Optical detection and quantification of radiocarbon dioxide 14

Optical detection and quantification of radiocarbon dioxide (14 CO 2) at and below ambient levels D. A. Long, A. J. Fleisher, Q. Liu, and J. T. Hodges National Institute of Standards and Technology Gaithersburg, Maryland HDR Architecture, Inc.

14 C • Long-lived isotope of carbon (half-life 5, 730 years) • Used in carbon dating • Present abundance is 1. 2 parts-per-trillion (ppt) • Fossil fuels and their products are nearly entirely depleted in 14 C

Why measure 14 C? • Bioplastics • Biofuels and biofuel blends • Identify illicitly traded specimens • Atmospheric CO 2 source apportionment • Radiocarbon dating

Current method – AMS • • Measurements of 14 C are extremely difficult due to low natural abundance (~1. 2 ppt) AMS uses an accelerator to mass separate the analyte Then analyzed using mass spectrometry High precision (generally 0. 2 -3%) • Disadvantages: -Expensive -Requires a large facility and highly trained staff -Very limited number of facilities -Can’t be performed in situ -Sample destructive image from LLNL
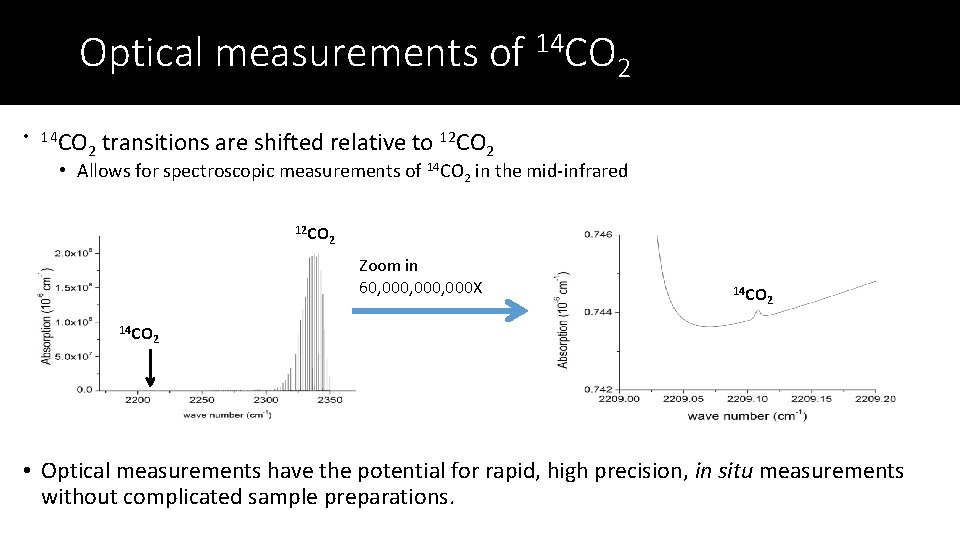
Optical measurements of 14 CO 2 • 14 CO 2 transitions are shifted relative to 12 CO 2 • Allows for spectroscopic measurements of 14 CO 2 in the mid-infrared 12 CO 2 Zoom in 60, 000, 000 X 14 CO 2 2 • Optical measurements have the potential for rapid, high precision, in situ measurements without complicated sample preparations.

Why is this so challenging? • The maximum 14 CO 2 concentration we will observe is 1. 2 ppt. • At our sample pressure of 7 Torr this corresponds to only 4× 106 14 CO 2 molecules in the laser beam. • Over a single pass of our 1. 5 m path at most 2. 7× 10 -8 of the light is absorbed by the 14 CO 2. • In addition, there are spectral interferences from a wide range of other CO 2 isotopes and N 2 O. • Finally, the mid-infrared laser technology is significantly less advanced than in the more common telecom region (i. e. , near 1. 5 µm). • Due to these challenges, optical detection at ambient levels has been demonstrated by only three research groups (CNR-Italy, NIST, LLNL).
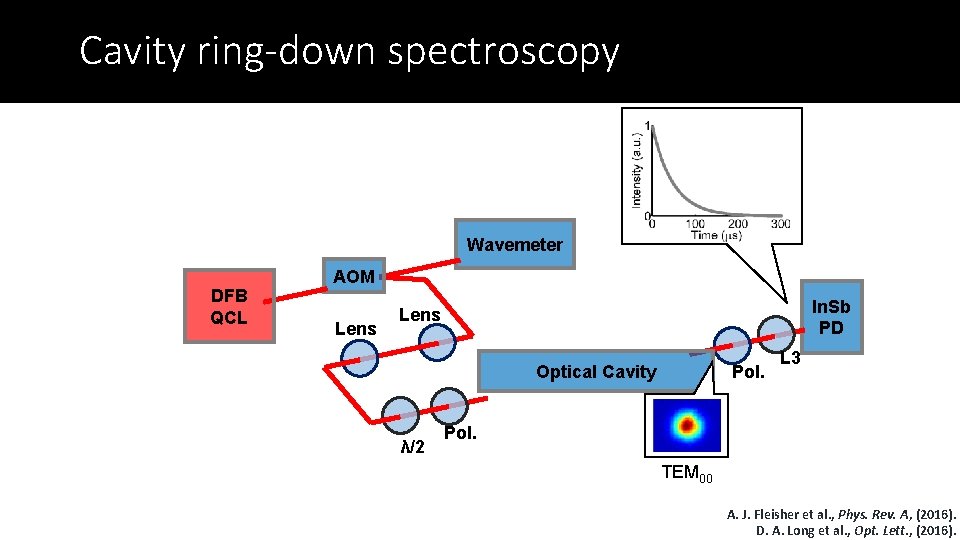
Cavity ring-down spectroscopy Wavemeter DFB QCL AOM Lens In. Sb PD Lens Pol. Optical Cavity λ/2 L 3 Pol. TEM 00 A. J. Fleisher et al. , Phys. Rev. A, (2016). D. A. Long et al. , Opt. Lett. , (2016).
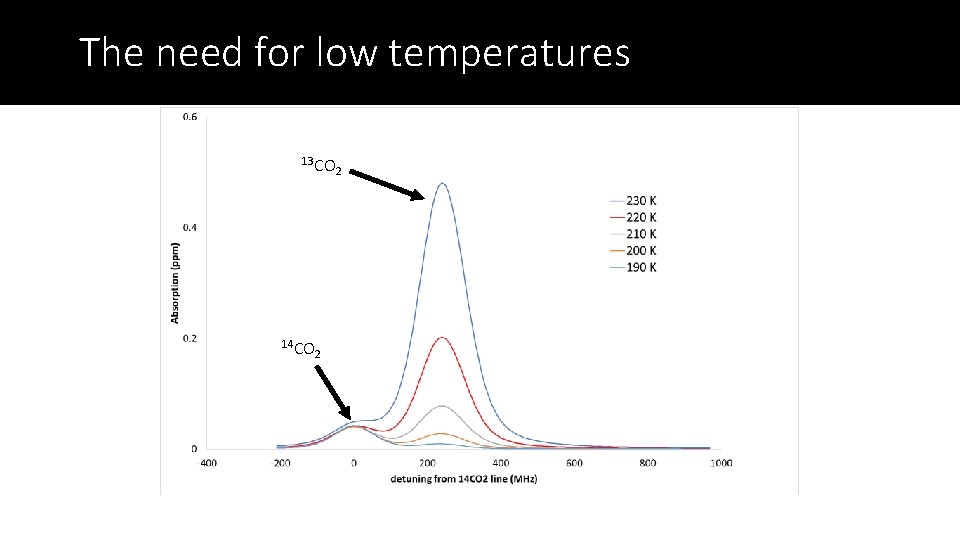
The need for low temperatures 13 CO 14 CO 2 2

CLR PR M 2
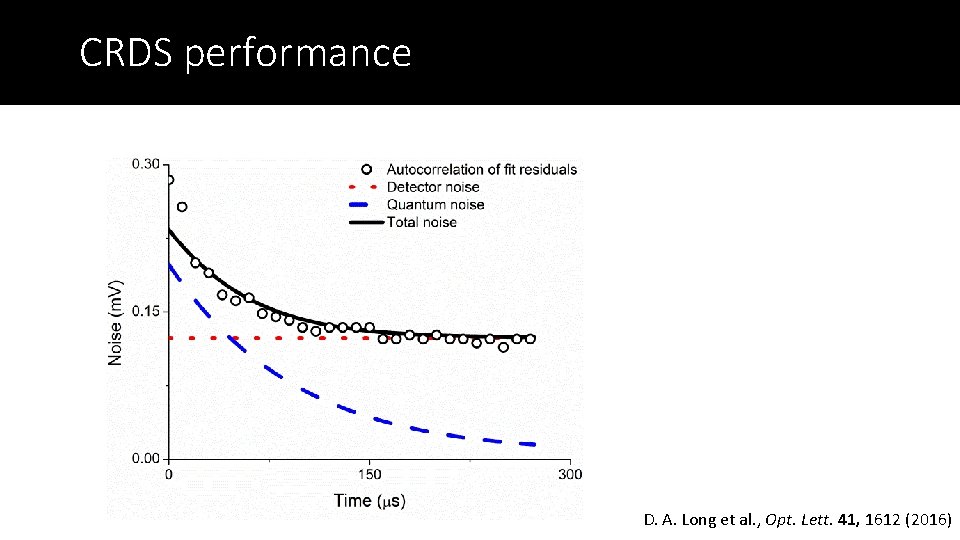
CRDS performance D. A. Long et al. , Opt. Lett. 41, 1612 (2016)
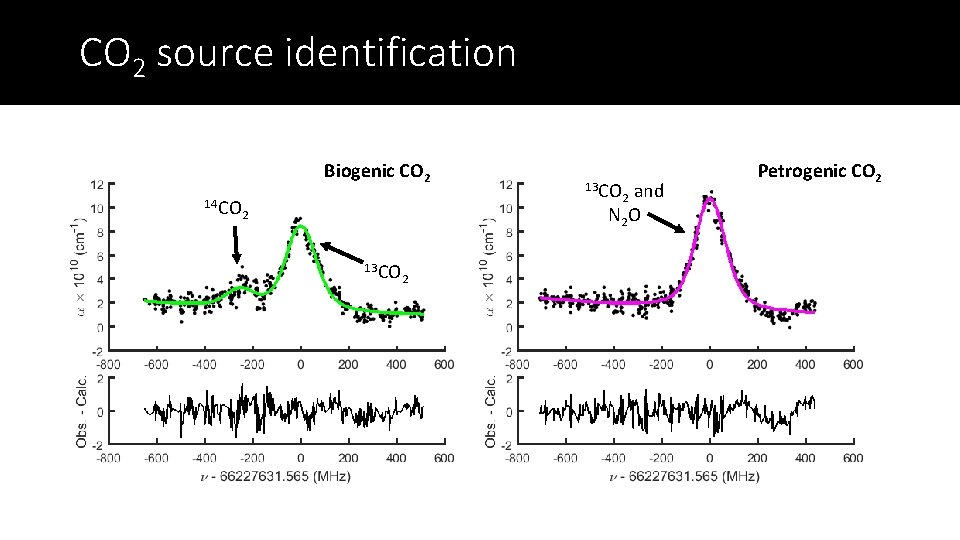
CO 2 source identification Biogenic CO 2 14 CO 13 CO 2 and N 2 O 2 13 CO 2 Petrogenic CO 2
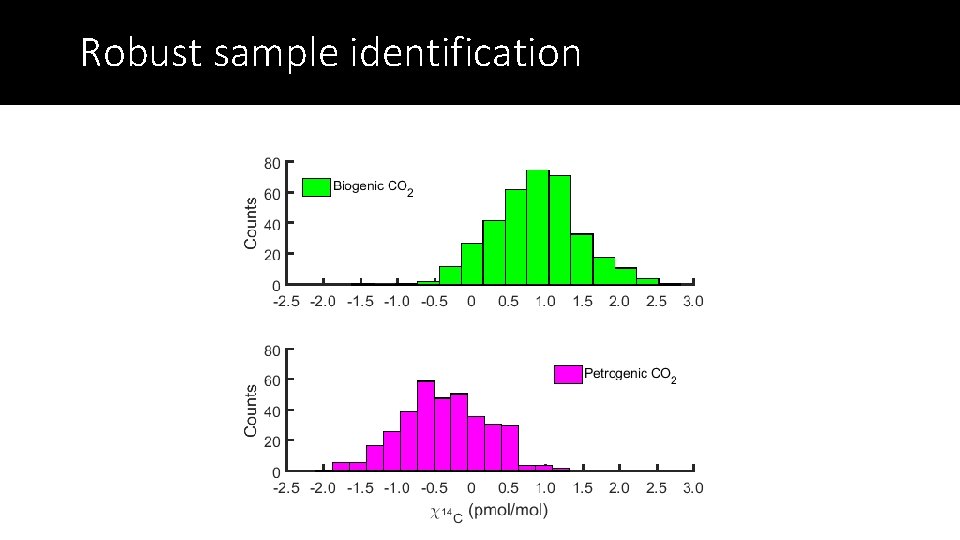
Robust sample identification
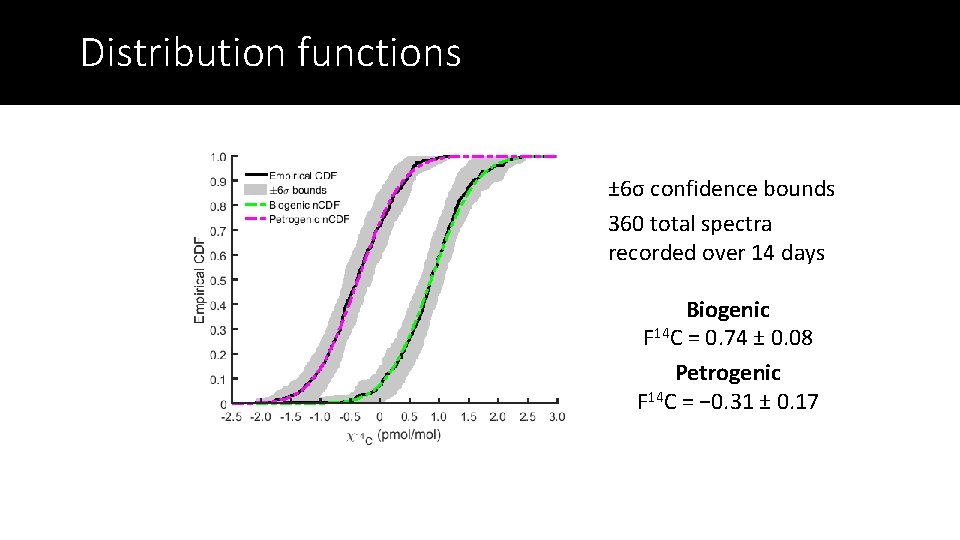
Distribution functions ± 6σ confidence bounds 360 total spectra recorded over 14 days Biogenic F 14 C = 0. 74 ± 0. 08 Petrogenic F 14 C = − 0. 31 ± 0. 17

Uncertainty budget and sensitivity Combined F 14 C = 8. 1 % N = 40 scans T = 47 minutes 14 C/12 C = 96 fmol/mol or 96 parts-per-quadrillion (ppq)

Conclusions • Developed a low temperature (200 K) cavity ring-down spectrometer in the mid-infrared. • Have measured the 14 C content of fossil fuel and biofuel samples. • After 1 hour can reach a measurement precision < 0. 1 ppt. • Instrument cost is < $100 k.

Acknowledgements • Roger van Zee, Keith Gillis (NIST) • Charles Miller (JPL) • NIST Innovation in Measurement Science (IMS) Award • NIST Greenhouse Gas Measurements and Climate Research Program


System improvements • Improved supermirrors with lower absorption and transmission losses • Lengthen averaging time and increase acquisition rate by improving laser frequency stability • Improved thermal isolation of cold cell from the optical table • New design of the cold cell with a significant reduction in the observed radial heat transfer • Gas separation methods to remove N 2 O interference
- Slides: 18