Opioids Gh Bahrami Professor of Pharmacology Kermanshah University

Opioids Gh. Bahrami Professor of Pharmacology Kermanshah University of Medical Sciences

Pain • Universal, complex, subjective experience • Number one reason people take medication • Generally is related to some type of tissue damage and serves as a warning signal

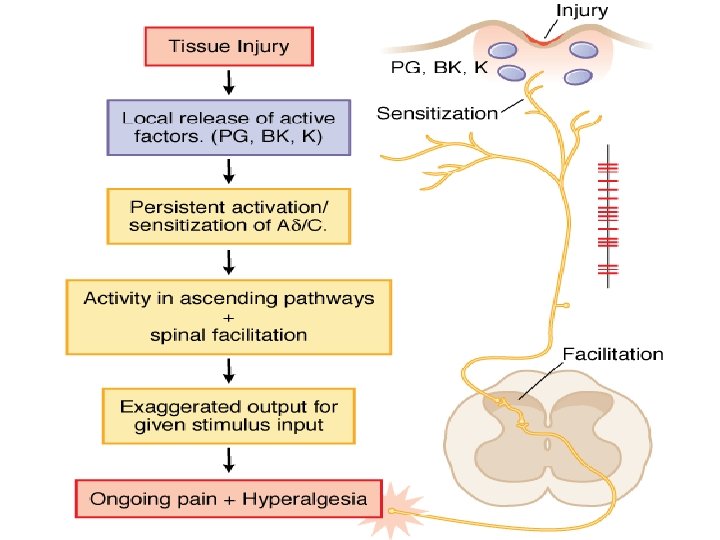
Gate Control Theory of Pain • Gate control theory of pain is the idea that physical pain is not a direct result of activation of pain receptor neurons, but rather, its perception is modulated by interaction between different neurons

Gate Control Theory of Pain • Nerve fibers (A delta (fast channels)) and C fibers (slow channels) transmit pain impulses from the periphery • Impulses are intercepted in the dorsal horns of the spinal cord, the substantia gelatinosa • In this region, cells can be inhibited or facilitated to the T-cells (trigger cells) • When cells in the substantia gelatinosa are inhibited, the ‘gate’ to the brain is closed • When facilitated, the ‘gate’ to the brain is open

Gate Control Theory of Pain • Similar gating mechanisms exist in the nerve fibers descending from the thalamus and the cortex. • These areas of the brain regulate thoughts and emotions.

Inhibitory and Facilitatory Mechanisms • Neurotransmitters chemicals that exert inhibitory or excitatory activity at post-synaptic nerve cell membranes. Examples include: acetylcholine, norepinehprine, epinephrine, dopamine, and serotonin. • Neuromodulators endogenous opiates help to relieve pain.

World Health Organization (WHO) Step Ladder Approach Severe Pain 7 -10/10 Moderate Pain 4 -6/10 Mild Pain 1 -3/10 ASA, Tylenol, NSAIDS Potent opioids (e. g. morphine) +/non-opioids Weak opioids +/- nonopioids (e. g. Tylenol #3®)

• Opioid – Compound with morphine-like activity • Opiate – Substance extracted from opium – Exudate of unripe seed capsule of Papaver somniferum – Contain 2 types of alkaloids Phenanthrene derivatives • Morphine (10% in opium) • Codeine (0. 5% in opium) • Thebaine (0. 2% in opium), (Nonanalgesic) Benzoisoquinoline derivatives • Papaverine (1%) Nonana. Igeslic • Noscapine (6%)

Opioids • Mordern definition of opioid – Molecule that interact with opioid receptor • Opioid compound – Opioid receptor agoninsts, antagonists, agonists-antagonists – Natural products, synthetic and semisynthetic compounds – peptides synthesized by neurone and other cell

CLASSIFICATION OF OPIOIDS Natural opium alkaloids: • Morphine • Codeine Semisynthetic opiates: • Diacetylmorphine (Heroin) • Pholcodeine Synthetic opioids: • Pethidine (Meperidine) • Fentanyl, Alfentanil, Sufentanil, Remifentanil • Methadone • Dextropropoxyphene • Tramadol

COMPLEX ACTION OPIOIDS AND OPIOID ANTAGONISTS Agonist-antagonists ( analgesics) • Nalorphine • Pentazocine • Butorphanol Partial/weak agonist + antagonist • Buprenorphine Pure antagonists • Naloxone • Naltrexone • Nalmefene
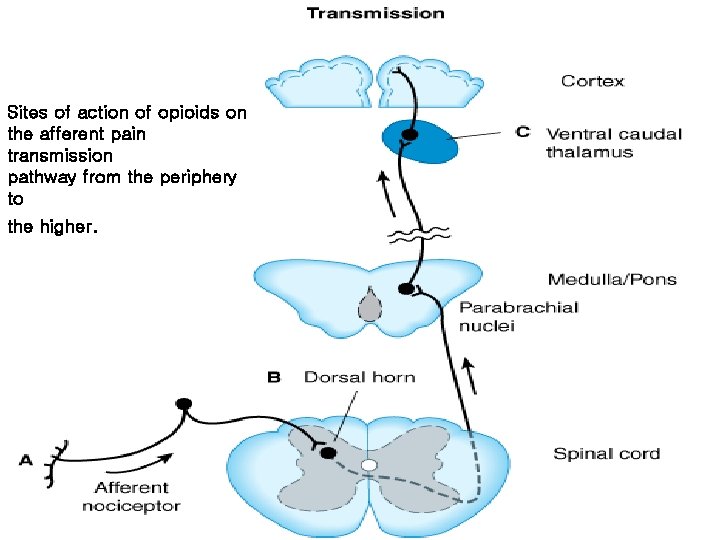
Sites of action of opioids on the afferent pain transmission pathway from the periphery to the higher.
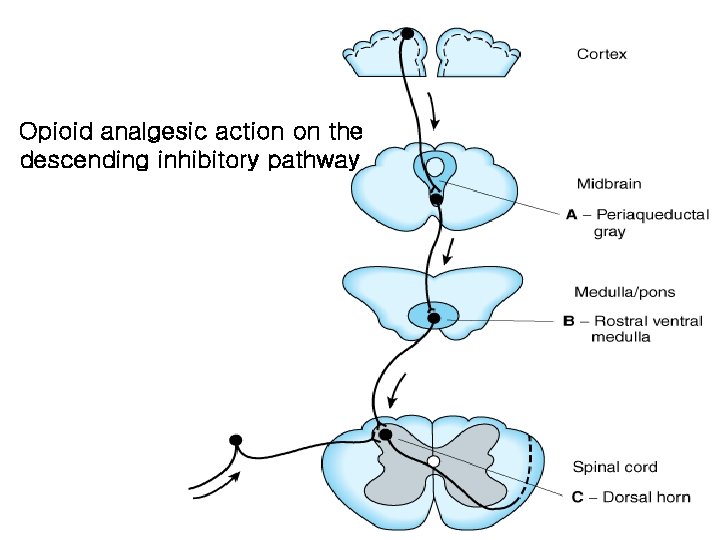
Opioid analgesic action on the descending inhibitory pathway
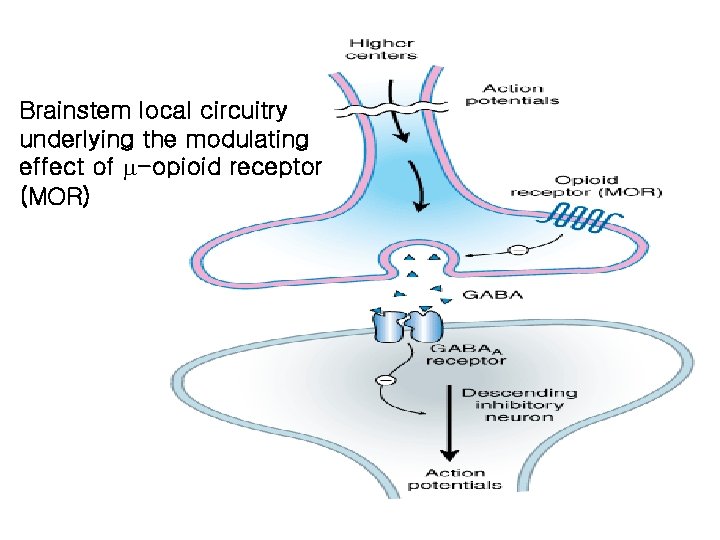
Brainstem local circuitry underlying the modulating effect of -opioid receptor (MOR)
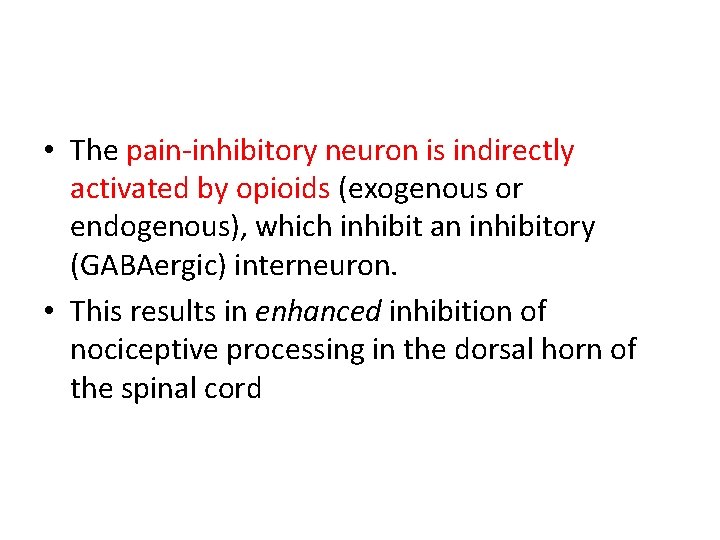
• The pain-inhibitory neuron is indirectly activated by opioids (exogenous or endogenous), which inhibit an inhibitory (GABAergic) interneuron. • This results in enhanced inhibition of nociceptive processing in the dorsal horn of the spinal cord

Opioid Receptor Transducer Mechanism • Agonist binding -conformational changes in the GPCR -Inhibition of adenylyl cyclase activity ( , ) -Stimulation of K+ current ( , ) -Inhibition of voltage-gated Ca 2+ channels ( ) -decreased release of neurotransmitter (substance-P, neurokinin A, neurokinin B, glutamate)

Opioid Receptor Activation • They are activated both by endogenously produced opioid peptides and by exogenously administered opiate compounds, some of which are not only among the most effective analgesics known but also highly addictive drugs of abuse.

Receptor subtypes • To date, four opioid receptors have been cloned: • The MOP ( µ=mu for morphine) • The KOP (κ= kappa for ketocyclazocine) • The DOP (δ= delta for deferens because it was first identified in mouse) • And the NOP-R [initially called LC 132, ORL 1, or nociceptin/orphanin FQ receptor].

The endogenous Opioid Ligands • The endogenous opioid peptides are mainly derived from four precursors. • Except for nociceptin/orphanin FQ, all peptides derived from the other precursors consist of a pentapeptide sequence Tyr. Gly. Phe. Met/Leu (YGGFM/L).

• Most of available opioid analgesics – Act at -opioid receptor • Activation of -opioid receptor → analgesia, euphoria, respiratory depress, nausea, vomiting, decreased gastrointestinal motility, tolerance, dependence • -, -opioid receptor – – analgesia dysphoria, Psychotomimetic ( ) Affective behaviour, proconvulsant ( ) Not cause respiratory depression or to decrease GI motility → Analgesia without -opioid side effect

Effects of opioids Central Nervous System Effects Analgesia • Pain consists of both sensory and affective (emotional) components. • Opioid analgesics reduce both aspects of the pain experience, especially the affective aspect. • In contrast, nonsteroidal anti-inflammatory analgesic drugs have no significant effect on the emotional aspects of pain. Euphoria • intravenous drug users experience a pleasant floating sensation with lessened anxiety and distress (DA release in nucleus accumbance). • However, dysphoria, an unpleasant state characterized by restlessness and malaise, may sometimes occur.

Sedation • • • Effects of Opioids Drowsiness clouding of mentation little or no amnesia No motor incoordination Sleep is induced in the elderly (can be easily aroused from this sleep) Respiratory Depression • by inhibiting brainstem respiratory mechanisms. • Alveolar PCO 2 may increase, and depressed response to a carbon dioxide. • In individuals with increased intracranial pressure, asthma, chronic obstructive pulmonary disease, this decrease in respiratory function may not be tolerated.

Effects of Opioids Cough Suppression • Codeine in particular (risk of atelectasis). Temperature regulating centre depression • chances of hypothermia Vasomotor centre depression • Fall in BP • CTZ (nausea, vomiting) • Edinger Westphal nucleus of III nerve is stimulated (miosis) • no tolerance develops and valuable in the diagnosis of opioid overdose • Vagal centre (bradycardia) • Truncal Rigidity-

Peripheral Effects Cardiovascular System • Bradycardia Meperidine is an exception (can result in tachycardia) • Hypotension - due to -peripheral arterial and venous dilation -depression of vasomotor centre -release of histamine. • Increased PCO 2 leads to cerebral vasodilation associated with a decrease in cerebral vascular resistance, an increase in cerebral blood flow, and an increase in intracranial pressure.

Peripheral Effects Gastrointestinal Tract Constipation • no tolerance • Opioid receptors exist in high density in the gastrointestinal tract • constipating effects of the opioids are mediated through an action on the enteric nervous system as well as the CNS • gastric secretion of hydrochloric acid is decreased • propulsive peristaltic waves are diminished • this delays passage of the fecal mass and allows increased absorption of water, which leads to constipation • so used in the management of diarrhea

Peripheral Effects Biliary Tract • sphincter of Oddi may constrict • contract biliary smooth muscle • result in biliary colic Renal • Renal function is depressed by opioids • decreased renal plasma flow • enhanced renal tubular sodium reabsorption • Ureteral and bladder tone are increased • Increased sphincter tone may precipitate urinary retention • ureteral colic caused by a renal calculus is made worse by opioid-induced increase in ureteral tone

Uterus • may prolong labor • both peripheral and central actions of the opioids can reduce uterine tone Neuroendocrine • stimulate the release of ADH, prolactin, and somatotropin • inhibit the release of luteinizing hormone Pruritus • CNS effects and peripheral histamine release may be responsible for these reactions • pruritus and occasionally urticaria (when administered parenterally)

Clinical Use of Opioid Analgesics • • • Analgesia Cough Diarrhea Acute Pulmonary Edema Balanced anaesthesia Preanaesthetic medication
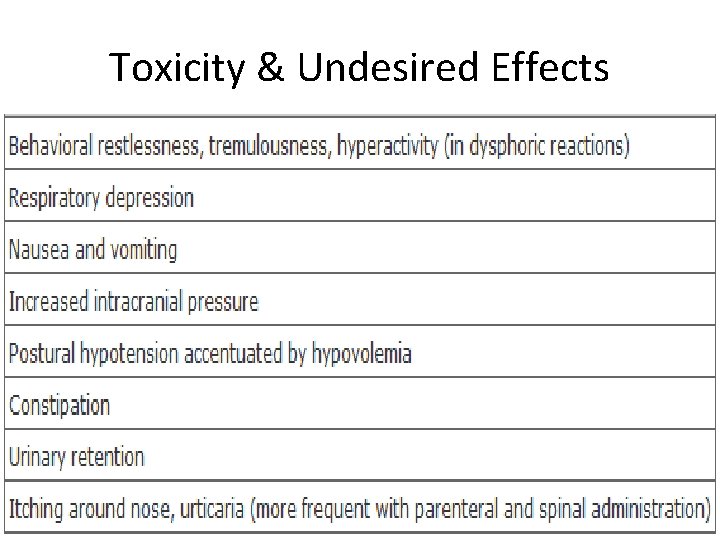
Toxicity & Undesired Effects

Acute morphine poisoning • >50 mg of morphine • Lethal dose is 250 mg • Stupor, coma, shallow breathing, cyanosis, pinpoint pupil, fall in BP, convulsions • Death due to respiratory failure Treatment • Positive pressure respiration • Iv fluids • Gastric lavage with potassium permagnate • Naloxone

The outcome of long-term Opioid usage • The major limitation to their long-term use is the development of physiological “tolerance, ” a profound decrease in analgesic effects observed in most patients during prolonged drug administration. • In addition to tolerance, physiological “dependence, ” which results in the necessity for continued administration of increasing doses of drug to prevent the development of symptoms of opioid withdrawal, can ensue in some patients.

Withdrawl Reactions Acute Action • • • Analgesia Respiratory Depression Euphoria Relaxation and sleep Tranquilization Decreased blood pressure Constipation Pupillary constriction Hypothermia Drying of secretions Reduced sex drive Flushed and warm skin Withdrawl Sign • • • Pain and irritability Hyperventilation Dysphoria and depression Restlessness and insomnia Fearfulness and hostility Increased blood pressure Diarrhea Pupillary dilation Hyperthermia Lacrimation, runny nose Spontaneous ejaculation Chilliness and “gooseflesh”

Tolerance and Dependence • Maintenance of normal sensitivity of receptors requires reactivation by endocytosis and recycling. • activation of receptors by endogenous ligands results in endocytosis followed by resensitization and recycling of the receptor to the plasma membrane. • But morphine fails to induce endocytosis of the -opioid receptor - tolerance and dependence. • In contrast, methadone, used for the treatment of opioid tolerance and dependence, does induce receptor endocytosis.

Tolerance and Dependence • NMDA receptor ion channel complex play a critical role in tolerance development and maintenance • NMDA-receptor antagonists such as ketamine can block tolerance development

DESENSITIZATION/ENDOCYTOSIS/DOWNREGUL ATION • Receptor desensitization is defined as any process that alters the functional coupling of a receptor to its G-protein/second messengersignaling pathway. • Endocytosis is defined as the translocation of receptors from the cell surface to an intracellular compartment. • Down regulation is defined as any process that decreases the number of ligand binding sites.

Desensitization • Following activation by alkaloid or peptide agonists, opioid receptors are regulated by multiple mechanisms, . • One of these is receptor phosphorylation by G protein -coupled receptor kinases (GRKs) and β-arrestin. • These processes contribute directly to rapid receptor desensitization and uncoupling of the receptor from its G protein.
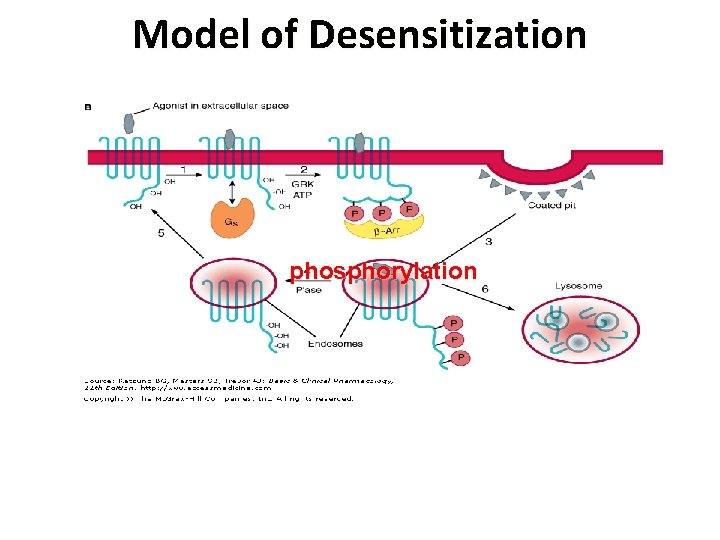
Model of Desensitization phosphorylation

Other Kinases that regulate (desensitize) Opioid Rs • Many kinases phosphorylate opioid receptors, a number of which have been shown to desensitize receptors as well. • These include not only GRK, but also protein kinase A (PKA), protein kinase C (PKC) and calcium/calmodulin dependent protein kinase II. • Endocytosis is one feature that distinguishes GRK/β –arrestin mediated desensitization from other phosphorylation-dependent desensitization mechanisms.
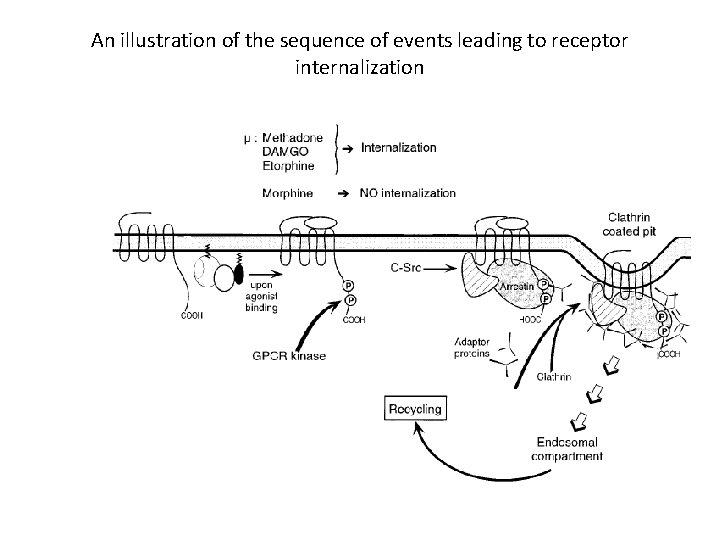
An illustration of the sequence of events leading to receptor internalization

• Following desensitization by GRKs and βarrestin, opioid receptors are rapidly endocytosed into an intracellular compartment. • This process occurs following even brief agonist exposure and independently of signal transduction. • The speed and conservation of this process is ideal for modulating signaling from endogenous ligands, such as neurotransmitters, that are released in a pulsatile manner. • Following their endocytosis, receptors can then be recycled back to the membrane, thereby restoring the functional complement of receptors, a process termed “resensitization. ”

Exogenous drug administration, and endocytosis • In contrast, chronic exposure of opioid receptors to agonist, can also lead to receptor desensitization/uncoupling. • This involves alternate mechanisms, including PKAand PKC-mediated phosphorylation. These desensitization processes most often require prolonged agonist treatment. • Importantly and in contrast to GRK-phosphorylated receptors, PKA/PKC-phosphorylated receptors are not necessarily rapidly endocytosed. • Therefore, receptors that have been desensitized by GRK-independent phosphorylation require a mechanism other than endocytosis to resensitize.

Different fate of MOP-R and DOP-R Endocytosis • Whereas MOP-Rs are recycled following their endocytosis, DOP-Rs are transported deeper into the endocytic pathway, are rapidly degraded by the lysosome, and hence “downregulated. ”

RAVE: Relative Activity Versus Endocytosis • Regulation of opioid receptors by endocytosis serves a protective role in reducing the development of tolerance and dependence to opioid drugs. Agonist activity and receptor endocytosis have opposing effects on receptor-mediated signaling. • The net amount of signal transmitted to the cell is a function of both processes. This “net signal” has been termed RAVE, for relative activity versus endocytosis.

RAVE: Relative Activity Versus Endocytosis • Morphine would have a particularly high RAVE value as a consequence of its inability to promote receptor desensitization and endocytosis. • In contrast, endorphins and opioid drugs that acutely signal with similar efficacy, yet induce receptor desensitization and endocytosis, would have lower RAVE values than morphine.

Contraindications and Cautions in Therapy Use of Pure Agonists with Weak Partial Agonists • morphine with pentazocine - risk of diminishing analgesia or even inducing a state of withdrawal Use in Patients with Head Injuries • Carbon dioxide retention caused by respiratory depression results in cerebral vasodilation. • In patients with elevated intracranial pressure, this may lead to lethal alterations in brain function. • Marked respi. depression • Vomiting, miosis, altered mentation by morphine interferes with assessment of pt condition

Use during Pregnancy • In pregnant women who are chronically using opioids, the fetus may become physically dependent in utero and manifest withdrawal symptoms in the early postpartum period. • A daily dose as small as 6 mg of heroin (or equivalent) taken by the mother can result in a mild withdrawal syndrome in the infant, and twice that much may result in severe signs and symptoms, including irritability, shrill crying, diarrhea, or even seizures. • When withdrawal symptoms are mild - diazepam • with more severe withdrawal - methadone

Use in Patients with Impaired Pulmonary Function • opioid analgesics may lead to acute respiratory failure. Use in Patients with Impaired Hepatic or Renal Function • morphine and its congeners are metabolized primarily in the liver • Half-life is prolonged in patients with impaired renal function Use in Patients with Endocrine Disease -adrenal insufficiency (Addison's disease) and hypothyroidism (myxedema) – -prolonged and exaggerated responses to opioids.

Opioid Use in the Elderly Educating staff is essential!! • Opioids produce higher plasma concentrations in older persons • Greater sensitivity in both analgesic properties and side effects • Smaller starting doses required • Consider duration of action, formulation availability, side-effect profile, and resident preference. • Review for drug interactions

Opioid Use in the Elderly, cont. • Older persons may have fluctuating pain levels. • Long-acting are generally suitable once steady pain levels have been achieved. • Once steady pain relief levels are achieved, controlled-released formulas can be used. • Fentanyl patches should not be placed on areas of the body that may receive excessive heat. Patches may be contraindicated with exceptionally low body fat.

Deciding to Convert From a Short-acting to a Long-acting Opioid 19 Short-acting Opioids Long-acting Opioids Advantages Fast-acting; appropriate for acute pain, breakthrough pain May be more appropriate for patients with a constant pain component; analgesic stability Disadvantages Need for repetitive dosing Initial delayed onset of action

Rationale for Opioid Rotation • • Ineffectiveness of initial opioid Adverse effects/toxicity of initial opioid Inter-patient variability of response Incomplete cross-tolerance Note: Conservative dose-conversion ratios are advised 20

Management of Opioid Side Effects 21 Side Effects Amelioration · Nausea and vomiting · Switch opioids; anti-emetics · Sedation · Lower dose (if possible); add co-analgesics; · Constipation · Treat prophylactically with stool softeners, bowel stimulants; non-pharmacological measures
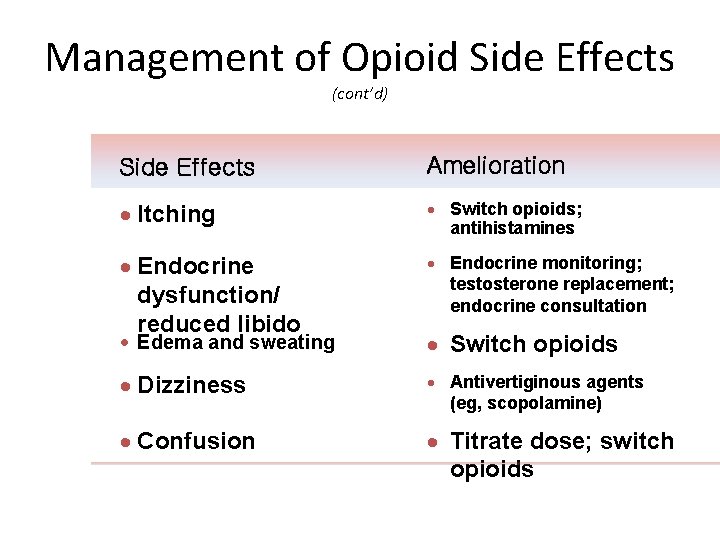
Management of Opioid Side Effects (cont’d) 22 Side Effects Amelioration · Itching · Switch opioids; antihistamines · Endocrine dysfunction/ reduced libido · Endocrine monitoring; testosterone replacement; endocrine consultation · Edema and sweating · Switch opioids · Dizziness · Antivertiginous agents (eg, scopolamine) · Confusion · Titrate dose; switch opioids

Related drugs Pethidine • 1/10 th in analgesic potency • Spasmodic action on smooth muscles is less • Tachycardia (antimuscarinic action)- it is related to atropine, even acts on opioid receptors • Safer in asthmatics (less histamine release) • Uses- analgesia, preanaesthetic medication • Preferred opioid analgesic during labour (less neonatal respi depression)

Fentanyl 80 -100 times more potent than morphine few cardiovascular effects little propensity to release histamine. Because of high lipid solubility, it enters brain rapidly and produces peak analgesia in 5 min after i. v. injection. • The duration of action is short: • Transdermal fentanyl has become available for use in cancer • •

Methadone • Slow & persistant action • Sedative & subjective effects are less intense • Less abuse potential • Use- as substitute therapy for opioid dependence • 1 mg methadone for 4 mg morphine.

Tramadol – Analgesic action mechanism • Weak affinity for -opioid receptor • norepinephrine & 5 -HT reuptake Inhibition – Advantage • Less respiratory depression, nausea, vomiting, constipation • Less abuse potential • Rapid psychomotor recovery – Labour pain, injury, surgery (other short lasting pain) – Moderate pain treatment : as effective as morphine – Severe pain treatment : less effective than morphine

Tramadol • Available 50 mg tabs • Sustained Release 100 mg, 200 mg, 300 mg • Dosage: 50 mg q 6 hr prn • May use 50 mg qid • Max dose 100 mg qid • Studied in osteoarthitis, back pain, others • Side effects: dizziness, nausea, constipation

Tramadol--warnings Seizure risk: reported in recommended dosage range, increased risk combined with TCA, SSRI, SNRI, opioids, MAO-I Serotonin Syndrome: other serotonergic drugs– triptans, Serotonin Syndrome management: stop agents, iv hydration, consider cyproheptadine

Pentazocine ( analgesic) • It has agonistic actions and weak opioid antagonistic activity • elicit dysphoric and psychotomimetic effects • increase in blood pressure and heart rate Uses • moderate to severe pain • as a preoperative medication and • as a supplement to anesthesia

Buprenorphine (weak agonist & antagonist) • 25 -50 times more potent than morphine • Sublingual route • Slower onset & longer duration of action • Postural hypotension is marked • Cannot be used during labour (respi dep not reversed by naloxone) Uses • Long lasting pain- cancer • Tt of morphine dependence

Naloxone ( , , antagonist) • Antagonizes all morphine actions • Sedation is less completely reversed • Blocks placebo, acupuncture, stress induced analgesia Use • Morphine poisoning • Diagnostic test for opioid addiction • Revert neonatal respi depression due to opioid use during labour
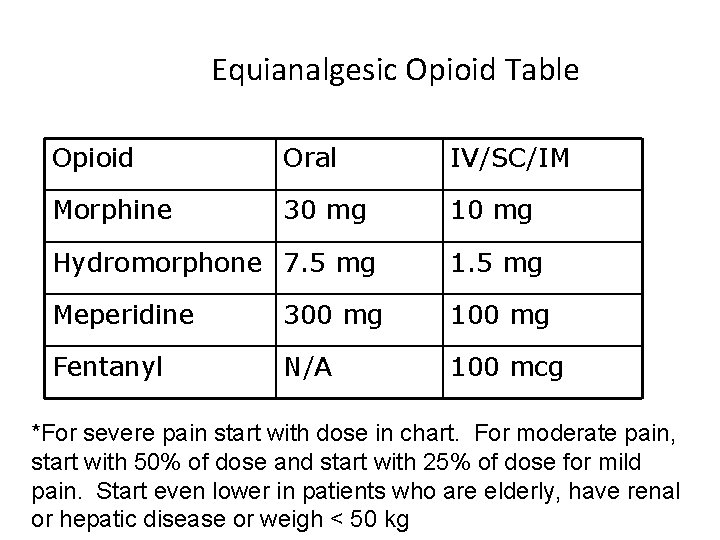
Equianalgesic Opioid Table Opioid Oral IV/SC/IM Morphine 30 mg 10 mg Hydromorphone 7. 5 mg 1. 5 mg Meperidine 300 mg 100 mg Fentanyl N/A 100 mcg *For severe pain start with dose in chart. For moderate pain, start with 50% of dose and start with 25% of dose for mild pain. Start even lower in patients who are elderly, have renal or hepatic disease or weigh < 50 kg

Peripherally Acting Opioid • Opioid receptor – outside central nerve system – Peripherally acting opioid agonist → analgesia without CNS side effect • Loperamide, Diphenoxylate – – -opioid receptor agonist Not cross blood-brain barrier Treatment : inflammation-induced hyperalgesia Relieve diarrhea • Alvimopan – – peri -opioid receptor antagonist Relieves constipation in opium addicts Without precipitating opioid withdrawl Treat postoperative paralytic ileus

Ion Channels & Novel Analgesic Targets Ziconotide is a powerful analgesic drug that has a unique mechanism of action involving potent and selective block of N-type calcium channels, which control neurotransmission at many synapses. The analgesic efficacy of ziconotide likely results from its ability to interrupt pain signaling at the level of the spinal cord.

Ion Channels & Novel Analgesic Targets Ziconotide is a peptidic drug and has been approved for the treatment of severe chronic pain in patients only when administered by the intrathecal route. Importantly, prolonged administration of ziconotide does not lead to the development of addiction or tolerance.

Opioid with Other Analgesics • Goal of using analgesics in combination – Achieve superior analgesia – Reduce dose of each drug – Minimizing side effect • NSAID – Synergistical action with systemic opioid to produce analgesia • Local anesthetics and opioid – Synergistical pain relief when intrathecal or epidural administration

Antidepressants • Tricyclic antidepressants – Analgesic effects separate from anti-depressant effects. – Amitriptyline: most studied, but most side effects – Nortriptyline & desipramine: better tolerated, less well studied • SSRIs: little evidence of analgesic effect. • SNRI’s – inhibit both norepinephrine and serotonin reuptake – efficacy in neuropathic pain syndromes or pain associated with depression duloxetine, venlafaxine

Anticonvulsants • Agents for neuropathic pain – gabapentin (Neurontin®) – pregabalin (Lyrica®) – clonazepam (Klonopin®) – Other newer agents • Start low, go slow • Watch for side effects • Monitor serum levels, if available

Thanks
- Slides: 71