Optical Fiber Communication Lecture 8 Optical Sources LASER

Optical Fiber Communication Lecture 8: Optical Sources LASER Dr. Ghusoon Mohsin Ali M. Sc. in Electronics & Communication Department of Electrical Engineering College of Engineering Al-Mustansiriya University 1

The word LASER brings to mind a highly coherent source of light with a high output power and a high directivity of optical output. As we already know, LASER stands for „Light Amplification by Stimulated Emission of Radiation‟. The very first characteristic of a LASER lies in its name itself that LASER is in actual principle, an optical amplifier and not an optical source by itself. Like any amplifier, it receives the optical output from a low output optical source and then amplifies it by its internal processes. If we compare it to that of an LED, LED is truly an optical source which emits light via radiative recombinations. However, by providing proper feedback to the LASER, it can be converted into an oscillator and can then be treated as an optical source.

Coherence There are two types of coherence 1. Temporal Coherence 2. Spatial Coherence Considering light as an electromagnetic wave, we find that light would be comprised of periodic variations of electric and magnetic field with respect to time and space. If we observe the behaviour of the amplitude of the electric field of the light-wave as a function of time, the quantity that explains this behaviour is called temporal coherence. On the other hand, if we observe the behaviour of the amplitude of the electric field in a plane perpendicular to the direction of propagation as a function of space, the quantity that explains this behaviour, is called spatial coherence. Thus a highly focussed radiation is very important to ensure high efficiency. So, a lower value of „θ‟ is desirable for an optical source. Since the angle „θ‟ is related to the spatial coherence function, higher the spatial coherence smaller is the width „θ‟ of the power radiation pattern and higher is the focus of the generated radiation. The temporal coherence is important to obtain high bandwidths of communication and spatial coherence is required for high efficiency of optical source.

Coherence So the optical source that is desired in a high speed, long distance optical communication link is one with high efficiency and with highly coherent radiation (both temporally and spatially). In our search for such an optical source, scientists devised a semiconductor based source named LASER which meets all the mentioned requirements of an optical source to be used for high speed, long distance optical communication. Two photons are said to be coherent if and only if they have: (a) Same Energy „E‟ (=hν) (i. e. same frequency). (b) Same phase (c) Same momentum vector (both in magnitude and direction) (d) Same Polarization.

Having understood the basic meaning and types of the phenomenon of coherence, we shall now look into the processes that take place inside a semiconductor material and the conditions and requirements to be met so that the radiation from the material is coherent. This understanding would henceforth help in grasping the fundamentals of LASER technology which would be discussed subsequently. Consider a simple system of two energy levels, E 1 and E 2 in a semiconductor material where we have E 2>E 1. A radiative recombination between these two energy levels would correspond to photon of energy „Eph‟ given by: The density of electrons at any energy level in a semiconductor can be determined from the Boltzmann‟s Distribution. If „N 1‟ and „N 2‟ respectively are thermally generated electron densities at E 1 and E 2, then: This relationship suggests that the density of electrons decreases exponentially as we move towards higher energy levels in the semiconductor at the same temperature. In order to have a quantitative feel of the situation,

let us assume some typical values for the quantities on the R. H. S. of above equation and calculate the ratio of the two densities. For our convenience, let 0. 7μm be the wavelength of the emitted photon at room temperature (300 K). Then: As seen from the result, the density of electrons in E 2 , for a photon of energy corresponding to 0. 7μm, is almost negligible which suggests that at room temperature, the higher energy levels are almost empty and unoccupied by electrons as compared to lower energy levels. At thermal equilibrium, electrons in a semiconductor material tend to occupy lower energy levels. If an electron occupies a higher energy level, it is said to be in the excited state otherwise it said to be in the ground state.

Laser Transition Processes (Stimulated and Spontaneous Emission) ØThe transition of an electron from the excited state to the ground state can happen as a result of the natural tendency of the electron without the action of any external agent. The radiation produced as a result of such transitions is called as spontaneous radiation. ØWhen light is incident on a semiconductor material, some photons get absorbed in the material which then transfer their energy to the electrons in the ground state and cause them to migrate to the excited state. This phenomenon is called absorption. ØIn addition to the two phenomena mentioned above, there exists another type of emission from a semiconductor material which takes place under the action of an we illuminated this semiconductor with external photons, the externally incident photons may cause the electron in the excited state to jump down to the ground state by releasing the excess energy in the form of a photon. The externally incident photon does not get absorbed in the process; it just initiates the generation of this new photon by causing the electron in the excited state to release its energy and come down to the ground state. this process of emission is called stimulated emission.

Laser Transition Processes (Stimulated and Spontaneous Emission) Energy absorbed from the incoming photon Random release of energy Coherent release of energy

In order to study all the three phenomena above, certain coefficients have been assigned to these phenomena which are actually defined by the transition probabilities and are called as Einstein coefficients. The phenomenon that occurs in the absence of external stimulus (spontaneous emission) is assigned the coefficient, A 21. The phenomena that occur in presence of external stimuli are denoted by „B‟; the absorption phenomenon is assigned B 12 and the stimulated emission is assigned B 21. Note that the subscripts in the coefficients indicate the direction of transition from the initial level to the final level. Using these coefficients, the three processes can now be expressed in mathematically.

Spontaneous Emission: The rate of spontaneous emission as a result of electron transitions from the excited state to the ground state is directly proportional to the number of electrons in the excited state. The proportionality constant would then be the Einstein coefficient, A 21 mentioned above. Mathematically, If we define some sort of an average life-time of an electron in the excited state before its spontaneous transition to the ground state takes place and denote this life-time by „τsp‟, then: Equation suggests that the transition of electrons from the excited state to the ground state takes place exponentially and the electrons have an average life-time of „τsp‟ in the excited state before they undergo spontaneous transition.

Since the remaining processes occur only in presence of external photons, we assume an input photon flux which can be denoted by a flux density function „ρ(ν)‟, incident on the semiconductor material („ν‟ is the frequency of the incident photons). With this input photonic flux, let us now express absorption and stimulated emission mathematically. Absorption: The rate of absorptive transitions of electrons from the ground state to the excited state is directly proportional to the density of electrons in the ground state. It is also directly proportional to the incident photon flux density function. The proportionality constant, in this case would be given by the Einstein coefficient, B 12. Mathematically,

Stimulated Emission: The rate of stimulated transitions of electrons from the excited state to the ground state is directly proportional to the density of electrons in the excited state. It is also directly proportional to the incident photon flux density function. The proportionality constant, in this case would be given by the Einstein coefficient, B 21. Mathematically,

Thus we have three phenomena that may simultaneously occur inside a material; two processes cause electron transition from excited state to the ground state and the third causes electron transition from ground state to the excited state. At thermal equilibrium and without the injection of external electrons, the electron densities in the two states have to be maintained and so the total number of emissive transitions must be equal to the total number of absorptive transitions. Mathematically, Rearranging the equation, we may obtain the expression for the photon flux density function as:

The three distinct phenomena that occur inside a semiconductor material are the absorption, spontaneous emission and the stimulated emission phenomena. Spontaneous emission occurs without the influence of any external stimulating agent and hence is named so. On the other hand, absorption and stimulated emission occur in the presence of external stimuli and, hence, are called as stimulated processes. This external stimulus is usually light, which is allowed to be incident on the material. However, even when the material is illuminated, spontaneous emissions may occur if there are electrons in the excited state. The terms in the two energy levels E 1 and E 2 (E 2>E 1). We know that, (8. 2) (8. 3)

The Rayleigh-Jean’s law describes the spectral energy distribution in the radiation emitted by a ‘Black-Body’. This law is mathematically expressed as: (8. 4) Here, ‘n’ is the refractive index of the medium in which the radiation travels. The photon flux emanating from the material under study which is at thermal equilibrium at temperature ‘T’ may be considered similar to the radiations from a black body at thermal equilibrium at the same temperature. This leads us to compare equation 8. 3 with 8. 4 and, by doing so we may write the following expressions: (8. 5) (8. 6)

Equation suggests that there is an equal probability of occurrence of absorption and stimulated emission in a semiconductor material when light is incident on it. However, in nature we do not see the occurrence of stimulated emission but absorption is a more common phenomena observed when light is incident on a material. The reason for this lies in the Boltzmann’s distribution of equation 8. 2. ØIn practical situations, at thermal equilibrium, the electron density in the excited state is negligible. So the rate of stimulated emission is almost negligible in comparison to the rate of absorption. That is why, we see absorption phenomenon occurring more often than an emission phenomena. ØEmission phenomenon can occur only if there is electron density in the excited state, which occurs only when the material is energized. Hence, although the absorption and the stimulated emission processes are equally probable, the absorption process dominates over the stimulated process at thermal equilibrium. ØThe above discussion initiates a basic thought in the mind that, what would be the outcome if, by some means, the population (density) of electrons in the excited state is made much more in comparison to that in the ground state? The answer would be- the stimulated emission process would dominate over the absorption process.

ØSo, for stimulated emission to occur, first, we have to create a considerable density of electrons in the excited state from the ground state so that the ground state density of electrons becomes negligible in comparison to that in the excited state. ØThat is, we have to invert thermal equilibrium distribution of electrons in the material. This process is called as population inversion. This would cause the emissive process to be dominant over the absorptive process and the net result would be occurrence of stimulated emission from the material when it is illuminated. However, the net radiation from the material is not only due to the stimulated emission but also due to the spontaneous emission which emits highly incoherent light. ØIf we roll back to our discussion on the stimulated phenomena occurring between two energy levels E 1 and E 2 (E 2>E 1), we would realize that the photon that is absorbed or emitted out, also has an energy which is equal to the energy difference between the two levels i. e. (E 2 -E 1). In other words, only those photons whose energy equals the above energy difference are either absorbed or given out. One can refer to this observation as some kind of a resonance phenomenon which involves the emission or absorption of photons with a particular frequency ‘ν’ where this corresponds to the frequency of the above energy difference (hν=E 2 -E 1).

Whenever, light is incident on a material, three simultaneous processes occur in the material, viz. absorption, spontaneous emission and the stimulated emission. However, the contribution of the spontaneous emission over the stimulated emission in the net radiation from the material can be ascertained from a quantitative calculation as shown below. We already know that the rate of stimulated emission is directly proportional to the product of the Einstein coefficient, B 21 and the incident photonic flux density, ρ(ν); whereas the spontaneous emission is proportional to the Einstein coefficient, A 21. Hence, to find the contribution of one over the other we have to find a ratio between these proportionalities. This ratio can be calculated from equation 8. 3 above, and is given as: (8. 7) For a radiation having wavelength, λ=1μm, the exponential term in the denominator of equation turns out to approximately e 50, at room temperature. Thus the ratio: (8. 8)

HW

ØSince e-50<<1, the equation 8. 8 suggests the fact that stimulated emission is practically non-existent at room temperature, under thermal equilibrium. Hence the net radiation from the material is highly incoherent since it takes place due to spontaneous emissions which are incoherent and are called as black-body radiations. ØThe stimulated emission process is a cumulative process which causes multiplication in the number of emitted photons. On the other hand, spontaneous emission process is indebted to the natural tendency of the excited electron to go down to the ground state by release of energy. ØThe carrier life-time against spontaneous emission is much larger than the carrier lifetime against stimulated emission which is a stimulated process. This fact causes the stimulated emission to dominate over the spontaneous process, as soon as photons are made incident onto the material and the emission predominantly is stimulated rather than spontaneous. Ø Therefore, in the subsequent discussions about emission of photons, we shall emphasize only on the stimulated emission process.

ØWe are already familiar with the fact that to have a stimulated emission of photons from a material, there has to a population inversion of the electrons created apriori. ØTo create population inversion, the electrons have to be supplied with the required amount of energy to cause them to migrate to the excited state. Ø The initial energy to be supplied to the electrons to migrate to the excited state thus equals the energy of the photon that is emitted out. In particular, the frequency of the incident photon (to cause absorption) has to be exactly equal to the energy of the emitted photon (by stimulated emission). ØThat is, to have a coherent emission we must already have coherent photons incident on to the material. This is practically not possible. In other words, this fact suggests that a simple two energy level system of E 1 and E 2 (E 2>E 1) would, rather, be inappropriate to realise a LASER action in practice. So, the obvious alternative that comes to the mind is to have an energy level system which has more than two energy levels. This type of a system is shown in figure 8. 1 below.

As seen from the figure, a beam of incoherent photons is made to illuminate the material, which causes the electrons in the ground state to migrate to an excited state (E 3) that is higher in energy than the required excited state (E 2). This excited electron then quickly jumps to E 2 by a spontaneous process of release of energy and then is released out as a photon of the required nature by decaying to the ground state as a result of stimulated emission. Figure 8. 1: System of more than two energy levels

ØIf the time constant against the spontaneous decay (τ32) of the electron from E 3 to E 2 is negligible small in comparison to the carrier lifetime against stimulated emission (τ21) then there would be majority electrons already available for stimulated emission in the excited state and, hence, would result in a population inversion in the material. ØSo, when a triggering photon of the required energy is incident on the material, the stimulated emission process would instantly take over and there would be a emission of photons from the material. The LASER mechanism to function in an appropriate manner, a material with the above energy level system (three energy level system) is necessary. ØAlthough, in principle, the three energy level system appears to be sufficient in producing a LASER, yet in practical scenario, the three energy level system is very inefficient. ØThe reason of this inefficiency lies in working of this system. According to its functioning, a population inversion is created by means of transporting electrons from the ground state to a excited state higher in energy than the required excited state and then triggering photons are allowed to be incident on to the material which would cause stimulated emissions thereafter. ØHowever, one very important observation to emphasize is that, when the triggering photon is incident on the material, it has equal probability to be absorbed as well as to stimulate an emission, as already discussed earlier.
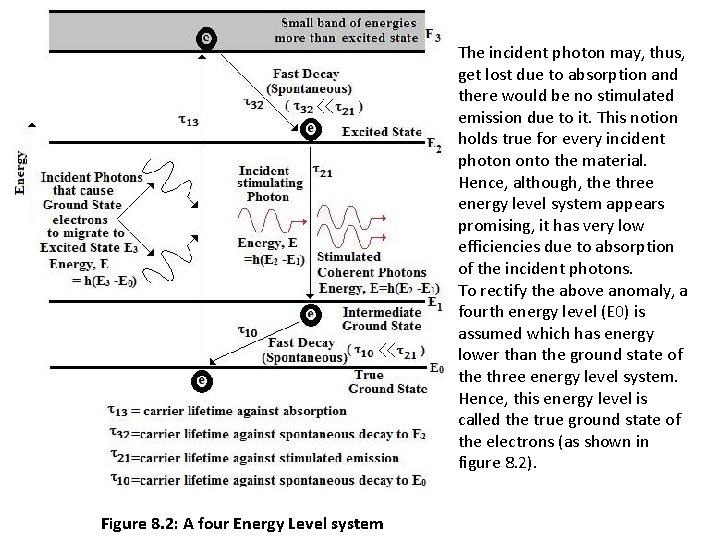
The incident photon may, thus, get lost due to absorption and there would be no stimulated emission due to it. This notion holds true for every incident photon onto the material. Hence, although, the three energy level system appears promising, it has very low efficiencies due to absorption of the incident photons. To rectify the above anomaly, a fourth energy level (E 0) is assumed which has energy lower than the ground state of the three energy level system. Hence, this energy level is called the true ground state of the electrons (as shown in figure 8. 2). Figure 8. 2: A four Energy Level system

ØIn order to create a population inversion, electrons are caused to migrate from this true ground state to E 3 and these electrons then undergo a fast spontaneous decay to E 2 and remain there until they jump down to E 1 by stimulated emission. After reaching E 1 these electrons again undergo a fast spontaneous decay to E 0 and remain there until they absorb energy to jump to E 3. ØThus a four energy level system is better in efficiency than a three energy level system. Thus for a LASER to function appropriately in practice, a material with a four energy level system is a basic necessity. In practical LASERs, however, materials with more complicated energy level systems are used for better performance and maximum efficiencies with the available power.

ØSince the stimulated emission dominates over the spontaneous emission in a material when photons are incident, it is only the stimulated processes that effectively constitute the net processes in a material after it is illuminated with photons. Therefore, the net downward transition is given by: This equation suggests that if N 2>N 1 (which is a case of population inversion), there would be greater number of downward transitions (stimulated emissions); whereas if N 2<N 1, the number of upward transitions (absorptions) would exceed the number of downward transitions. If we assume ‘Np’ be the number of photons emitted from the material over an interval of ‘t’ seconds, then:

If we multiply both sides of the equation by the energy of an emitted photon ‘hν’, we find that the quantity ‘hνNp’ is actually the photon flux density function ‘ρ(ν)’ and the equation modifies to: If we substitute the value of the coefficient B 21, calculated from equation 8. 5 (where A 21 is the reciprocal of the carrier lifetime against spontaneous emission ‘τsp’) then the above equation may be re-written as: (8. 13)

The equation 8. 13 shows that the rate of generation of photons with time. However, a photon is actually an electromagnetic wave travelling with the speed of light as soon as it is generated. The distance travelled by the photon in time ‘t’ is, hence, equal to the distance travelled by light in the same time. In the differential form we may write this as: (8. 14) Here ‘n’ is the refractive index of the medium in which the light travels. Equation 16. 14 helps us to transform equation 8. 13 from the time domain to the space domain which may be done by substituting the value ‘dt’ from equation 8. 14 into 8. 13. That is (8. 15)

Equation 8. 15 is a differential equation which can be solved by classical methods and the solution is of the form: (8. 16) Equation 8. 16 suggests that ‘G’ be termed as a Gain constant of the system. Although there are various parameters on which the value of G depends (as seen above), the significant parameter on which the value of G depends is the term (N 2 -N 1).

This term, hence generates three distinct cases for the value of G which may be state as: Case 1: N 2<N 1; G is Negative (G<0) Since G is negative, the photon flux decays exponentially as it travels inside the material and so, G , in this case, may be termed as the attenuation constant. Case 2: N 2=N 1; G=0 Since G=0, there is neither decay nor growth of the photon flux as it travels inside the material. Hence, this condition may be termed as the condition of transparency of the material. Case 3: N 2>N 1; G is Positive (G>0) This is the case of population inversion. In this case, we see that the value of G is positive and so the photon flux grows exponentially as it travels inside the material. Thus, if a condition of population inversion is maintained and the emitted photon flux is caused to travel repeatedly inside the material (before it escapes the material), it grows exponentially. Thus the goal is to make the generated radiation travel more and more inside the material while maintaining the population inversion in order to have an amplified coherent stimulated emission from the material.

The variation of photonic flux generated from a material, stimulated by an external illumination, as a function of the distance moved by the photons in the material may be described by the following differential equation: (8. 17) (8. 17 b) The quantity within the square brackets is referred to as the gain-constant (G) of the photonic flux density function. Out of the three distinct above cases, our discussion shall be centred around the case which signifies the population inversion scenario (N 2>N 1), because in this case, not only does the lasing action successfully take place but also the photonic flux grows exponentially as it spends more and more time inside the material by traveling repeatedly inside the material while the population inversion condition continues.

If we observe the expression of the gain constant G as shown in the equation 8. 17 above, we may derive the following proportionalities for the value of G. (8. 18) (8. 19) (8. 20) ØAs it is obvious from the above equations, G is directly proportional to the difference in electron concentrations between the two lasing levels. ØEquation 8. 19 suggests that G varies inversely with the square of frequency of the photon. In other words, the gain at longer wavelengths is higher than that at shorter wavelengths. This means, when a material having multiple excited and ground state energy levels is stimulated to emit radiations, the longer wavelength radiations get amplified faster than the shorter wavelengths and the population inversion condition then gets more involved in the amplification of the longer wavelength signals. That is, it is easy to manufacture a LASER for longer wavelengths than for shorter wavelengths.

ØAlso, the value of G varies inversely as the square of the refractive index of the material. So, if we use a material of low refractive index, the gain would be higher than that with a material with higher refractive index. ØHowever, one of the important parameters on which the gain depends, is the carrier lifetime against spontaneous emission „τsp‟. The value of gain varies inversely with „τsp‟. This means, a material with very low carrier lifetime against spontaneous emission would exhibit a high photonic flux gain. In other words, in a material where the electrons in the excited state are more ready to jump to the ground state via spontaneous emission rather than stimulated emission, shows a better exponential increase in the photonic flux density. The ease of causing a stimulated emission increases when the electrons themselves are in a tendency to jump down to the ground state. Shorter the lifetime more is the number of spontaneous emissions. However, higher number of spontaneous emissions causes the population inversion condition to die out faster and so we have to keep on replenishing electrons in the excited state to maintain the population inversion condition to achieve higher gain.

ØThe process of stimulated emission requires externally incident photon flux, of precisely the same energy as that need to be generated, to have an amplified output optical intensity from the material. ØHowever, the LASER has to be used as a source of light rather than an amplifier. This indicates the absence of an externally incident photon flux for the LASER because “to generate light we cannot already have a light”. Ø If there is no incident photon flux on the LASER, in principle there is no input to an amplifier and so the LASER seems to be of little use to serve as an optical source. ØHowever, one should note that once a population inversion condition is created inside the material of the LASER, the small spontaneous carrier lifetime initiates some spontaneous emission flux which serves as the triggering flux for the lasing action of the LASER to start. ØOnce the stimulated emission process starts, due to the very low carrier lifetime against stimulated emission (which is in fact lower than the spontaneous carrier emission lifetime „τsp‟) the stimulated emission process starts dominating over the spontaneous process and we obtain a net output photonic flux as a result of stimulated emissions.
- Slides: 34