Optical Isomerism Same molecular formula but different arrangement

Optical Isomerism

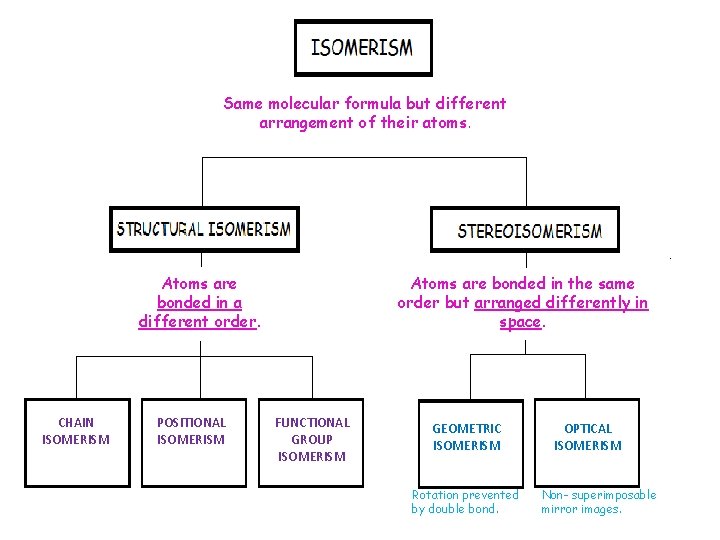
Same molecular formula but different arrangement of their atoms. Atoms are bonded in a different order. CHAIN ISOMERISM POSITIONAL ISOMERISM Atoms are bonded in the same order but arranged differently in space. FUNCTIONAL GROUP ISOMERISM GEOMETRIC ISOMERISM Rotation prevented by double bond. OPTICAL ISOMERISM Non- superimposable mirror images.

E-Z vs cis-trans - Draw cis and trans 1, 2 -dichloroethane
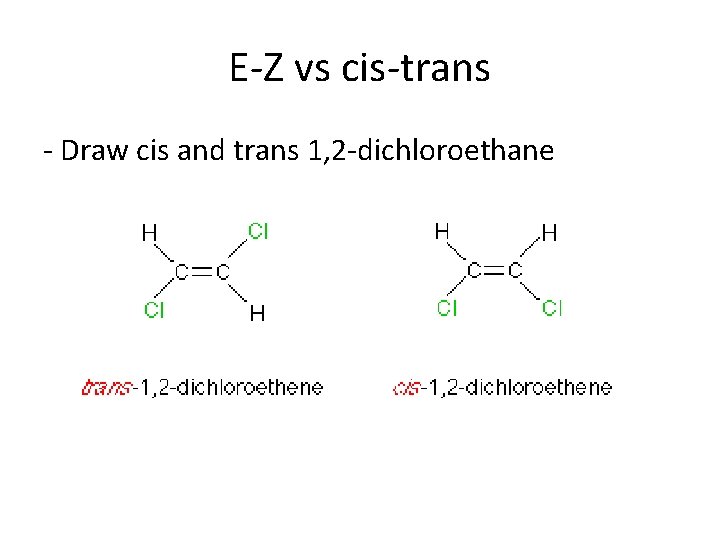
E-Z vs cis-trans - Draw cis and trans 1, 2 -dichloroethane

E-Z vs cis-trans • Now try and name these using the cis-trans system

E-Z naming system 1. You look at what is attached to each end of the double bond in turn, and give the two groups a "priority" according to a set of rules. The atoms with the higher atomic number take priority.

E-Z naming system If the two groups with the higher priorities are on the same side of the double bond, that is described as the (Z)- isomer. So you would write it as (Z)-name of compound. The symbol Z comes from a German word which means together.

E-Z naming system If the two groups with the higher priorities are on opposite sides of the double bond, then this is the (E)- isomer. E comes from the German word which means opposite.
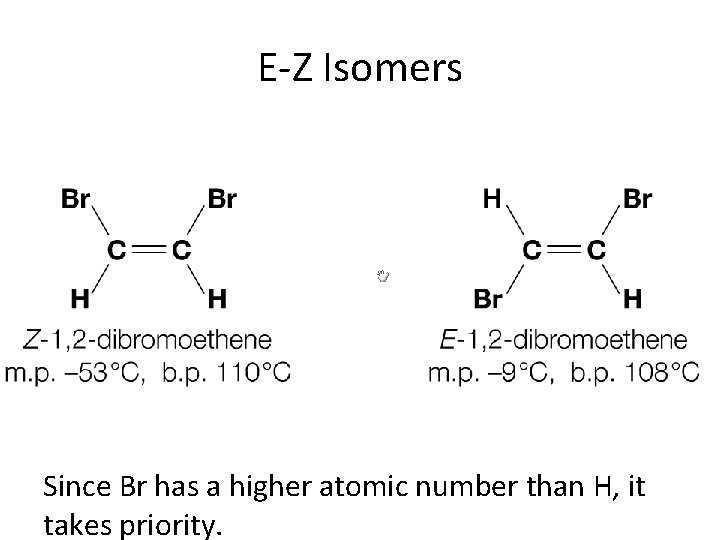
E-Z Isomers Since Br has a higher atomic number than H, it takes priority.
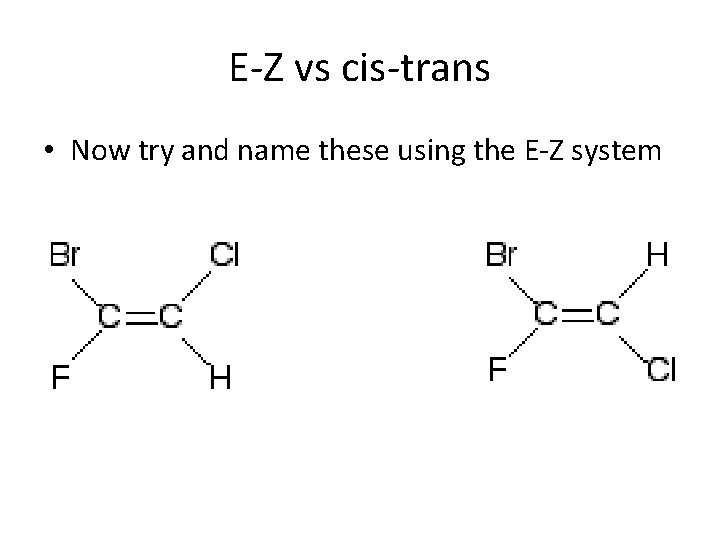
E-Z vs cis-trans • Now try and name these using the E-Z system
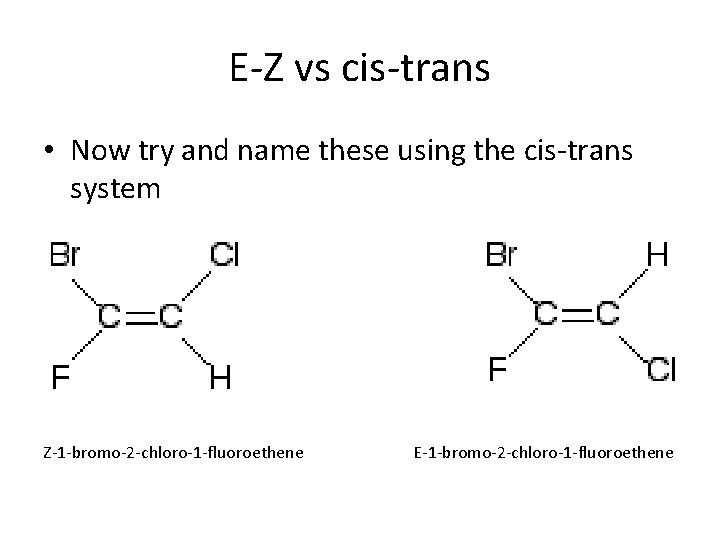
E-Z vs cis-trans • Now try and name these using the cis-trans system Z-1 -bromo-2 -chloro-1 -fluoroethene E-1 -bromo-2 -chloro-1 -fluoroethene
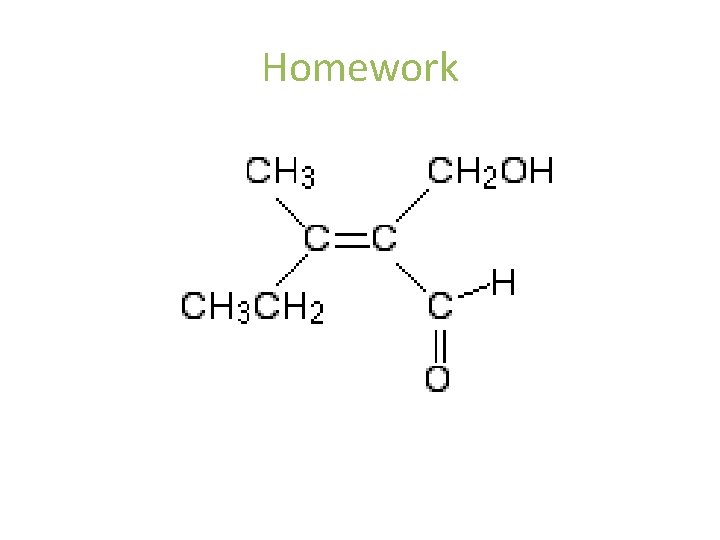
Homework

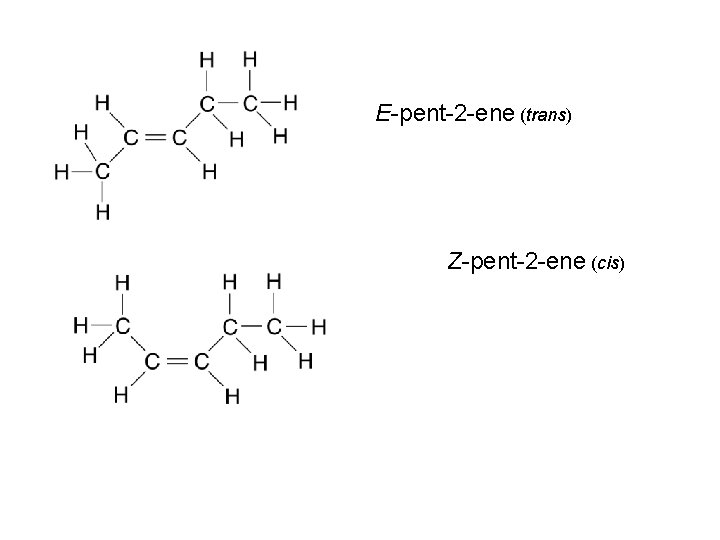
E-pent-2 -ene (trans) Z-pent-2 -ene (cis)
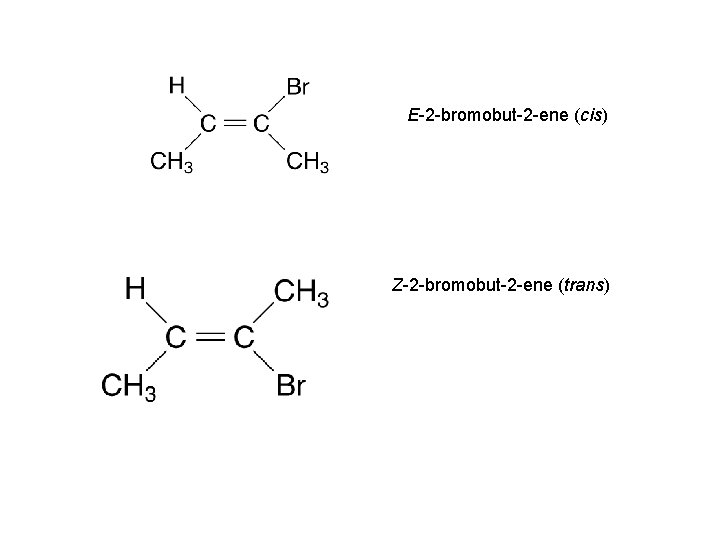
E-2 -bromobut-2 -ene (cis) Z-2 -bromobut-2 -ene (trans)
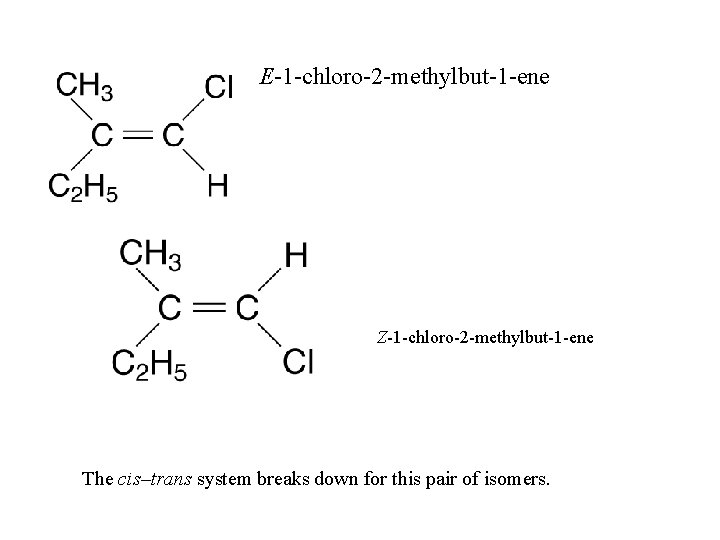
E-1 -chloro-2 -methylbut-1 -ene Z-1 -chloro-2 -methylbut-1 -ene The cis–trans system breaks down for this pair of isomers.

Optical isomers are: Mirror images; Non superimposable; Chiral*; Exclusive to asymmetrical compounds; No plane of symmetry; Simplest example has 4 different groups attached to a central carbon atom; • This carbon atom is known as the chiral centre; • Referred to as enantimers; • Optically active. * Chiral means hand in greek* • • •
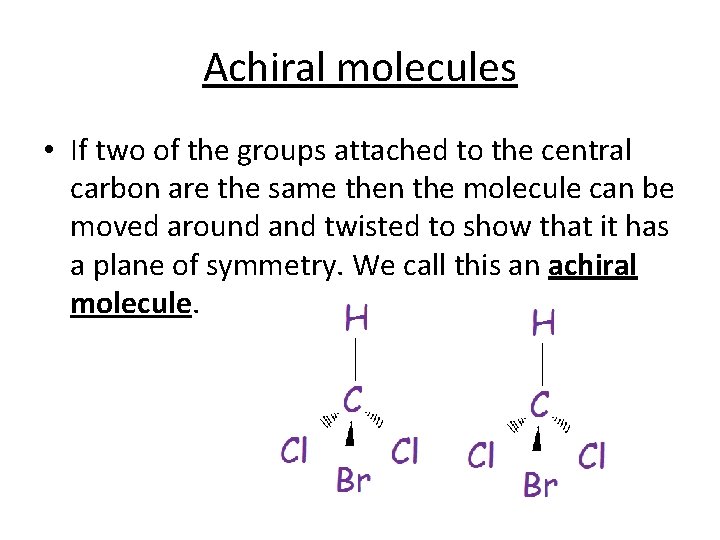
Achiral molecules • If two of the groups attached to the central carbon are the same then the molecule can be moved around and twisted to show that it has a plane of symmetry. We call this an achiral molecule.
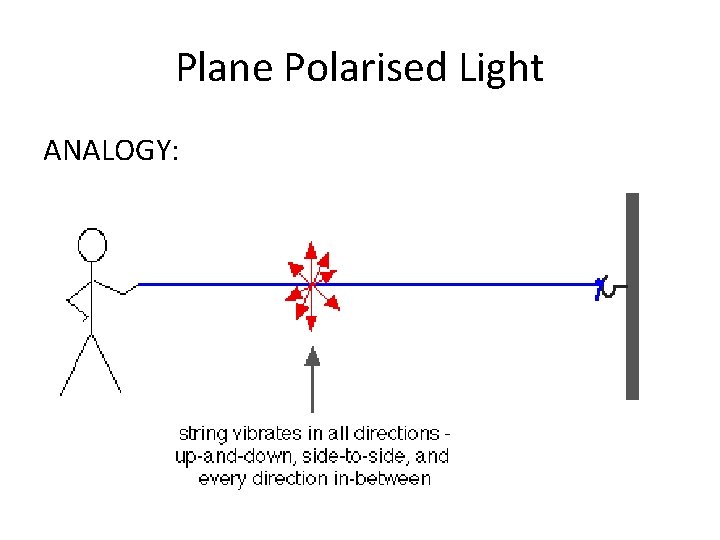
Plane Polarised Light ANALOGY:
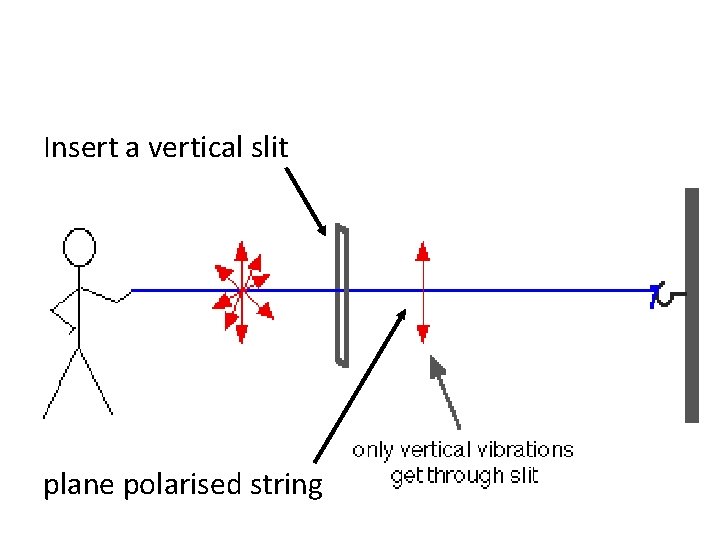
Insert a vertical slit plane polarised string
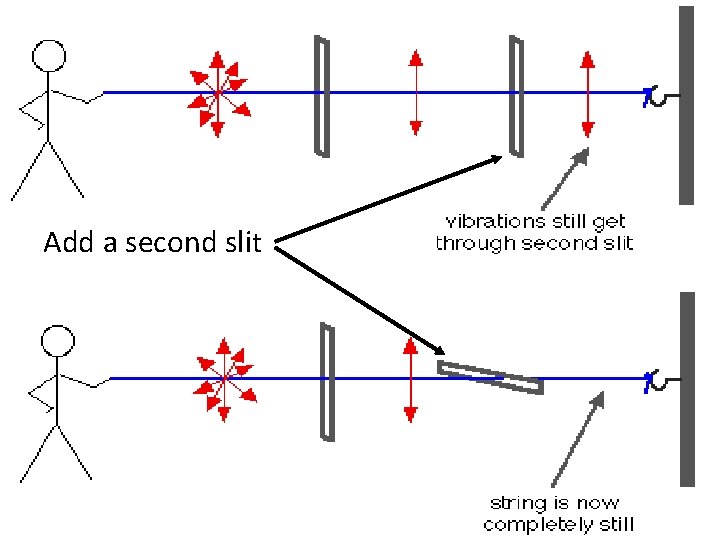
Add a second slit

The real thing - plane polarised light • Light is also made up of vibrations - this time, electromagnetic. • Polaroid has the ability to screen out all the vibrations apart from those in one plane and so produce plane polarised light. • If you wear one pair of Polaroid sunglasses and hold another pair up in front of them so that the glasses are held vertically rather than horizontally, you'll find that no light gets through - you will just see darkness. This is equivalent to the two slits at right angles in the string analogy. The polaroids are described as being "crossed".

Chiral Molecules • Enantiomers: – are optically active; – have different effects on plane polarised light; • A solution of one enantiomer rotates the plane of polarisation in a clockwise direction named the (+) form. • The other enantiomer will rotate the plane of polarisation in the other direction, named the (-) form. • A racemic mixture (racemate) has equal quantities of both the enantiomers.
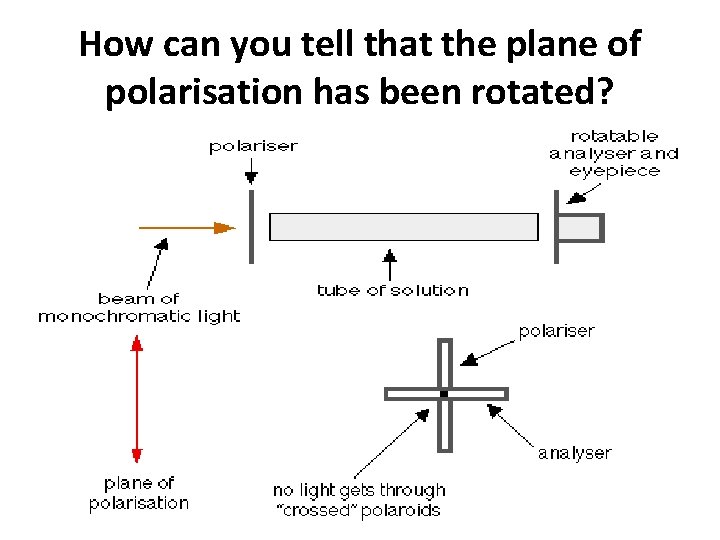
How can you tell that the plane of polarisation has been rotated?

Optically active sample: You can easily tell whether the plane of polarisation has been rotated clockwise or anti-clockwise, and by how much.
- Slides: 27