Ongoing collaborative projects Prospective for collaborative projects Kirsten

Ongoing collaborative projects Prospective for collaborative projects Kirsten Sundby Hall Oslo CTOS 4. Nov - 06
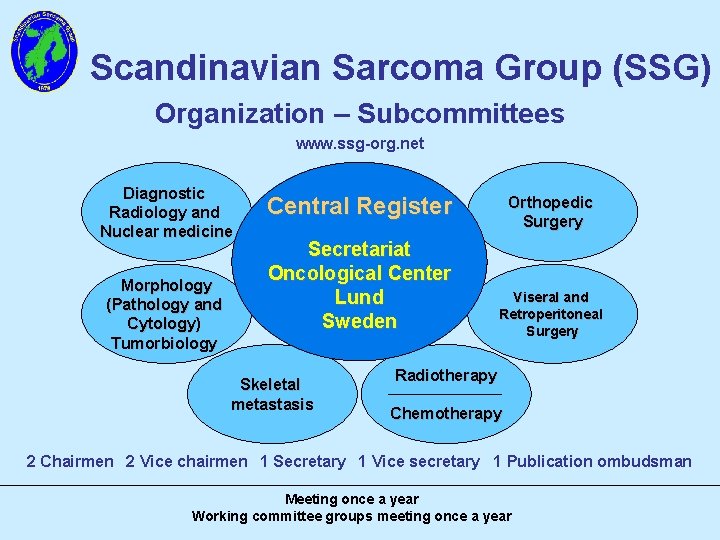
Scandinavian Sarcoma Group (SSG) Organization – Subcommittees www. ssg-org. net Diagnostic Radiology and Nuclear medicine Morphology (Pathology and Cytology) Tumorbiology Central Register Secretariat Oncological Center Lund Sweden Skeletal metastasis Orthopedic Surgery Viseral and Retroperitoneal Surgery Radiotherapy Chemotherapy 2 Chairmen 2 Vice chairmen 1 Secretary 1 Vice secretary 1 Publication ombudsman Meeting once a year Working committee groups meeting once a year

SSG Registry 1986– 2005: 8 126 patients All sarcoma centers in Sweden (6) and Norway (3) register patients, Finland (1) Population based for Norway and Sweden. Referral pattern, treatment, prognosis www. ssg-org. net

www. ssg-org. net

Current thesis projects based on SSG registry Leiomyosarcoma C. Svarvar, Helsinki Liposarcoma K. Engström, Gothenburg Chondrosarcoma B. Widhe, Stockholm Osteosarcoma C. Müller, Oslo Quality of life L. H. Aksnes, Oslo Skeletal metastases B. H. Hansen, Århus Radiation therapy and local recurrence in STS N. L. Jebsen, Bergen www. ssg-org. net

Guidelines for treatment of metastatic soft-tissue sarcomas in adult patients Proposals for treatment • With a curative intent • With a palliative intent • Chemotherapy-Surgery-Radiotherapy

Guidelines for treatment of abdominal sarcomas • Preoperative diagnosis and planning • Surgery • Chemotherapy, radiotherapy • Follow-up • Centralized management!

Osteosarcoma SSG II Combination therapy in OS 1982 – 1990 SSG VIII Therapy of OS 1990 – 1998 ISG/SSG I An Italian-Scandinavian treatment and 1997 – 2000 research protocol for high-grade osteosarcoma of the extremities ISG/SSG II Treatment of metastatic and pelvic 1998 – 2003 osteosarcoma SSG XIV A Scandinavian treatment research 2000 – 2004 protocol for extemity localized highgrade osteosarcoma Euroboss I A European treatment protocol for bone 2003 sarcoma in patients older than 40 years Euramos I An American/European treatment protocol 2004 for OS in patients <40 years www. ssg-org. net
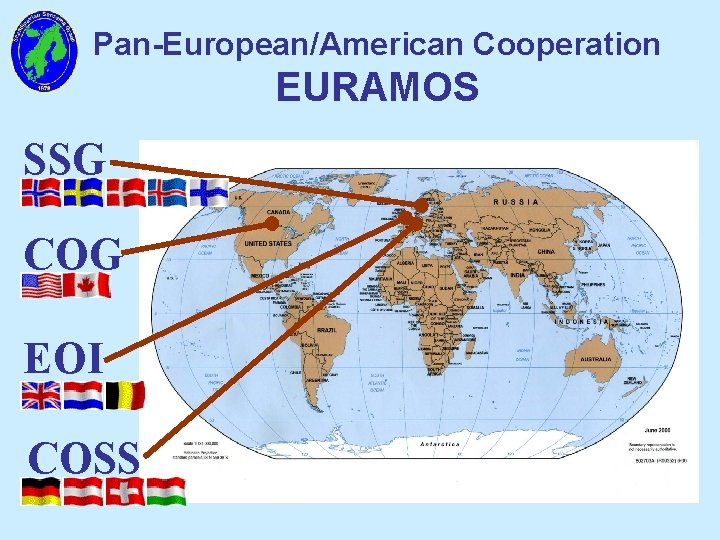
Pan-European/American Cooperation EURAMOS SSG COG EOI COSS
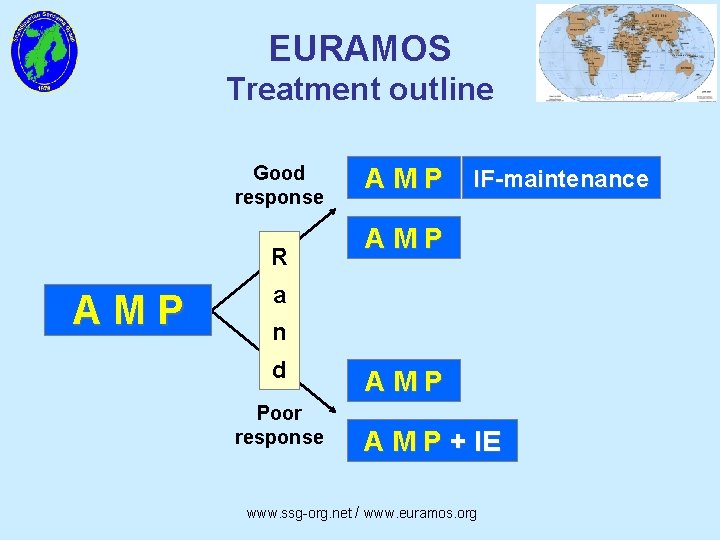
EURAMOS Treatment outline Good response R AMP IF-maintenance AMP a n d Poor response AMP A M P + IE www. ssg-org. net / www. euramos. org

Current status of SSG contribution to EURAMOS I Total of registered patients 18 Total of randomized patients 13 30/9 2006

Ewing´s sarcoma SSG IV 90 Combined modality therapy in EWS SSG IX Therapy of EWS 1984 – 1990 – 99 ISG/SSG III ISG/SSG treatment protocol for metastatic EWS 1999 non- ISG/SSG IV ISG/SSG treatment protocol for high-risk EWS 1999 www. ssg-org. net

SSG/AIO (SSG XVIII) Adjuvant study for high-risk GIST R A N D O M I Z E Arm 1 Imatinib 12 months Follow-up 48 months Imatinib 36 months Follow-up 24 months Arm 2 Primary objective: To investigate the recurrence-free survival in GIST patients with a high (>50%) risk of disease recurrence within the first 5 years following the diagnosis and treated with adjuvant imatinib mesylate either for 12 or 36 months. www. ssg-org. net
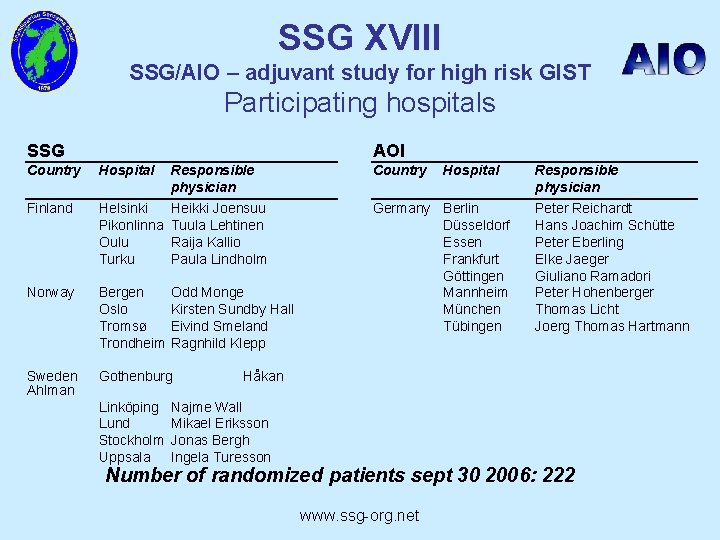
SSG XVIII SSG/AIO – adjuvant study for high risk GIST Participating hospitals SSG AOI Country Hospital Responsible physician Country Finland Helsinki Pikonlinna Oulu Turku Heikki Joensuu Tuula Lehtinen Raija Kallio Paula Lindholm Norway Bergen Oslo Tromsø Trondheim Odd Monge Kirsten Sundby Hall Eivind Smeland Ragnhild Klepp Germany Berlin Düsseldorf Essen Frankfurt Göttingen Mannheim München Tübingen Sweden Ahlman Gothenburg Linköping Lund Stockholm Uppsala Hospital Responsible physician Peter Reichardt Hans Joachim Schütte Peter Eberling Elke Jaeger Giuliano Ramadori Peter Hohenberger Thomas Licht Joerg Thomas Hartmann Håkan Najme Wall Mikael Eriksson Jonas Bergh Ingela Turesson Number of randomized patients sept 30 2006: 222 www. ssg-org. net

SSGXX-a new adjuvant STS protocol 2007 “A Scandinavian Sarcoma Group (SSG) treatment protocol for adult patients with non-metastatic high-risk STS of the extremities and trunk wall”

(Opened 1998 -closed 31. 12. 05) * 2 or 3 risk (SIN) factors • Size>8 cm • Presence of necrosis • Presence of vascular invasion
Introduction of a novel risk factor assessment • Peripheral tumor growth pattern Infiltrative growth of a STS is a strong factor for both local recurrence and metastases
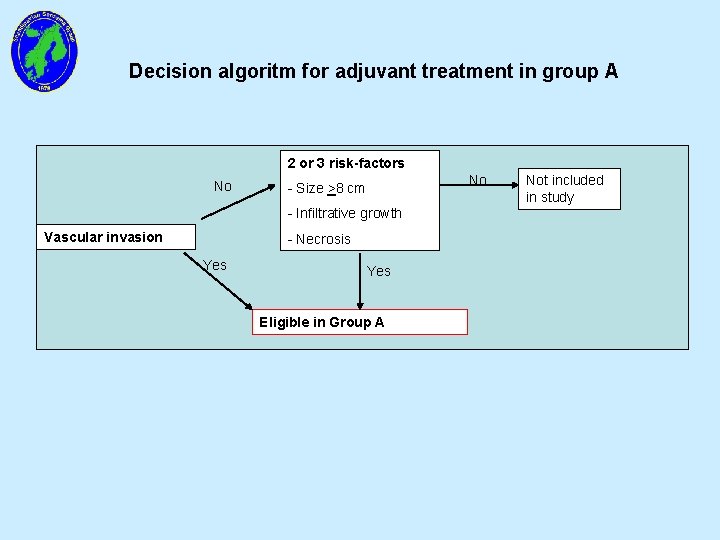
Decision algoritm for adjuvant treatment in group A 2 or 3 risk-factors No No - Size >8 cm - Infiltrative growth Vascular invasion - Necrosis Yes Eligible in Group A Not included in study
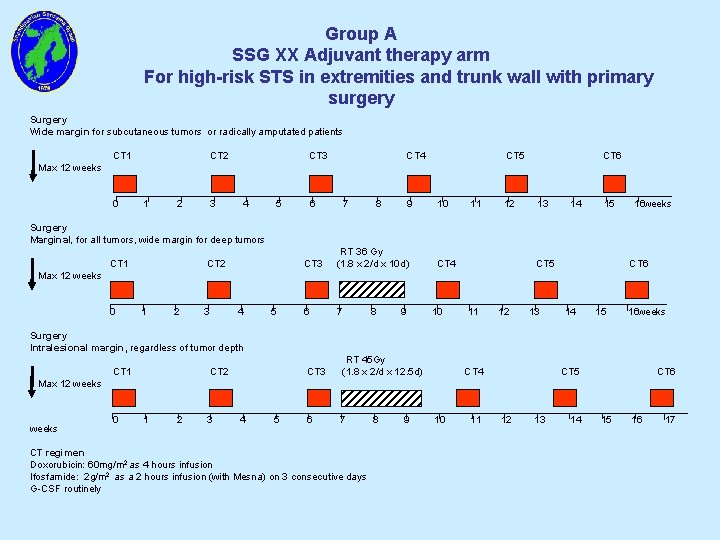
Group A SSG XX Adjuvant therapy arm For high-risk STS in extremities and trunk wall with primary surgery Surgery Wide margin for subcutaneous tumors or radically amputated patients CT 1 CT 2 CT 3 CT 4 CT 5 CT 6 Max 12 weeks 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 weeks Surgery Marginal, for all tumors, wide margin for deep tumors CT 1 CT 2 CT 3 RT 36 Gy (1. 8 x 2/d x 10 d) 6 7 CT 4 CT 5 CT 6 Max 12 weeks 0 1 2 3 4 5 8 9 10 11 12 13 14 15 16 weeks Surgery Intralesional margin, regardless of tumor depth CT 1 CT 2 CT 3 RT 45 Gy (1. 8 x 2/d x 12. 5 d) 6 7 CT 4 CT 5 CT 6 Max 12 weeks 0 1 2 3 4 5 CT regimen Doxorubicin: 60 mg/m 2 as 4 hours infusion Ifosfamide: 2 g/m 2 as a 2 hours infusion (with Mesna) on 3 consecutive days G-CSF routinely 8 9 10 11 12 13 14 15 16 17
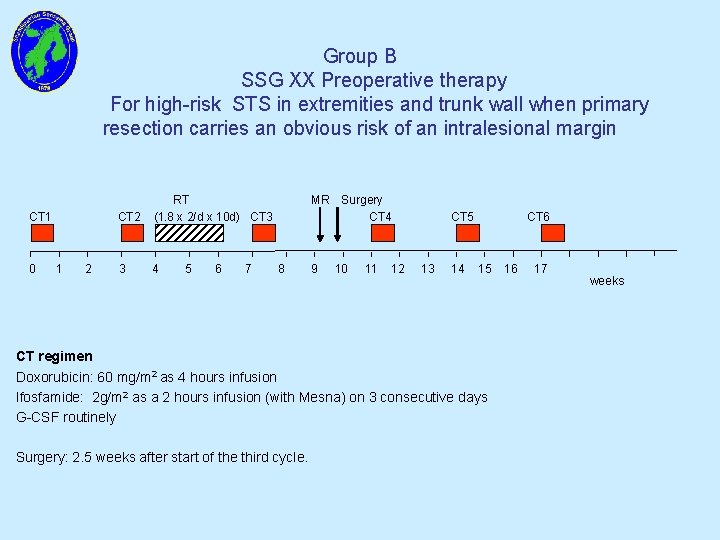
Group B SSG XX Preoperative therapy For high-risk STS in extremities and trunk wall when primary resection carries an obvious risk of an intralesional margin CT 1 0 1 2 CT 2 RT (1. 8 x 2/d x 10 d) CT 3 3 4 5 6 7 MR Surgery CT 4 8 9 10 11 12 CT 5 13 14 CT 6 15 CT regimen Doxorubicin: 60 mg/m 2 as 4 hours infusion Ifosfamide: 2 g/m 2 as a 2 hours infusion (with Mesna) on 3 consecutive days G-CSF routinely Surgery: 2. 5 weeks after start of the third cycle. 16 17 weeks

SSG policy - to continuously work for more international collaboration on treatment protocols and research www. ssg-org. net

• End
- Slides: 22