Opioid Agonists Nancy BrutonMaree CRNA MS Opioid Agonists

Opioid Agonists Nancy Bruton-Maree, CRNA, MS

Opioid Agonists - Introduction • • Opium – derived from the Greek word opion (poppy juice)Opium poppy is Papaver somniferum Opiates – Drugs derived from opium Narcotic – derived from the Greek word for stupor – traditionally used to refer to potent morphine-like analgesics • Opioid – refers to all exogenous substances, natural and synthetic, that bind specifically to several subpopulations of opioid receptors – produce at least some agonistic effects

Opioid Agonists - Introduction • Opioids produce analgesia • The do not produce the following: – Loss of touch – Loss of proprioception – Loss of consciousness

Opioids – Chemical Structure • Two distinct chemical classes – Phenanthrene • Morphine • Codeine • Thebaine – Benzylisoquinoline • Papaverine • Noscapine
Basic Chemical Structures
Opioids – Chemical Structure

Opioids – Synthetic • Contain the phenanthrene ring • Manufactured by synthesis rather than chemical modification of morphine • Types are – Morphine derived – levorphanol – Methadone derived – – Benzomorphan derived – pentazocine – Phenylpiperidine derived – meperidine, fentanyl

Opioids – Synthetic • Fentanyl • Sufentanil • Remifentanil • Alfentanil Major pharmacodynamic differences between these drugs are: Potency & rate of equilibrium between plasma and the site of action (Biophase)

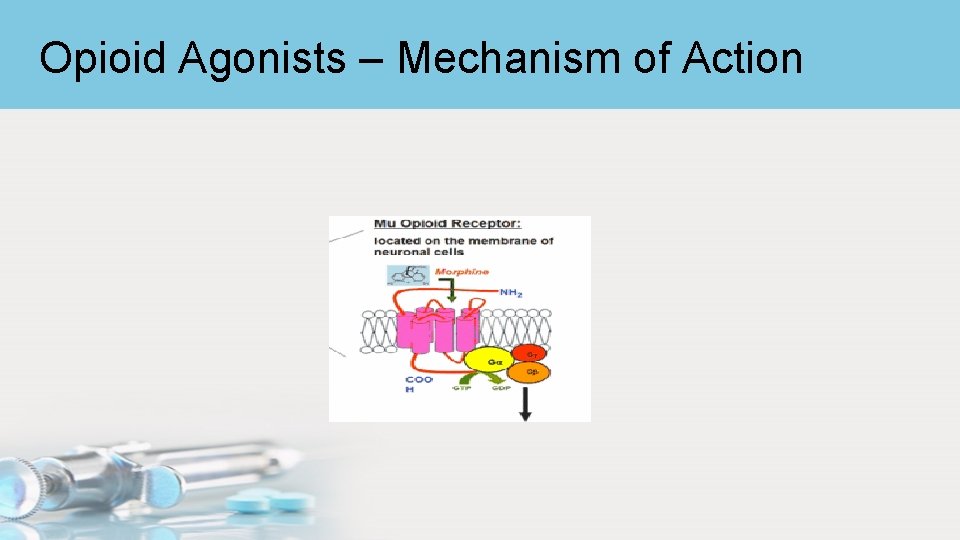
Opioid Agonists – Mechanism of Action • Opioids act at specific opioid receptors at both pre- & postsynaptic sites in the CNS and periphery. • CNS sites are primarily in the brain & spinal cord • Endogenous transmitters for these receptors are: – Enkephalins – Endorphins – Dynorphins • Opioids mimic the actions of these endogenous ligands

Opioid Agonists – Mechanism of Action • Opioids bind to opioid receptors & activate the painmodulating system (antinociceptive system) • Opioids appear to be ionized for strong binding at the anionic opioid receptor site • Only the levorotatory forms exhibit agonistic activity • Affinity for the receptors correlates with analgesic potency

Opioid Agonists – Mechanism of Action • Activation of the receptors decreases neurotransmission • This occurs largely by presynaptic inhibition of neurotransmitter release – Acetylcholine – Dopamine – Norepinephrine – Substance P • Postsynaptic inhibition of evoked activity may also occur

Opioid Agonists – Mechanism of Action • Intracellular biochemical events initiated by occupation of the opioid receptors by an opioid agonist include: – Increased potassium conductance – causes hyperpolarization – Increased calcium channel inactivation – produces an immediate decrease in neurotransmitter release – Or both action

Opioid Agonists – Mechanism of Action • Receptors are guanine (G) proteins • The receptors belong to a superfamily of 7 transmembranesegment G protein-coupled receptors • Binding of an opioid agonists to the EC domain changes the shape of the receptor which activated the G protein bound to the IC domain • The G protein replaces it guanine diphosphate (GDP) with guanine triphosphate (GTP) & dissociates into two active subunit
Opioid Agonists – Mechanism of Action

Opioid Agonists – Mechanism of Action • Subsequent mechanisms include: – Inhibition of adenylate cyclase – Decreased conductance of voltage-gated calcium channels or – Opening of inward flowing potassium channels • Any of these effects ultimately results in decreased neuronal activity • Opioid receptors also modulate the phosphoinositidesignaling cascade and phospholipase C

Opioid Agonists – Mechanism of Action • Prevention of calcium ion inflow results in suppression of neurotransmitter release (substance P) in many neuronal systems • Hyperpolarization results from actions at potassium channels – prevents excitation or propagation of action potentials • Opioid receptors may also regulate the functions of other ion channels including excitatory postsynaptic currents evoked by N-methyl-D-aspartate (NMDA) receptors

Opioid Agonists – Mechanism of Action • Opioid-mediated inhibition of adenylate cyclase is not responsible for an immediate effect but may have a delayed effect, possibly by reduction on cyclic adenosine monophosphate (c. AMP)-responsive neuropeptide genes as well as reduction in neuropeptide messenger RNA concentration • Opioid-induced inhibition of acetylcholine release at nerve endings in the CNS depresses cholinergic transmission may play a role in analgesia & other opioid effects

Opioid Agonists – Mechanism of Action • Opioid agonists do not alter responsiveness of afferent nerve endings to noxious stimulation • They also do not block conduction of nerve impulses along peripheral nerves
Classification of Opioid Receptors Mμ 1 Mμ 2 Kappa Delta Effect Supraspinal & Spinal Analgesia Euphoria Low Abuse Potential Miosis Bradycardia Hypothermia Urinary retention Analgesia (Spinal) Ventilatory Depression Physical Dependence Marked Constipation Analgesia (Supraspinal, Spinal) Dysphoria Low Abuse Potential Miosis Diuresis Analgesia (Supraspinal, Spinal) Ventilatory Depression Physical Dependence Minimal Constipation Urinary Retention Agonist Endorphins Morphine Synthetic Opioids Dynorphins Enkephalins Antagonists Naloxone Naltrexone Nalmefene

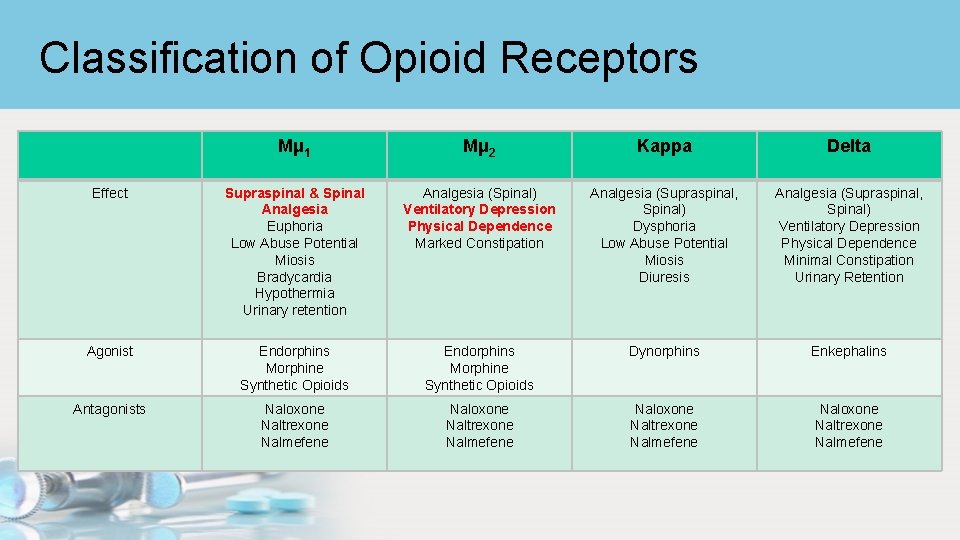
Opioid Agonists – Opioid Receptors • Names of endogenous neurotransmitters came from – Mu – morphine – Kappa – ketocyclazocine – Delta – isolated from mouse vas deferens • A single μ-receptor gene has been identified and 6 distinct μ receptors subtypes • Opioid receptors in the brain – Periaqueductal gray – Locus ceruleus – Rostral ventral medulla

Opioid Agonists – Opioid Receptors • Opioid receptors in the spinal cord – – Interneurons – Primary afferent neurons in the dorsal horn • Consequently direct application of opioids to the spine produces intense analgesia • Outside the CNS opioid receptors are found on – Sensory neurons – Immune cells

Opioid Agonists – Opioid Receptors • Kappa receptors – activation results in inhibition of neurotransmitter release of N-type calcium channels • Dysphoria & diuresis may accompany activation of these receptors • κ-mediated analgesia may be less effective for high-intensity painful stimulation • Opioid-agonist antagonist act predominantly on κ receptors • Delta receptors respond to enkephalins – These receptors may modulate μ-receptors activity

Opioid Agonists – Opioid Receptors • Functional & physical interactions between the receptor subtypes have been identified – Heteromerization between μ & δ receptors leads to distinct receptor pharmacology in that doses of δ receptor ligands (agonist & antagonists) too low to trigger signaling can potentiate the binding and signaling of μ receptor agonists. – Chronic (not acute) morphine treatment results in an increase in μδ heteromers in key areas of the CNS – these key areas are implicated in pain processing

Opioid Agonists – Endogenous Pain Modulating Mechanisms • Pain perception integration of pain impulses and responses to pain– for this opioid receptors are located in: – Areas of the brain • • Periaqueductal gray matter of the brain Amygdala Corpus striatum Hypothalamus – Area of the spinal cord • Substantia gelatinosa

Opioid Agonists – Endogenous Pain Modulating Mechanism • Speculation – endorphins inhibit the release of excitatory neurotransmitters from nerve terminals carrying nociceptive impulses • This results in neuron hyperpolarization which suppresses spontaneous discharges and evoked potentials • Sustained pain and stress induces regional release of endogenous opioids that interact with μ receptors in many of cortical & subcortical bran areas

Opioid Agonists – Endogenous Pain Modulating Mechanisms • μ-opioid receptor system activation is associated with reduction in the sensory and affective ratings of pain experience with distinct neuroanatomic involvement • Expectations of positive treatment substantially enhances the analgesic benefit of remifentanil; negative treatment expectation abolishes remifentanil analgesia (hippocampus modulates this latter part)

Common Side Effects - Opioids • The focus for this is morphine (M/S) – the prototypical opioid • CV system – In healthy supine normovolemic people – no direct myocardial depression or hypotension – may experience orthostatic hypotension and syncope is he/she stands • May reflect M/S-induced impairment of compensatory SNS responses – Decreased SNS tone to peripheral veins – causes venous pooling and subsequent decreases in venous return, CO, and BP

Opioid Agonists – Opioid Receptors • CV system – M/S can cause decrease BP because of drug-induced bradycardia and histamine release – Increased activity of the vagal nerves induced by M/S causes bradycardia – this probably reflects stimulation of the vagal nuclei in the medulla – M/S may exert a direct depressant effect on the SA node which acts to slow conduction of cardiac impulses through the AV node – This may explain, in part, decreased vulnerability to ventricular fibrillation when M/S is being used

Opioid Agonists – Common Side Effects • CV System – M/S evoked histamine release and associated hypotension vary in incidence and severity – Expect variability – The magnitude of histamine release & decrease in BP can be minimized by • Limiting the rate of a M/S infusion to 5 mg/minute IV • Maintaining the patient in supine position to slightly head-down position • Optimizing intravascular volume

Opioid Agonists – Common Side Effects • CV System – Using H 1 & H 2 blockers does not alter histamine release but does prevent changes in systemic BP and SVR – There is no sensitization of the heart to catecholamines or otherwise predisposing the heart to dysrhythmias as long as hypercarbia or arterial hypoxemia does not result from ventilatory depression – Hypertension and tachycardia are responses to pain that is not being suppressed by M/S – SNS and RAA contribute to these responses

Opioid Agonists – Common Side Effects • CV System – Opioid agonists + nitrous oxide • Results in CV depression from decreased CO and systemic BP plus increased cardiac filling pressures – Opioid agonists + benzodiazepine • May result in decreased SVR and systemic BP – Protecting the myocardium from ischemia – • δ & κ receptors – opioids enhance the resistance of the myocardium to oxidation and ischemic stress. Mitochondrial ATP-regulated potassium channels appear to be important in this signaling pathway

Opioid Agonists – Common Side Effects • Ventilatory Effects – Dose-dependent gender-specific ventilatory depression – primarily a μ 2 effect causing direct depression of the brainstem respiratory centers – Characteristics include decreased responsiveness to CO 2 – this is evident by increased Pa. CO 2 and displacement of the CO 2 response curve to the right – There is also interference with the pontine and medullary centers that regulate rhythm – see prolonged pauses between breaths and periodic breathing

Opioid Agonists – Common Side Effects • Ventilatory Effects – M/S may produce some of its effects on ventilation by decreases the release of acetylcholine from neurons in the medullary ventilatory center area which would normally occur with hypercarbia – Ventilatory depression occurs rapidly and persists for hours – this is reflected by the decreased response to increasing CO 2 and a right shift of the CO 2 curve – Due to the above, physostigmine may reverse some of the ventilatory depression but not the analgesia

Opioid Agonists – Common Side Effects • Ventilatory Effects – High doses may cause apnea – Patient remains conscious and can initiate a breath if requested to do so – Death – ventilatory depression – Ventilatory depression manifests as decreased frequency accompanied by compensatory increased TV – compensation is incomplete as evidenced by increased Pa. CO 2

Opioid Agonists – Common Side Effects • Ventilatory Effects – Influences on magnitude and duration of depression • Aging increases • Natural sleep increases • Pain from surgical stimulation decreases – Dose-dependent decrease in ciliary activity • Also increased airway resistance probably due to direct effect on bronchial smooth muscle and indirect action due to release of histamine

Opioid Agonists – Common Side Effects • Cough suppression – Mechanism is depression is effects on the medullary cough centers that are not the same as other effects caused by opioids on the ventilatory center. – The best cough suppression comes from opioids that have a bulky substitution on the number 3 carbon – Codeine and dextromethorphan – Dextromethorphan has no analgesic properties

Opioid Agonists – Common Side Effects • CNS Effects – In the absence of hypoventilation • Decrease CBF • Decrease CMRO 2 – Reasons for careful use in head injury • Effects on wakefulness • Production of miosis – due to an excitatory action on the ANS component of the Edinger-Westphal nucleus of the oculomotor nerve – tolerance to this effect is not prominent – can antagonized by atropine and profound hypoxemia • Depression of ventilation with associated increases in ICP

Opioid Agonists – Common Side Effects • CNS Effects – EEG – effects resemble sleep – see slow δ waves – no evidence of seizures – No alteration in NMBA – Large doses cause skeletal muscle rigidity particularly of the thoracic and abdominal muscles – may be due to actions at opioid receptors by dopamine and GABA

Opioid Agonist – Common Side Effects • Rigidity – Large doses of opioid (fentanyl & derivatives) can lead to generalized skeletal muscle rigidity – Majority of resistance to ventilation is laryngeal musculature contraction – Reason is probably inhibition of striatal release of GABA and increased dopamine production – Used NMBA or naloxone

Opioid Agonists – Common Side Effects • Sedation – Often get sedation prior to analgesia – Recommendation for titrated M/S • A short interval between boluses (5 – 7 min. ) – Sedation is a reason to discontinue M/S titration for postoperative analgesia – Sedation should not be used as an indication that appropriate analgesia has occurred

Opioid Agonists – Common Side Effects • Biliary Tract – Spasm of biliary smooth muscle – results in increased biliary pressure – epigastric distress or biliary colic – sphincter of Oddi – May be confused with angina pectoris – Naloxone – nitroglycerine – Equal analgesic doses of fentanyl, morphine, meperidine, and pentazocine increase biliary pressure in the following order: 99%, 53%, 61%, and 15% above predrug levels – Treatment • Naloxone • Glucagon (2 mg/IV)

Opioid Agonists – Common Side Effects • GI Tract – Spasm of GI smooth muscle – Side effects include: • Constipation, biliary colic, & delayed gastric emptying – Decreased propulsive peristaltic contractions of the small & large intestines & enhances tone of the pyloric sphincter, ileocecal valve, & anal sphincter – increases absorption of water – constipation – Delayed passage of gastric content into the proximal duodenum occurs when the gallbladder contracts against a closed or narrowed sphincter of Oddi – there is an increased tone at the gastroduodenal junction • Premeds containing opioids could increase the incidence of delayed absorption of oral drugs & potentially increase risk of aspiration due to delayed gastric emptying

Opioid Agonists – Common Side Effects • Nausea & Vomiting – Direct stimulation of the chemoreceptor trigger zone on the floor of the 4 th ventricle – Maybe partial dopamine agonists – M/S may increase GI secretions and delay passage of intestinal content toward the colon – IV M/S also depresses the vomiting center in the medulla, thus producing less N & V than IM M/S – A vestibular component may also be involved since N&V rarely occurs in supine patients

Opioid Agonists – Common Side Effects • Genitourinary Tract – M/S increases the tone and peristaltic activity of the ureter – Urinary urgency is produced by opioid-induced augmentation of the detrusor muscle tone, at the same time, the tone of the urinary sphincter is enhanced, making voiding difficult – M/S does not provoke the release of ADH in humans in the absence of painful stimulation & if intravascular volume is adequate, there is no change in urinary output

Opioid Agonists – Common Side Effects • Cutaneous Changes – M/S causes – Cutaneous blood vessels dilate – Flushing of the head, neck and upper chest; warmth also occurs – These are in part due to histamine release – Urticaria & erythema at injection site – Conjunctival erythema & pruritus – Evidence of histamine release along the injected vessel – this is not an allergic reaction

Opioid Agonists – Common Side Effects • Placental Transfer – Opioids are readily transferred across the placental barrier – therefore, ventilatory depression can occur in the neonate (the neonatal depression is greater with M/S than meperidine) – May reflect the immaturity of the neonate’s BBB – Chronic use of opioid in the mother can result in physical dependence of the fetus – use of naloxone can precipitate neonatal abstinence syndrome

Opioid Agonists – Common Side Effects • Drug interactions: – Amphetamines, phenothiazines, MAOIs, and tricyclic antidepressants – increased ventilatory depression – Exaggerated CNS depression & hyperpyrexia with MAOIs – Sympathomimetics – enhance analgesia – Physostigmine enhances analgesia – Atropine antagonizes analgesia

Opioid Agonists – Common Side Effects • Hormonal Changes – Prolonged opioid therapy • May influence the hypothalamic-pituitary-adrenal axis & the hypothalamicpituitary-gonadal axis – causes endocrine & immune effects – M/S may cause progressive decreases in plasma cortisol concentrations – Increased prolactin & decreased luteinizing hormone, follicle-stimulating hormone, testosterone, and estrogen concentrations

Opioid Agonists – Common Side Effects • Overdose – Depression of ventilation - characterized by slow breathing frequency which may progress to apnea – Symmetrical pupils & miotic unless severe arterial hypoxemia is present – mydriasis – Flaccid skeletal muscles – Upper airway obstruction – Pulmonary edema – Hypotension – Seizures

Opioid Agonists – Common Side Effects • Overdose • The Triad of overdose – Miosis – Hypoventilation – Coma – Treatment • Mechanical ventilation • Opioid antagonist – may precipitate withdrawal

Opioid Agonists – Common Side Effects • Provocation of Cough – Paradoxically, preinduction administration of fentanyl, sufentanil, or alfentanil may associate with significant reflex coughing – ? Due to imbalance between stimulation of juxtacapillary irritant receptors. – Not caused by M/S or hydromorphone

Opioid Agonists – Common Side Effects • Tolerance & Physical Dependency – Tolerance & physical dependence – characteristics of all opioids and is a reason for limiting clinical use – Tolerance – Development of increased doses of a drug to achieve the same effect once received by a smaller dose – Tolerance develops to all but miosis and bowel motility – When opioid agonist actions predominate, repeated doses can result in physical and psychological dependence

Opioid Agonists – Signs of Withdrawal • • • Yawning Diaphoresis Tearing or coryza Insomnia Restlessness • • Abdominal cramps Nausea Vomiting Diarrhea

Opioid Agonists – Common Side Effects • Physical Dependence – Pharmacodynamic tolerance • Related to neurological changes that occur after long-term exposure to opioids • Involves changes at the level of the receptor and involves receptor desensitization – the receptors reduce transcription and subsequent decreases in the absolute number of receptors (downregulation) • May also be upregulation of the c. AMP system • Long-term pharmacodynamic tolerance – characterized by opioid insensitivity – most likely represents persistent neural adaptation.

Opioid Agonists - Morphine • Introduction – Increases the threshold for pain; modifies the perception of noxious stimulation – Relieves continuous dull pain better than sharp, intermittent pain – Affective against pain arising from the viscera, skeletal muscle, joints, and integumental structures – Analgesia is more prominent when administered before the pain – In the absence of pain, dysphoria may be experienced

Opioid Agonists - Morphine • Pharmacokinetics – Protein binding – 35% – Clearance – 1, 050 m. L/min – Vd – 224 L – Elimination half-time – 1. 7 – 3. 3 hours – Well absorbed by IM and is administered IV – effect after IM is onset 15 – 30 min. , peak 45 – 90 min. , clinical duration 4 hr. – Bioavailability of orally administered is ~ 25% – IV bioavailable is delayed compared to newer opioids – peak 15 – 30 min.

Opioid Agonists - Morphine • Pharmacokinetics – Aerosol – can be used for lung cancer and associated pleural effusion - ? Act on afferent nerve pathways in the airway – ventilatory depression may occur – analgesic effects are the same as if delivered IV or by a pulmonary drug delivery system – Plasma morphine concentration following IV administration do not correlate closely with pharmacological activity – this is most likely due to delay in transit across the BBB – only a small amount of morphine gains access to the CNS

Opioid Agonists - Morphine • Pharmacokinetics – Alkalinization of the blood – increases the nonionized fraction of morphine – enhances passage through the BBB – Respiratory acidosis – decreases the nonionized fraction but a higher plasma and brain concentration results than would in a normocarbic patient – This suggests the increase in CBF induced by hypercarbia enhances the delivery of morphine to the brain – enhanced delivery is more important than the fraction of nonionized drug – Morphine accumulates in the kidney, liver and skeletal muscle

Opioid Agonists - Morphine • Pharmacokinetics – Metabolism • Primarily by conjugation with glucuronic acid by the liver and antihepatic sites • Metabolites are morphine-3 -glucuronide (inactive), morphine-6 glucuronide (active), normorphine, & codeine • Analgesic potency of morphine-6 -glucuronid is 650 -fold higher than morphine • Renal clearance also occurs

Opioid Agonists - Morphine • Elimination Half-Time – – Elimination of morphine-3 -glucuronide is longer than morphine Initial redistribution is due to hepatic metabolism Plasma concentrations are higher in the elderly Clearance is decreased in the first 4 days of life, thus the elimination half-life is prolonged – Patients with renal failure exhibit higher plasma and CNS concentrations than do normal patients – reflects a smaller volume of distribution – It is unlikely that significants amount of this drug will be found in breast milk

Opioid Agonists - Morphine • Gender – Greater analgesic potency and slower onset in women – Decreases the slope of the ventilatory response to carbon dioxide more in women but there is no significant effect – Increases apneic threshold in men – Hypoxic threshold is decreased in women

Opioid Agonists - Meperidine • • • Pethidine Synthetic Agonist for the μ & κ receptors Analogues are fentanyl, sufentanil, alfentanil remifentanil Structure similar to local anesthetics – Also blocked sodium channel to a degree similar to these anesthetics • Structurally it is also similar to atropine

Opioid Agonists – Meperidine • Pharmacokinetics – 1/10 the potency of M/S – Duration of action – 2 to 4 hours – It produces similar sedation. euphoria, nausea, vomiting and ventilatory depression in equipotent doses – 80% first-pass hepatic effect – limits oral use

Opioid Agonists – Meperidine • Metabolism – Extensive hepatic metabolism – Demethylated to normeperidine – hydrolyzed to meperidinic acid – Renal elimination and it is p. H dependent – acidification speed urinary elimination – Normeperidine can accumulate in renal failure – Normeperidine has an elimination half-time of 15 hrs. – has ½ the potency of meperidine as an analgesic – produces CNS stimulation

Opioid Agonists – Meperidine • Metabolism – Normeperidine toxicity • Myoclonus and seizures – Normeperidine may be important in meperidine-induced delirium

Opioid Agonists – Meperidine • Elimination-Half-Time – 3 to 5 hrs. – A large dose could potentially saturate hepatic enzymes prolonging duration of action – 60% protein binding – Elderly patients – decreased protein binding – Alcoholics have tolerance - ? Increased volume of distribution

Opioid Agonists – Meperidine • Clinical Uses – Only opioid adequate for surgery administered intrathecally – because of ability to block sodium channels + its μ-mediated opioid activity – Postop analgesia – IM can have plasma concentrations that vary 3 - to 5 -fold and peak concentration that vary from 3 - to 7 -fold – Minimal analgesic plasma concentration is highly variable among patients – Normeperidine toxicity has occurred during patient-controlled analgesia

Opioid Agonists – Meperidine • Clinical Uses – Postoperative shivering – Potent α 2 -agonist – Is not useful to treat diarrhea or cough – Not used in high doses because of significant negative cardiac inotropic effects plus histamine release

Opioid Agonists – Meperidine • Side Effects – – – – – Rarely causes bradycardia Decreased myocardial contractility Delirium & seizures – normeperidine Serotonin syndrome Ventilatory depression Crosses the placenta Less constipation and urinary retention Less of an affect on biliary spasm Mydriasis, dry mouth, increased HR Transient neurological symptoms when given intratheacally

Opioid Agonists – Meperidine • Serotonin Syndrome – can occur when combined with MOAIs and fluoxetine) – Autonomic instability characterized by • • Hypertension Tachycardia Diaphoresis Confusion Agitation Behavioral changes Coagulopathy Metabolic acidosis

Opioid Agonists - Meperidine • Withdrawal Symptoms – Related to those seen with morphine but with less ANS effects – Symptoms develop more rapidly and are of shorter duration

Opioid Agonists - Fentanyl • General Information – Phenylpiperidine derivative synthetic opioid agonist – Structurally resembles morphine – It is 75 -125 times more potent than morphine – A meperidine deriviative

Opioid Agonists - Fentanyl • Pharmacokinetics – Single dose IV – more rapid onset & shorter duration of action the M/S – There is time lag between peak plasma concentration & peak slowing of EEG – reflects effect-site equilibration time between brain and blood which is 6. 4 min. – High lipid solubility – Rapid distribution to inactive tissue sites – Lungs serve as a large inactive tissue site – undergoes first-pass pulmonary uptake

Opioid Agonists - Fentanyl • Pharmacokinetics – Multiple IV doses or continuous infusion progressively saturates inactive tissue sites and plasma concentrations do not decrease as rapidly – duration of analgesia and ventilatory depression can be prolonged – CPB has little effect on pharmacokinetics of this drug

Opioid Agonists - Fentanyl • Metabolism – N-demethylation to norfentanyl, hydroxyproprionyl-fentanyl, and hydroxypropironyl-norfentanyl – Substrate of CYP 3 A 4

Opioid Agonists - Fentanyl • Elimination Half-Time – Protein binding – 84% – Clearance – 1, 530 m. L/min – Volume of distribution – 335 L – Elimination half-time – 3. 1 – 6. 6 hr. – Context-sensitive half-time – 260 min – Effect-site equilibration time – 6. 8 min

Opioid Agonists - Fentanyl • Elimination Half-Time – Plasma concentrations maintained by slow reuptake from nonactive tissue sites – give persistent drug effects that parallel prolonged elimination half-time – Prolonged elimination half-time in the elderly, patients undergoing abdominal aortic surgery requiring infrarenal aortic cross-clamping – Significant cirrhosis does not prolong elimination half-time

Opioid Agonists - Fentanyl • Context-sensitive half-time – Beyond 2 hr. becomes longer than sufentanil – Reflects saturation of inactive tissue sites during prolonged infusion & return of opioid from the peripheral tissues sites

Opioid Agonists - Fentanyl • CPB – Initiation of – all opioids show a decrease in plasma concentrationthis is greater with fentanyl – it is the least with opioids that have large volumes of distribution – Elimination of alfentanil and fentanyl – prolonged by

Opioid Agonists - Fentanyl • Clinical Uses – Low dose – analgesia – 1 to 2 μg/kg/IV – Adjunct to inhaled anesthesia – 2 μg/kg/IV to 20 – Must consider the prolonged effect-site equilibration – Use of before painful stimuli may decrease subsequent amounts needed – 1. 5 to 3 μg/kg/IV 5 minutes before induction reduces the amount of isoflurane and desflurane with 60% nitrous oxide

Opioid Agonists – Fentanyl • Clinical Uses – 50 to 100 μg/kg/IV – surgical anesthesia • Advantages – Lack of direct myocardial depression – Absence of histamine release – Suppression of the stress response to surgery • Disadvantages – Failure to prevent SNS response to painful surgical stimulation – Unpredictable amnesia – Postop ventilatory depression

Opioid Agonist – Fentanyl • Clinical Uses – Labor epidurals – Transmucosal preparation – designed to deliver 5 to 20 μg/kg • Can have PONV • Decreased ventilation frequency • Arterial hypoxemia – Transdermal preparation • Delivers 75 to 100 μg/hr

• Side Effects – Similar to morphine – Potential postoperative problem is persistent & recurrent ventilatory depression

Opioid Agonists – Fentanyl • Cardiovascular Effects – No release of histamine – Depressed carotid sinus baroreceptor reflex – neonates – Bardycardia

Opioid Agonists – Fentanyl • Seizure Activity – This type activity has followed rapid injection of fentanyl, sufentanil, and alfentanil – may be secondary to depression of inhibitory neurons

Opioid Agonists - Fentanyl • Somatosensory Evoked Potentials and EEG – >30 μg/kg IV – produce changes in somatosensory evoked potentials that do not interfere with the use & interpretation of monitoring during anesthesia – Attenuate skeletal muscle movement at doses that have little effect on EEG

Opioid Agonist - Fentanyl • ICP – Fentanyl & sufentanil – modest increases in ICP in patients with head injury regardless of maintenance of unchanged Pa. CO 2 – Usually accompanied with decreased MAP and CPP – Increase ICP does not occur with sufentanil if MAP decreases are prevented.

Opioid Agonists – Fentanyl • Drug Interactions – Potentiation of benzodiazepine – synergistic as to hyponosis and ventilatory depression – Decrease dose of propofol

Opioid Agonist - Sufentanil • General Facts – 5 to 10 times more potent than fentanyl – The parallels the greater affinity this drug has for the receptors – It is 50% more potent at slowing the EEG as fentanyl – 1, 000 -fold difference in the analgesic dose & the dose that causes seizures in animals – Accidental intrathecal injections have been associated with transient skeletal muscle spasm

Opioid Agonist - Sufentanil • Pharmacokinetics – Protein binding – 93% – Clearance – 900 m. L/min – Volume of distribution – 123 L – Elimination half-time – 2. 2 – 4. 6 h – Context-sensitive half-time – 30 min – Effect-site equilibration-time – 6. 2 min

Opioid Agonists – Sufentanil • Prolonged elimination half-time – elderly having abdominal aortic surgery • Vd and elimination half-time is increased in obesity • High tissue affinity is consistent with its lipid solubility • Penetration of BBB and onset of CNS effects associates with its lipid solubility • Rapidly redistributes to inactive tissue sites • 60% first-pass pulmonary uptake

Opioid Agonists – Sufentanil • Smaller Vd is due to increased protein binding • Binds primarily to α 1 -acid glycoprotein • Because of decreased α 1 -acid glycoprotein in neonates, ventilation depression occurs at much lower doses than those for an adult

Opioid Agonists - Sufentanil • Metabolism – N-dealkylation by the liver – metabolites are inactive – O-demethylation produces a weakly active metabolite – Metabolized by microsomal enzymes – Metabolites are excreted equally by feces and kidney – Sensitive to changes in HBF and not enzymatic activity

Opioid Agonists - Sufentanil • Context-Sensitive Half-Time – Less than alfentanil for continuous infusions up to 8 hours – Partially explained by the larger Vd of sufentanil – Compared to alfentanil, sufentanil may have a more favorable recovery profile than alfentanil when used over a longer period of time. – However, alfentanil has the advantage for treating discrete and transient noxious stimuli because of its short effect-site equilibration time which allows rapid access to the brain and facilitates titration

Opioid Agonists – Sufentanil • Clinical Uses – Single dose of 0. 1 to 0. 4 μg/kg IV produces longer period of analgesia and less ventilatory depression than a comparable dose of fentanyl – Compared to fentanyl or M/S, 18. 9 μgkg IV results in a more rapid induction of anesthesia – It decreases or does not change CMRO 2 and CBF – Bradycardia may decrease CO – Delayed ventilatory depression has happened

Opioid Agonists – Sufentanil • Clinical Uses – Large doses produce minimal CVS effects with good left ventricular function – Systemic BP and hormonal reaction to painful stimuli may not be predictable prevented – Can produce IV induction of anesthesia – Can get chest wall rigidity

Opioids - Alfentanil • Introduction: – Less potent than fentanyl (1/5 to 1/10) – 1/3 the duration of action – Advantages over both fentanyl and sufentanil • More rapid onset of action (rapid effect-site equilibrium = 1. 4 minutes) • Rapid effect-sight equilibrium results because of low p. Ka – 90% is in the nonionized form.

Opioid - Alfentanil • Shorter elimination half-time than fentanyl or sufentanil • Half-time is prolonged by cirrhosis • Elimination half-time is shorter in children (4 to 8 years) May be due to small Vd ? due to decreased adipose tissue • Vd os 4 to 6 times smaller than fentanyl due to lower lipid solubility & high protein binding • Renal failure does not alter elimination half-time or clearance • Primarily bound to α 1 -acid glycoprotein

Opioid - Alfentanil • Metabolism – Two independent pathways • Piperidine N-dealkylation to noralfentanil • Amide N-dealkylation to N-phenylpropionamide – Individual variability – variability in the P 450(CYP 3 A 4) – 96% cleared from the plasma in 60 minutes

Opioid - Alfentanil • Context-sensitive half-time – Longer than sufentanil for infusion up to 8 hours – partly due to the larger Vd for sufentanil

Opioid - Alfentanil • Clinical Uses – Acute pain – but transient – 15 μg/kg IV 90 seconds before intubation & performance of retrobulbar block – 30 μg/kg IV blunts catecholamine response to noxious stimulation – 10 to 20 μg/kg blunts circulatory but not catecholamine response t high inhaled concentrations of desflurane – Induction dose = 150 -300μg/kg IV – Maintenance Infusion = 25 to 150 μg/kg/hr IV + inhaled agent

Opioids - Alfentanil • Supplemental doses seem more likely to reduce blood pressure due to painful stimuli • Increases biliary tract pressure = fentanyl • Associated with lower incidence of PONV than equipotent doses of fentanyl and sufentanil • Untreated Parkinson’s - dystonia

Opioids - Remifentanil • • Selective μ-opioid agonist Analgesic potency similar to fentanyl Effect-site equilibration similar to alfentanil Short-acting phenylpiperidine with an ester linkage – makes it susceptible to hydrolysis by nonspecific plasma and tissue esterases to inactive metabolites – Brief action – Precise and rapidly titratable effect due to rapid onset and rapid offset – Lack of accumulation

Opioid - Remifentanil • Pharmacokinetics – Small Vd – Rapid clearance = ~ 3 L/minute – Low interindividual variability – Contest-sensitive half-time will be nearly independent of infusion duration – reaches steady-state plasma concentration in about 10 minutes – About 19 times more potent than alfentanil – Has an alfentanil-like onset

Opioid - Remifentanil • Metabolism – Nonspecific plasma and tissue esterases to inactive metabolites – Remifentanil acid – less potent than remifentanil – excreted by the kidney – Likely pharmacokinetics will not be affected by renal or hepatic disease – Hypothermia cardiopulmonary bypass decreases clearance by 20%

Opioids - Remifentanil • Elimination half-time – 98. 8% eliminated during the distribution (0. 9 minute) and elimination (6. 3 minutes) half-time – Behaves like a drug with an elimination half-time of ≤ 6 minutes • Context-sensitive half-time – Independent of duration of infusion – estimated to be about 4 minutes

Opioids - Remifentanil • Unique characteristics – Rapid onset – Precise titration to desired effect – Ability to maintain sufficient effect-site concentration to suppress the stress response – Rapid recovery

Opioids – Remifentanil • Ventilation – 0. 5 μg/kg IV – decreased slope & downward shift of the carbon dioxide ventilatory response curve – reaches its nadir after about 150 seconds – Recovery is complete in 15 minutes – Remifentanil + propofol = synergism – severe respiratory depression

Opioids - Remifentanil • Clinical Uses – – – Profound analgesia – retrobulbar blocks Suppression of transient SNS response to tracheal intubation in at-risk patients Intermittent dosing – pain during labor & delivery Can be used for long surgeries Induction doses – 1 μg/kg Iv over 60 – 90 seconds with a gradual infusion of 0. 5 to 1. 0 μg/kg IV for about 10 minutes before hypnotic dose prior to intubation – Analgesic component for general anesthesias – 0. 25 – 1. 00 μg/kg Iv or 0. 05 – 2. 00 μg/kg/minute IV – MAC – midazolam 2 mg IV with 0. 05 – 0. 10 μg/kg/minute – EEG – 100 μg IV
Opioids - Remifentanil • Side effects – Long duration opioid for postoperative pain – “seizure-like” activity – PONV – Ventilatory depression – Mild decreased B/P & HR – Decreased CBF & CMRO 2 – Delayed drainage of dye from gallbladder into duodenum

Opioids - Remifentanil • Hyperalgesia – May cause acute opioid tolerance – Mechanism may be alterations in the NMDA receptors and its IC second messenger systems – Ketamine and magnesium block opioid tolerance

Opioid Antagonists • Naloxone, naltrexone, & nalmefene are pure μ antagonists with no agonistic effects • Naloxone – Nonselective for all 3 receptors – Is selective when used to treat • Treat opioid-induced ventilatory depression • Treat neonatal depression • Facilitate treatment of deliberate opioid overdose – Dose 1 to 4 μg/kg/IV – Continuous infusion – 5 μg/kg/hour

Opioid Antagonists • Naloxone – Side effects • • Reversal of analgesia N&V CV stimulation Crosses placental barrier – Antagonism of General Anesthesia • Drug-induced activation of the cholinergic arousal system – Shock • Dose-related improvement in cardiac contractility in septic shock

Opioid Antagonists • Naltrexone – Oral – Sustained antagonism – 24 hours • Nalmefene – Equipotent to naloxone – Can use to decrease the need for antiemetics and antipruritic with IV patientcontrolled analges

Opioid Antagonists • Methylnaltrexone – Does not cross BBB – Active in peripheral antagonism – Attenuates morphine changes in the GI tract – Decreases N & V • Alvimopan – μ-selective oral peripheral opioid antagonists – Metabolized by gut flora

Opioid Allergy • True opioid allergy is rare • The histamine release from morphine is nonimmune release • 4 cases of fentanyl allergy induced by anaphylaxis

Tamper- or Abuse Resistant Opioids • Suboxone (buprenorphine + Naloxone) • Embeda (extended release morphine + naltrexone) • Oxy. Nal (oxycodone + naltrexone)

- Slides: 118