Oncometabolites linking altered metabolism with cancer BY Marwa

Oncometabolites linking altered metabolism with cancer BY: Marwa Mostafa Nourhan Hossam
Outlines Definition Members Metabolic Origins of Oncometabolites in Normal and Cancer Cells Epigenetic link with metabolism Mechanism of action of oncometabolite Examples of cancers with dysregulated oncometabolites Environmental cues regulates oncometabolite production laboratory methodologies that can be used to quantify these metabolites Targeted Therapies Future Directions Metabolic structure of oncometabolites Previously Identified in Oncometabolites and tumor repopulation

Definition The term oncometabolites refers to intermediates of metabolism that abnormally accumulate in cancer cells upstream (e. g. , L‐ 2‐hydroxyglutarate, succinate, fumarate or downstream (e. g. , D‐ 2‐hydroxyglutarate) of metabolic defects, often through loss‐of‐function or gain‐of function mutations, respectively, of genes encoding the corresponding enzymes.

Members The currently recognized metabolites include: 1. Succinate 2. Fumarate 3. 2 -hydroxyglutarate. They share metabolic proximity in the TCA cycle. As a result, they promote tumorigenesis in cancer cells through nearly similar mechanisms.
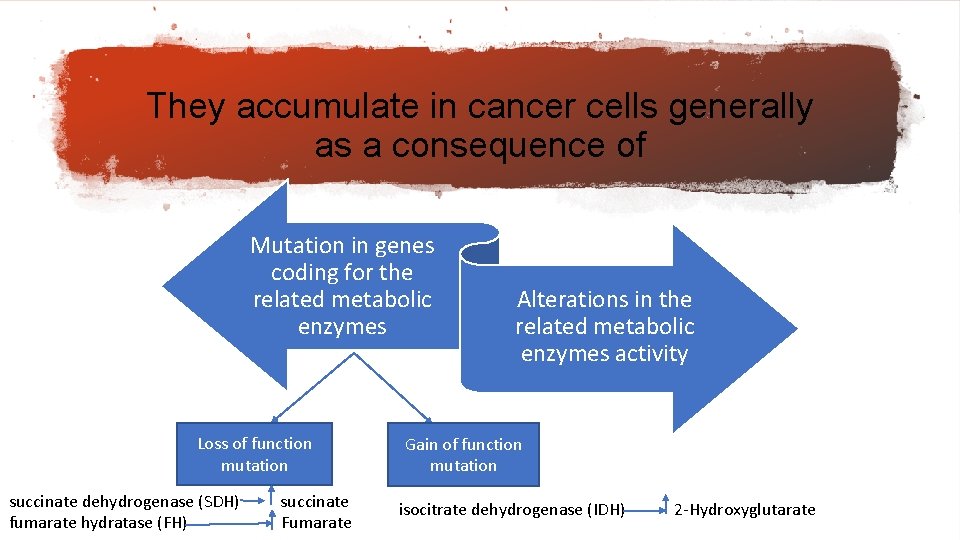
They accumulate in cancer cells generally as a consequence of Mutation in genes coding for the related metabolic enzymes Loss of function mutation succinate dehydrogenase (SDH) fumarate hydratase (FH) succinate Fumarate Alterations in the related metabolic enzymes activity Gain of function mutation isocitrate dehydrogenase (IDH) 2‐Hydroxyglutarate

Metabolic Origins of Oncometabolites in Normal and Cancer Cells: The concept of glycolytic fueling of cancer cells known as The Warburg effect has received renewed attention. The ability of cancer cells to undergo aerobic glycolysis allows for the production of glycolytic and TCA cycle intermediates that support biosynthetic pathways required for cell growth and proliferation. Accordingly, certain cancers are associated with genetic aberrations in TCA cycle enzymes.
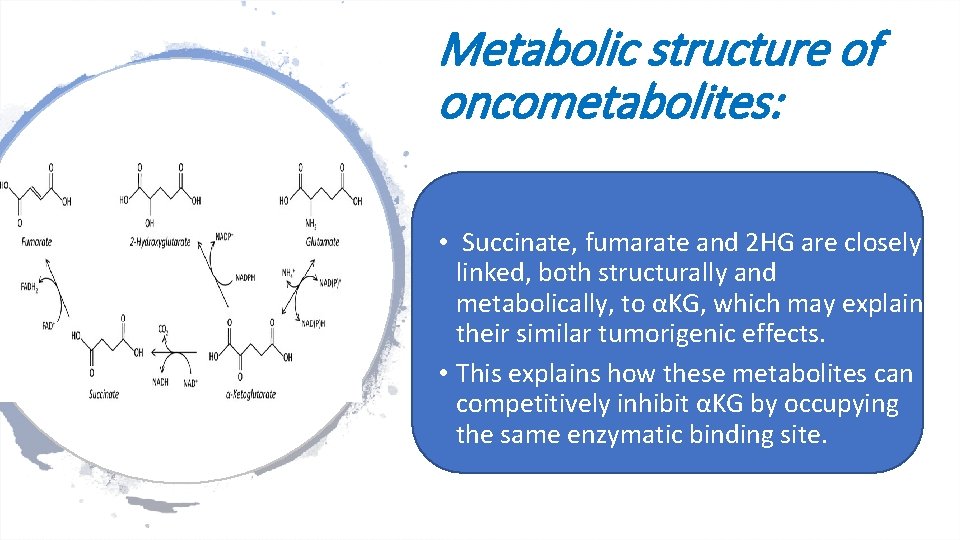
Metabolic structure of oncometabolites: • Succinate, fumarate and 2 HG are closely linked, both structurally and metabolically, to αKG, which may explain their similar tumorigenic effects. • This explains how these metabolites can competitively inhibit αKG by occupying the same enzymatic binding site.

Those oncometabolites were Previously Identified in: Pediatric clinical laboratories commonly measure TCA CYCLE intermediates for screening, diagnosis, and monitoring of specific inborn errors of metabolism, such as organic acidurias including: • D-2‐ hydroxyglutaric aciduria. • L-2‐ hydroxyglutaric aciduria. • Fumaric aciduria.
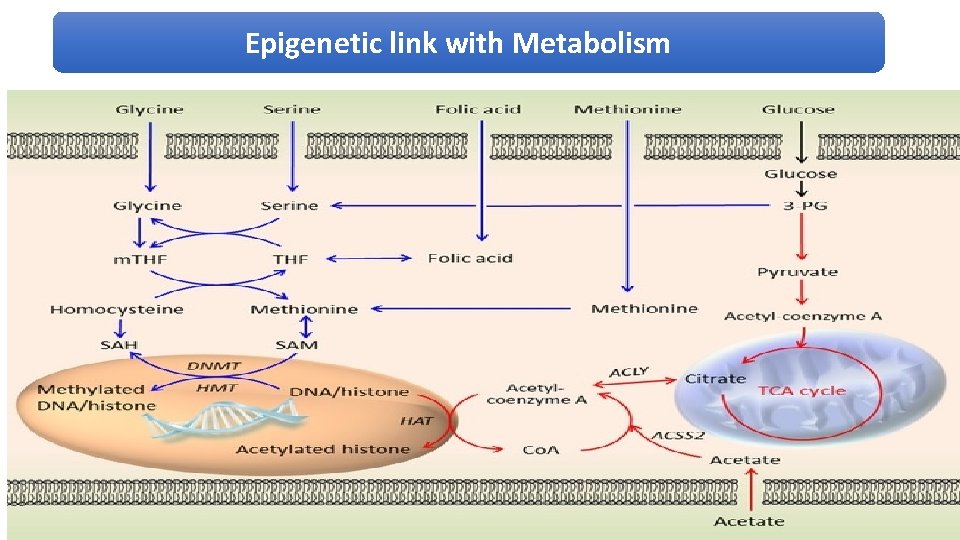
Epigenetic link with Metabolism
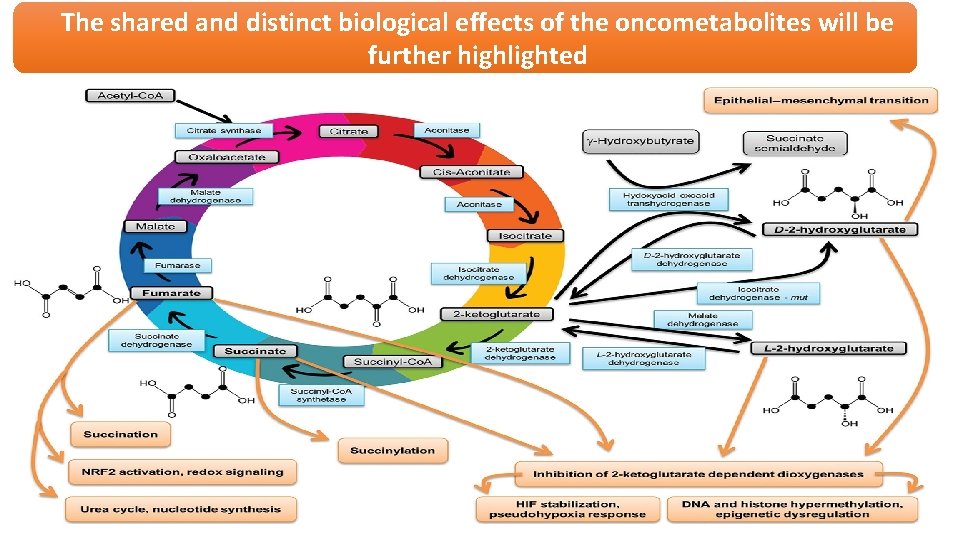
The shared and distinct biological effects of the oncometabolites will be further highlighted
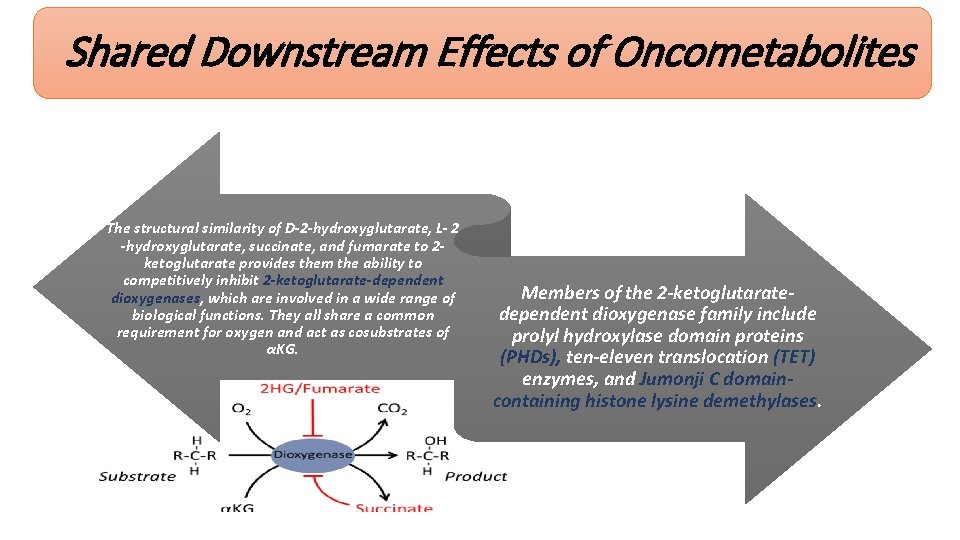
Shared Downstream Effects of Oncometabolites The structural similarity of D-2 -hydroxyglutarate, L- 2 -hydroxyglutarate, succinate, and fumarate to 2 ketoglutarate provides them the ability to competitively inhibit 2 -ketoglutarate-dependent dioxygenases, which are involved in a wide range of biological functions. They all share a common requirement for oxygen and act as cosubstrates of αKG. Members of the 2 -ketoglutaratedependent dioxygenase family include prolyl hydroxylase domain proteins (PHDs), ten-eleven translocation (TET) enzymes, and Jumonji C domaincontaining histone lysine demethylases.

Shared Downstream Effects of Oncometabolites (pseudohypoxic state) Under normoxic conditions, PHDs hydroxylate specific proline residues in the hypoxia-inducible factors (HIFs), which are then polyubiquitinated by von Hippel-Lindau E 3 ubiquitin ligase complex and degraded by the proteasome. Hypoxia reverses this process, resulting in stabilization of HIFs that are transcription factors for genes involved in angiogenesis, cell growth, and cell migration. Inhibition of PHDs by oncometabolites causes aberrant stabilization of HIFs (termed pseudohypoxia), which then promote angiogenesis and cancer cell growth.

Shared Downstream Effects of Oncometabolites (hypermethylation) TET enzymes hydroxylate 5 -methylcytosine in DNA Cp. G dinucleotides as a step toward cytosine demethylation, whereas lysine demethylases demethylate lysine residues in histones. Inhibition of ten-eleven translocation enzymes and lysine demethylases by oncometabolites leads to aberrant DNA and histone hypermethylation, respectively, effecting epigenetic changes in cancer cells. Indeed, cancers with high levels of oncometabolites have a hypermethylation phenotype.

Shared Downstream Effects of Oncometabolites (EMT) EMT is a biological process that involves multiple biochemical changes that enable an epithelial cell to assume a mesenchymal cell phenotype. EMT is involved in many physiological events and is now considered a key mechanism for the acquisition of malignant features by epithelial cancer cells. Through the circulatory system, mesenchymal cancer cells have the ability to extravasate, increasing cancer aggressiveness, invasiveness, metastatic potential and resistance to chemotherapy. All features that are closely associated with stemness and with the presence of CSCs cancer stem cells.

Succinate and succinate Dehydrogenase (SDH) in cancer Succinate is a mitochondrial metabolite of TCA cycle Oxidized to fumarate by SDH enzyme SDH is a mitochondrial enzyme that acts in two metabolic processes: TCA cycle and Electron transport chain The most commonly mutated SDH genes are: SDHB and SDHD SDH enzyme is composed of 4 subunits SDHA, SDHB, SDHC, SDHD And 2 associated complex factors SDHF 1 and SDHF 2

Succinate and succinate Dehydrogenase (SDH) in cancer(cont. ) Besides the mutational status of SDH subunits, it is emerging that SDH expression/activity might also be regulated: At a transcriptional or post transcriptional level By epigenetic suppression of SDH expression At a post-translational level By the inhibition of SDH activity • By RNA non-sense mutation due to C-to-U RNA editing. • The binding of the chaperone tumor necrosis factor receptor-associated protein 1 (TRAP-1). • By the expression of micro. RNAs (mi. RNAs) targeting • The regulation of SDH phosphorylation through protein SDH messenger RNAs (m. RNAs). tyrosine phosphatase mitochondrial 1 (PTPMT 1).

Succinate and succinate Dehydrogenase (SDH) in cancer(cont. ) The inhibition of SDH functionality can result in a marked accumulation of succinate in plasmaof patients, Ranging from the physiological concentration of about 5 μM to the pathological millimolar levels. These elevated concentrations of succinate may: Suggesting the marked influence of this oncometabolite cancer cell modification of proteins succinate has been recently identified to participate ininpost-translational through succinylation of lysine residues. Some identified targets of succinylation are: energetic metabolism • Induce metabolic reprogramming of the tumour microenvironment to promote cancer cell growth. • L-lactate dehydrogenase. A, glyceraldehyde 3 -phosphate dehydrogenase and malate dehydrogenase. • Succinate is also involved in the γ –aminobutyric acid (GABA) shunt, which consists of a cycle of reactions ensuring the production and conservation of GABA.

Fumarate and Fumarate Hydratase (FH) in cancer Fumarate is another physiological metabolic intermediate of TCA cycle. It is transformed to malate by Fumarate Hydratase(FH) enzyme. FH is a homotetrameric protein present in 2 isoforms A mitochondrial isoform involved in the conversion of fumarate to malate in the TCA cycle, A cytosolic isoform involved in amino acid catabolism and the urea cycle

Fumarate and Fumarate Hydratase (FH) in cancer(cont. ) FH is regulated at the m. RNA level by mi. RNA 378 which increases during breast cancer progression. FH is regulated at The post translational level by 5 Adenosine monophosphate activated protein kinase(AMPK).

Fumarate and Fumarate Hydratase (FH) in cancer(cont. ): Fumarate is present in normal cells at micromolar levels and inhibition of FH functionality leads to an abnormal accumulation of fumarate to millimolar levels, sufficient to drive the neoplastic transformation of cells. Exposure to high levels of fumarate is: • ROS production and redox homeostasis dysregulation. • Urea cycle reversion. which have been associated with tumor growth.

2 -Hydroxyglutarate and Isocitrate dehydrogenase(IDH) in cancer 2 -HG exists in 2 forms: D-2 -hydroxyglutarate L-2 -hydroxyglutarate *D-2 -HG is mainly produced by mutated IDH in cancer cells. IDH are a class of enzymes that convert isocitrate to α-ketoglutarate (α-KG) in 2 steps during TCA cycle. They exist in 3 different isoforms: IDH 1, localized in the cytosol and in peroxisomes. IDH 2 and IDH 3 are present in the mitochondria.

2 -Hydroxyglutarate and Isocitrate dehydrogenase(IDH) in cancer(cont. ): D-2 -HG was the first molecule described as an oncometabolite for its effects on tumour progression. D-2 -HG is involved in: • Inhibition of α-ketoglutarate-dependent dioxygenases, including members of the alkylated DNA repair protein (Alk. B) family, such as ALKBH 2 and ALKBH 3. • Metabolic dysregulation. • Impairment of cell differentiation. Thus the detection of this oncometabolite in urine can be considered a valuable diagnostic tool to identify patients at risk for cancer development.

Examples of cancers with dysregulated oncometabolites: 1. Succinate dehydrogenase (SDH) Pheochromocytoma/paraganglioma (20%), GIST , RCC 2. Cytoplasmic and mitochondrial isocitrate dehydrogenase (IDH) Glioma (65– 85%), glioblastoma multiforme (5– 10%), acute myeloid leukemia (20%), chondrosarcoma (50%), cholangiocarcinoma (20%) 3. Fumarate hydratase (FH) Papillary kidney cancer (very rare), uterine leiomyoma (very rare), skin leiomyoma (very rare), pheochromocytoma/paraganglioma (1%)
Oncometabolites and tumor repopulation TUMOUR REPOPULATION: Repopulation of tumour cells following therapy is one of the major reasons for treatment failure. It means that the dying tumour cells release several: proteins, non-coding RNAs, and bioactive lipids, which in turn stimulate therapy-surviving cells to proliferate rapidly, sustaining the tumour-repopulation process.

Oncometabolites and tumor repopulation(cont. ) To our knowledge, no experimental studies have directly explored the role of oncometabolites in the repopulation of tumors after therapy. These are just several pivotal findings that strongly support a causal link between oncometabolites and the induction of tumor repopulation after therapy Thus opening the way to new discoveries on the mechanisms underlying tumor repopulation.

Oncometabolites and tumor repopulation(cont. ) It is suggested that oncometabolites may support tumor repopulation in two different ways: (i) by acting as signal molecules released by dying cells and/or by cells of the tumor microenvironment, stimulating the proliferation of ‘recipient’ cells. and/or (ii) by accumulating in therapy-resistant cancer cells after the uptake of mi. RNAs or other molecules able to regulate the functionality of SDH, FH, or IDH enzymes.

Oncometabolites and tumor repopulation(cont. ) It is important to note that the induction of tumour repopulation by oncometabolites may not only necessarily be relevant to tumours harbouring SDH, FH, or IDH mutations, but also to other tumour types that may express alterations in proteins, signalling pathways, non-coding RNAs, or bioactive lipids, which could alter the expression or activity of the SDH, FH, or IDH enzymes. For example: therapy-resistant or dormant cells may repopulate the tumoral mass by receiving secreted or exosome-encapsulated succinate or by enhancing succinate production after receiving signals from other tumour cells, immune cells, or cancer-associated fibroblasts (CAFs).

Oncometabolites and tumor repopulation(cont. ) Released mi. RNAs targeting SDH and FH in tumour repopulation: • The expression levels of about 1100 mi. RNAs known to be involved in the regulation of cellular processes, including apoptosis, proliferation, invasion, local immune response, and radioresistance, display two‐ to threefold changes after irradiation • Interestingly, mi. RNAs are present both in the cytosol of the irradiated cells and within exosomes released by those cells, where they are encapsulated to deliver information to neighboring cells and thus playing an important role in cancer metastasis. • Once tumor-derived micro-vesicles have been released, they are harvested by recipient cells that have survived therapy, inducing alterations that lead to tumor repopulation via the regulation of crucial signaling pathways.

Released mi. RNAs targeting SDH and FH in tumor repopulation:
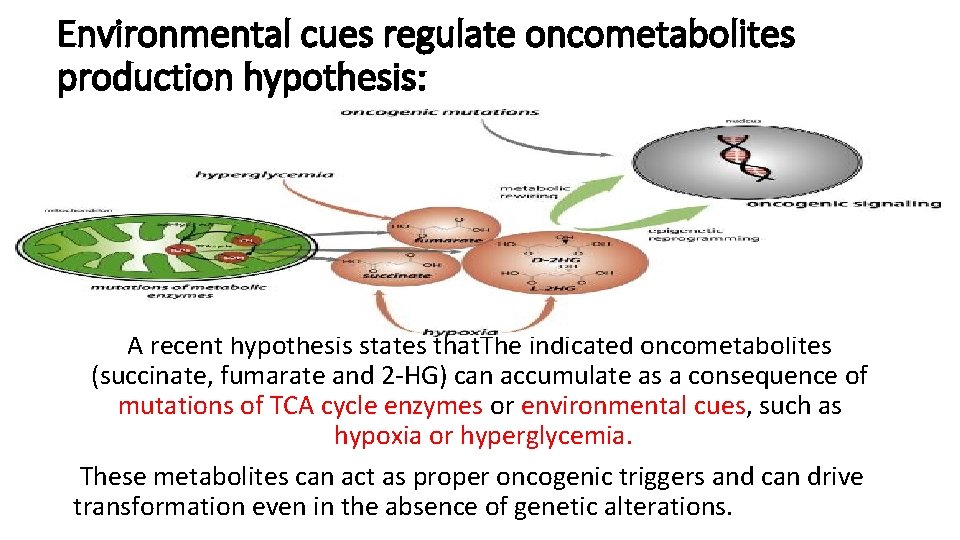
Environmental cues regulate oncometabolites production hypothesis: A recent hypothesis states that. The indicated oncometabolites (succinate, fumarate and 2‐HG) can accumulate as a consequence of mutations of TCA cycle enzymes or environmental cues, such as hypoxia or hyperglycemia. These metabolites can act as proper oncogenic triggers and can drive transformation even in the absence of genetic alterations.

Environmental cues regulate oncometabolites production hypothesis (cont. ): In support to this hypothesis, it has been shown that hypoxia leads to the production of 2 HG either via reductive carboxylation or via promiscuous substrate usage of LDHA and Malic Dehydrogenase. Since hypoxia is a common feature of solid tumours, it is possible that hypoxia‐driven production of 2 HG elicits genetic and epigenetic changes that drive or amplify the process of tumorigenesis.

Gerometabolites Likewise, gerometabolites refer to small‐molecule components of normal metabolism whose depletion causes signaling dysregulation to establish a milieu that drives aging.

Different methods to quantify these oncometabolites and their downstream targets: • Non‐laboratory‐based techniques , such as magnetic resonance spectroscopy. • Laboratory based techniques in labs that are typically equipped with hybrid instruments, such as GC‐MS, LC‐MS instruments. • Allele specific digital PCR for monitoring mutant IDH
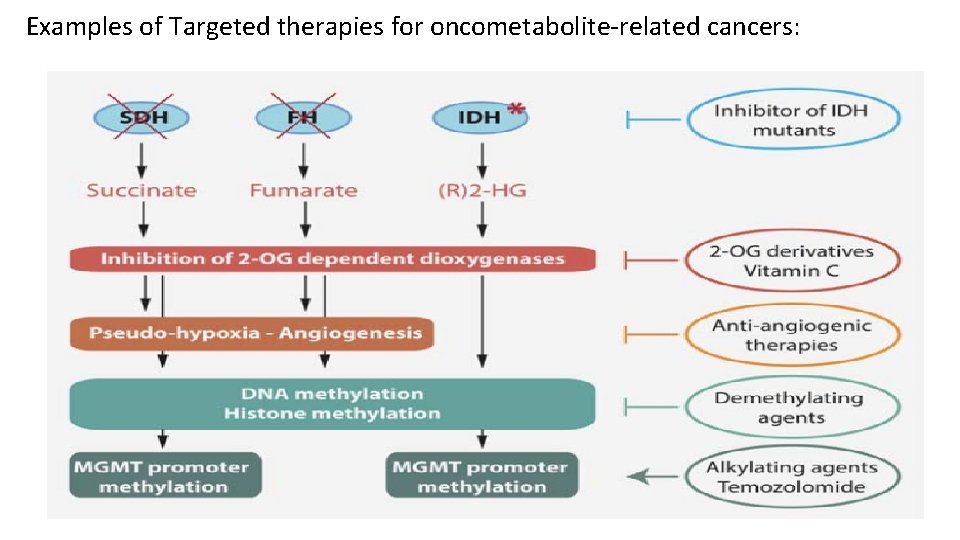
Examples of Targeted therapies for oncometabolite‐related cancers:

Future directions: • Increased production of these oncometabolites in cancer cells makes them ideal potential cancer biomarkers for prognosis, monitoring tumor burden , assessing the effectiveness of treatment and early recurrence. • Moreover, the genetic defects that lead to high levels of these metabolites as well as the downstream signaling pathways are attractive targets for therapeutic interventions. • The discovery of metabolic enzymes that can alter the epigenome has opened up a new, exciting area for drug development. • However, the difficulty in drug development (drugs targeting epigenetic enzymes )may be attributed to the broad activity of these enzymes. • Targeting the metabolic causes of these epigenetic changes may therefore have greater clinical potential.


References: 1. Dando, I. , Pozza, E. D. , Ambrosini, G. , Torrens‐Mas, M. , Butera, G. , Mullappilly, N. , . . . & Donadelli, M. (2019). Oncometabolites in cancer aggressiveness and tumour repopulation. Biological Reviews. 2. Collins RRJ, Patel K, Putnam WC, Kapur P, Rakheja D. Oncometabolites: A New Paradigm for Oncology, Metabolism, and the Clinical Laboratory. Clin Chem. 2017 Dec; 63(12): 1812‐ 1820. 3. Nowicki S, Gottlieb E. Oncometabolites: tailoring our genes. FEBS J. 2015; 282(15): 2796 – 2805. doi: 10. 1111/febs. 13295 4. Morin A, Letouzé E, Favier J, Oncometabolites‐driven tumorigenesis: From genetics to targeted therapy, International journal of cancer , Volume 135, Issue 10, 15 November 2014; 2237‐ 2248 5. De. Berardinis RJ, Chandel NS. Fundamentals of cancer metabolism. Sci Adv. 2016; 2(5): e 1600200. Published 2016 May 27. doi: 10. 1126/sciadv. 1600200 6. JAVIERABEL MENENDEZ, TOMÁS ALARCÓN & Jorge Joven(2014) Gerometabolites: The pseudohypoxic aging side of cancer oncometabolites, Cell Cycle, 13: 5, 699‐ 709. 7. Ming Yang , Tomoyoshi Soga, Patrick J. Pollard, Oncometabolites: linking altered metabolism with cancer, J Clin Invest. 2013; 123(9): 3652‐ 3658.

- Slides: 38