Optical isomerism Learning Objectives LO Be able to

Optical isomerism Learning Objectives (LO) • Be able to explain how racemic mixtures are formed • Be able to describe how enantiomers affect plane polarised light • Be able to identify molecules that exhibit optical isomerism Keywords: Stereoisomer, chiral, plane-polarised light, enantiomer, racemate, optically-active

SHOW CLIP
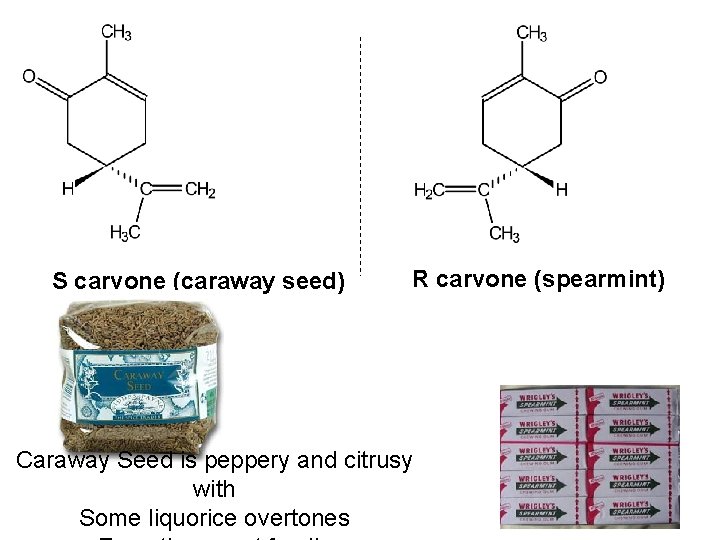
S carvone (caraway seed) R carvone (spearmint) Caraway Seed is peppery and citrusy with Some liquorice overtones
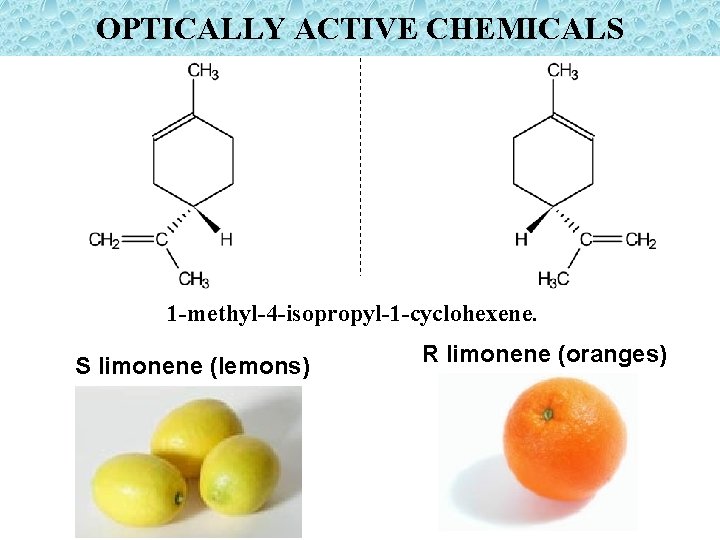
OPTICALLY ACTIVE CHEMICALS 1 -methyl-4 -isopropyl-1 -cyclohexene. S limonene (lemons) R limonene (oranges)

PLANE POLARISED LIGHT Light travels in waves in all directions from the source. It is like tying a piece of thick string to a hook in a wall, and then shaking the string vigorously; up and down and side to side. Now, suppose you passed the string through a vertical slit. The string is a really snug fit in the slit. The only vibrations still happening is one direction (up and down). What you have is "plane polarised string“. Well, light does the same thing, after it emerges from such slits it is called PLANE POLARISED LIGHT
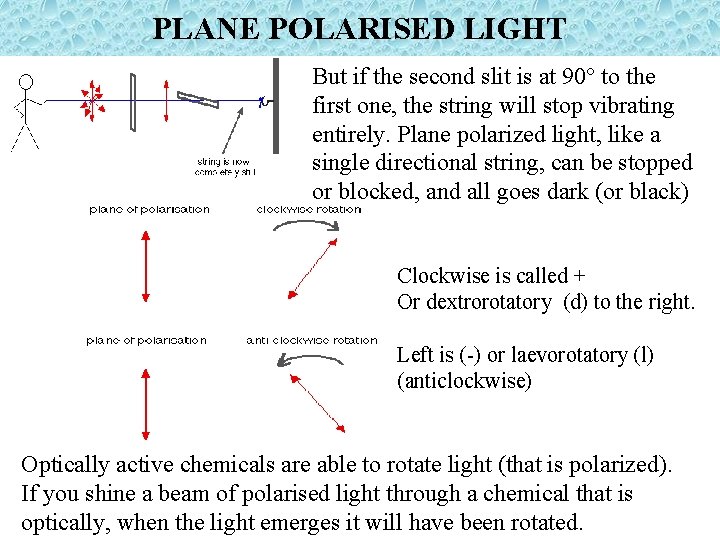
PLANE POLARISED LIGHT But if the second slit is at 90° to the first one, the string will stop vibrating entirely. Plane polarized light, like a single directional string, can be stopped or blocked, and all goes dark (or black) Clockwise is called + Or dextrorotatory (d) to the right. Left is (-) or laevorotatory (l) (anticlockwise) Optically active chemicals are able to rotate light (that is polarized). If you shine a beam of polarised light through a chemical that is optically, when the light emerges it will have been rotated.
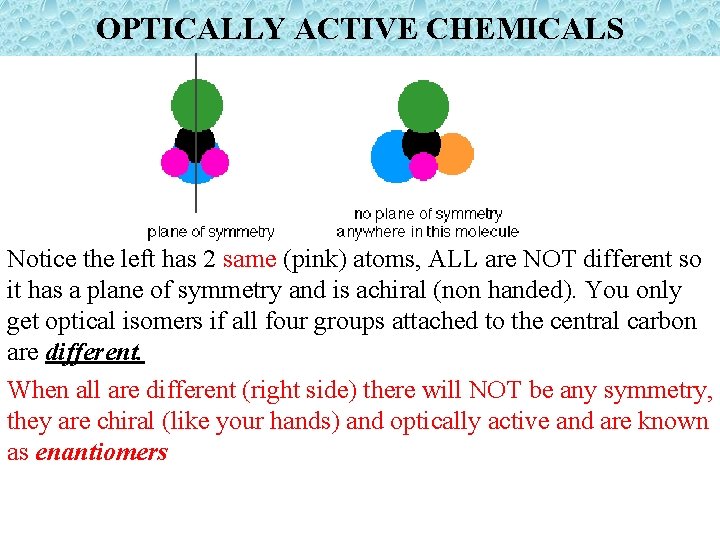
OPTICALLY ACTIVE CHEMICALS Notice the left has 2 same (pink) atoms, ALL are NOT different so it has a plane of symmetry and is achiral (non handed). You only get optical isomers if all four groups attached to the central carbon are different. When all are different (right side) there will NOT be any symmetry, they are chiral (like your hands) and optically active and are known as enantiomers

Chirality Chiral means handedness. Hands are mirror images and NOT superimposable. Try it. Optical isomers (enantiomers) like hands, are molecules that are mirror images and are non-superimposable

CHEMICALS and MIRROR IMAGES All molecules have a mirror image – but they are NOT optically active if they are not ALL different (notice 2 hydrogen's, so all atoms not different)

OPTICALLY ACTIVE CHEMICALS Chiral, all 4 groups are different, have mirror images which are not non-superimposable

Non Superimposable Mirror Image What is a non-superimposable mirror image? Wait a few seconds Animation doesn’t work in old versions of Powerpoint

Chirality and mirror images explained Film clip Khan Video ( 4 min) https: //www. youtube. com/watch? v=y. Ch. PRfm 4 yvg

CHEMICALS and MIRROR IMAGES Prove all this for yourself, use your molymods to build the following molecules: Build 2 of each • Methane • Bromochloromethane • Bromochlorofluoromethane Try to superimpose them onto one another, can it be done. mirror images of the following compounds: Which ones are superimposable? Which ones aren’t? What is the difference between them? Which are chiral, which achiral? Which are optically active?

OPTICALLY ACTIVE AMINO ACIDS Do amino acids (alanine, if R = CH 3 ) have 4 different groups? If Glycine, where R = H Answer : Yes, no Are they therefore chiral? Answer : Yes, no Is the amino acid a mirror images? Answer : Yes, no Are they superimposable? Answer : Yes, no Are they optically active, or will they rotate plane polarized light? Answer : Yes, no

SUMMARY • These are chiral molecules and so are optically active or are able to rotate light right (+) or left (-). They are called optical isomers or enantiomers • They are asymmetric carbons with four different groups arranged tetrahedrally around it. v A 50/50 mixture of the two enantiomers is a racemic mixture and is NOT optically active as the rotations cancel if present in equal amounts

SPOTTING CHIRAL CENTRES Look at each carbon atom in the chain and see what is attached to it. For a chiral centre you need an asymmetric carbon with four different atoms/groups) arranged tetrahedrally around it. IF A CARBON HAS MORE THAN ONE OF ANY ATOM/GROUP ATTACHED, IT CAN’T BE CHIRAL CH 3 CH 2 CH 2 Cl 1 -chlorobutane CH 3 CH 2 CHCl. CH 3 2 -chlorobutane (CH 3)2 CHCH 2 Cl 1 -chloro-2 -methylpropanane (CH 3)3 CCl 2 -chloro-2 -methylpropanane C C 3 H’s around it 2 H’s around it NOT chiral C C 3 H’s around it 2 H’s around it H, CH 3, Cl, C 2 H 5 around it 3 H’s around it NOT chiral CHIRAL NOT chiral C 3 H’s around it C 2 CH 3’s around it C 2 H’s around it NOT chiral C 3 H’s around it C 3 CH 3’s around it NOT chiral

TASK s e t u n i m 0 1 Are each of the molecules optically active? Draw them and identify the chiral carbon a) propan-2 -ol e) butanone b) 2 -chlorobutane f) 2 -methylbutanoic acid c) 1 -chlorobutane g) butan-2 -ol d) 3 -methylhexane h) 1 -chloro-3 -methylpentane
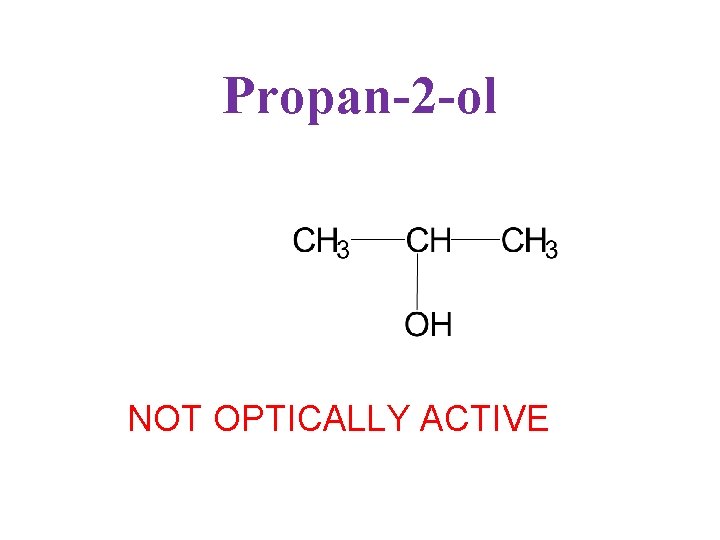
Propan-2 -ol NOT OPTICALLY ACTIVE

2 -chlorobutane OPTICALLY ACTIVE
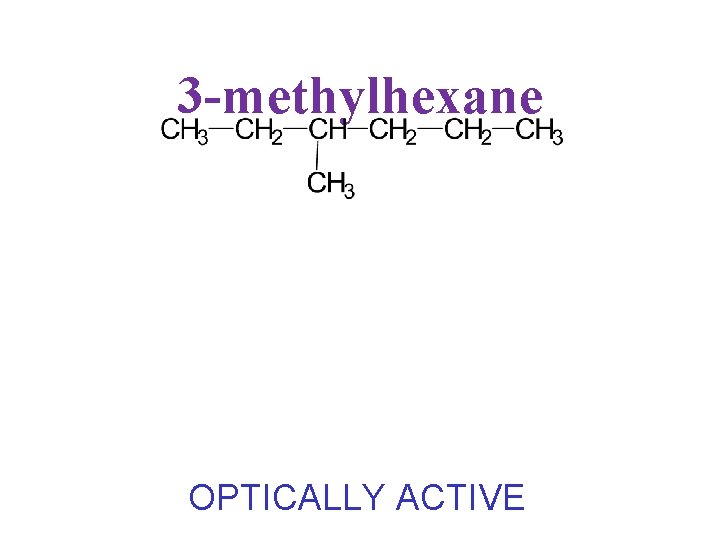
3 -methylhexane OPTICALLY ACTIVE
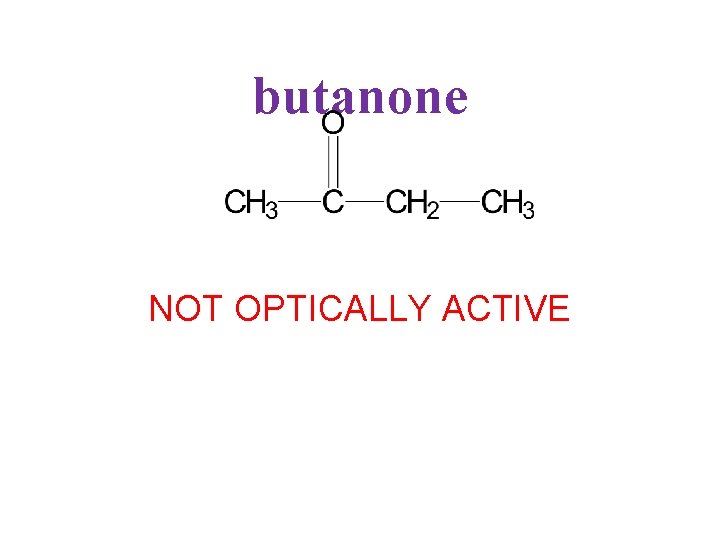
butanone NOT OPTICALLY ACTIVE
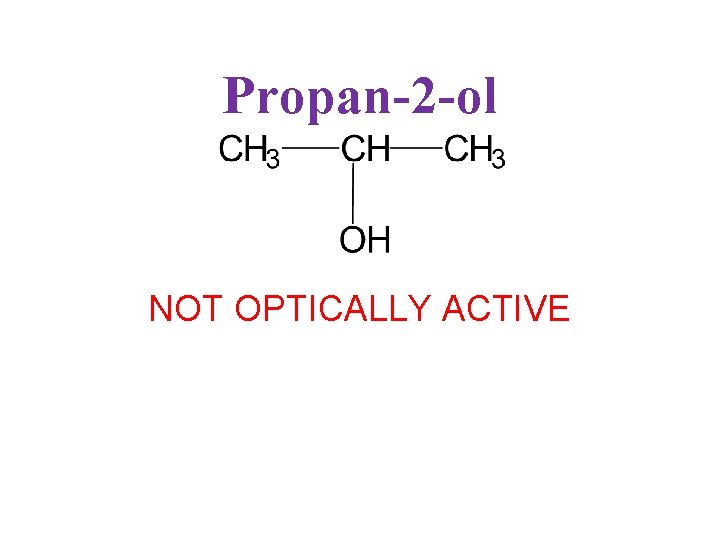
Propan-2 -ol NOT OPTICALLY ACTIVE
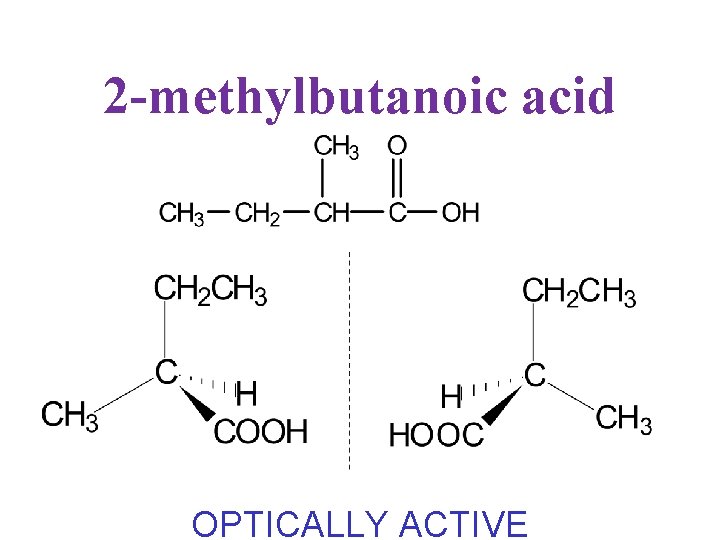
2 -methylbutanoic acid OPTICALLY ACTIVE
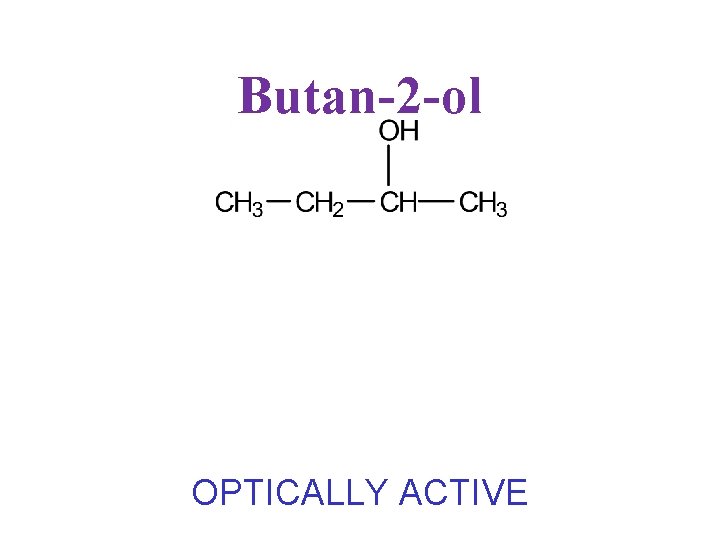
Butan-2 -ol OPTICALLY ACTIVE

1 -chloro-3 -methylpentane OPTICALLY ACTIVE
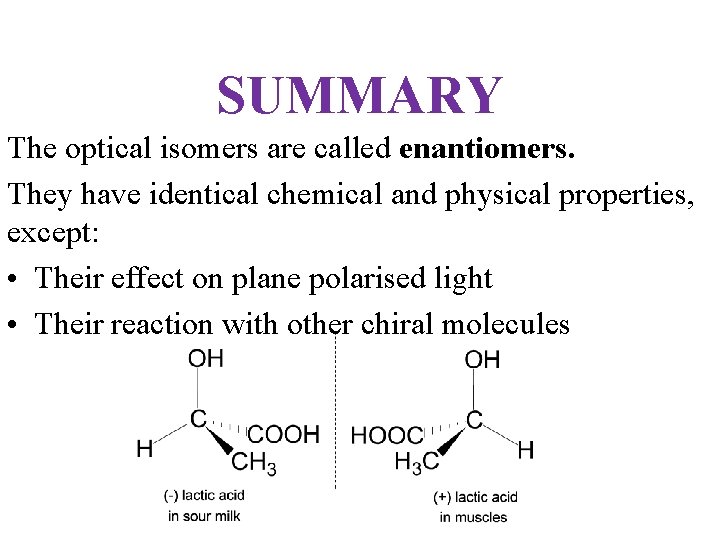
SUMMARY The optical isomers are called enantiomers. They have identical chemical and physical properties, except: • Their effect on plane polarised light • Their reaction with other chiral molecules
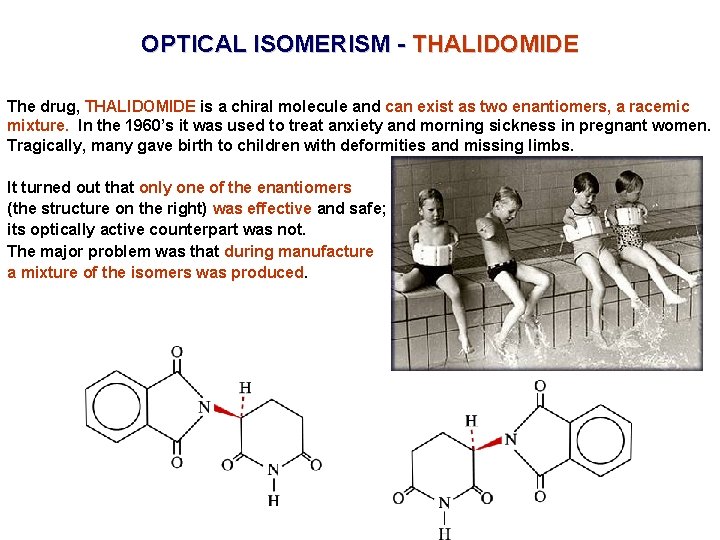
OPTICAL ISOMERISM - THALIDOMIDE The drug, THALIDOMIDE is a chiral molecule and can exist as two enantiomers, a racemic mixture. In the 1960’s it was used to treat anxiety and morning sickness in pregnant women. Tragically, many gave birth to children with deformities and missing limbs. It turned out that only one of the enantiomers (the structure on the right) was effective and safe; its optically active counterpart was not. The major problem was that during manufacture a mixture of the isomers was produced.

Thalidomide (2 min) https: //www. youtube. com/watch? v=n. Yn. LXz. Wwdf. U

S methamphetamine (illegal psychotic drug) R methamphetamine (nasal decongestant)

ALAN BAXTER • won silver in skiing • stripped of medal after failed drugs test • had used US Vicks inhaler • contained R-methamphetamine (not in UK version) – but original test did not distinguish this from enantiomer (which is stimulant & illegal) • despite the R-methamphetamine Winter Olympics 2002 in USA

Spot the chiral carbon (there may be one, none or many)

Check your answers… * 2 -bromobutane
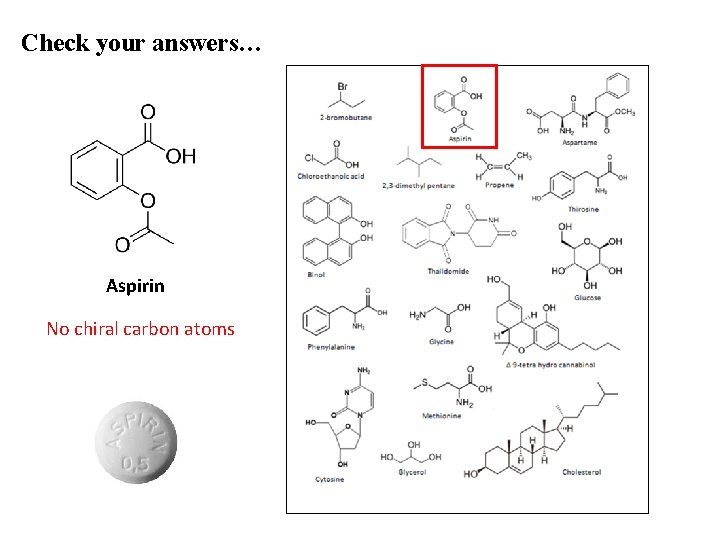
Check your answers… Aspirin No chiral carbon atoms
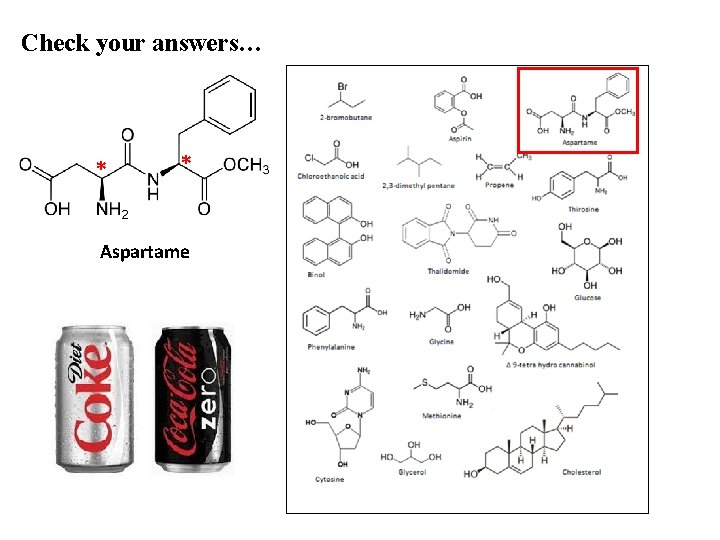
Check your answers… * * Aspartame
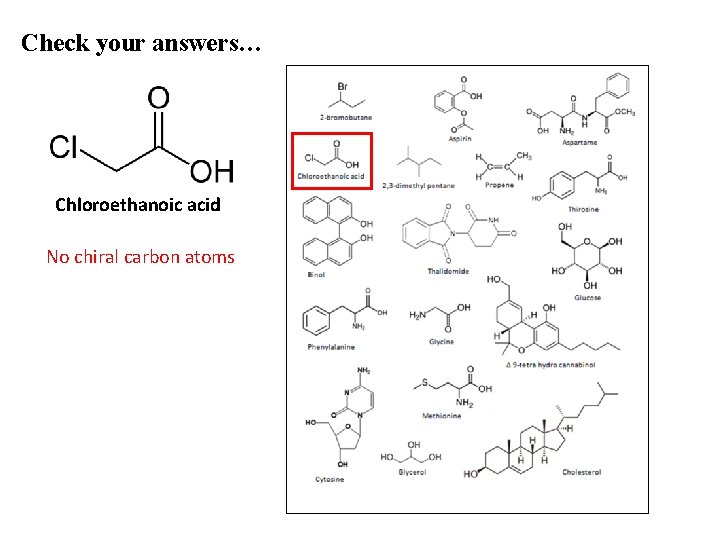
Check your answers… Chloroethanoic acid No chiral carbon atoms
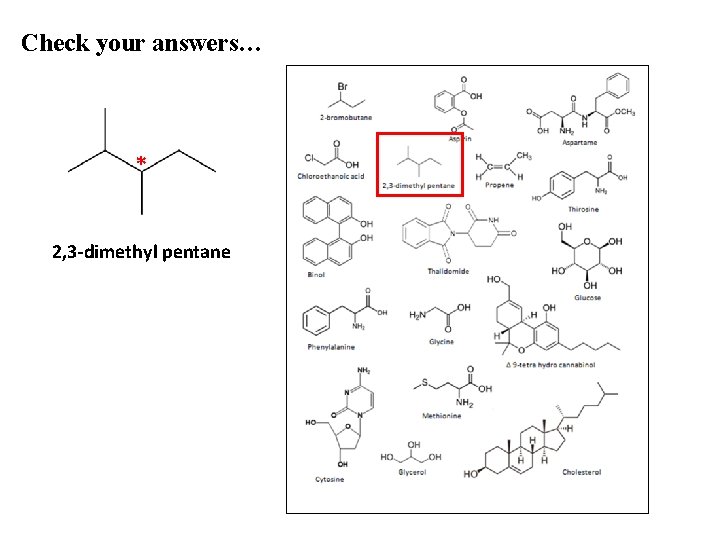
Check your answers… * 2, 3 -dimethyl pentane
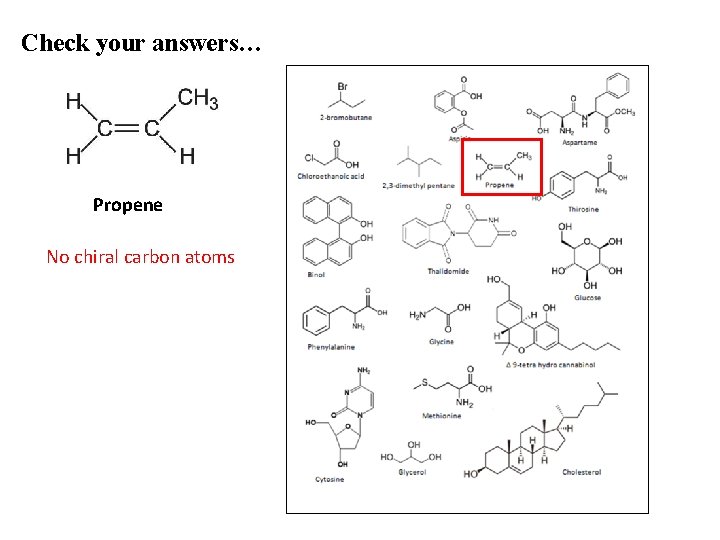
Check your answers… Propene No chiral carbon atoms
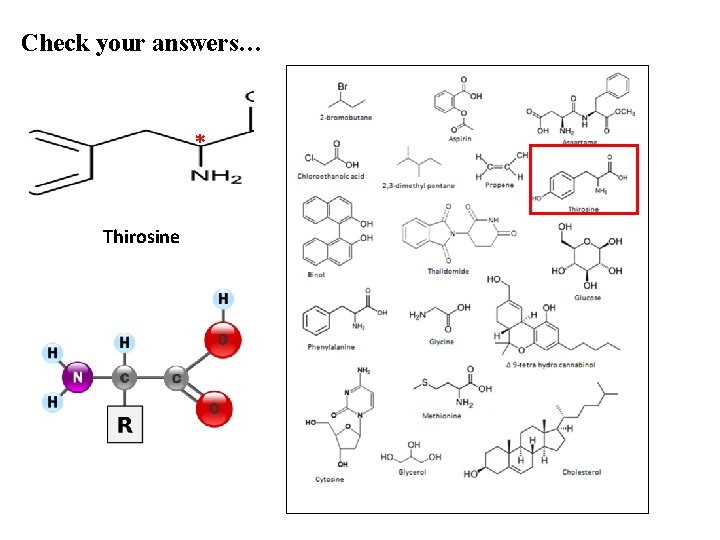
Check your answers… * Thirosine
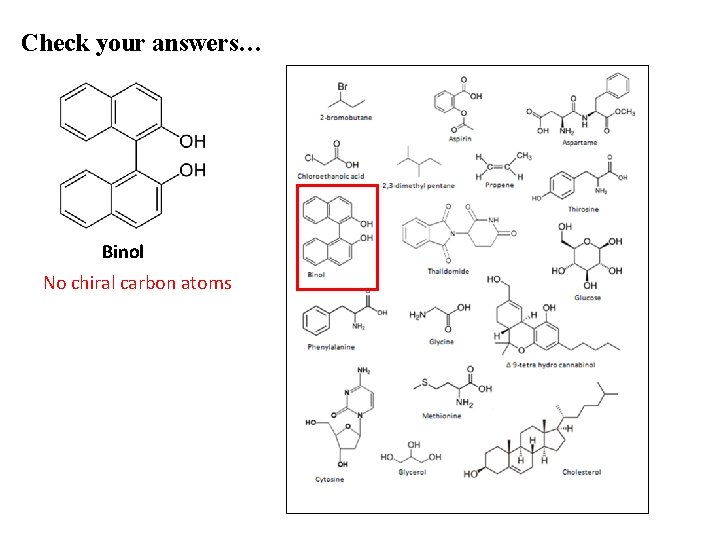
Check your answers… Binol No chiral carbon atoms
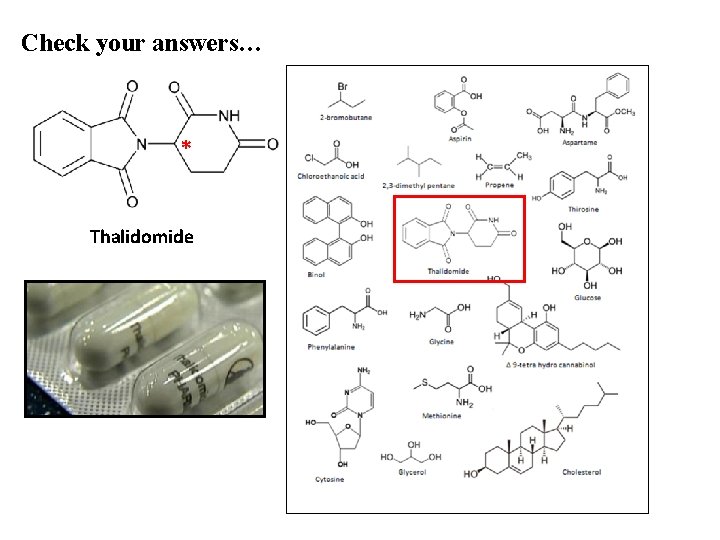
Check your answers… * Thalidomide
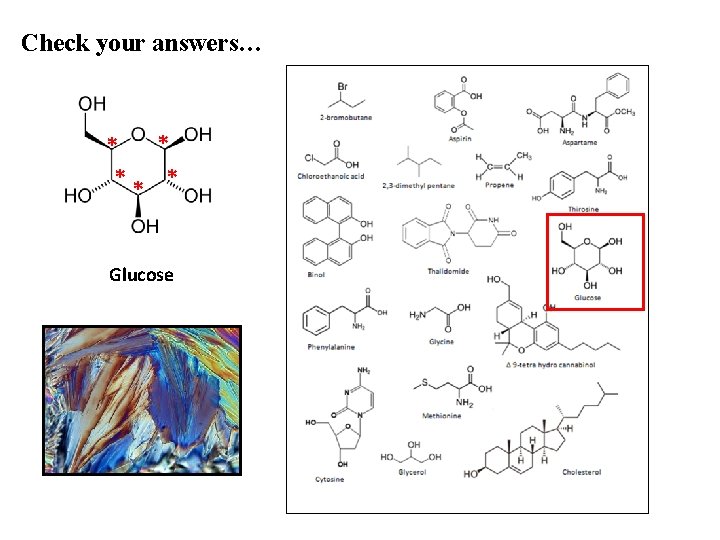
Check your answers… * * ** * Glucose
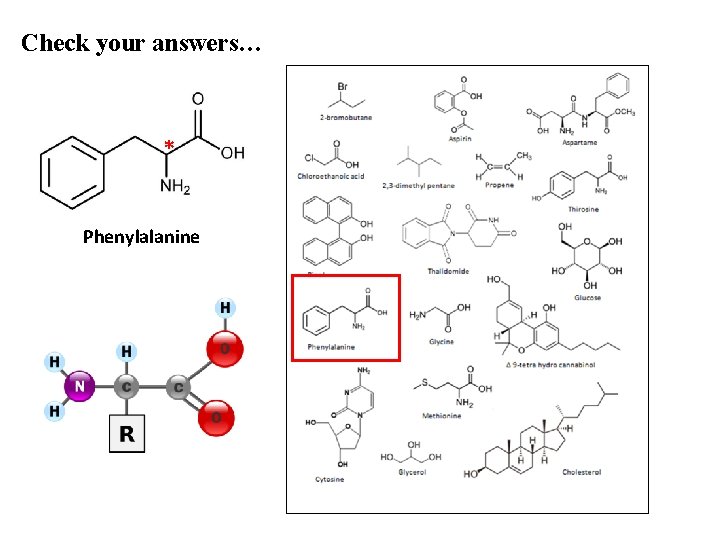
Check your answers… * Phenylalanine
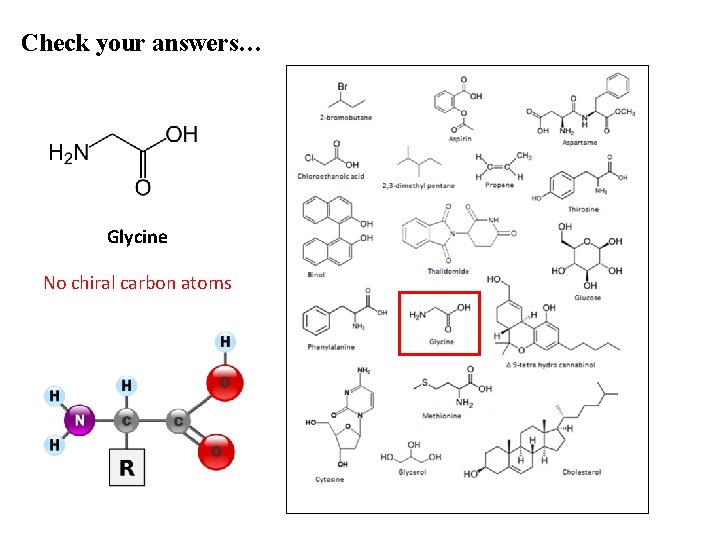
Check your answers… Glycine No chiral carbon atoms
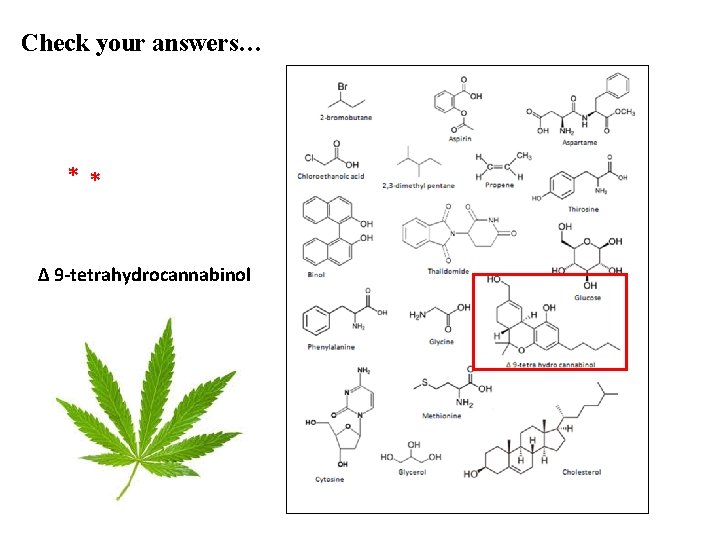
Check your answers… * * ∆ 9 -tetrahydrocannabinol
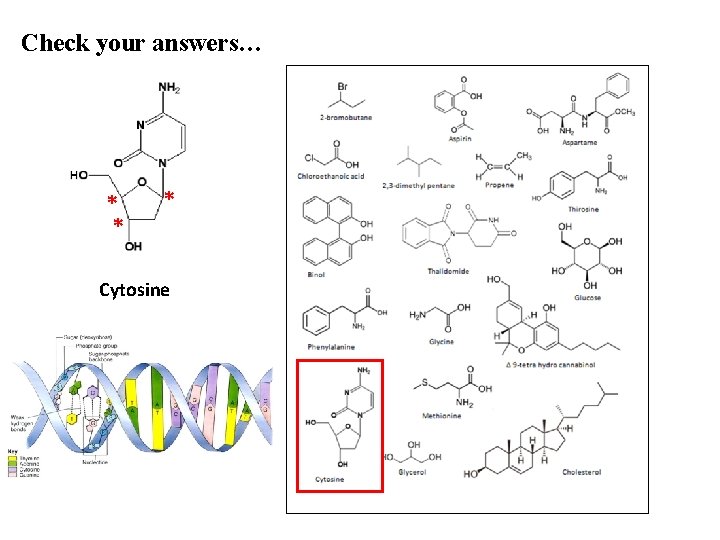
Check your answers… * * * Cytosine
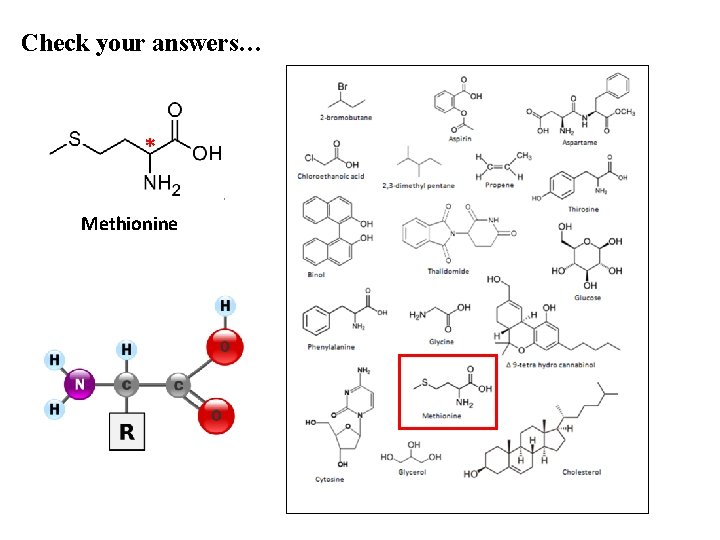
Check your answers… * Methionine
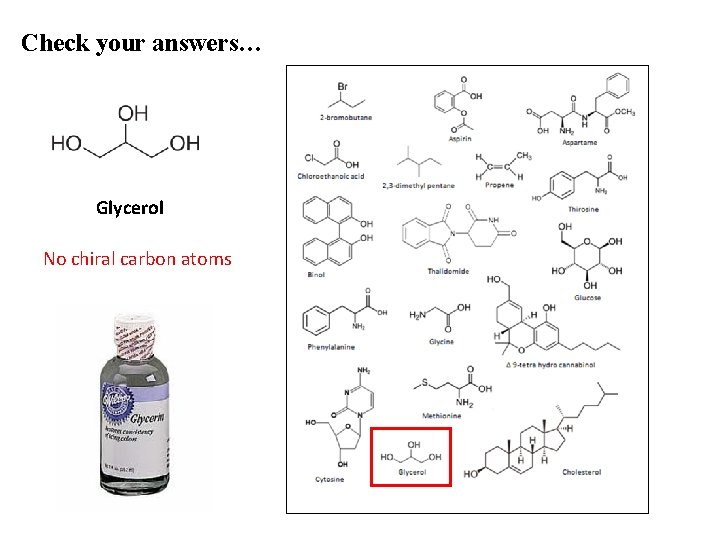
Check your answers… Glycerol No chiral carbon atoms
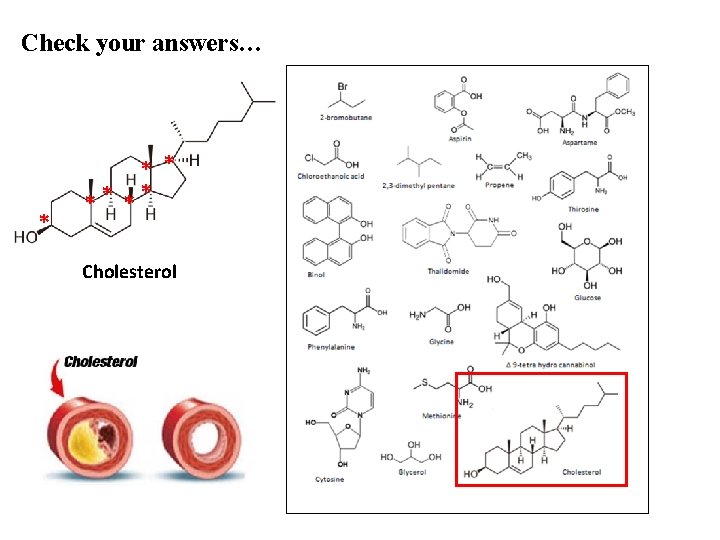
Check your answers… * ** ** Cholesterol
- Slides: 48