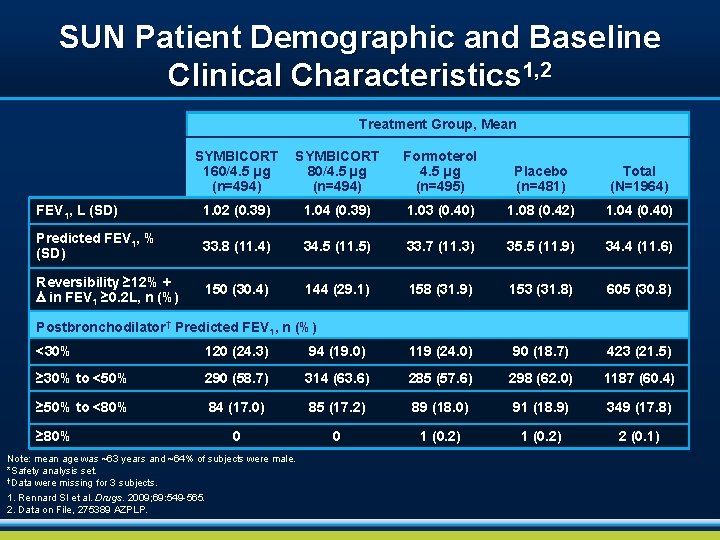
SUN Patient Demographic and Baseline Clinical Characteristics 1, 2 Treatment Group, Mean SYMBICORT 160/4. 5 µg (n=494) SYMBICORT 80/4. 5 µg (n=494) Formoterol 4. 5 µg (n=495) Placebo (n=481) Total (N=1964) FEV 1, L (SD) 1. 02 (0. 39) 1. 04 (0. 39) 1. 03 (0. 40) 1. 08 (0. 42) 1. 04 (0. 40) Predicted FEV 1, % (SD) 33. 8 (11. 4) 34. 5 (11. 5) 33. 7 (11. 3) 35. 5 (11. 9) 34. 4 (11. 6) Reversibility ≥ 12% + D in FEV 1 ≥ 0. 2 L, n (%) 150 (30. 4) 144 (29. 1) 158 (31. 9) 153 (31. 8) 605 (30. 8) Postbronchodilator† Predicted FEV 1, n (%) <30% 120 (24. 3) 94 (19. 0) 119 (24. 0) 90 (18. 7) 423 (21. 5) ≥ 30% to <50% 290 (58. 7) 314 (63. 6) 285 (57. 6) 298 (62. 0) 1187 (60. 4) ≥ 50% to <80% 84 (17. 0) 85 (17. 2) 89 (18. 0) 91 (18. 9) 349 (17. 8) 0 0 1 (0. 2) 2 (0. 1) ≥ 80% Note: mean age was ~63 years and ~64% of subjects were male. *Safety analysis set. †Data were missing for 3 subjects. 1. Rennard SI et al. Drugs. 2009; 69: 549 -565. 2. Data on File, 275389 AZPLP.
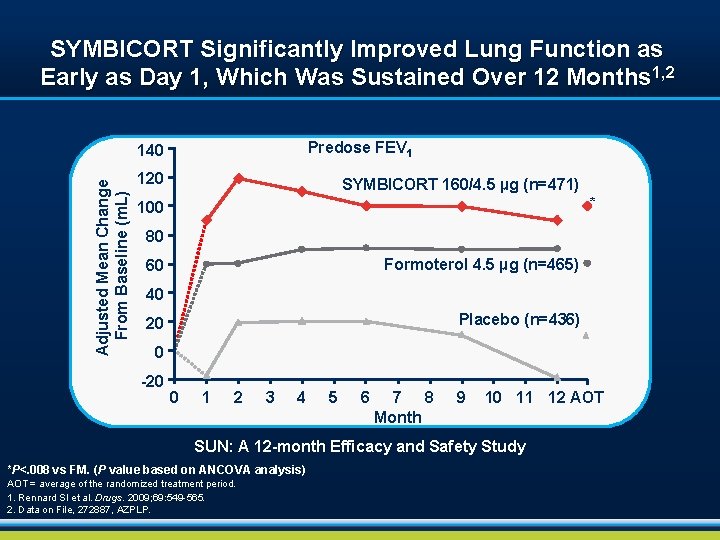
SYMBICORT Significantly Improved Lung Function as Early as Day 1, Which Was Sustained Over 12 Months 1, 2 Predose FEV 1 Adjusted Mean Change From Baseline (m. L) 140 120 SYMBICORT 160/4. 5 µg (n=471) * 100 80 Formoterol 4. 5 µg (n=465) 60 40 Placebo (n=436) 20 0 -20 0 1 2 3 4 5 6 7 8 Month 9 10 11 12 AOT SUN: A 12 -month Efficacy and Safety Study *P<. 008 vs FM. (P value based on ANCOVA analysis) AOT = average of the randomized treatment period. 1. Rennard SI et al. Drugs. 2009; 69: 549 -565. 2. Data on File, 272887, AZPLP.
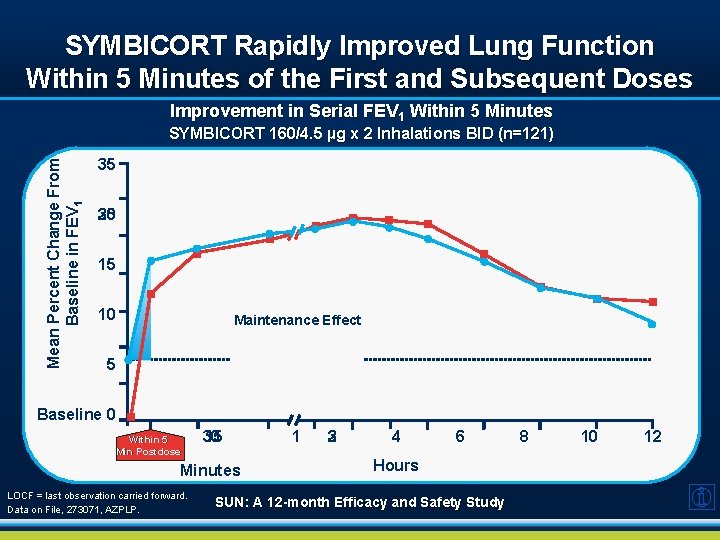
SYMBICORT Rapidly Improved Lung Function Within 5 Minutes of the First and Subsequent Doses Improvement in Serial FEV 1 Within 5 Minutes Mean Percent Change From Baseline in FEV 1 SYMBICORT 160/4. 5 µg x 2 Inhalations BID (n=121) 35 20 30 25 15 10 Maintenance Effect 5 Baseline 0 Within 5 Min Postdose 30 15 Minutes LOCF = last observation carried forward. Data on File, 273071, AZPLP. 1 2 3 4 6 Hours SUN: A 12 -month Efficacy and Safety Study 8 10 12
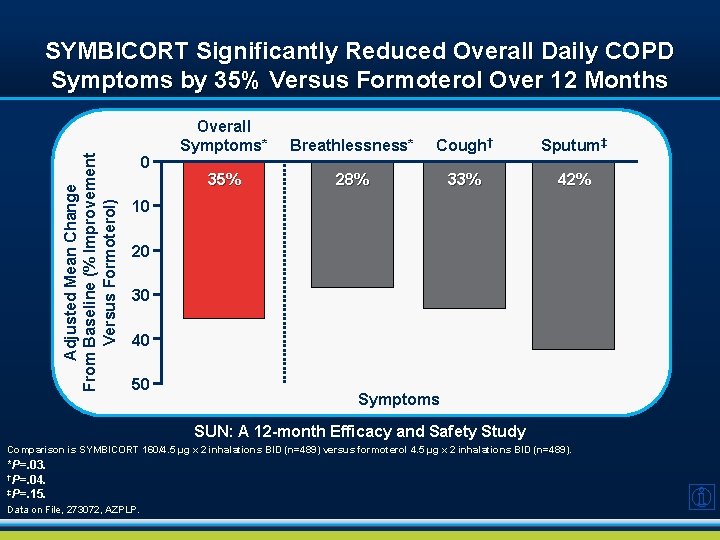
Adjusted Mean Change From Baseline (% Improvement Versus Formoterol) SYMBICORT Significantly Reduced Overall Daily COPD Symptoms by 35% Versus Formoterol Over 12 Months 0 Overall Symptoms* Breathlessness* Cough† Sputum‡ 35% 28% 33% 42% 10 20 30 40 50 Symptoms SUN: A 12 -month Efficacy and Safety Study Comparison is SYMBICORT 160/4. 5 µg x 2 inhalations BID (n=489) versus formoterol 4. 5 µg x 2 inhalations BID (n=489). *P=. 03. †P=. 04. ‡P=. 15. Data on File, 273072, AZPLP.
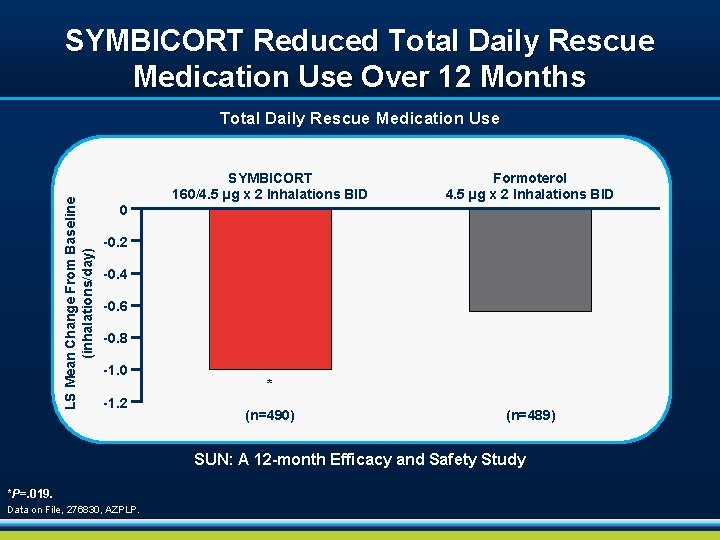
SYMBICORT Reduced Total Daily Rescue Medication Use Over 12 Months LS Mean Change From Baseline (inhalations/day) Total Daily Rescue Medication Use 0 SYMBICORT 160/4. 5 µg x 2 Inhalations BID Formoterol 4. 5 µg x 2 Inhalations BID -0. 2 -0. 4 -0. 6 -0. 8 -1. 0 -1. 2 * (n=490) (n=489) SUN: A 12 -month Efficacy and Safety Study *P=. 019. Data on File, 276830, AZPLP.
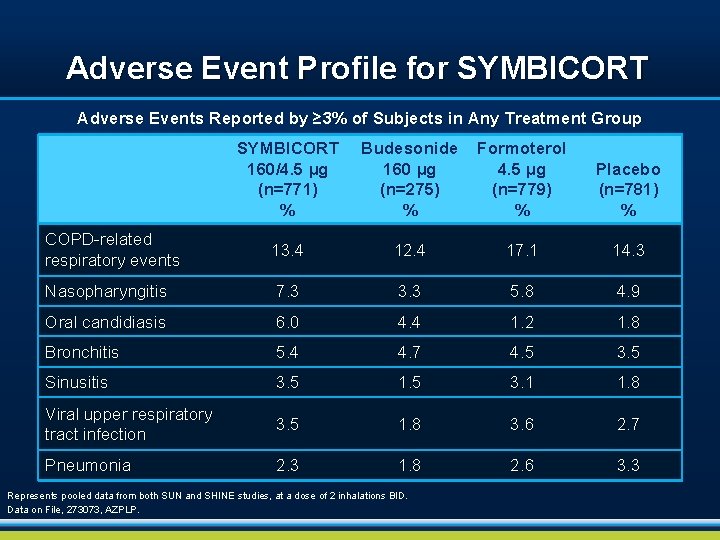
Adverse Event Profile for SYMBICORT Adverse Events Reported by ≥ 3% of Subjects in Any Treatment Group SYMBICORT 160/4. 5 µg (n=771) % Budesonide 160 µg (n=275) % Formoterol 4. 5 µg (n=779) % Placebo (n=781) % COPD-related respiratory events 13. 4 12. 4 17. 1 14. 3 Nasopharyngitis 7. 3 3. 3 5. 8 4. 9 Oral candidiasis 6. 0 4. 4 1. 2 1. 8 Bronchitis 5. 4 4. 7 4. 5 3. 5 Sinusitis 3. 5 1. 5 3. 1 1. 8 Viral upper respiratory tract infection 3. 5 1. 8 3. 6 2. 7 Pneumonia 2. 3 1. 8 2. 6 3. 3 Represents pooled data from both SUN and SHINE studies, at a dose of 2 inhalations BID. Data on File, 273073, AZPLP.

Other Safety Findings for SYMBICORT in COPD No Clinically Important Differences Between SYMBICORT p. MDI 160/4. 5 µg and Placebo Were Observed in the Following Parameters 1 -3 • Vital signs • 24 -hour urinary cortisol • Physical examination • Bone mineral density* • ECG • Lenticular opacities and intraocular pressure* *As reported in SUN only. 1. Rennard SI et al. Poster 308 presented at: Chest 2008; October 25 -30, 2008; Philadelphia, PA. . 2. Tashkin DP et al. Drugs. 2008; 68: 1975 -2000. 3. Rennard SI et al. Drugs. 2009; 69: 549 -565.

SYMBICORT: Important Safety Information • Particular care is needed for patients being transferred from systemically active corticosteroids to inhaled corticosteroids • Patients who are receiving SYMBICORT should not use additional formoterol or other long-acting inhaled beta 2 -agonists for any reason • Lower respiratory tract infections, including pneumonia, have been reported following the inhaled administration of corticosteroids, including budesonide, a component of SYMBICORT • Long-term use of orally inhaled corticosteroids, such as budesonide, a component of SYMBICORT, may affect normal bone metabolism resulting in a loss of bone mineral density • Glaucoma, increased intraocular pressure, and cataracts have been reported in patients following the long -term administration of inhaled corticosteroids, including budesonide, a component of SYMBICORT • Caution should be exercised when considering the coadministration of SYMBICORT with long-term ketoconazole and other known potent CYP 3 A 4 inhibitors • SYMBICORT should be administered with caution in patients being treated with MAO inhibitors or tricyclic antidepressants, or within 2 weeks of discontinuation of such agents • WARNING: Long-acting beta 2 -adrenergic agonists may increase the risk of asthma-related death. Therefore, when treating patients with asthma, SYMBICORT should only be used for patients with asthma not adequately controlled on other asthma-controller medications (eg, low- to medium-dose inhaled corticosteroids) or whose disease severity clearly warrants initiation of treatment with 2 maintenance therapies. Data from a large placebo-controlled US study that compared the safety of another long-acting beta 2 -adrenergic agonist (salmeterol) or placebo added to usual asthma therapy showed an increase in asthma-related deaths in patients receiving salmeterol. This finding with salmeterol may apply to formoterol (a long-acting beta 2 -adrenergic agonist), one of the active ingredients in SYMBICORT (see WARNINGS in full Prescribing Information)
Approved Dosage for Patients With COPD 160/4. 5 µg, 2 inhalations twice daily* • SYMBICORT 160/4. 5 μg is indicated for the twice-daily maintenance treatment of airflow obstruction in patients with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and emphysema • SYMBICORT is not a rescue medication and does NOT replace fast-acting inhalers *Administered in the morning and in the evening. SYMBICORT [package insert]. Wilmington, DE: Astra. Zeneca LP.

Rapid Improvement in Lung Function and Long-lasting Control* With SYMBICORT Can Help Patients Do More • SYMBICORT significantly improved lung function as early as day 1, which was sustained over 12 months 1 • SYMBICORT rapidly improved lung function within 5 minutes† of the first and subsequent doses 2 • SYMBICORT significantly reduced overall daily COPD symptoms by 35% vs formoterol over 12 months 3 *In a 12 -month clinical study, SYMBICORT improved predose FEV 1, 1 -hour postdose FEV 1, serial FEV 1 (to measure onset of effect), AM and PM peak expiratory flow (PEF), and reduced COPD symptoms and rescue albuterol use. †Significant bronchodilation (>15% improvement in FEV ) occurred within 5 minutes. 1 1. Data on File, 272887, AZPLP. 2. Data on File, 273071, AZPLP. 3. Data on File, 273072, AZPLP.

Comments to Astra. Zeneca is committed to conducting business with the highest standards of integrity and professionalism. If you have any comments that could improve the delivery of our promotional educational programs, please contact Astra. Zeneca at 1 -800 -236 -9933 (C) 2009 Astra. Zeneca Pharmaceuticals LP. All rights reserved. 277388 3/09.
- Slides: 28