Optimization and Testing of Nucleic Acid and PCR










































- Slides: 53

Optimization and Testing of Nucleic Acid and PCR Carry-over Contamination Detection in the Clinical Virology Laboratory William Rivera Public Health Internship Program School of Biological Sciences The University of Texas at Austin Mentor: Ana Maria Valle-Rivera Texas Department of State Health Services

Introduction 2

What is a clinical virology laboratory? • A specialized laboratory for the isolation and diagnosis of viral diseases • Locations: large hospitals, reference laboratories, public health laboratories • Report: – Physicians or patients – CDC and WHO 3

Texas Department of State Health Services (DSHS) • Exists as a resource to help in many sorts of public health issues for the entire state of Texas • Aids in disease outbreaks • Helps track and prevent epidemics • Informs the public about health care options and resources Source: http: //www. dshs. state. tx. us/ 4

DSHS Laboratory Services Section • • • Newborn screening DNA analysis lab Clinical chemistry Prenatal testing Consumer microbiology Clinical bacteriology • • • Medical parisitology Rabies Viral isolation Arbovirus/Entomology Mycobacteriology Molecular/ Serology Source: http: //www. dshs. state. tx. us/ 5

Viral Isolation Team • Receives specimens from a variety of sources- hospitals, clinics, public, CDC (proficiency testing) • Identifies most viral species with exception of those identified by specialized groups (e. g. , rabies) • Some of the most commonly isolated viruses are influenza, adenovirus, and enterovirus 6

Adenovirus and Enterovirus Adenovirus Enterovirus • Family- Adenoviridae • Double stranded DNA virus • Mainly causes upper respiratory tract infections • Spread primarily through respiratory droplets • Family- Picornaviridae • Single stranded positive sense RNA viruses • Enteroviruses are a large group • Can cause a wide range of diseases 7

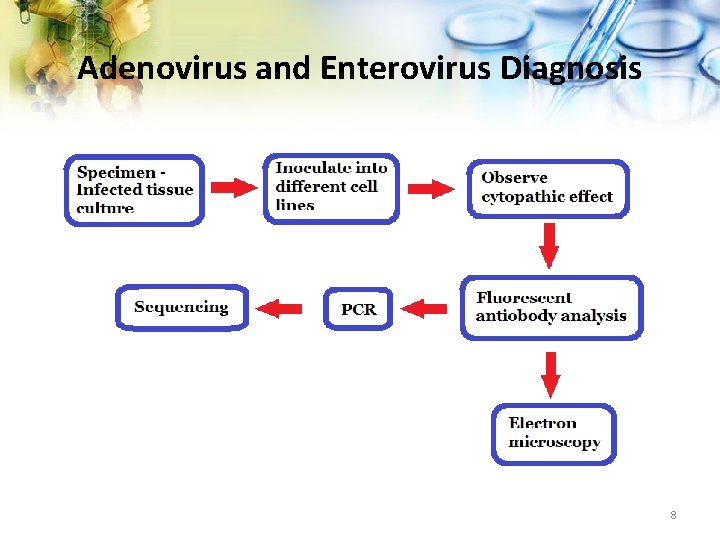
Adenovirus and Enterovirus Diagnosis 8

Molecular Diagnostics & PCR • Molecular diagnostics is the identification of a genetic disease or of a pathogen using methods that specifically analyze the organisms DNA or RNA • Polymerase Chain Reaction (PCR) amplifies trace amounts of nucleic acid to identify the presence of a virus Source: http: //www. researchandmarkets. com/reports/28452/molecular_diagnostics_revolution_or_hype 9

Advantages & Disadvantages of PCR Advantages Disadvantages • Avoid the need for tissue culture • Detection of fastidious viruses • Quicker results • Extraordinary sensitivity • Live virus is not necessary • Carryover contamination • Build up of amplicons in laboratory environment • Possibility of false positive results from DNA contamination 10

Amplicons? • Amplified segments of target DNA • Generated from PCR • Due to constant amplification of the same target sequence, amplicons can accumulate in laboratory environment • Perfect piece of DNA to contaminate a negative specimen and yield a false positive 11

Sources of DNA contamination in a clinical virology laboratory • Aerosols produced from opening PCR reaction vessels • Careless handling of specimens • Contaminated gloves • Pipettes and other lab equipment • Contaminated media and reagents 12

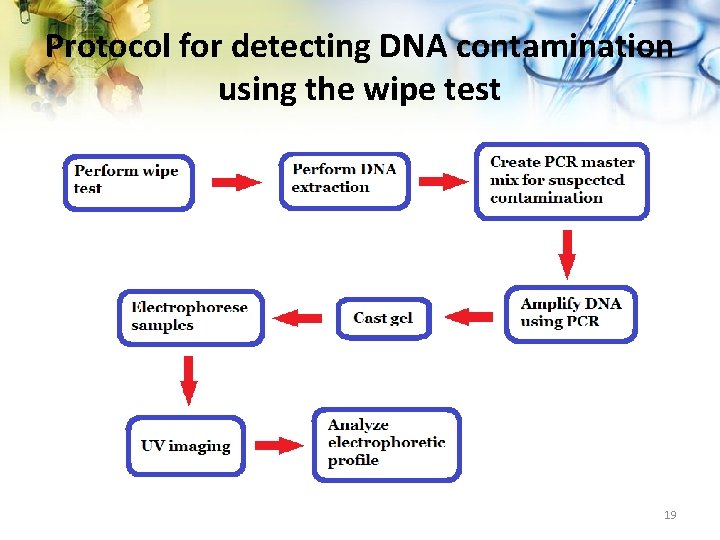
The Wipe Test • Method used to check for DNA contamination • Relied upon for quality control • Sensitivity unspecified • Reliability of results unknown 13

Purpose • To assess the validity of the methods employed to test for and eradicate potential amplified carry-over DNA contamination from environmental surfaces in the virology laboratory • To make suggestions about how to improve the current methodology used to detect DNA contamination 14

Methods 15

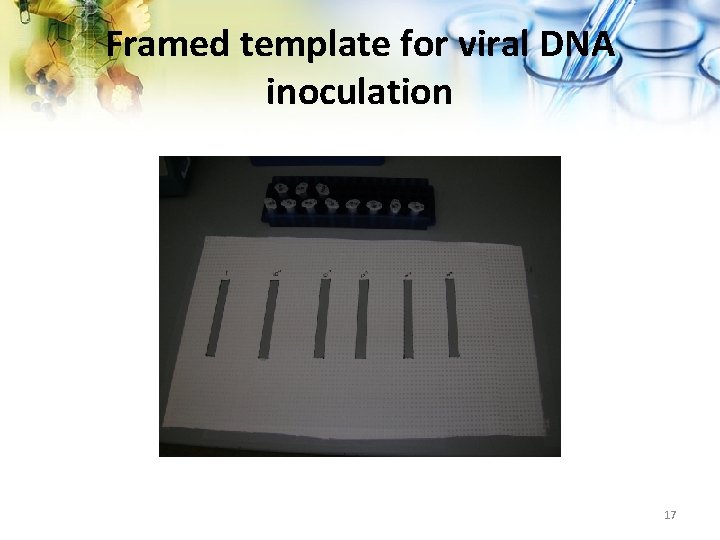
Experimental Design 1. Deliberately inoculate laboratory surfaces with 10 fold dilutions of adenovirus and enterovirus. DNA 2. Framed template used to define the areas for inoculation 3. DNA applied and allowed to dry 4. Perform wipe test to retrieve, amplify and detect DNA 5. Analyze results 16

Framed template for viral DNA inoculation 17

Inoculation of bench top 18

Protocol for detecting DNA contamination using the wipe test 19

Results 20

Experiment 1 21

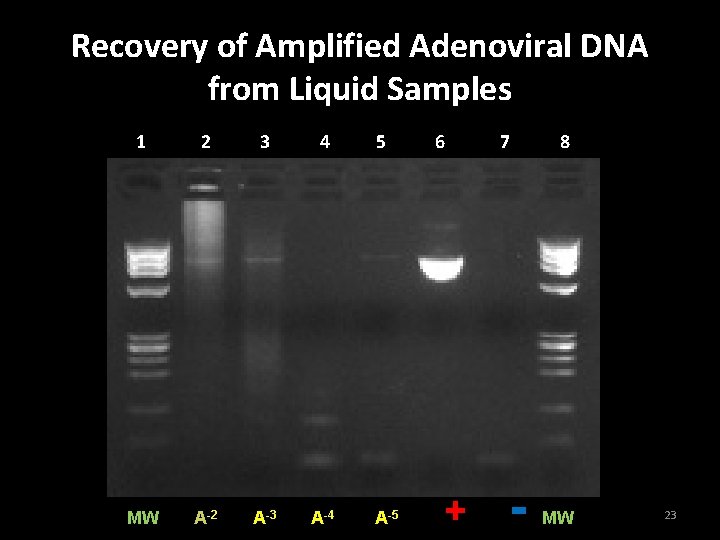
Purpose: Preliminary Assessment of the wipe test sensitivity in liquid samples • Method: • Prepare stock solution of adenovirus DNA; amplify the DNA by PCR • Dilute stock solution of amplified DNA – 10 -2, 10 -3, 10 -4, and 10 -5 in microcentrifuge tubes • Sample from tubes using wipe test swabs • Identify DNA using wipe test protocol 22

Recovery of Amplified Adenoviral DNA from Liquid Samples 1 2 3 4 MW A-2 A-3 A-4 5 A-5 6 + 7 - 8 MW 23

Conclusion Experiment 1 • Carryover contamination of at least 10 -5 dilution from adenovirus PCR product is capable of yielding a false positive result 24

Experiment 2 25

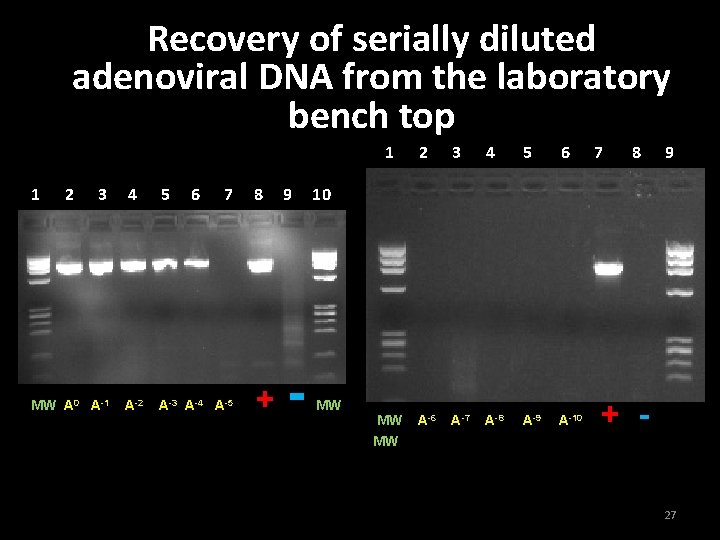
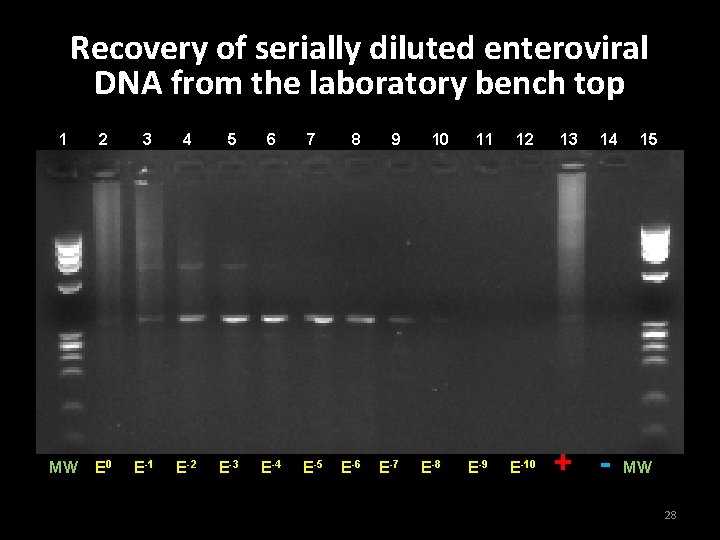
Evaluation of wipe test sensitivity on a laboratory bench top 1. PCR product from adenovirus and enterovirus were used to make serial 1/10 dilutions from 10 -1 to 10 -10 2. Bench top was contaminated with each sample and allowed to dry for 30 min 3. Wipe test was performed, swabbing surface for approx. 45 sec. to retrieve DNA 26

Recovery of serially diluted the laboratory adenoviral DNA from bench top 1 2 3 MW A 0 A-1 4 5 6 7 A-2 A-3 A-4 A-5 8 9 + - 1 2 3 4 5 6 MW MW A-6 A-7 A-8 A-9 A-10 7 8 9 10 MW + 27

Recovery of serially diluted enteroviral DNA from the laboratory bench top 1 2 3 4 5 6 7 MW E 0 E-1 E-2 E-3 E-4 E-5 8 9 E-6 E-7 10 E-8 11 E-9 12 E-10 13 14 + - 15 MW 28

Conclusion for Experiment 2 • Detection of adenoviral DNA - 10 -4 • Detection of enteroviral DNA - 10 -8 29

Experiment 3 30

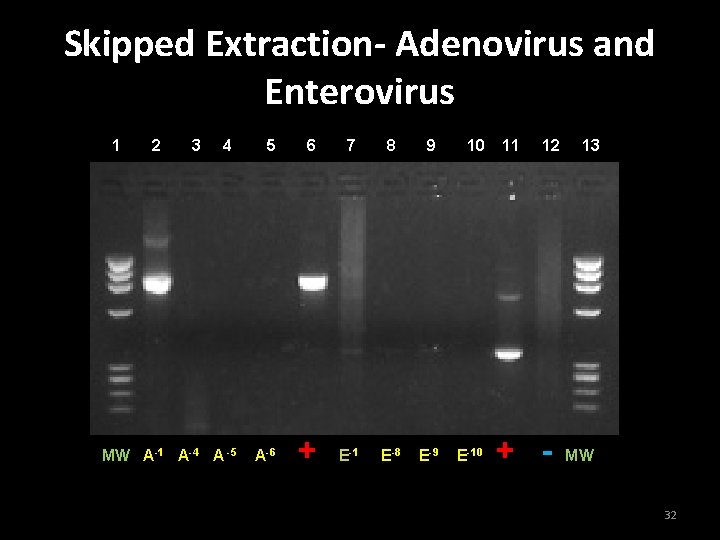
Skipping extraction in wipe test • The wipe test was modified to skip extraction step • Dilutions used: – Adenoviral DNA dilutions: 10 -1, 10 -4, 10 -5, 10 -6 – Enteroviral DNA dilutions: 10 -1, 10 -8, 10 -9, 10 -10 • Testing to see if skipping extraction improved DNA retrieval sensitivity 31

Skipped Extraction- Adenovirus and Enterovirus 1 2 3 4 MW A-1 A-4 A -5 5 6 7 8 9 10 A-6 + E-1 E-8 E-9 E-10 11 12 + - 13 MW 32

Conclusion for Experiment 3 • Skipping extraction does not necessarily improve sensitivity of wipe test • Results are inconclusive • More data needed for statistically significant results 33

Experiment 4 34

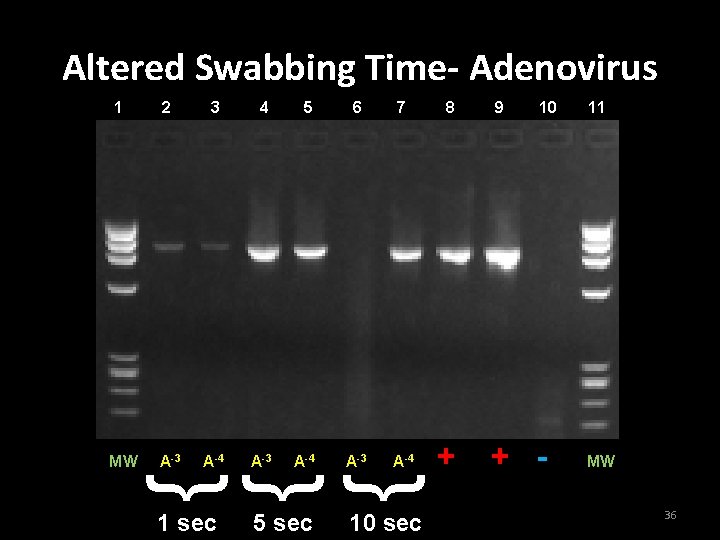
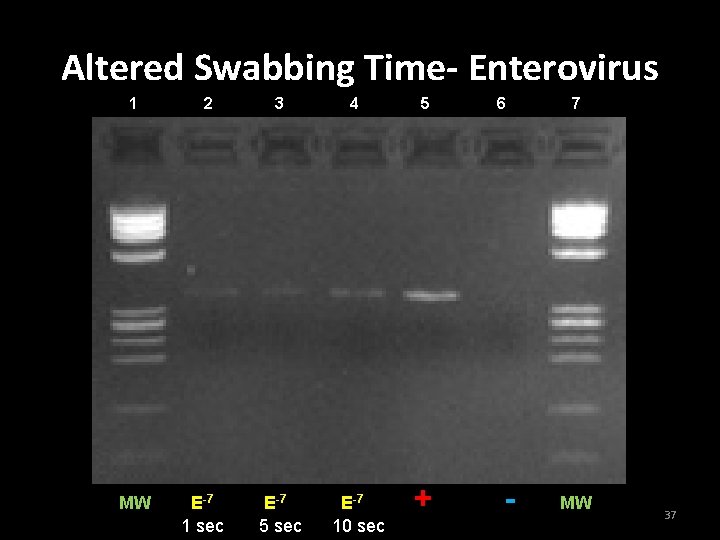
Alteration of swabbing time • Purpose • To determine if the amount of time spent swabbing the surface affected the results of the wipe test • Swab surfaces – Adenoviral DNA: 10 -3, 10 -4 dilutions – Enteroviral DNA: 10 -7 or 10 -8 dilution • Each area was either swabbed for 1 sec. , 5 sec. , or 10 sec to retrieve the DNA. 35

Altered Swabbing Time- Adenovirus 2 3 4 5 6 7 MW A-3 A-4 8 + 9 10 + - 11 MW { { { 1 1 sec 5 sec 10 sec 36

Altered Swabbing Time- Enterovirus 1 MW 2 E-7 1 sec 3 4 E-7 5 sec E-7 10 sec 5 + 6 - 7 MW 37

Conclusion for Experiment 4 • Increasing the swabbing time increased the ability to retrieve DNA from the laboratory surfaces. 38

Experiment 5 39

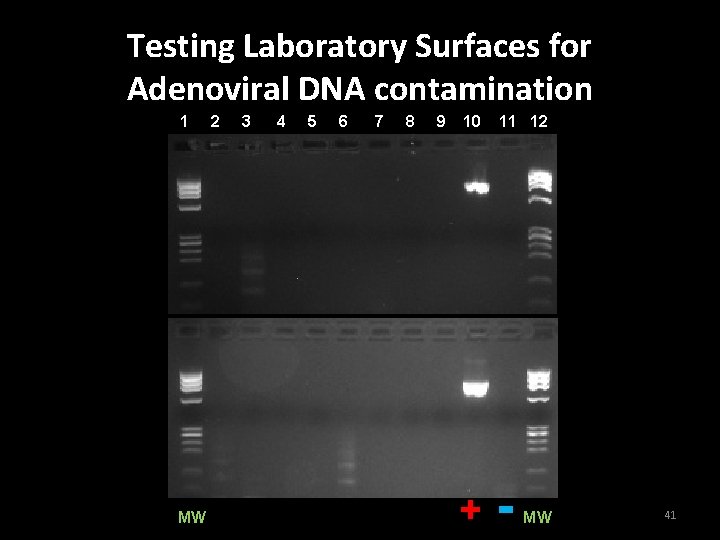
Checking laboratory surfaces for DNA contamination • The lab was systematically checked for adenovirus DNA contamination • 16 locations were identified as being at high risk for accumulation of DNA • Standard wipe test protocol was used with a 10 second swab time 40

Testing Laboratory Surfaces for Adenoviral DNA contamination 1 MW 2 3 4 5 6 7 8 9 10 11 12 + - MW 41

Conclusion of Experiment 5 • All areas tested in laboratory were found to be free of adenoviral DNA contamination 42

Experiment 6 43

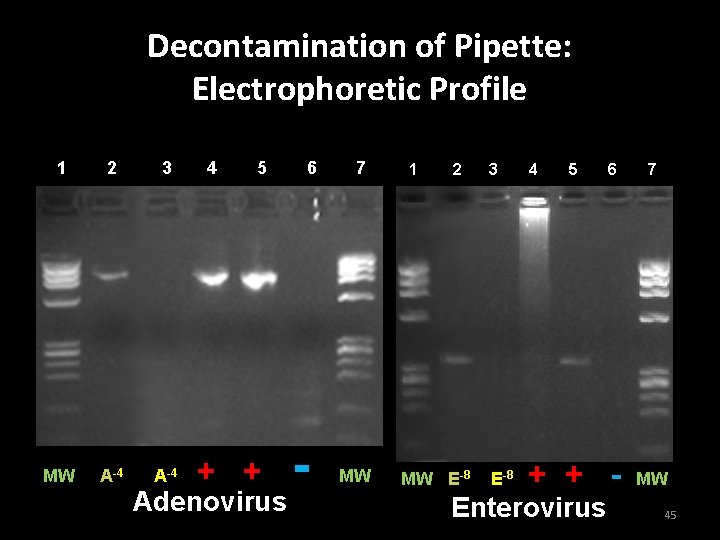
Validation of decontamination procedures for equipment • A pipette was contaminated with amplified DNA dilution and used due to its textured surface • Wipe test was performed • Decontamination performed: – RNase AWAY – Bleach – 70% ethanol • Second wipe test was performed after decontamination 44

Decontamination of Pipette: Electrophoretic Profile 1 2 3 MW A-4 4 5 + + Adenovirus 6 - 7 MW 1 2 MW E-8 3 4 5 E-8 + + - Enterovirus 6 7 MW 45

Conclusion of Experiment 6 • Decontamination procedures used at the DSHS are sufficient to eliminate amplified DNA contamination from laboratory equipment surfaces 46

Conclusion 47

Evaluation of Wipe Test Sensitivity • Detected up to 1/100, 000 th dilution of amplified viral DNA • Very sensitive • Suitable for detecting DNA contamination from laboratory surfaces produced as PCR product 48

Findings on Swabbing Time Alteration • Increasing the swabbing time improves DNA detection • 10 seconds should be sufficient to detect threatening levels of contamination, if they exist 49

Evaluation of Decontamination Procedures • Successful at eliminating amplified DNA contamination from laboratory surfaces • Sufficient for keeping laboratory surfaces free of detectable levels of DNA contamination 50

Future Studies • Quantification of wipe test sensitivity • Further testing to improve wipe test sensitivity – Skipping DNA extraction step – Altering the diluent used for the wipe test – Altering the swab used for the wipe test 51

Acknowledgements • • • Ana Maria Valle-Rivera, Ph. D. Martha Thompson Members of the Viral Isolation Team Dr. Kneeland Ms. Nancy Elder Leanne Field, Ph. D. 52

Special thanks for: • Funding generously provided by – The Centers for Disease Control and Prevention through an Association of Schools of Public Health/Association of Public Health Laboratories: "Pathways to Public Health Careers and Internships Grant. " 53