Optical Isomerism Written by A Bourne MChem 2014

Optical Isomerism Written by A Bourne (MChem), 2014

Aims of this Lesson • Be able to recognise and explain optical isomerism and its importance in Chemistry • Increase your spatial skills and awareness using software designed by the CCDC

What are spatial skills? • The ability to mentally view and change 2 -dimensional and 3 -dimensional figures. • Important in chemistry, biology, medicine, mathematics, physics and everyday life. These are both the same molecule, viewed in 2 -D and 3 -D. What is the name of this simple molecule?

You should already know • The term isomer and its definition • How to calculate ‘priority’ of functional groups (i. e. higher atomic number = higher priority) – the C-I-P Rules • Some common organic functional groups
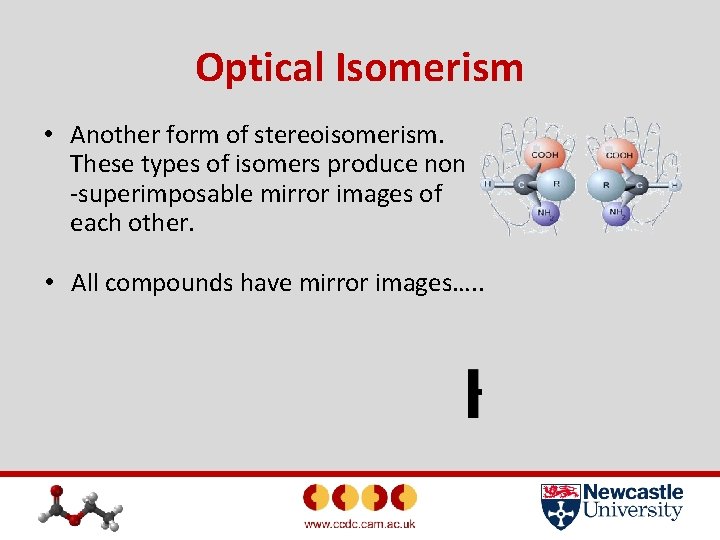
Optical Isomerism • Another form of stereoisomerism. These types of isomers produce non -superimposable mirror images of each other. • All compounds have mirror images…. .

…. only a special few are optical isomers Lactic acid, how on the hand will never superimpose No matter youother rotate fluoromethane, you always onto its mirror image. Think of a pair of hands. These have the same molecule. Try using some Molymods™ cannot be exactly the same as each other. to proveever to yourself. Why not see for yourself? Use some Molymods to make models of lactic acid and fluoromethane Lactic acid (+ and – isomers) Fluoromethane
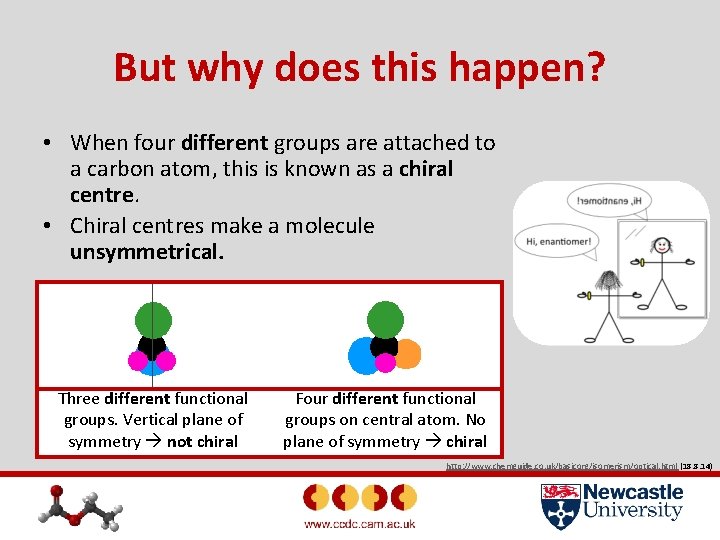
But why does this happen? • When four different groups are attached to a carbon atom, this is known as a chiral centre. • Chiral centres make a molecule unsymmetrical. Three different functional groups. Vertical plane of symmetry not chiral Four different functional groups on central atom. No plane of symmetry chiral http: //www. chemguide. co. uk/basicorg/isomerism/optical. html (18. 8. 14)

But why does this happen? Each chiral centre can produce up to two enantiomers (the actual compound and its mirror image) Two stereoisomers which are not mirror images of each other are known as diastereomers If there is more than one chiral centre, there can be up to 2 n diastereomers (n = number of chiral centres) e. g. 2 chiral centres can produce up to 4 diastereomers

But why does this happen? See the 3 -D projections of these compounds here, or try making them using Molymods yourself. http: //employees. csbsju. edu/cschaller/principles%20 chem/stereo_diastereo. htm
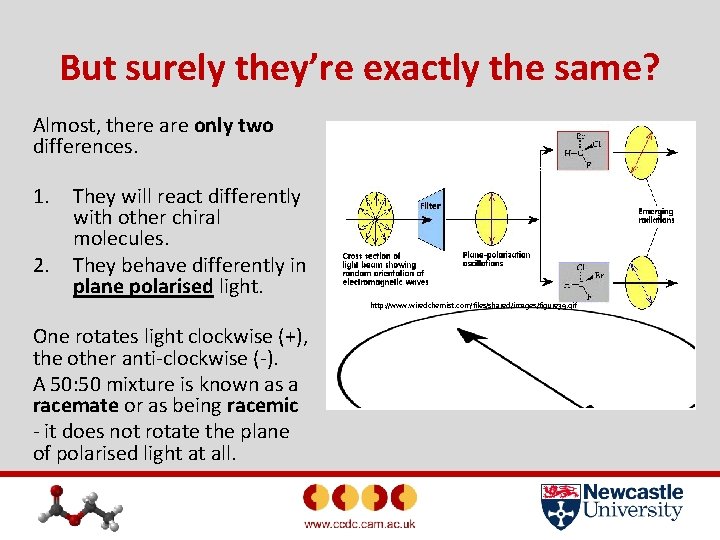
But surely they’re exactly the same? Almost, there are only two differences. 1. 2. They will react differently with other chiral molecules. They behave differently in plane polarised light. http: //www. wiredchemist. com/files/shared/images/figure 39. gif One rotates light clockwise (+), the other anti-clockwise (-). A 50: 50 mixture is known as a racemate or as being racemic - it does not rotate the plane of polarised light at all.

How do you identify chiral centres? 1. Draw the displayed or skeletal formula of the compound. 2. Check each carbon atom - do any have four different groups bonded? 3. Draw one isomer of the molecule using the ‘wedge-dash’ notation to show the tetrahedral carbon atom. 4. Draw a dotted/dashed line to represent the mirror plane. 5. Draw the mirror image on the other side of the mirror line.

Task • Do worksheet F 2 – Optical Isomers • If you haven’t used these resources before, you must download and follow sheet A 2 – Introduction to Access Structures first!

Why does it matter? • Thalidomide was a drug prescribed in the early 1960’s as a remedy for morning sickness in pregnant women. • It has a chiral centre (red circle), so exists as two optical isomers. • The drug was given as a racemic mixture of both isomers. • Around half of all women given the drug had children with birth defects. Unfortunately, even if pure (S) thalidomide were prescribed, the body can still change this into the dangerous teratogenic (R) form
- Slides: 13