Optical Properties Introduction By optical property is meant

Optical Properties

Introduction By “optical property” is meant a material’s response to exposure to electromagnetic radiation and, in particular, to visible light. In classical sense, electromagnetic radiation is considered to be wave-like, wave-like consisting of electric and magnetic field components that are perpendicular to each other and also to the direction of propagation. 2
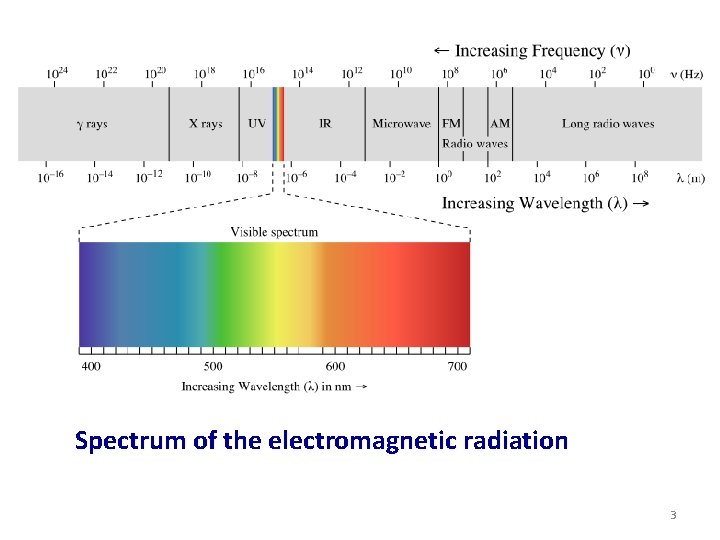
Spectrum of the electromagnetic radiation 3

Light and Electromagnetic Spectrum • Visible light: Electromagnetic radiation with wavelength 0. 4 to 0. 75 micrometers. Ø Ultraviolet : 0. 01 – 0. 4 micrometers Ø Infrared: 0. 75 – 1000 micrometers • Light is in form of waves and consist of particles called photons. ΔE = hν = h. C/λ ΔE = Energy λ = wavelength ν = frequency C = speed of light = 3 x 108 m/s H = plank’s constant = 6. 62 x 10 -34 J. s 15 -2

Electron Transitions The absorption and emission of electromagnetic radiation may involve electron transitions from one energy state to another. An electron may be excited from an occupied state at energy E 2 to a vacant and higher-lying one, denoted E 4, by the absorption of a photon of energy. The change in energy experienced by the electron, ΔE, depends on the radiation frequency as follows: Δ E = hν 5

Important concepts: 1. Since the energy states for the atom are discrete, only specific ΔE exist between the energy levels; thus, only photons of frequencies corresponding to the possible ΔE for the atom can be absorbed by electron transitions. 2. A stimulated electron cannot remain in an excited state indefinitely; after a short time, it falls or decays back into its ground state, state or unexcited level, with a reemission of electromagnetic radiation. 6

• A photon in a Zn. S semiconductor drops from an impurity energy level at 1. 38 e. V below its conduction band to its valence band. If Zn. S has an energy band gap of 3. 54 e. V, what is the wavelength of the radiation given off by the photon? What is the color of the radiation? The energy difference =3. 54 e. V-1. 38 e. V= 2. 16 e. V =hc/ E =574. 7 nm Visible yellow region

Wavelength vs. Band Gap Example: What is the minimum wavelength absorbed by Ge? (Given Eg = 0. 67 e. V, h = 6. 62 x 10 -34 J. s, c = 3. 0 x 108 m/s) Answer: 8

Corrosion

Types of Corrosion • Uniform or general attack corrosion: Reaction proceeds uniformly on the entire surface. Ø Controlled by protective coatings, cathodic protection. • Galvanic or two metal corrosion: Electrochemical reaction leads to corrosion of on metal. Ø Zinc coatings on steel protects steel as zinc is anodic to steel and corrodes. Ø Large cathode area to small anode area should be avoided. 13 -19

Pitting Corrosion • Pitting: Localized corrosive attacks that produces holes or pits in a metal. • Results in sudden unexpected failure as pits go undetected (covered by corrosion products). • Pitting requires an initiation period and grows in direction of gravity. • Pits initiate at structural and compositional heterogeneities. Figure 12. 20 Pitting of stainless steel 13 -20 Courtesy of La. Que Center for Corrosion Technology, Inc.

Growth of Pit • Growth of pit involves dissolution of metal in pit maintaining high acidity at the bottom. • Anodic reaction at the bottom and cathodic reaction at the metal Figure 12. 21 surface. • At bottom, metal chloride + water Metal hydroxide + free acid. • Some metals (stainless steel) have better resistance than others (titanium). 13 -21 After M. G. Fontana and N. D. Greene, “ Corrosion Engineering, ” 2 nd ed. , Mc. Graw-Hill, 1978

Crevice Corrosion • Localized electrochemical corrosion in crevices and under shielded surfaces where stagnant solutions can exist. • Occurs under valve gaskets, rivets and bolts in alloy systems like steel, titanium and copper alloys. • Anode: M M+ + e • Cathode: O 2 + 2 H 2 O + 4 e- 4 OH • As the solution is stagnant, oxygen is used up and not replaced. • Chloride ions migrate to crevice to balance positive charge and form metal Figure 12. 23 hydroxide and free acid that causes corrosion. 13 -22 After M. G. Fontana and N. D. Greene, “ Corrosion Engineering, ” 2 nd ed. , Mc. Graw-Hill, 1978.

Intergranular Corrosion • Localized corrosion at and/or adjacent to highly reactive grain boundaries resulting in disintegration. • When stainless steels are heated to or cooled through sensitizing temperature range (500 -8000 C) chromium carbide precipitate along grain boundaries. • When exposed to corrosive environment, the region next to grain boundaries become anodic and corrode. 13 -23

Stress Corrosion • Stress corrosion cracking (SCC): Cracking caused by combined effect of tensile stress and corrosive environment. • Stress might be residual and applied. • Only certain combination of alloy and environment causes SCC. • Crack initiates at pit or other discontinuity. • Crack propagates perpendicular to stress • Crack growth stops if either stress or corrosive environment is removed.

Erosion Corrosion and Cavitation Damage • Erosion corrosion: Acceleration in rate of corrosion due to relative motion between corrosive fluid and surface. • Pits, grooves, valleys appear on surface in direction of flow. • Corrosion is due to abrasive action and removal of protective film. • Cavitation damage: Caused by collapse of air bubbles or vapor filled cavities in a liquid near metal surface. • Rapidly collapsing air bubbles produce very high pressure (60, 000 PSI) and damage the surface. • Occurs at metal surface when high velocity flow and pressure are present. 13 -25
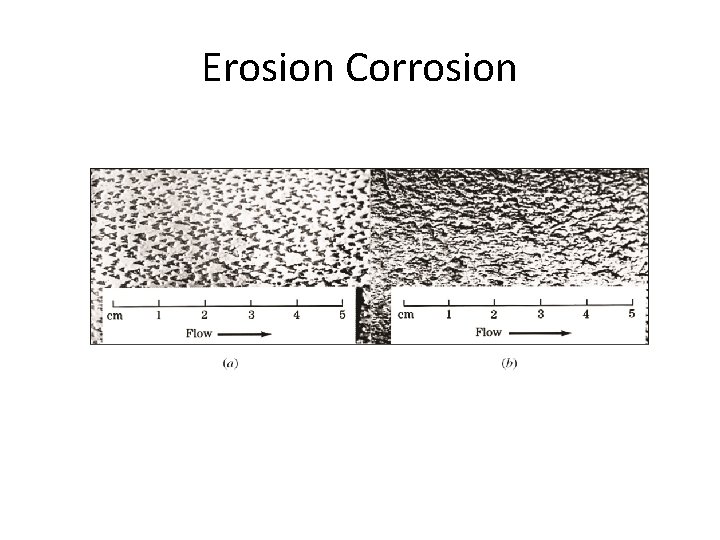
Erosion Corrosion

Fretting Corrosion and Selective Leaching • Fretting corrosion: Occurs at interface between materials under load subjected to vibration and slip. Ø Metal fragments get oxidized and act as abrasives between the surfaces. • Selective leaching: Selective removal of one element of alloy by corrosion. Ø Example: Dezincification Selective removal of zinc from copper and brasses. Ø Weakens the alloy as single metal might not have same strength as the alloy. 13 -26

Given the atomic mass for Al is 26. 98 g/mol, and the density for Al=2. 70 g/cm 3
- Slides: 19