Oncology Nursing 2016 Temozolomide resistance in glioblastoma multiforme

Oncology Nursing 2016 Temozolomide resistance in glioblastoma multiforme (GBM) and its application for drug development Sang Y. Lee, Ph. D. Assistant Professor Pennsylvania State University College of Medicine

Conflict of interest ■ None

Contents ■ Brain tumor & GBM ■ Temozolomide (TMZ) & its resistance ■ TMZ resistant GBM cell lines (intrinsic, acquired) ■ Drug development for TMZ resistant GBM

Top 10 leading causes of death in the US (2014) 1. Heart disease: 614, 348 (23. 5% of total deaths) 2. Cancer: 591, 699 (22. 5% of total deaths) 3. Chronic lower respiratory diseases: 147, 101 (5. 7% of total deaths) 4. Accidents (unintentional injuries): 136, 053 (5. 0% of total deaths) 5. Stroke (cerebrovascular diseases): 133, 103 (5. 0% of total deaths) 6. Alzheimer's disease: 93, 541 (3. 3% of total deaths) 7. Diabetes: 76, 488 (2. 9% of total deaths) 8. Influenza and pneumonia: 55, 227 (2. 2% of total deaths) 9. Kidney disease (Nephritis, nephrotic syndrome, and nephrosis): 48, 146 (1. 8% of total deaths) 10. Intentional self-harm (suicide): 42, 773 (1. 6% of total deaths) Centers for Disease Control and Prevention (www. cdc. gov)
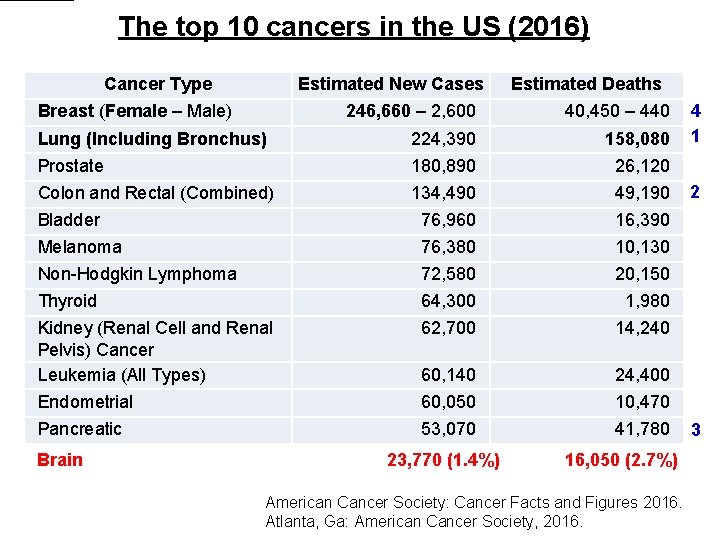
The top 10 cancers in the US (2016) Cancer Type Estimated New Cases Breast (Female – Male) Estimated Deaths 246, 660 – 2, 600 40, 450 – 440 Lung (Including Bronchus) 224, 390 158, 080 Prostate 180, 890 26, 120 Colon and Rectal (Combined) 134, 490 49, 190 Bladder 76, 960 16, 390 Melanoma 76, 380 10, 130 Non-Hodgkin Lymphoma 72, 580 20, 150 Thyroid 64, 300 1, 980 Kidney (Renal Cell and Renal Pelvis) Cancer Leukemia (All Types) 62, 700 14, 240 60, 140 24, 400 Endometrial 60, 050 10, 470 Pancreatic 53, 070 41, 780 4 1 2 3 Brain 23, 770 (1. 4%) 16, 050 (2. 7%) American Cancer Society: Cancer Facts and Figures 2016. Atlanta, Ga: American Cancer Society, 2016.

Brain tumor (BT) : statistics (incidence, death) - BTs are the most common cancer among those age 0 -19 (leukemia is the second) - BTs are the second leading cause of cancer-related deaths in children (males and females) under age 20 (leukemia is the first) American Brain Tumor Association (www. abta. org)

Brain tumor (BT) : general information - BT is an abnormal growth of tissue in the brain or central spine that can disrupt proper brain function - BTs may be primary (starting in the brain) or secondary (spreading to the brain from another area of body). Secondary BT (Metastatic BT) is about 5 -10 times more common than primary BT - Primary BT may be benign or malignant. Benign BT grows slowly, has distinct boundaries, and rarely spreads. Malignant (cancerous) BT grows quickly, has irregular boundaries, and spreads to nearby brain areas - BTs are named after the cell type which they grow. There are more than 120 histologically distinct types of primary brain and central nervous system tumors
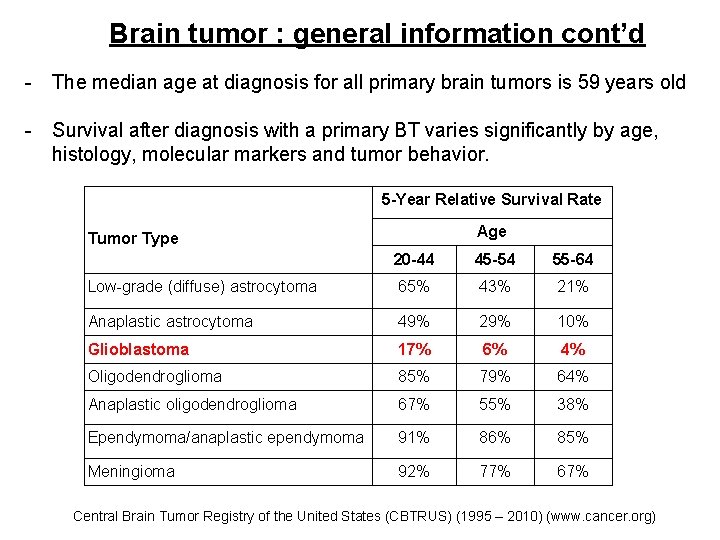
Brain tumor : general information cont’d - The median age at diagnosis for all primary brain tumors is 59 years old - Survival after diagnosis with a primary BT varies significantly by age, histology, molecular markers and tumor behavior. 5 -Year Relative Survival Rate Age Tumor Type 20 -44 45 -54 55 -64 Low-grade (diffuse) astrocytoma 65% 43% 21% Anaplastic astrocytoma 49% 29% 10% Glioblastoma 17% 6% 4% Oligodendroglioma 85% 79% 64% Anaplastic oligodendroglioma 67% 55% 38% Ependymoma/anaplastic ependymoma 91% 86% 85% Meningioma 92% 77% 67% Central Brain Tumor Registry of the United States (CBTRUS) (1995 – 2010) (www. cancer. org)

Brain tumor : statistics (cancer type) - Meningiomas represent 34 -36% of all primary brain tumors, making them the most common primary brain tumor. There will be an estimated 24, 880 new cases in 2016 - Gliomas, a broad term which includes all tumors arising from the gluey or supportive tissue of the brain, represent 27 -30% of all brain tumors and 80% of all malignant tumors - Glioblastomas represent 15. 1% of all primary brain tumors, and 55. 1% of all gliomas - Glioblastoma has the highest number of cases of all malignant tumors - Astrocytomas, including glioblastoma, represent approximately 75% of all gliomas American Brain Tumor Association (www. abta. org)

Brain tumor treatments Brain tumor treatment depends on a number of factors, including the type, location, size, and grade of the tumor, as well as the age and health of the patient. - (Image guided) Surgery - Radiation therapy (e. g. , conventional radiation therapy, stereotactic radiosurgery) - Chemotherapy - Targeted therapy (e. g. , anti-angiogenesis therapy) - Alternating electric field therapy (tumor treating fields) for recurrent GBM www. cancer. net
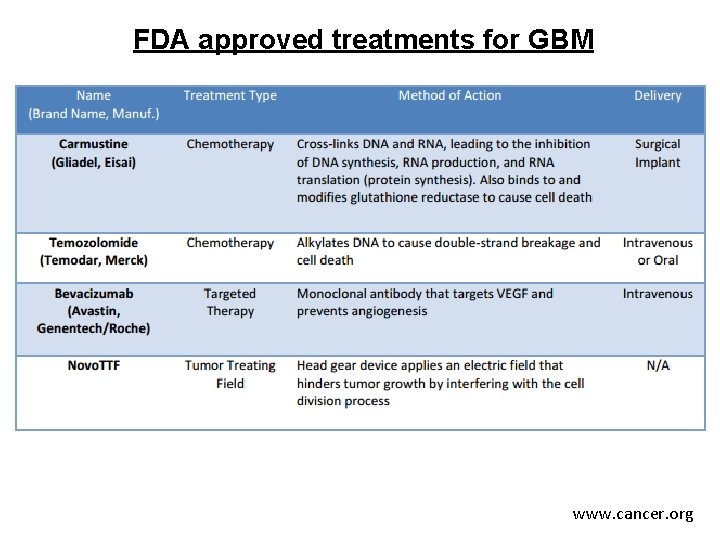
FDA approved treatments for GBM www. cancer. org

Stupp R et al. N Engl J Med 2005
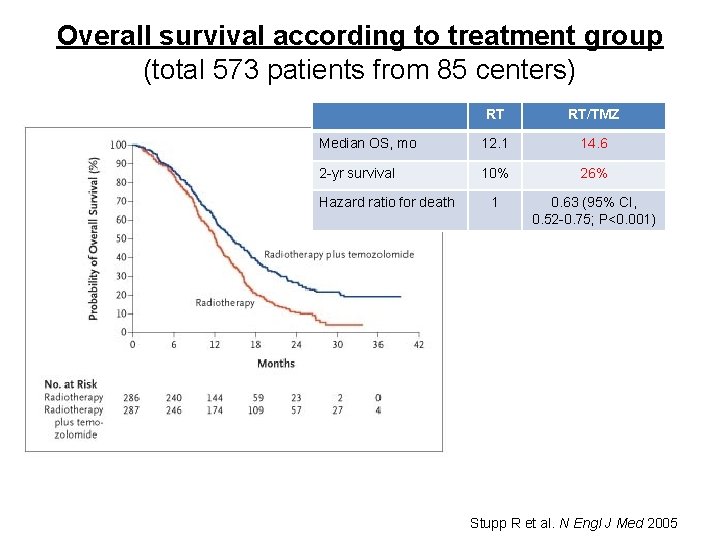
Overall survival according to treatment group (total 573 patients from 85 centers) RT RT/TMZ Median OS, mo 12. 1 14. 6 2 -yr survival 10% 26% 1 0. 63 (95% CI, 0. 52 -0. 75; P<0. 001) Hazard ratio for death Stupp R et al. N Engl J Med 2005
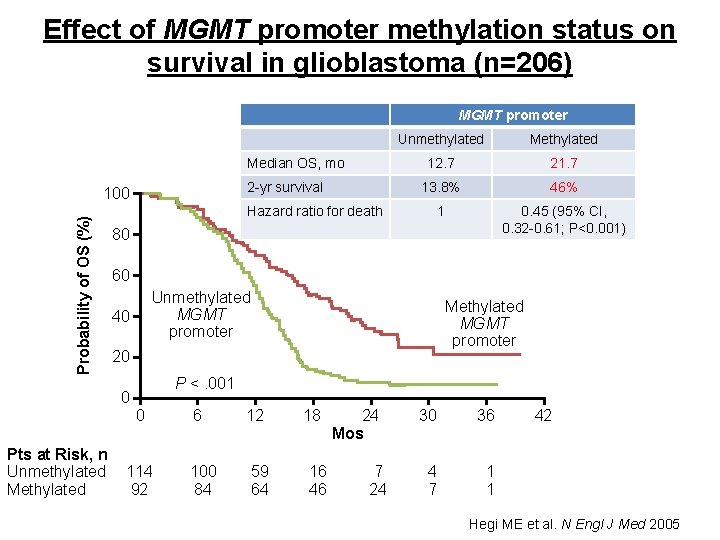
Effect of MGMT promoter methylation status on survival in glioblastoma (n=206) MGMT promoter EORTC/NCIC Trial Median OS, mo 2 -yr survival Probability of OS (%) 100 Methylated 12. 7 21. 7 13. 8% 46% 1 0. 45 (95% CI, 0. 32 -0. 61; P<0. 001) Hazard ratio for death 80 60 Unmethylated MGMT promoter 40 Methylated MGMT promoter 20 0 Pts at Risk, n Unmethylated Methylated Unmethylated P <. 001 0 114 92 6 100 84 12 59 64 18 16 46 24 Mos 7 24 30 36 4 7 1 1 42 Hegi ME et al. N Engl J Med 2005

Standard care for glioblastoma For people with glioblastoma, the latest standard of care is radiation therapy with daily low-dose temozolomide (Temodar), followed by monthly doses of temozolomide after radiation therapy for six months to one year However, the development of TMZ resistance during adjuvant therapy occurs in >30% of patients In addition, it is a challenge in overcoming doselimiting myelo-suppressive toxicity of TMZ while maintaining its efficacy
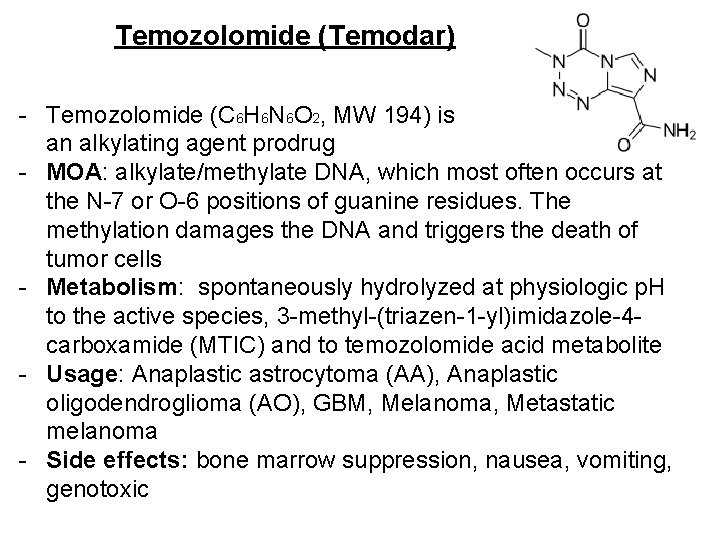
Temozolomide (Temodar) - Temozolomide (C 6 H 6 N 6 O 2, MW 194) is an alkylating agent prodrug - MOA: alkylate/methylate DNA, which most often occurs at the N-7 or O-6 positions of guanine residues. The methylation damages the DNA and triggers the death of tumor cells - Metabolism: spontaneously hydrolyzed at physiologic p. H to the active species, 3 -methyl-(triazen-1 -yl)imidazole-4 carboxamide (MTIC) and to temozolomide acid metabolite - Usage: Anaplastic astrocytoma (AA), Anaplastic oligodendroglioma (AO), GBM, Melanoma, Metastatic melanoma - Side effects: bone marrow suppression, nausea, vomiting, genotoxic
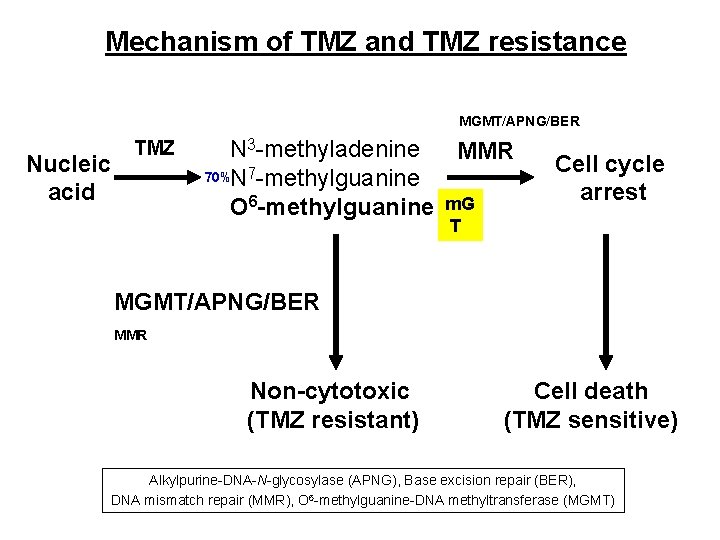
Mechanism of TMZ and TMZ resistance MGMT/APNG/BER Nucleic acid TMZ N 3 -methyladenine MMR 70%N 7 -methylguanine O 6 -methylguanine m. G Cell cycle arrest T MGMT/APNG/BER MMR Non-cytotoxic (TMZ resistant) Cell death (TMZ sensitive) Alkylpurine-DNA-N-glycosylase (APNG), Base excision repair (BER), DNA mismatch repair (MMR), O 6 -methylguanine-DNA methyltransferase (MGMT)
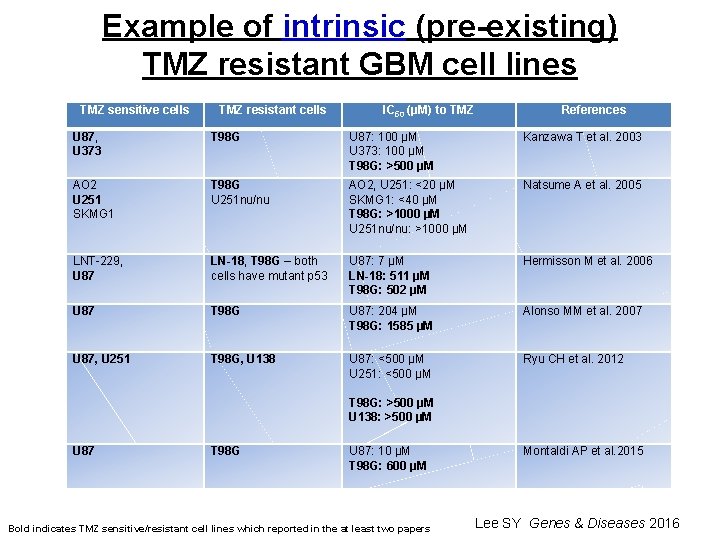
Example of intrinsic (pre-existing) TMZ resistant GBM cell lines TMZ sensitive cells TMZ resistant cells IC 50 (µM) to TMZ References U 87, U 373 T 98 G U 87: 100 µM U 373: 100 µM T 98 G: >500 µM Kanzawa T et al. 2003 AO 2 U 251 SKMG 1 T 98 G U 251 nu/nu AO 2, U 251: <20 µM SKMG 1: <40 µM T 98 G: >1000 µM U 251 nu/nu: >1000 µM Natsume A et al. 2005 LNT-229, U 87 LN-18, T 98 G – both cells have mutant p 53 U 87: 7 µM LN-18: 511 µM T 98 G: 502 µM Hermisson M et al. 2006 U 87 T 98 G U 87: 204 µM T 98 G: 1585 µM Alonso MM et al. 2007 U 87, U 251 T 98 G, U 138 U 87: <500 µM U 251: <500 µM T 98 G: >500 µM U 138: >500 µM Ryu CH et al. 2012 U 87 T 98 G U 87: 10 µM T 98 G: 600 µM Montaldi AP et al. 2015 Bold indicates TMZ sensitive/resistant cell lines which reported in the at least two papers Lee SY Genes & Diseases 2016
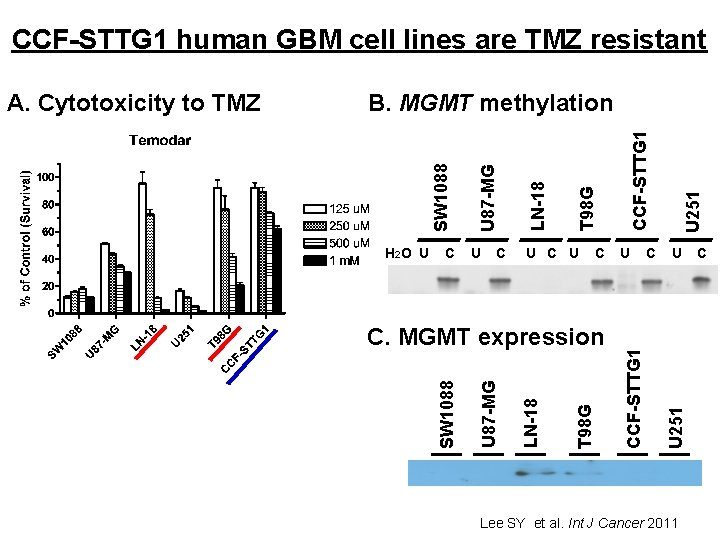
CCF-STTG 1 human GBM cell lines are TMZ resistant U 251 CCF-STTG 1 T 98 G LN-18 U 87 -MG SW 1088 B. MGMT methylation U 251 T 98 G LN-18 U 87 -MG C. MGMT expression CCF-STTG 1 H 2 O U C U C SW 1088 A. Cytotoxicity to TMZ Lee SY et al. Int J Cancer 2011
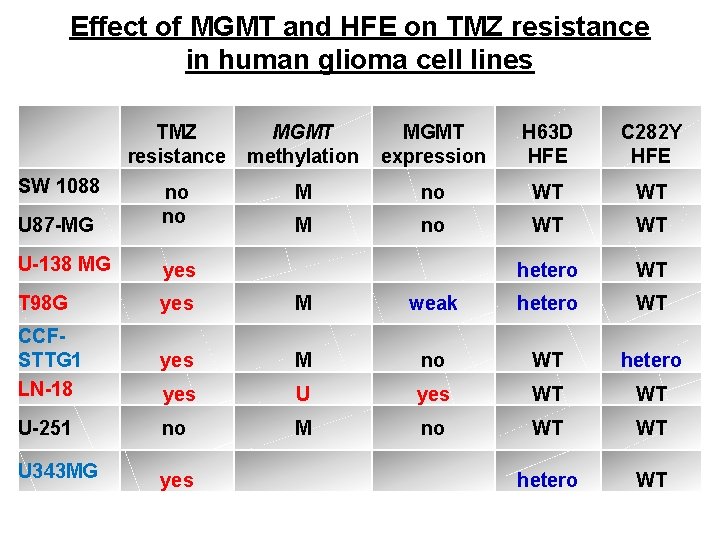
Effect of MGMT and HFE on TMZ resistance in human glioma cell lines TMZ resistance MGMT methylation MGMT expression H 63 D HFE C 282 Y HFE M no WT WT U 87 -MG no no M no WT WT U-138 MG yes hetero WT T 98 G yes M weak hetero WT CCFSTTG 1 LN-18 yes M no WT hetero yes U yes WT WT U-251 no M no WT WT U 343 MG yes hetero WT SW 1088
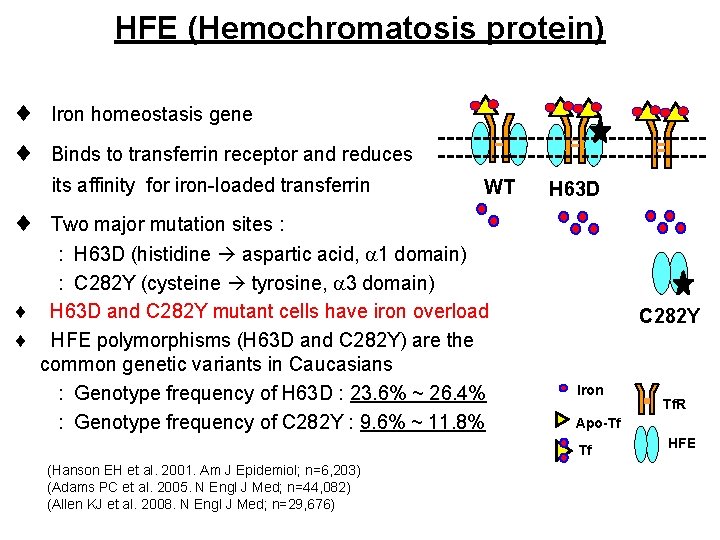
HFE (Hemochromatosis protein) Iron homeostasis gene Binds to transferrin receptor and reduces its affinity for iron-loaded transferrin WT H 63 D Two major mutation sites : H 63 D (histidine aspartic acid, 1 domain) : C 282 Y (cysteine tyrosine, 3 domain) H 63 D and C 282 Y mutant cells have iron overload HFE polymorphisms (H 63 D and C 282 Y) are the common genetic variants in Caucasians : Genotype frequency of H 63 D : 23. 6% ~ 26. 4% : Genotype frequency of C 282 Y : 9. 6% ~ 11. 8% C 282 Y Iron Apo-Tf Tf (Hanson EH et al. 2001. Am J Epidemiol; n=6, 203) (Adams PC et al. 2005. N Engl J Med; n=44, 082) (Allen KJ et al. 2008. N Engl J Med; n=29, 676) Tf. R HFE
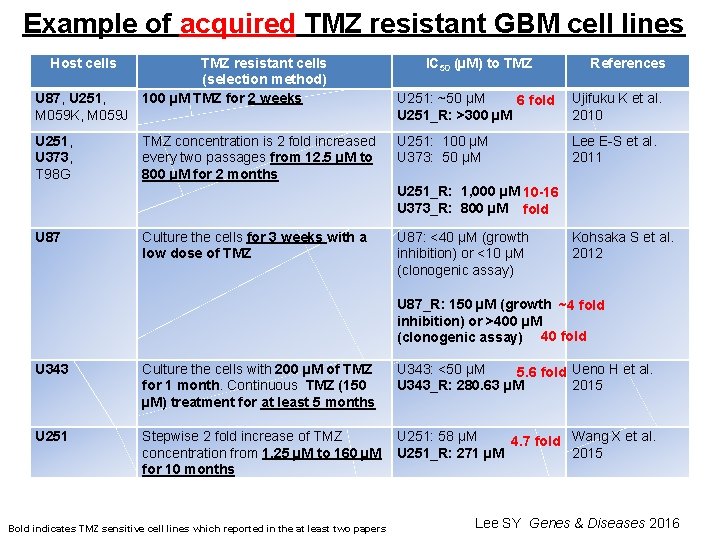
Example of acquired TMZ resistant GBM cell lines Host cells U 87, U 251, M 059 K, M 059 J TMZ resistant cells (selection method) 100 µM TMZ for 2 weeks IC 50 (µM) to TMZ U 251: ~50 µM 6 fold U 251_R: >300 µM References Ujifuku K et al. 2010 U 251, U 373, T 98 G TMZ concentration is 2 fold increased every two passages from 12. 5 µM to 800 µM for 2 months U 251: 100 µM Lee E-S et al. U 373: 50 µM 2011 U 251_R: 1, 000 µM 10 -16 U 373_R: 800 µM fold U 87 Culture the cells for 3 weeks with a low dose of TMZ U 87: <40 µM (growth Kohsaka S et al. inhibition) or <10 µM 2012 (clonogenic assay) U 87_R: 150 µM (growth ~4 fold inhibition) or >400 µM (clonogenic assay) 40 fold U 343 Culture the cells with 200 µM of TMZ for 1 month. Continuous TMZ (150 µM) treatment for at least 5 months U 343: <50 µM 5. 6 fold Ueno H et al. U 343_R: 280. 63 µM 2015 U 251 Stepwise 2 fold increase of TMZ U 251: 58 µM 4. 7 fold Wang X et al. concentration from 1. 25 µM to 160 µM U 251_R: 271 µM 2015 for 10 months Bold indicates TMZ sensitive cell lines which reported in the at least two papers Lee SY Genes & Diseases 2016
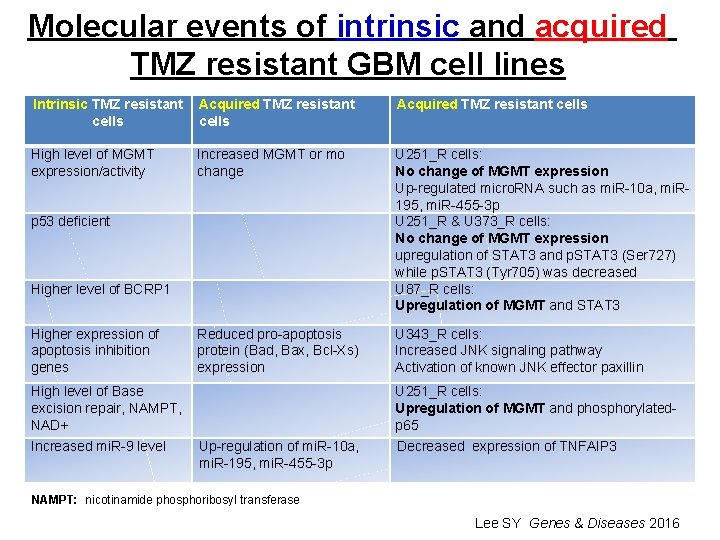
Molecular events of intrinsic and acquired TMZ resistant GBM cell lines Intrinsic TMZ resistant Acquired TMZ resistant cells High level of MGMT expression/activity Increased MGMT or mo change U 251_R cells: No change of MGMT expression Up-regulated micro. RNA such as mi. R-10 a, mi. R 195, mi. R-455 -3 p U 251_R & U 373_R cells: No change of MGMT expression upregulation of STAT 3 and p. STAT 3 (Ser 727) while p. STAT 3 (Tyr 705) was decreased U 87_R cells: Upregulation of MGMT and STAT 3 Reduced pro-apoptosis protein (Bad, Bax, Bcl-Xs) expression U 343_R cells: Increased JNK signaling pathway Activation of known JNK effector paxillin p 53 deficient Higher level of BCRP 1 Higher expression of apoptosis inhibition genes High level of Base excision repair, NAMPT, NAD+ Increased mi. R-9 level U 251_R cells: Upregulation of MGMT and phosphorylatedp 65 Up-regulation of mi. R-10 a, mi. R-195, mi. R-455 -3 p Decreased expression of TNFAIP 3 NAMPT: nicotinamide phosphoribosyl transferase Lee SY Genes & Diseases 2016
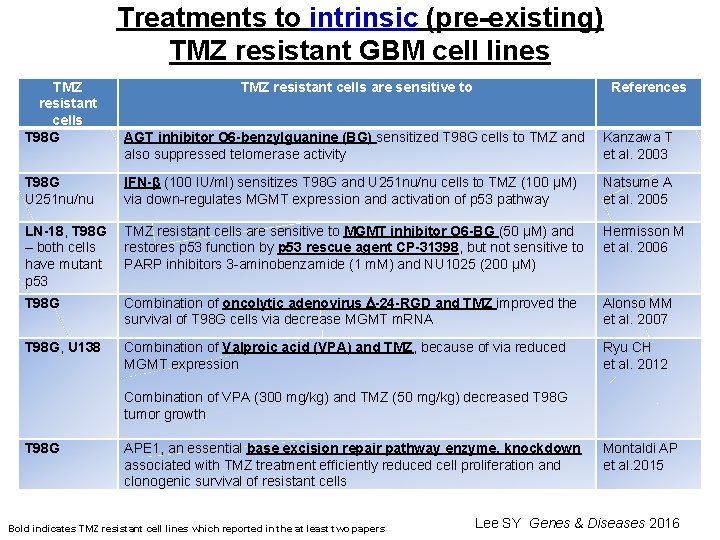
Treatments to intrinsic (pre-existing) TMZ resistant GBM cell lines TMZ resistant cells T 98 G U 251 nu/nu TMZ resistant cells are sensitive to References AGT inhibitor O 6 -benzylguanine (BG) sensitized T 98 G cells to TMZ and also suppressed telomerase activity Kanzawa T et al. 2003 IFN-β (100 IU/ml) sensitizes T 98 G and U 251 nu/nu cells to TMZ (100 µM) via down-regulates MGMT expression and activation of p 53 pathway Natsume A et al. 2005 LN-18, T 98 G TMZ resistant cells are sensitive to MGMT inhibitor O 6 -BG (50 µM) and – both cells restores p 53 function by p 53 rescue agent CP-31398, but not sensitive to have mutant PARP inhibitors 3 -aminobenzamide (1 m. M) and NU 1025 (200 µM) p 53 Hermisson M et al. 2006 T 98 G Combination of oncolytic adenovirus -24 -RGD and TMZ improved the survival of T 98 G cells via decrease MGMT m. RNA Alonso MM et al. 2007 T 98 G, U 138 Combination of Valproic acid (VPA) and TMZ, because of via reduced MGMT expression Combination of VPA (300 mg/kg) and TMZ (50 mg/kg) decreased T 98 G tumor growth APE 1, an essential base excision repair pathway enzyme, knockdown associated with TMZ treatment efficiently reduced cell proliferation and clonogenic survival of resistant cells Ryu CH et al. 2012 T 98 G Bold indicates TMZ resistant cell lines which reported in the at least two papers Montaldi AP et al. 2015 Lee SY Genes & Diseases 2016

Treatments of acquired TMZ resistant GBM cell lines TMZ resistant cells are sensitive to References U 251_R mi. R-195 knockdown showed moderate growth inhibition to U 251_R cells combined treatment with both mi. R-10 a or mi. R-455 -3 p inhibitors with TMZ showed better cytotoxicity Ujifuku K et al. 2010 U 251_R U 373_R STAT 3 si. RNA sensitize TMZ resistant cells Lee E-S et al. 2011 U 87_R STAT 3 inhibitor Kohsaka S et al. 2012 U 343_R JNK inhibitor SP 600125 or JNK si. RNA suppressed upregulation of invasiveness Ueno H et al. 2015 U 251_R Ik. Bα inhibitor BAY 11 -7082 sensitize TMZ resistant cells growth Combination effect of Ik. Bα inhibitor and TMZ on TMZ resistant cells Wang X et al. 2015 Bold indicates TMZ sensitive cell lines which reported in the at least two papers Lee SY Genes & Diseases 2016
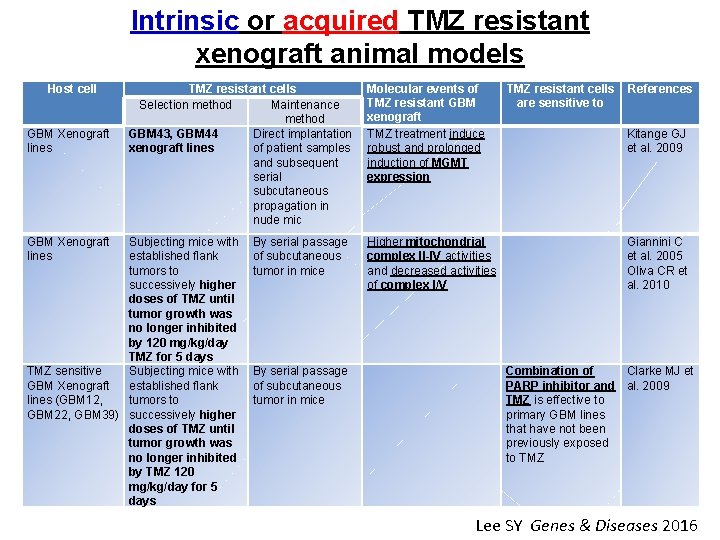
Intrinsic or acquired TMZ resistant xenograft animal models Host cell GBM Xenograft lines TMZ resistant cells Selection method Maintenance method GBM 43, GBM 44 Direct implantation xenograft lines of patient samples and subsequent serial subcutaneous propagation in nude mic Subjecting mice with established flank tumors to successively higher doses of TMZ until tumor growth was no longer inhibited by 120 mg/kg/day TMZ for 5 days TMZ sensitive Subjecting mice with GBM Xenograft established flank lines (GBM 12, tumors to GBM 22, GBM 39) successively higher doses of TMZ until tumor growth was no longer inhibited by TMZ 120 mg/kg/day for 5 days Molecular events of TMZ resistant GBM xenograft TMZ treatment induce robust and prolonged induction of MGMT expression TMZ resistant cells References are sensitive to Kitange GJ et al. 2009 By serial passage of subcutaneous tumor in mice Higher mitochondrial complex II-IV activities and decreased activities of complex I/V Giannini C et al. 2005 Oliva CR et al. 2010 By serial passage of subcutaneous tumor in mice Combination of Clarke MJ et PARP inhibitor and al. 2009 TMZ is effective to primary GBM lines that have not been previously exposed to TMZ Lee SY Genes & Diseases 2016
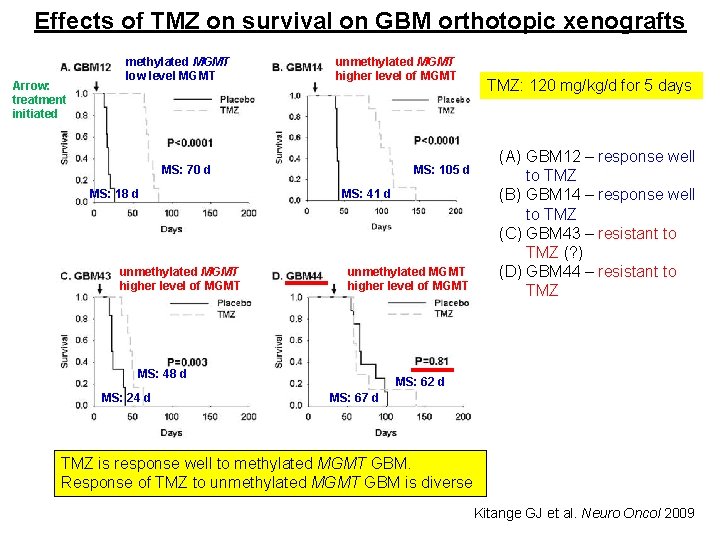
Effects of TMZ on survival on GBM orthotopic xenografts Arrow: treatment initiated methylated MGMT low level MGMT unmethylated MGMT higher level of MGMT MS: 70 d MS: 18 d unmethylated MGMT higher level of MGMT MS: 105 d MS: 41 d unmethylated MGMT higher level of MGMT MS: 48 d MS: 24 d TMZ: 120 mg/kg/d for 5 days (A) GBM 12 – response well to TMZ (B) GBM 14 – response well to TMZ (C) GBM 43 – resistant to TMZ (? ) (D) GBM 44 – resistant to TMZ MS: 62 d MS: 67 d TMZ is response well to methylated MGMT GBM. Response of TMZ to unmethylated MGMT GBM is diverse Kitange GJ et al. Neuro Oncol 2009
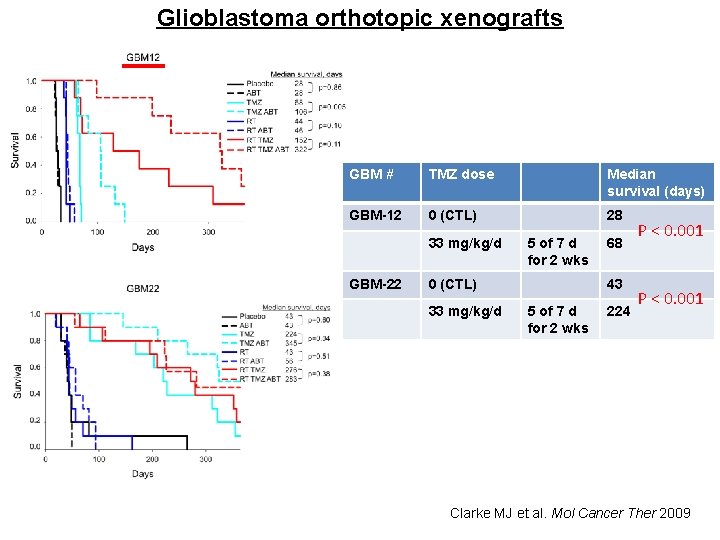
Glioblastoma orthotopic xenografts GBM # TMZ dose Median survival (days) GBM-12 0 (CTL) 28 33 mg/kg/d GBM-22 5 of 7 d for 2 wks 0 (CTL) 33 mg/kg/d 68 43 5 of 7 d for 2 wks 224 P < 0. 001 Clarke MJ et al. Mol Cancer Ther 2009
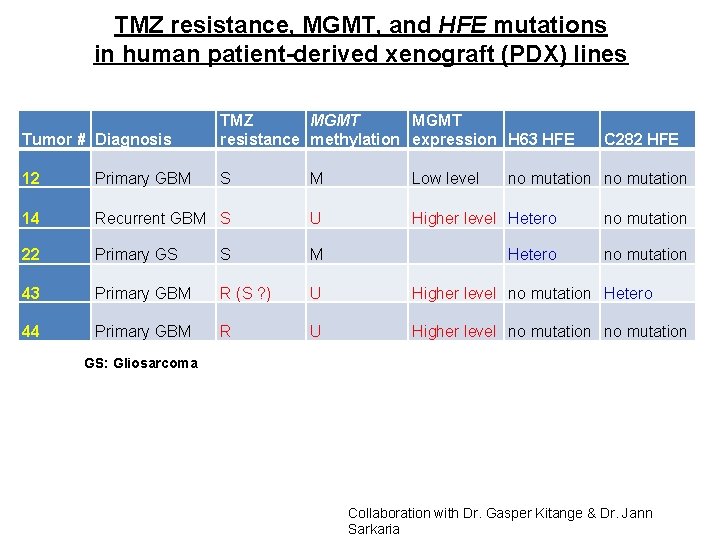
TMZ resistance, MGMT, and HFE mutations in human patient-derived xenograft (PDX) lines Tumor # Diagnosis TMZ MGMT resistance methylation expression H 63 HFE 12 Primary GBM S M Low level 14 Recurrent GBM S U Higher level Hetero no mutation 22 Primary GS S M Hetero no mutation 43 Primary GBM R (S ? ) U Higher level no mutation Hetero 44 Primary GBM R U Higher level no mutation C 282 HFE no mutation GS: Gliosarcoma Collaboration with Dr. Gasper Kitange & Dr. Jann Sarkaria
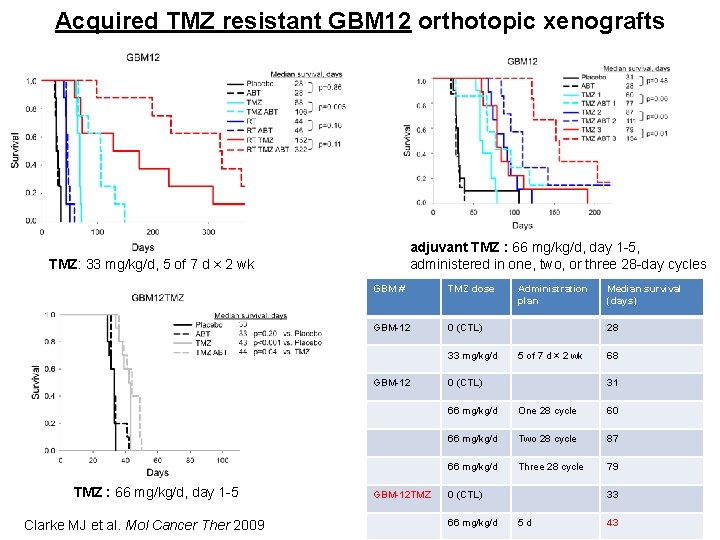
Acquired TMZ resistant GBM 12 orthotopic xenografts adjuvant TMZ : 66 mg/kg/d, day 1 -5, administered in one, two, or three 28 -day cycles TMZ: 33 mg/kg/d, 5 of 7 d × 2 wk GBM # TMZ dose GBM-12 0 (CTL) 33 mg/kg/d GBM-12 TMZ : 66 mg/kg/d, day 1 -5 Clarke MJ et al. Mol Cancer Ther 2009 GBM-12 TMZ Administration plan Median survival (days) 28 5 of 7 d × 2 wk 0 (CTL) 68 31 66 mg/kg/d One 28 cycle 60 66 mg/kg/d Two 28 cycle 87 66 mg/kg/d Three 28 cycle 79 0 (CTL) 66 mg/kg/d 33 5 d 43
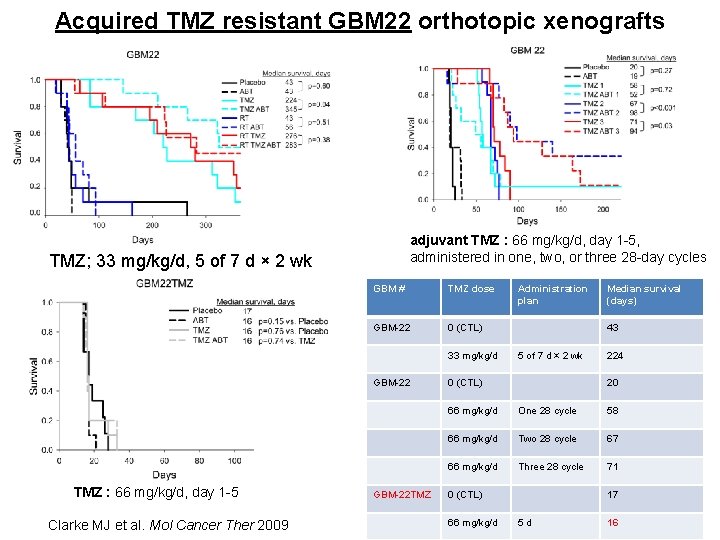
Acquired TMZ resistant GBM 22 orthotopic xenografts adjuvant TMZ : 66 mg/kg/d, day 1 -5, administered in one, two, or three 28 -day cycles TMZ; 33 mg/kg/d, 5 of 7 d × 2 wk GBM # TMZ dose GBM-22 0 (CTL) 33 mg/kg/d GBM-22 TMZ : 66 mg/kg/d, day 1 -5 Clarke MJ et al. Mol Cancer Ther 2009 GBM-22 TMZ Administration plan Median survival (days) 43 5 of 7 d × 2 wk 0 (CTL) 224 20 66 mg/kg/d One 28 cycle 58 66 mg/kg/d Two 28 cycle 67 66 mg/kg/d Three 28 cycle 71 0 (CTL) 66 mg/kg/d 17 5 d 16

Testing TMZ in animal models of glioma: 60 papers up to 2013 Rat Gliosarcoma Human Glioblastoma Rat Glioblastoma Human Glioma Rat Glioblastoma Human Glioblastoma Red line: TMZ resistant Blue line: TMZ sensitive Human Glioblastoma Hirst TC et al. Br J Cancer 2013
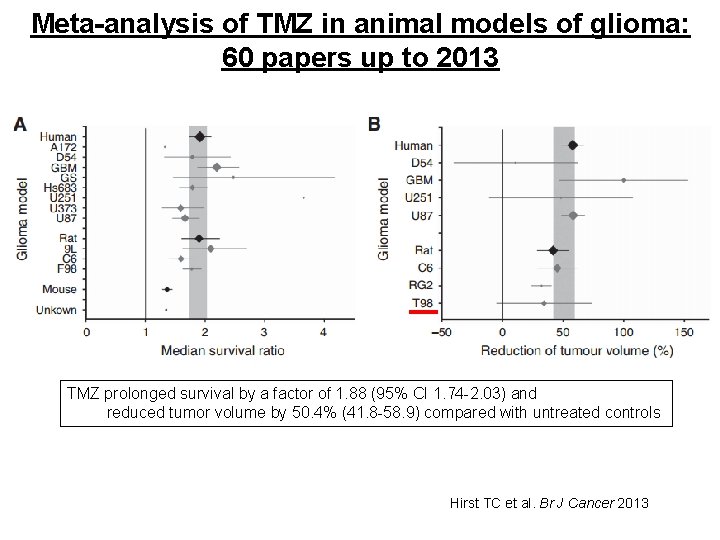
Meta-analysis of TMZ in animal models of glioma: 60 papers up to 2013 TMZ prolonged survival by a factor of 1. 88 (95% CI 1. 74 -2. 03) and reduced tumor volume by 50. 4% (41. 8 -58. 9) compared with untreated controls Hirst TC et al. Br J Cancer 2013
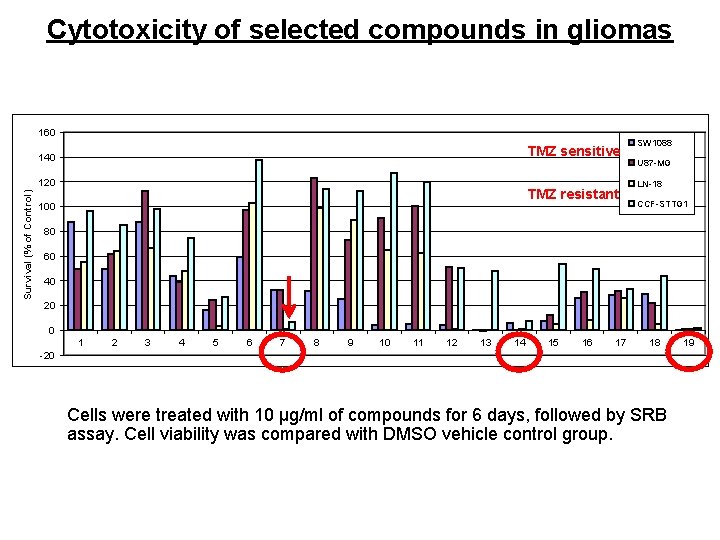
Cytotoxicity of selected compounds in gliomas 160 TMZ sensitive 140 Survival (% of Control) 120 TMZ resistant 100 SW 1088 U 87 -MG LN-18 CCF-STTG 1 80 60 40 20 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 -20 Cells were treated with 10 µg/ml of compounds for 6 days, followed by SRB assay. Cell viability was compared with DMSO vehicle control group. 19
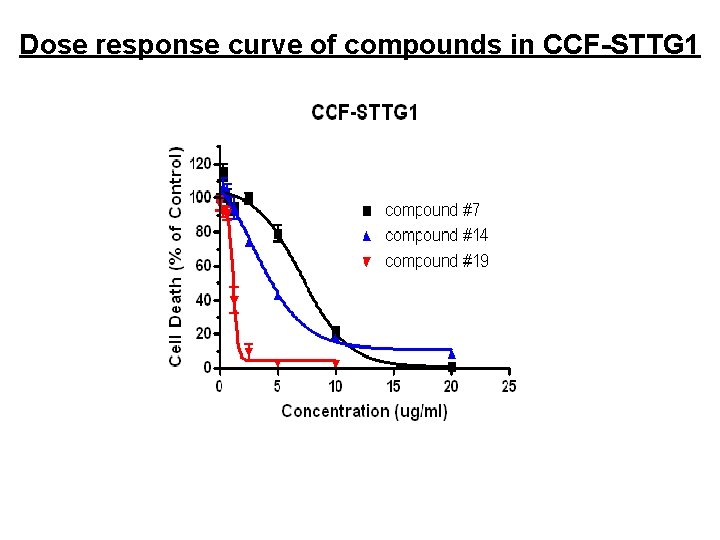
Dose response curve of compounds in CCF-STTG 1
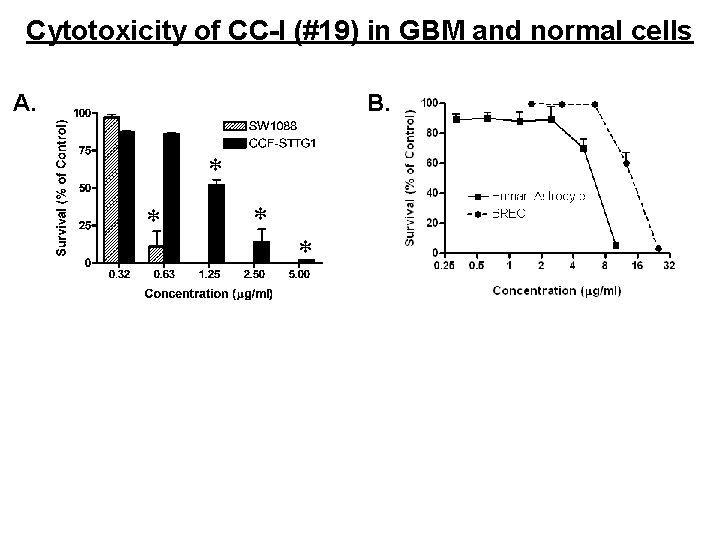
Cytotoxicity of CC-I (#19) in GBM and normal cells A. B.
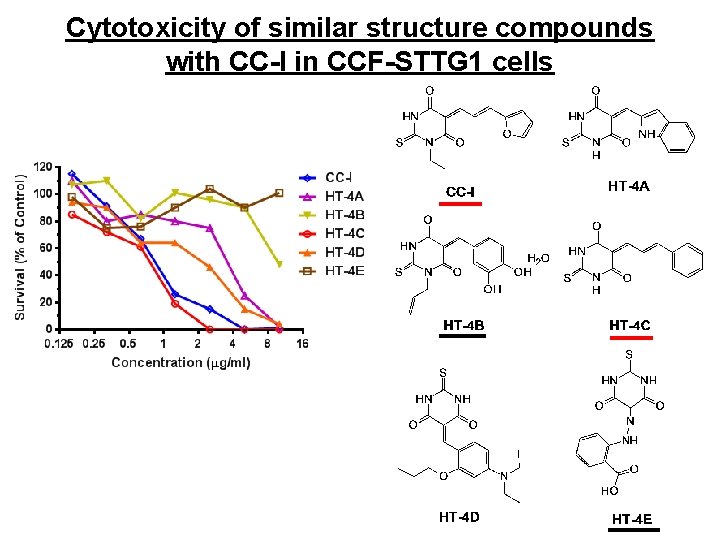
Cytotoxicity of similar structure compounds with CC-I in CCF-STTG 1 cells
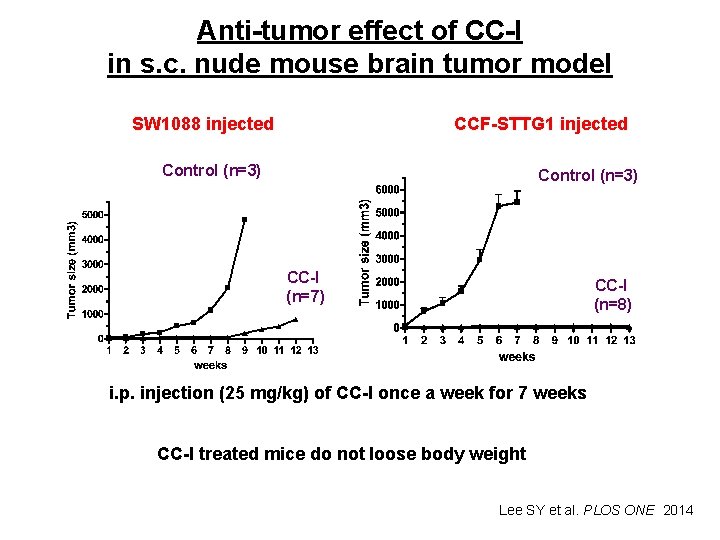
Anti-tumor effect of CC-I in s. c. nude mouse brain tumor model SW 1088 injected CCF-STTG 1 injected Control (n=3) CC-I (n=7) CC-I (n=8) i. p. injection (25 mg/kg) of CC-I once a week for 7 weeks CC-I treated mice do not loose body weight Lee SY et al. PLOS ONE 2014
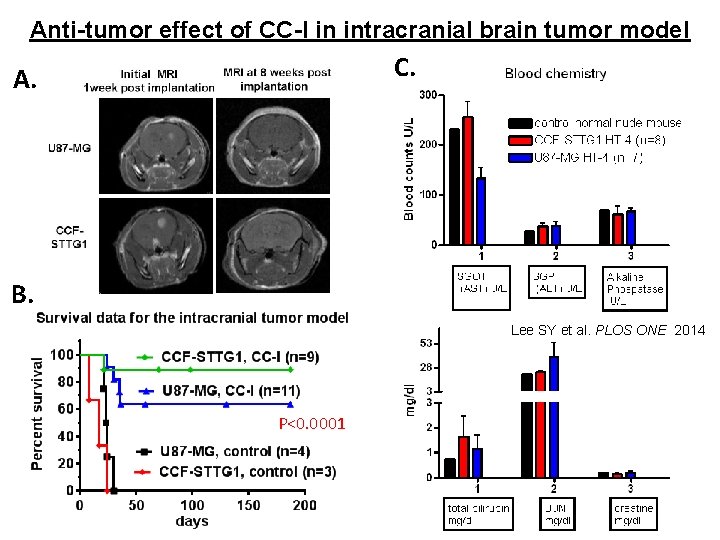
Anti-tumor effect of CC-I in intracranial brain tumor model C. A. B. Lee SY et al. PLOS ONE 2014 P<0. 0001
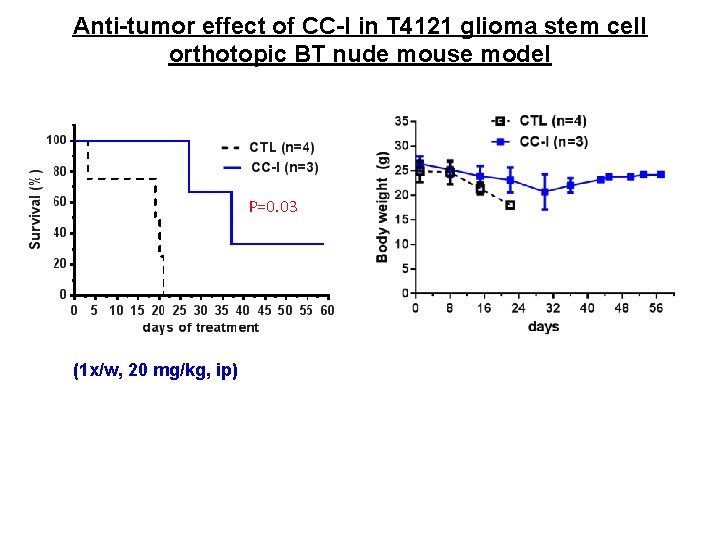
Anti-tumor effect of CC-I in T 4121 glioma stem cell orthotopic BT nude mouse model P=0. 03 (1 x/w, 20 mg/kg, ip)

Summary ■ We need a better understanding for the TMZ resistant (recurrent) GBM ■ There are distinct molecular events between intrinsic TMZ resistant GBM and acquired (recurrent) TMZ resistant GBM. Therefore, we should treat them different way. ■ TMZ resistance is not mediated by a single molecular event but by multiple ones. Therefore, identification of GBM patients based on the patient’s gene/protein profiling data could be beneficial for selecting drugs for their treatment ■ CC-I showed strong anti-tumor effect on GBM in in vivo tumor models

Acknowledgements Dr. James Connor Ms. Becky Webb Dr. Mohammed Alsaidi Drug Discovery, Development and Delivery Core MRI Imaging Core Facility, Solution Phase NMR Core Facility Dr. Gasper Kitange & Dr. Jann Sarkaria (Mayo Clinic) Penn State Hershey Cancer Institute Experimental Therapeutics Research initiative NIH/NCI R 21 CA 167406 Elsa U. Pardee Foundation Tara Leah Witmer Endowment

Questions
- Slides: 43